Abstract
Introduction
This review aims to evaluate smart wound dressings (SWDs) that integrate sensor technology for real-time monitoring of acute and chronic wounds, with emphasis on clinical relevance, validation status, and translational limitations.
Methods
A structured literature search was performed to identify preclinical, translational, and human studies evaluating sensor-enabled wound dressings, including technologies measuring temperature, pH, moisture, and infection-associated parameters. Relevant studies were selected based on applicability to sensor-integrated wound monitoring. Key features extracted included sensing modality, validation model, regulatory context, and reported clinical applicability.
Results
Sensor-enabled wound dressings demonstrate promising noninvasive approaches to wound monitoring; however, the level of clinical validation varies widely. While some systems have advanced to limited human use supported by observational data, many remain at the preclinical or early feasibility stage. Direct comparisons with established diagnostic standards and robust patient-centered outcome data are largely lacking.
Conclusions
SWDs represent a rapidly evolving field with significant potential to improve wound monitoring. Current clinical adoption is constrained by limited comparative validation and regulatory pathways that do not require demonstrated efficacy. Future studies benchmarking these technologies against accepted diagnostic standards will be essential to defining their clinical role.
Introduction
Over 6 million people in the United States are estimated to be affected by acute and chronic wounds yearly, costing roughly $25 billion annually. 1 Chronic wound incidence is expected to rise with the increasing prevalence of diabetes and ageing status of the U.S. population. 2 Chronic wounds can be persistent, progressive, complicated by sepsis and osteomyelitis, and lead to amputation and/or death. Similarly, acute wounds from trauma and surgeries are common, painful, and debilitating. Clinically, wound dressings are among the main strategies employed in the management of chronic and acute wounds. 3
Wound dressings can be categorized into 3 groups: traditional, modern, and smart. Smart wound dressings (SWDs) are an advancement of the last decade. These feature built-in sensors which indicate aspects of the underlying wound such as temperature, pH, uric acid concentrations, glucose levels, pressure, electrical conductance, and shear force.4,5 Some SWDs not only sense but respond to indications in closed-loop fashion with release of antibiotics or bioactive factors. 4
Biosafety demonstration limits the widespread commercialization of SWDs. 4 Thus, they are limited in clinical practice, with a “major gap” dividing use of SWDs in research with clinical application. 2 Despite rapid innovation in sensor-enabled wound dressings, clinical validation varies widely across technologies. While some systems have progressed to limited clinical use with human observational data, many remain at the preclinical or early feasibility stage, where studies primarily assess sensor performance rather than patient outcomes. Accordingly, this review distinguishes technologies supported by human use data from those that remain experimental and considers their relevance within current diagnostic practice. The purpose of this article is to support clinical use of SWDs by detailing pertinent aspects of those that are currently approved for use nationally and internationally, to review smart dressing features currently in development, and to describe the criteria of an ideal SWD.
Literature Search Strategy
A structured literature search was conducted to identify studies relevant to smart and sensor-enabled wound dressings. Searches were performed in PubMed/MEDLINE, Scopus, and Web of Science for articles published between January 1, 2010, and July 31, 2024, a period selected to capture the emergence and maturation of sensor-integrated and wearable wound monitoring technologies.
For PubMed, the search combined wound-specific terms with sensor-related parameters using Boolean operators. Articles were identified if the title or abstract contained “wound dressing,” “smart wound dressing,” “sensor-enabled wound dressing,” “smart dressing,” “wound sensor,” “wound biosensor,” or “wearable wound sensor,” in combination with at least one of the following terms in the title or abstract: “pH,” “temperature,” “moisture,” “infection,” “bacterial,” or “biosensor.” Equivalent keyword strategies were adapted for Scopus and Web of Science to account for database-specific indexing.
Searches were limited to English-language publications. Eligible studies included original research articles, clinical studies, observational studies, feasibility studies, and relevant review articles describing the development, analytical performance, or clinical application of sensor-integrated wound dressings. Given the emerging and translational nature of the field, both preclinical and human studies were included. Studies focused on nonwound wearable sensors, implantable devices unrelated to wound care, or dressings without integrated sensing capability were excluded. Reference lists of included articles were manually screened to identify additional relevant publications.
