Abstract
Purpose:
The aim of our study is to investigate the bone ongrowth of two different alternative surfaces and the effect of nonsteroidal anti-inflammatory drugs (NSAIDs) on osseointegration.
Methods:
Hips of 40 New Zealand white rabbits were operated bilaterally. Hydroxyapatite (HA)-coated titanium rods were implanted into the right femur, and grit-blasted titanium rods were implanted into the left femur. They were divided into three groups. At the end of 8 weeks, both femora of the rabbits were removed and investigated biomechanically and histologically.
Results:
HA-coated implants had a significantly better failure load and “percentage of bone–implant contact” than grit-blasted implants. There was no significant difference between the medication groups as a result of the biomechanical and histologic investigations.
Conclusions:
Our results indicate that NSAIDs did not have any negative effect on the osseointegration. HA-coated implants may provide more tensile strength and greater bone–implant contact rate in comparison with grit-blasted implants.
Introduction
Long-term survival of total hip arthroplasty relies on a stable bone–implant interface. Primary stability is provided with press-fit implantation of cementless stems, while secondary stability is achieved with successful osseointegration. The term osseointegration refers to the direct structural and functional connection, without intervening tissue, between living bone and the surface of the implants. Many factors can affect the osseointegration process, such as primary stability of the stem and stem design, the bisphosphonates used during the early postoperative period, and bone density. In this study, we analyzed the effect of two controversial issues on osseointegration; hydroxyapatite (HA) coating and nonsteroidal anti-inflammatory drug (NSAID) use.
It has already been shown that porous-coated and grit-blasted surfaces with press-fit implantation improved osseointegration. 1,2,3 The coatings of implant surfaces with osseoconductive materials, for example, HA, are being increasingly investigated, and the positive effects of HA coating have also been demonstrated in many experimental studies in the last 20 years. 4,5,6 However, some of the studies investigating possible clinical or radiological effects of hydroxyapatite coating on femoral components failed to show any significant benefit. 7,8 One of the most important points that our study focuses on is the effect of HA coating, by means of the comparison of two different types of implant surface in the same rabbit model.
NSAIDs are commonly prescribed for painful and inflammatory conditions, including traumatic injuries, arthritis, and postsurgical pain because of their well-known anti-inflammatory, antipyretic, and analgesic capabilities. The effects of these drugs are due to their inhibition of prostaglandin synthesis, through two cyclooxygenase enzymes (COX-1 and COX-2). Recently, more specific NSAIDs that are primarily COX-2 inhibitors have been introduced to the market in order to prevent the adverse effects of COX-1 inhibition that impair the normal cellular functions. Although the exact physiological role of the prostaglandins in bone homeostasis remains unclear, investigators tried to show the importance of the prostaglandins and COX enzyme in preclinical scientific studies. Studies conducted on the null mutation in the COX-2 gene and EP4 receptor knockout mice also gave us important information about the role of the prostaglandins and COX-2 enzyme on bone metabolism.
The effect of these drugs on the osseointegration after press-fit implantation has remained controversial. Studies examining osseointegration have mainly focused on the comparison of selective and nonselective NSAIDs treatment, as well as the duration of treatment. İn the experimental studies, the effects of NSAIDs on osseointegration can vary both positively and negatively. The negative effects of NSAIDs on osseointegration were shown in experimental studies 9 –13 ; however, in other studies, no negative effects were shown. 14,15 Recently, Cai et al. show that postoperatively 7-day regimen of appropriate doses, selective and nonselective NSAID’s dose, not adversely effect osseointegration. 16
This study investigated the effect of NSAID use on osseointegration in a rabbit model using both HA- and grit-blasted titanium implants. The quality of osseointegration is investigated quantitatively, using biomechanical and histologic parameters.
Materials and methods
Implants
Solid, cylindrical, titanium-alloy (Ti-6Al-4V extra-low interstitial) rods, 3 mm in diameter and 55 mm in length, were used in the study (Figure 1). The proximal portion of the implants had a hook-shaped design for easy manipulation during biomechanical testing. The proximal 45 mm of all implants was blasted with aluminum oxide particles to achieve a 3-µm mean surface roughness (Sayan Medical Equipment Company, Izmir, Turkey). Half of the implants were coated with HA powder on grit-blasted surface (HAP XPT-D-703), with 88.4 µm thickness (Bio-vac, Espana S.A, Spain). Gamma irradiation was used for implant sterilization.