SWD Technologies
Temperature Sensors
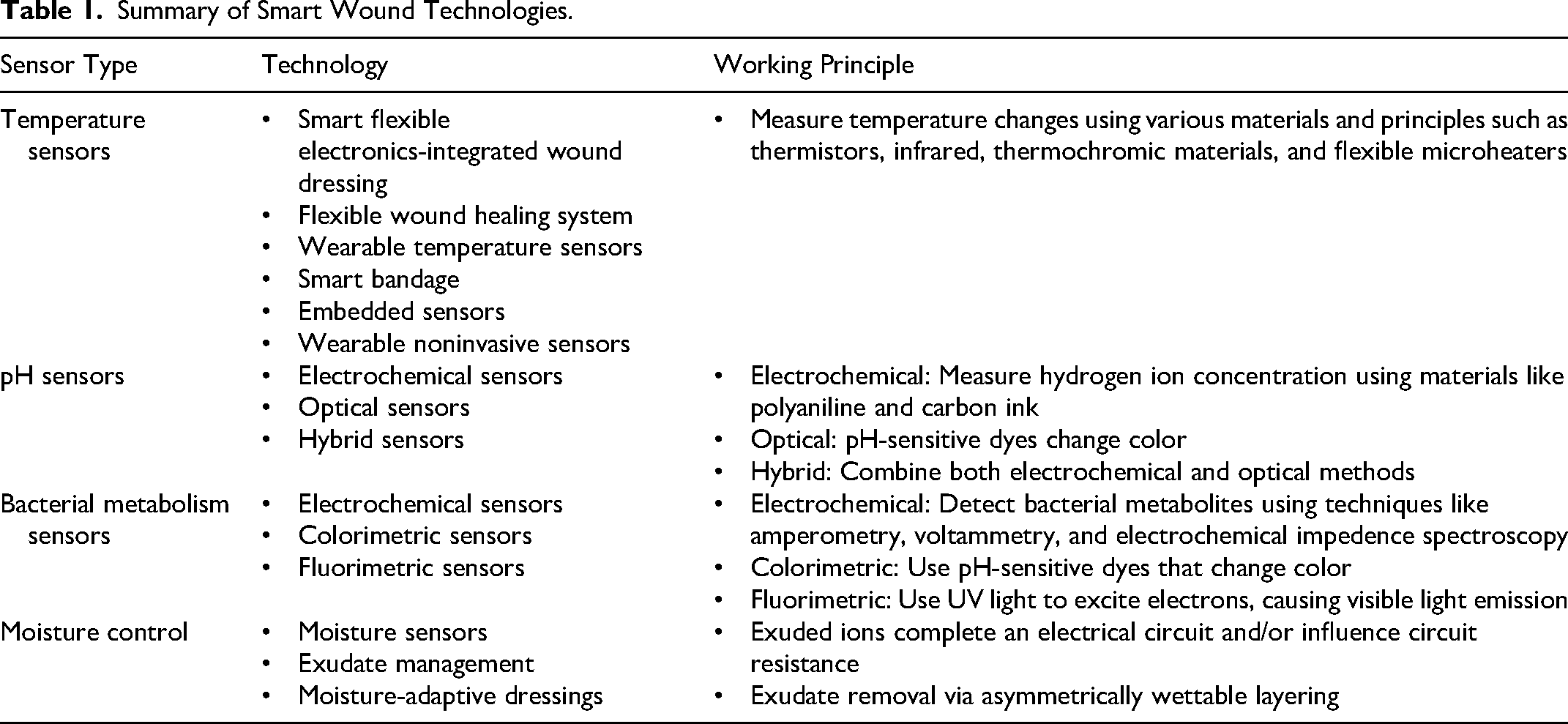
Temperature is a key indicator of infection and inflammation, making temperature-sensing SWDs essential for wound monitoring and management (Table 1). Healing wounds demonstrate a predictable temporal pattern, with an initial increase in local temperature during the inflammatory phase followed by gradual decline as inflammation resolves and tissue repair progresses. In contrast, infected wounds often exhibit sustained temperature elevation or secondary temperature increases. Human observational studies have demonstrated statistically significant temperature differences between infected wounds versus control sites, particularly in cases of deep or surrounding infection, supporting the use of temperature trends as a clinically relevant signal over isolated thresholds. 6 These temperature differences fall within the physiologic range targeted by the flexible temperature sensors incorporated in SWDs. 7 By integrating temperature sensors, SWDs address the limitations of traditional methods, which rely on frequent dressing changes and subjective assessments.
Summary of Smart Wound Technologies.
Design and Integration of Temperature Sensors in SWDs
SWDs integrate temperature sensors within flexible and wearable electronic architectures to enable continuous, noninvasive monitoring of local wound conditions. Temperature serves as a key biomarker of inflammation and infection, and real-time sensing allows early identification of abnormal deviations from baseline. In some experimental systems, temperature thresholds are used to trigger localized therapeutic responses, such as controlled release of antimicrobial agents, effectively integrating monitoring and intervention within a single platform. 3
Several designs incorporate embedded or wearable temperature sensors using thermistors, resistive sensors, infrared-based approaches, or thermochromic materials, each offering distinct advantages and limitations with respect to sensitivity, durability, and suitability for continuous monitoring. Thermistors and resistive sensors allow continuous electronic monitoring when embedded within flexible substrates, whereas infrared approaches may require dressing exposure, limiting long-term use. Thermochromic materials provide visual temperature indication as a nonelectronic alternative but lack continuous quantitative output.
Representative systems demonstrate the clinical relevance of this approach. Lou et al developed a temperature-sensing SWD embedded within a collagen–chitosan dermal scaffold that wirelessly transmits data via Bluetooth Low Energy, enabling continuous monitoring across inflammatory, proliferative, and remodeling phases of healing. Distinct temperature patterns corresponding to underlying biological processes were observed, and validation in infection models supported the utility of temperature monitoring for early detection of wound complications.4,8 Similarly, Mostafalu et al described a smart bandage integrating temperature and pH sensors with a thermoresponsive drug delivery system, demonstrating closed-loop modulation of localized therapy in response to infection-associated temperature changes. 9
Wearable and embedded temperature sensors further support continuous monitoring without repeated dressing removal, reducing reliance on subjective visual assessment and minimizing wound disturbance. Integration with wireless data transmission enables remote longitudinal oversight of wound progression, potentially decreasing the need for frequent in-person evaluations while maintaining clinical vigilance.10–12
Overall, temperature-sensing smart dressings provide objective, trend-based assessment of wound status that may facilitate earlier identification of inflammatory or infectious complications, support data-driven clinical decision-making, and reduce the clinical burden associated with repeated dressing changes and prolonged wound surveillance.
pH Sensors in Wound Dressings
Wound pH exhibits characteristic changes during healing and infection that are relevant to sensor-based monitoring. Acute and chronic wounds typically present with alkaline pH at baseline, which progressively shifts toward neutral or mildly acidic levels during successful healing. Human observational studies have reported mean pH reductions of approximately 0.5 to 0.7 units over several weeks in healing wounds, with greater acidification correlating with wound size reduction. 6 In contrast, infected or nonhealing wounds tend to maintain persistently elevated alkaline pH values, often exceeding pH 7.4 to 8.0, although the magnitude of elevation varies by wound type and microbial burden. While pH-sensitive smart dressings can detect changes across this range, pH alone lacks sufficient specificity to reliably distinguish infection from colonization, underscoring the need for integration with additional physiologic or clinical parameters. 6
pH sensors in wound dressings are typically based on electrochemical or optical principles. Electrochemical sensors measure the concentration of hydrogen ions (H+) through electrochemical reactions, while optical sensors use pH-sensitive dyes that change color in response to pH variations.
Design and Integration of pH Sensors in SWDs
pH-sensing SWDs enable continuous assessment of the local wound environment by monitoring hydrogen ion activity, a key indicator of inflammation, bacterial metabolism, and tissue repair. Changes in wound pH have been associated with delayed healing and infection, establishing pH as a clinically relevant biomarker for longitudinal wound monitoring.