(a) Grit-blasted (uncoated) titanium implant. (b) Grid-blasted + HA-coated (HA coated) titanium implant. (c) Scanning electron microscope imaging of uncoated titanium implant surface (100× magnification). (d) Scanning electron microscope imaging of HA-coated titanium implant surface (100× magnification). HA: hydroxyapatite.
Operative technique
Forty skeletally mature adult New Zealand white rabbits with a body mass between 3.5 kg and 4 kg were operated under sterile conditions. The animals were anesthetized with an intramuscular injection of 8 mg/kg of xylazine and 40 mg/kg of ketamine. The skin and subcutaneous tissues were injected with 1% lidocaine to obtain intra- and postoperative pain control. A prophylactic dose of 10 mg/kg of cefazolin was given by intramuscular injection before the operation and also on the first postoperative day.
Following skin preparation, a small incision was made just proximal to the greater trochanter. The edges of the skin were retracted, and the fascia lata was split to expose the gluteus medius muscle. The anterior fibers of the gluteus medius were split to expose the intertrochanteric fossa. Medullary cavities were prepared sequentially with two size reamers (2.8 and 3 mm in diameter). A grit-blasted titanium implant with HA coating (HA coated) was implanted into the right femur, and a grit-blasted titanium implant without HA coating (uncoated) was implanted into the left femur through an antegrade method during the same operative setting. Meticulous soft-tissue closure was performed. Animals were isolated in separate cages to ensure that each received only the prescribed treatment.
Groups
Animals were randomly divided into three groups: control group, diclofenac group, and celecoxib group. Permuted block randomization was used for randomization. Each block was consisted three rabbits. Six different permutations were determined and were ordered on Medcalc (version 11.3.6) software using generate random sample. Control group was fed only with regular food. The diclofenac group received 5 mg/kg/day diclofenac Na (Voltaren; Novartis Pharma AG, Basel, Switzerland) intramuscularly. 17 The celecoxib group received 3 mg/kg/day celecoxib (Celebrex; Pfizer Inc. New York, USA) in addition to regular food during 8-week follow-up period. Celecoxib was supplied in powdered form and suspended in a delivery vehicle containing 0.5% methylcellulose (catalog number MO-512; Sigma Chemical, St Louis, Missouri, USA) and 0.1% Tween 80 (Sigma Chemical) in sterile deionized water and was inserted directly into the rabbit’s mouth to ensure consistent and accurate dosing. There were no restrictions on walking and all of the wounds healed without complications.
Follow-up
Radiographs were made within 72 h after the operation to confirm the position of the implant and the absence of fractures (Figure 2). There was fracture in four femora of four rabbits on the X-rays; these broken specimens were excluded from the study, while their unaffected contralateral sides were included. A total of eight rabbits were lost during the all study (Table 1).

X-ray image of a rabbit after implantation. Right side HA coating; left side uncoated titanium implant. HA: hydroxyapatite.
All data and distribution of rabbits.

*: Fractured femurs, which are excluded from study; Co. Pm: contact perimeter; I. Pm: implant perimeter; N: Newton.
Animals were killed 8 weeks after the operation under high-dose isoflurane inhaler general anesthesia and both femora were harvested. Removed femurs were put into the plastic bag and transferred to the laboratory in the ice for the mechanical testing. Femoral specimens were sectioned perpendicular to the long axis of the femur with use of a diamond blade on a precision water-cooled sectioning saw (Exakt 300 CL, ExaktApparatbau, Norderstad, Germany) at a 30 mm distance relative to proximal end of the implant (Figure 3). The proximal parts of the specimens, which also contained the proximal 30 mm of metallic implant, were used for mechanical testing. A second cut was placed 15 mm distal to the proximal cut, producing a femoral segment of 15 mm length, containing the distal 15 mm of the implant. These femoral segments were immersed in 4% neutral buffered formaldehyde for histologic evaluation.

Section levels of the samples. Section (a) was used for biomechanical analysis; section (b) was used for histological analysis.
Mechanical testing
Pullout strength of implants was tested within 24 h after harvesting of the specimens. After placement of two parallel Kirschner wires at the intertrochanteric region (Figure 4(a)), the specimens were embedded in acrylic cement in a metal cylinder (Figure 4(b) and (c)), which was attached to the lower jaw of the testing device. The upper end of the implant was fixed to the upper jaw using a hook-shaped adapter. All specimens were subjected to a uniaxial tension test (Testometric M500-25kN; Testometric Co, Lancashire, UK) at a crosshead speed of 1.0 mm/min. Mechanical failure of the interface was defined as a sudden, marked decrease in load. Interface failure load was recorded from the load-cell transducer (Figure 5).

(a) Two Kirschner wires were inserted both side of the specimens to improve stabilization in the cement. (b) and (c) Specimens were embedded in to the test cylinders.

Interface strength–time graphic of a sample from the control group.
Histological evaluation
The specimens were dehydrated in a graded series of ethanols and embedded in methylmethacrylate-based resin (Technovit 7200 VLC, Kulzer, Wehrheim, Germany). Ground sections without decalcification from the implants and surrounding bone were taken in accordance with the method described by Donath and Breuner.
18
A section was taken from the femur, from the proximal end of the middle segment, perpendicular to the long axis (Figure 3). A sample about 100 μm from the proximal end was cut using a diamond saw. Each sample was ground to a thickness of 40 μm using an Exakt 300 CL. One section was prepared from each block. The sections were stained with toluidine blue. Images of the sections were obtained using a digital camera (Olympus DP 20-SE, Tokyo, Japan) attached to a microscope (Olympus BX51). The images were transferred to a computer to quantitative analysis using BioQuant Osteo software (version 14.1.6; BIOQUANT Image Analysis Corporation, Nashville, TN, USA). Light microscopy was used for evaluation of histological specimens. In the qualitative histological analysis, bone quality, bone–implant interface, and host reaction were investigated. All specimens were also examined under polarized light in order to qualitatively assess the formation and remodeling of bone around the implant. For quantitative evaluation, osseointegration protocol of the BioQuant Osteo software on 20× magnification was used. Quantitative parameters measured during this analysis were as follows:
Total bone area: The total bone area (TBA) was defined as the total amount of cortical bone within a predefined circular area around the implant. This area was defined as the distance between the surface of the implant and a virtual circle with the same center as the implant and 700 µm longer radius for each sample (Figure 6(a)). Borders of the newly formed bone were marked on the histological sections using the BioQuant Osteo software. The TBA, within the defined circular area, was calculated by the software (Figure 6(b)).
Bone–implant contact rate: Integration of implant with new bone originating from the nearest cortical bone was observed in all sections. Microscopic evaluation of the bone–implant contact areas revealed an intermittent integration between bone and metal, and soft-tissue islands were observed within the macroscopic contact areas. The bone–implant contact rate was measured using seg assign measurement tool of BioQuant Osteo software. Contact rate was represented as a proportion of the circumference of the implant (contact perimeter/implant perimeter, %; Figure 7).
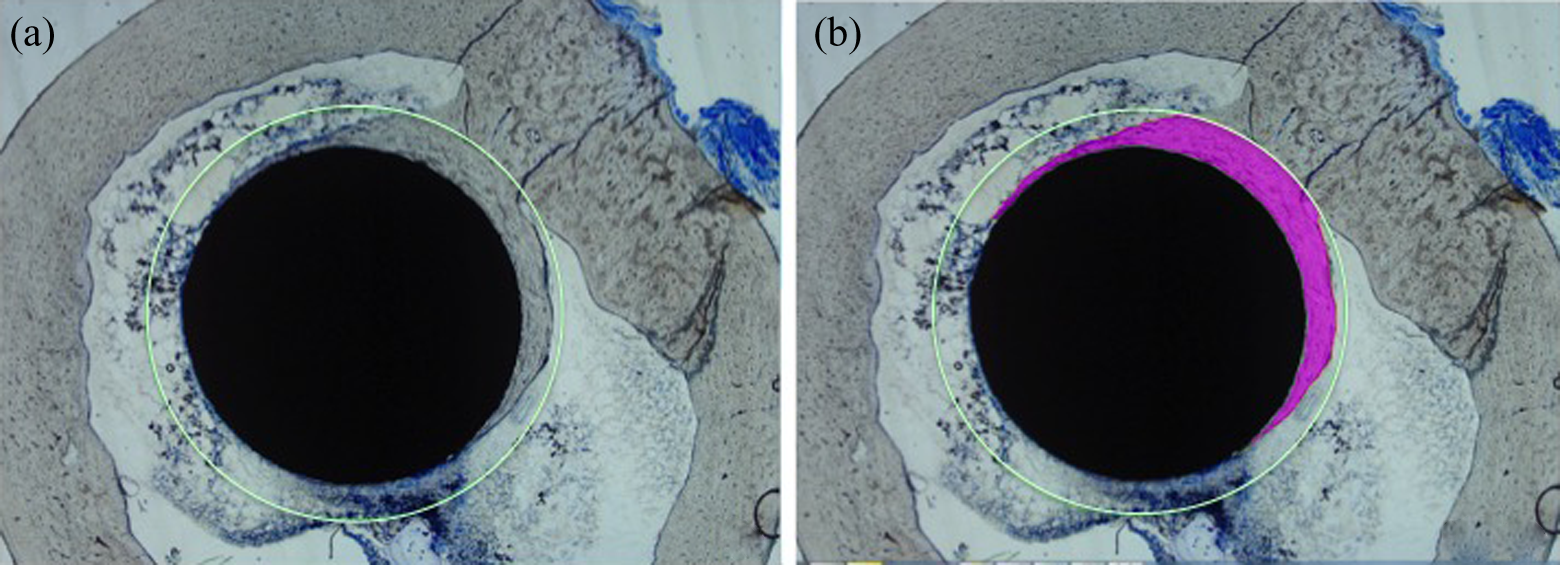
(a) The total area of bone between implant surface and green circle, which is 700-μm distances from the implant surface, was measured at 20× magnification using osseointegration protocol of the Bio Quant Osteo software. (b) Cross section showing bone borders, which is defined manually and bone area represented pink color (20× magnification).