Electrochemical pH sensors detect hydrogen ion concentration through changes in electrical potential or conductivity relative to a reference electrode. Common materials, including polyaniline and carbon-based composites, demonstrate rapid and reproducible responses to pH variation and have been successfully integrated into flexible and thread-based wearable dressings for continuous monitoring. 13 Optical pH sensors use pH-sensitive dyes embedded within polymer matrices such as polydimethylsiloxane, producing color or spectral shifts that can be quantified visually or through camera-based analysis, enabling noncontact pH assessment without electronic circuitry at the wound interface. 14 Hybrid approaches combine electrochemical and optical modalities to improve measurement robustness and signal stability across dynamic wound environments. 15
Clinically, continuous pH monitoring provides objective insight into wound status and may facilitate early identification of deviations associated with infection or impaired healing. The integration of pH sensors into wound dressings reduces reliance on subjective visual assessment and minimizes the need for frequent dressing removal. This supports uninterrupted longitudinal monitoring, particularly in chronic wound care settings.9,16,17
Bacterial Metabolism Sensors
Bacterial sensors integrated into wound dressings aim to detect infection-related biomarkers associated with bacterial colonization and biofilm formation. Traditional diagnostic approaches, including visual inspection and microbiological assays, are limited by subjectivity, delayed results, and the need for dressing removal. Sensor-based systems enable continuous, localized monitoring without disrupting the wound environment. 18
Types of Bacterial Metabolism Sensors
Bacterial metabolism sensors used in SWDs can be broadly categorized by detection modality, with the most common being electrochemical, colorimetric, or fluorimetric sensors. These approaches differ in mechanism, sensitivity, and integration feasibility.
Electrochemical sensors detect infection-associated metabolites or redox-active compounds using amperometry, voltammetry, or electrochemical impedance spectroscopy. Dual-analyte strategies, including detection of uric acid and pyocyanin associated with Pseudomonas aeruginosa, improve specificity. Inkjet-printed and nanomaterial-based electrodes have demonstrated stability in simulated wound environments, supporting feasibility for SWD integration.18,19
Colorimetric sensors rely on indicator dyes that undergo color changes in response to bacterial metabolites, enabling visual or camera-based assessment without specialized instrumentation. These sensors are commonly embedded within dressing matrices and provide semiquantitative information regarding bacterial activity. Volatile analyte-responsive films have been used to detect bacterial metabolism in wound headspace models, illustrating a low-cost approach to infection monitoring.18,20
Fluorimetric sensors detect bacterial activity through fluorescence changes triggered by bacterial enzymes or toxins. While these approaches offer high analytical sensitivity, they often require external excitation sources, limiting practicality for continuous monitoring. 21
Recent experimental systems have combined bacterial sensing with flexible electronics to enable wireless data transmission and, in some cases, multimodal monitoring alongside parameters such as pH and temperature, demonstrating the feasibility of integrated sensing platforms for real-time wound assessment. 2
Moisture Control
Moisture Sensors
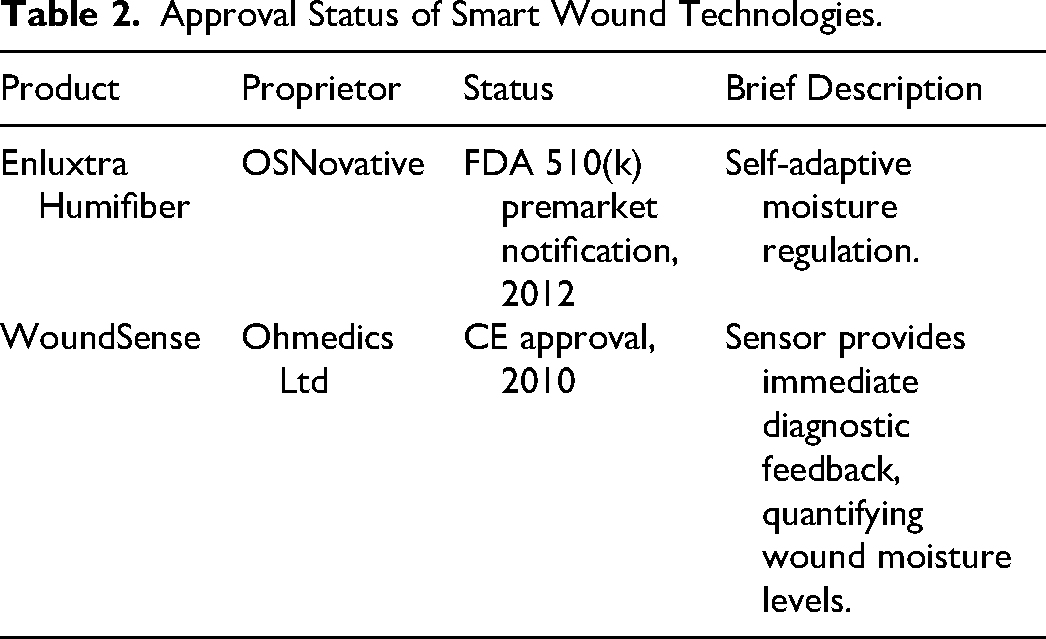