Sample demonstrating the calculation for percentage of bone–implant interface. Contact perimeter proportioned implant perimeter. Only areas with a direct bone–implant interface were selected (green line) (20× magnification).
Statistical analysis
Data were analyzed using SPSS software (version 20.0; IBM Corp., Armonk, NY, USA) for the personnel computers. Assessment of differences between uncoated and HA-coated implants was performed with paired Student’s t-test. The one-way analysis of variance (ANOVA) test was used for assessment of differences between medication groups. Statistical significance level was set at 0.05.
Results
The results were evaluated and presented according to two main points of view: one of them medication groups and other surface properties (HA coated and uncoated) of the implants (Table 1).
Results of mechanical tests
When groups with different types of medication were compared, failure loads showed no significant difference between the celecoxib, diclofenac, and control groups, both in HA-coated and uncoated sides (Table 2). When specimens were grouped according to surface coating, omitting the type of the medication, mean failure load of HA-coated specimens was significantly higher than uncoated implants (443 ± 165 N, 347 ± 140 N, respectively; p = 0.0052; Table 3).
The samples (included left and right side) of each group were compared with ANOVA test in terms of medication groups.a
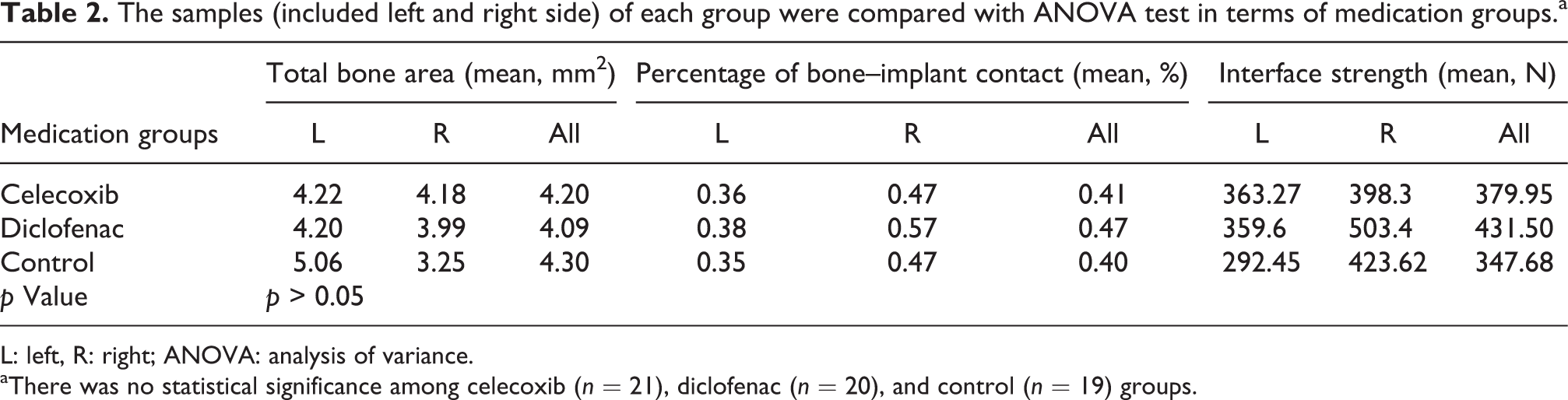
L: left, R: right; ANOVA: analysis of variance.
aThere was no statistical significance among celecoxib (n = 21), diclofenac (n = 20), and control (n = 19) groups.
Results are shown regarding surface properties.a

TBA: total bone area; HA: hydroxyapatite.
aPaired 28 HA-coated and non-coated implants were compared with paired Student’s t-test in terms of TBA, percentage of bone–implant contact, and interface strength on biomechanics test.
Histologic findings
In the qualitative histologic analysis, lamellar bone formation was observed around all implants at the end of the 8 weeks (Figure 8). There was no host immune reaction to the implant in the medullary cavity and at the bone–implant interface. Osseointegration proved a common crescent-shaped ongrowth pattern, partially enveloping the implant. Although the new bone formation appeared to have partially enveloped the implant, the new bone–implant interface presented a patchy pattern (Figure 9).

A section of lamellar bone formation at bone–implant interfaces and around the implant under (a) polarized light and (b) light microscopy (100× magnification). (c) Fibrous membrane (red arrow) around the implant and absence of bone tissue in the space around the implant (200× magnification).

Cross section of HA-coated implant. (a) 400× magnification, polarized light; (b) 100× magnification of the same section. Note good incorporation of lamellar bone with the HA-coated implant. HA: hydroxyapatite.
We investigate the effect of medication groups on the osseointegration parameters for both sides with ANOVA test. There were no significant changes among diclofenac, celecoxib, and control groups in terms of TBA and percentage of bone–implant contact parameters (Table 2).
When the specimens were grouped using only the type of the surface coating, omitting the medication type, there were no significant differences between HA-coated and uncoated implants in terms of TBA (mean 3.85 ± 2.16 mm2 and 4.41 ± 2.83 mm2, respectively; p > 0.05). However, percentage of bone–implant contact was significantly higher in HA-coated implants, when we compared contralateral non-coated implants (mean 0.51 ± 0.17 and 0.35 ± 0.13, respectively; p = 0.0002; Figure 10; Table 3).