A handful of moisture-sensing and/or moisture-responsive SWDs are in different stages of development and marketing.22–25 To our knowledge, only 2 are commercially available (Table 2). The first is a sensor (WoundSense; Ohmedics, Glasgow, Scotland) that is worn against the wound for objective moisture analysis without removal of the overlying dressing. Milne et al studied the use of this sensor to determine how often wound dressing changes were taking place outside the optimal timeframe. 26 The researchers took moisture measurements prior to each dressing change and included a total of 588 dressing changes from 30 patients for final data analysis. Patients with pressure ulcers had their dressings changed daily, and patients with diabetic foot ulcers had their dressings changed every 3 days. The researchers found that 44.9% (264/588) of the dressing changes were done while the dressing was within the optimum moisture range and didn’t need changing. On the other hand, 29.1% (171/588) of dressing changes were dry indicating that the wound may benefit from alternative dressing type, and 26% (153/588) of changes were wet. They concluded that a high percentage of unnecessary dressing changes were occuring while within the optimal moisture range, and this was likely contributing to high nursing and wound dressing costs, increased patient pain, and potential delayed healing due to overly frequent wound disturbance. 26
Approval Status of Smart Wound Technologies.
Moisture-Responsive Dressings
While the stimulus response component is less relevant to the moisture parameter, advances have been made in moisture control with or without wound sensors. Reyzelman and Vartivarian reported 2 cases treated with a self-adaptive wound dressing (Enluxtra; OSNovative Systems, California, USA) designed to respond to underlying wound moisture needs and offer either hydration or exudate removal. 27 This SWD may be pairable with a moisture sensor allowing for moisture quantification and response. On this vein, Ge et al designed and tested a SWD capable of wireless humidity and temperature sensing, thermosensitive drug release, and advanced exudate management utilizing coated Janus films for self-pumping and fluid storage. This SWD produced favorable results in animal testing, with future goals including additional improvements and clinical trials. 28
Clinical Relevance
The clinical application of moisture-sensing and moisture-responsive SWDs has potential to decrease dressing change frequency, thereby reducing costs, time investments, pain, and wound disruption. Moisture control supports the healing process and fosters an environment conducive to cell migration and proliferation. 29 These dressings may also be used to identify wounds that frequently fall outside of the ideal moisture parameters, informing dressing type changes. 26 In the clinic, wounds with suboptimal moisture levels could be identified without dressing removal by pairing with the moisture sensor. Self-adaptive wound dressings may be used to treat complex wounds with different moisture requirements by sublocation, preventing simultaneous wound edge maceration and wound desiccation. 27
Challenges and Future Directions
The clinical use of SWDs for the treatment of complex wounds remains severely limited by product availability and absent high-quality evidence. While numerous SWDs have been developed and tested in vitro or with animals, the vast majority remain commercially unavailable. Despite promising advancements, several challenges remain in developing and clinically implementing bacteria sensors in wound dressings. These include ensuring biocompatibility and stability, power source miniaturization and data transmission, reusability, sensor instability and drift, mechanical durability, topical agent incompatibility, and humidity and sweat interference.18,30 Additionally, given the potential wound-healing benefits of certain bacterial skin commensals, human trials demonstrating the link between broad bacterial metabolism sensors, responsive antibiotic treatment, and ultimate wound healing would be appropriate. 31 Furthermore, solutions aimed at addressing wound-level heterogeneity in temperature and bacterial density should be considered. Future research should address these points and overcome these challenges to create fully autonomous, easy-to-use, and cost-effective SWDs.