Comparison of TBA between (a) HA-coated and (b) uncoated implants in the same rabbit in the celecoxib group. Note the greater peri-implant bone area and bone–implant contact rate of the HA-coated implant versus the uncoated implant (20× magnification). TBA: total bone area; HA: hydroxyapatite.
Discussion
In order to increase the survival of prostheses, the effects of several parameters on osseointegration have been investigated. Although considerable progress has been achieved on this topic, there is still no consensus on the effect of a variety of interventions on osseointegration. Results of the presented study provide important conclusions for two of these controversial subjects, namely HA coating and postoperative NSAID use.
We used intramedullary implants as without weight sharing in the proximal femur. Most of the methods used in the study, including intramedullary implants and histological methods, were reported in the previous studies. 2,19 This setup does not represent total hip arthroplasty condition. However, it has advantages to analyze osseointegration only on press-fit implanting, so it eliminates mechanical reason of failure.
The width of the virtual circle for TBA measurement was determined as 700 µm by the authors, in order to avoid inclusion of cortices of the diaphysis. It was known that the osseointegration finishes around 6 weeks after the press-fit implantation. Similarly, in this study, new lamellar bone tissue with sufficient osseointegration was observed in the histologic sections at the 8-week follow-up period. Histological results were obtained using BioQuant Osteo software, which has specific osseointegration protocol.
The advantages of HA have been shown in experimental studies, the results of which were similar to our study. 4,6,20,21 However, the results of clinical studies are divided, and some have reported that HA-coated femoral components had equal or better clinical results than uncoated femoral components in mid- and long-term follow-up periods. 22 –24 Conversely, Goosen et al., after reviewing eight randomized controlled studies, concluded that HA coating had no positive effect when clinically and radiologically compared with porous surfaces. 7 In a meta-analysis, Gandhi et al. also show that there are no clinical benefits in the use of HA/porous coating over porous coating alone in primary hip arthroplasty. 25 Results of this study proved the significant positive effect of HA coating on interface strength and percentage of the bone–implant contact. However, new bone formation around implants was not affected by the HA coating. These results may encourage us to provide early and secure osseointegration. We believe that HA coating plays a role especially in early stage of the osseointegration, but these effects are not necessary for late-stage osseointegration and survival of the stem.
This study proved no effect of medication on interface strength and osseointegration, although celecoxib and diclofenac were used continuously during 8 weeks. The negative effects of NSAIDs on osseointegration have been shown in experimental studies. 9 –13 On the other hand, some researchers have disproved any negative effects of NSAİDs on osseointegration. 15,26,27 Cai et al. show that postoperatively 7-day regimen of appropriate doses, diclofenac and parecoxib dose, not adversely effect osseointegration. 16 There are just a few prospective human studies about the relationship between implant failure and use of NSAIDs. Lionberger and Noble investigated patients after total hip replacement in terms of bone markers, radiographic prosthetic subsidence, and periprosthetic bone mineral density, finding no significant difference between the celecoxib and control groups. 28 Hofmann et al. show that celecoxib does not seem to inhibit bone ingrowth or bone formation. 14 In order to completely understand the effect of NSAIDs on osseointegration, we should focus on the bone healing process. The early stages of fracture healing are characterized by inflammation. Peri-implant healing shows similarities with primary (direct) bone healing, but there is no endochondral ossification. There is also no consensus in the literature about the effect of NSAIDs on bone healing. 26 Based on these literatures, osseointegration seems not affected by the NSAİDs in the clinical results, which is in contrast to the experimental studies.
Although many studies have been conducted about this topic, the relevance of the available preclinical data regarding the effects of NSAIDs on bone metabolism in humans is not fully understood. Reasons for this were explained in a study published by Radi and Khan 29 : (a) no replicable data in humans from large randomized, controlled clinical reports; (b) inconsistent results from preclinical studies; (c) intra- and interspecies dose and sensitivity differences; (d) undefined compensating systemic and local factors that may also regulate bone repair; (e) the unknown effect of underlying conditions (age, rate of bone turnover, or disease affliction) that may influence bone healing; and (f) differences in dosing duration and levels between experimental animal models and clinical applications. This means that the experimental studies are not comparable with each other and clinical conditions. 26 In this study, we try to minimize the variability, such as age and weight of the rabbits, the same surgical technique, and standard implants to analyze osseointegration. The doses of the drugs were determined based on the literature. 10,12,17,30 On the other hand, different surfaces in the same rabbit provide controlled study to evaluate the effect of HA.
As a result of these findings, we had insufficient evidence to conclude that there is a relationship between early implant failure and NSAID use. It is clear that further research is required, especially when we consider the few number of prospective human studies.
Main limitation is that the study was conducted with a relatively small number of samples. Another limitation of our study is number of the histologic sectioning. Only one section was able to prepare on the exact cutting and grinding system. More sections might have provided a three-dimensional analysis. Our intramedullary models allow weight bearing; however, it is not subjected to the load as far as femoral stem. This can be another limitation of this study because of the relevance to total hip arthroplasty not directly applicable.
In conclusion, the effect of the celecoxib and diclofenac on osseointegration depends on the dose, time, and individual. However, it was shown that, these dosing regimen of the celecoxib and diclofenac, which are used in this study, are not jeopardize osseointegration in rabbit, although they were used continuously and long term. HA coating provides tight connection between bones and implant with good interface strength and good bone and implant contact rate.
Footnotes
Authors’ Note
All authors have participated in the research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was approved by the Institutional Review Board of the Istanbul University and supported by the Istanbul University Department of Scientific Research Projects (no: 1974).