Regulatory Considerations
The regulatory pathway for SWDs helps explain the gap between commercial availability and clinical efficacy data. In the United States, many wound dressings and sensor-enabled devices are cleared through the FDA 510(k) process, which requires demonstration of safety and substantial equivalence to a predicate device, rather than proof of clinical superiority or improved patient outcomes. As a result, regulatory clearance does not necessarily reflect evidence of improved healing, infection reduction, or cost-effectiveness. This distinction is particularly relevant for smart dressings, where technical feasibility and biosafety may be established well before robust clinical trials are performed. Understanding this regulatory framework is important for interpreting the current evidence base and for setting appropriate expectations regarding clinical adoption.
For wound infection assessment, quantitative microbiologic analysis of deep tissue biopsy or aspirate is widely regarded as the diagnostic gold standard. 32 However, these methods are invasive, episodic, and impractical for continuous or longitudinal monitoring. 33 To date, few studies have directly compared sensor-enabled wound dressings with established microbiologic standards using formal sensitivity and specificity analyses. Most smart dressing technologies have been evaluated in proof-of-concept studies, animal models, or against clinical assessment rather than validated reference methods. 34 This gap highlights an important limitation of the current evidence base and underscores the need for future studies that benchmark emerging technologies against accepted diagnostic methods.
Suggestions for Smart Dressing Production
System Design
Temperature Sensors
Temperature sensing can be incorporated using flexible, printable thermistors embedded within the dressing material to enable continuous monitoring of local wound temperature (Figure 1; Table 3). Longitudinal temperature trends, rather than isolated thresholds, may reflect evolving inflammatory or infectious processes while minimizing unnecessary intervention. Embedded sensors reduce the need for frequent dressing removal, preserving the wound environment. Temperature data may be transmitted wirelessly to an external monitoring device, allowing periodic review by clinicians without requiring constant real-time alerts.
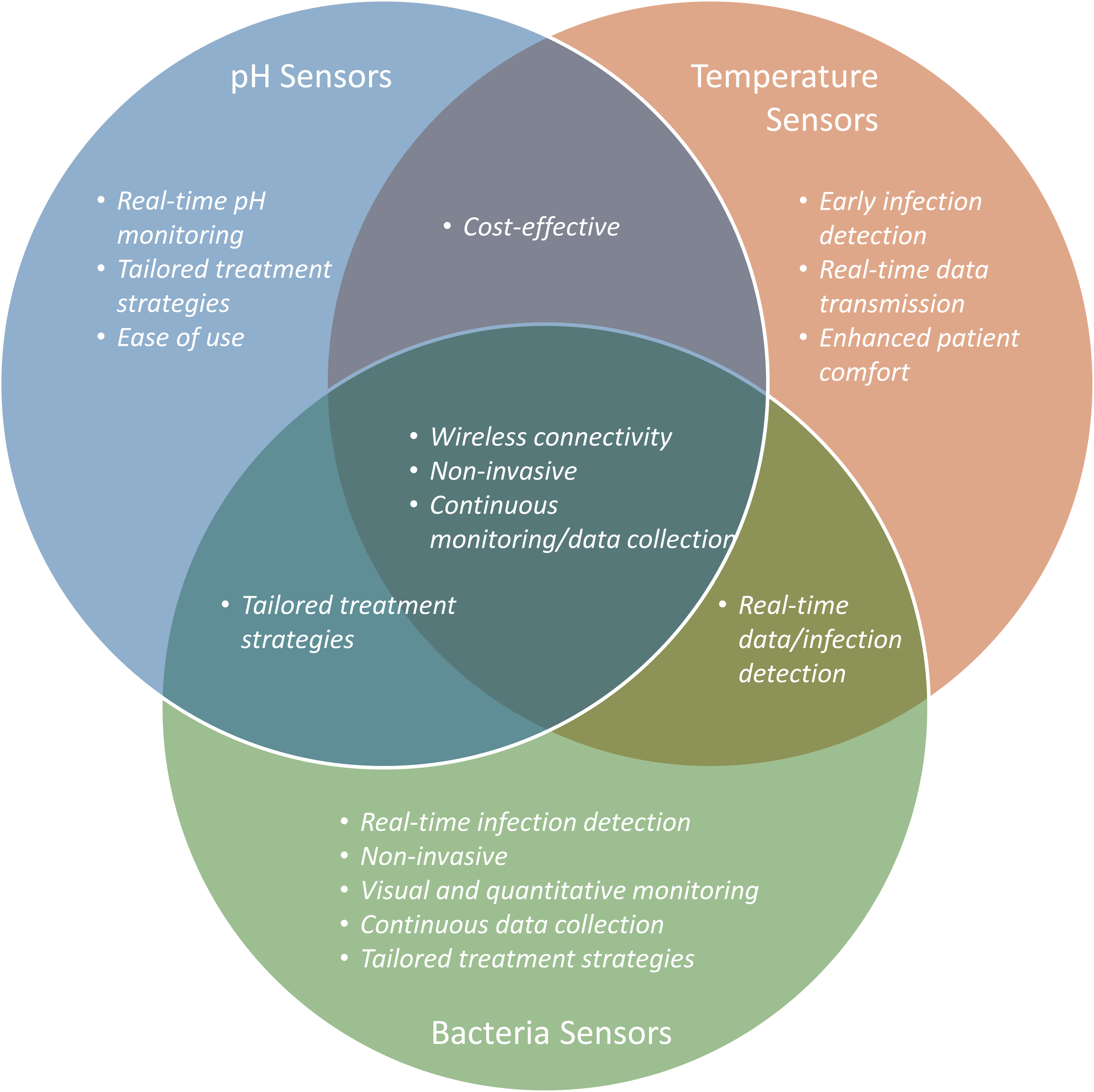
Venn diagram comparison of different sensor type advantages.
Ideal Smart Wound Dressing Features.

pH Sensors
pH monitoring can be achieved through hybrid sensor designs that combine electrochemical and optical detection methods. Electrochemical components quantify hydrogen ion concentration using conductive materials such as polyaniline or carbon-based inks, while optical components rely on pH-sensitive dyes that undergo color changes in response to environmental shifts. Deviations from physiologic wound pH ranges may indicate delayed healing or infection, making longitudinal pH trends clinically informative. Periodic wireless data transmission enables external review while prioritizing trend interpretation over continuous notification.
Bacteria Sensors
Bacterial sensing may be implemented using integrated electrochemical and colorimetric approaches designed to detect infection-associated metabolites. Electrochemical sensors can identify bacterial byproducts such as pyocyanin or uric acid through amperometric or voltammetric techniques, while colorimetric sensors provide visual or semiquantitative confirmation through metabolite-induced color changes. Fluorimetric strategies may further enhance sensitivity by detecting bacterial activity through fluorescence activation. These approaches support localized infection assessment without repeated wound sampling or dressing disruption.
Moisture Sensors
Moisture sensors can be incorporated to monitor hydration at the wound–dressing interface, which is critical for maintaining optimal healing conditions. Continuous moisture assessment may help identify sustained deviations such as excessive dryness or exudate accumulation, reducing unnecessary dressing changes. Embedded sensor design should preserve absorbent and barrier properties of the dressing while enabling periodic data transmission for longitudinal assessment.
Wireless Connectivity and Data Integration
Low-power wireless communication allows intermittent transfer of sensor data to an external device for storage and analysis. Data integration strategies should emphasize trend-based interpretation rather than frequent alerts to reduce information overload. Automated data logging supports longitudinal wound assessment, while selective notification may be reserved for sustained or clinically meaningful deviations. System design should prioritize reliability, low energy consumption, and secure data handling.
Conclusion
This review highlights the emerging role of SWDs that integrates pH, temperature, moisture, and bacterial sensors to address key limitations in current wound care. By enabling continuous, noninvasive monitoring of the wound microenvironment, these technologies may support earlier detection of infection, improved assessment of healing trajectories, and reduced reliance on frequent dressing changes. Although further validation and standardization are required, smart sensing platforms represent a promising adjunct to existing wound care strategies with potential to improve monitoring precision and clinical decision-making.
Supplemental Material
Footnotes
Author Contributions
Claudy Sarpong: conceptualization, methodology, writing—original draft, writing—review and editing; Joseph Larson: conceptualization, writing—original draft, writing—review and editing; Isabella Oh: writing—original draft; Nitya Devisetti: writing—original draft; Subhas Gupta: conceptualization, methodology, project administration, and supervision.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This project was funded internally by the Department of Plastic Surgery, Loma Linda University.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
