Abstract
Vertical rectus abdominis myocutaneous (VRAM) flaps remain a reliable option for perineal reconstruction following abdominoperineal resection (APR). Despite their robust vascularity, postoperative factors can compromise flap perfusion. We present a two-case series of patients who developed flap congestion secondary to postoperative bladder distension after APR with VRAM reconstruction. In both cases, removal of the Foley catheter led to urinary retention, abdominal distension, and venous congestion of the flap. Imaging in one patient demonstrated direct pedicle compression by the distended bladder. Prompt reinsertion of the Foley catheter and decompression restored flap perfusion, with both patients ultimately achieving full healing. To our knowledge, bladder distension as a cause of VRAM flap compromise has not been previously reported. These cases highlight the importance of vigilant postoperative urinary monitoring and consideration of prolonged catheterization protocols to prevent reversible, avoidable flap congestion in perineal reconstructions using pedicled VRAM flaps.
Introduction
Abdominoperineal resection (APR) is a critical surgical procedure primarily indicated for colorectal adenocarcinomas and other cancers where sphincter-preserving surgery is not feasible. 1 Pedicled locoregional flaps are commonly used to reduce complications such as wound dehiscence and infection, particularly in irradiated fields.2,3
The vertical rectus abdominis myocutaneous (VRAM) flap, with a relatively low rate of donor and recipient site complications, is a reliable method for immediate pelvic reconstruction after APR.4–6 Its robust vascular supply and bulk make it a preferred option for perineal reconstruction. Despite meticulous intraoperative technique, postoperative factors may compromise pedicle perfusion. Here, we describe two cases of bladder distension causing pedicle compression and VRAM flap congestion following APR.
Case Description #1
A 64-year-old male with stage IIIb rectal cancer underwent robotic-assisted APR with end colostomy and right VRAM perineal reconstruction, with a skin paddle for monitoring (Figure 1(a)). The Foley catheter was removed on POD 9. On POD 10, examination revealed superficial epidermolysis and partial necrosis of the distal skin paddle (Figure 1(b)).
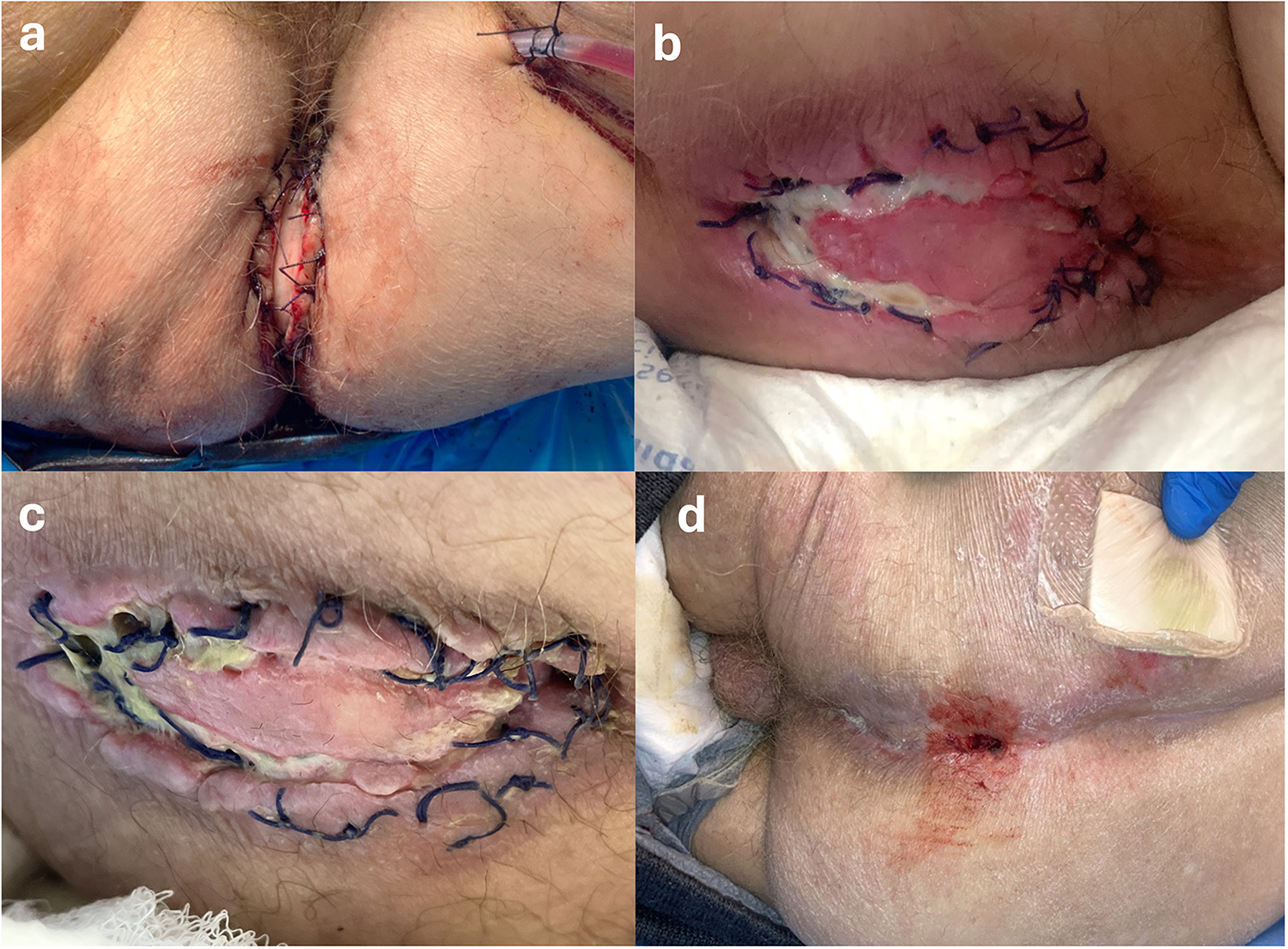
Sequential photographs demonstrating perineal reconstruction with a VRAM flap. (a) Postoperative day (POD) 0: surgical closure with Jackson-Pratt drain showing serosanguinous output. (b) POD 12: superficial epidermolysis with areas of necrotic tissue. (c) POD 15: progressive necrosis involving the distal aspect of the flap. (d) Three months postoperatively: complete wound closure and flap healing.
The patient developed abdominal distension and pain. CT imaging demonstrated ileus, bilateral hydronephrosis, and significant bladder distension compressing the VRAM pedicle (Figure 2(a–e)). A Foley catheter was reinserted on POD 14 for decompression. After bedside debridement of necrotic tissue, negative pressure wound therapy and intravenous antibiotics were initiated. Conservative management resulted in wound improvement and healing by secondary intent. The patient was discharged to a continuing care facility with interval improvement (Figure 1(d)).
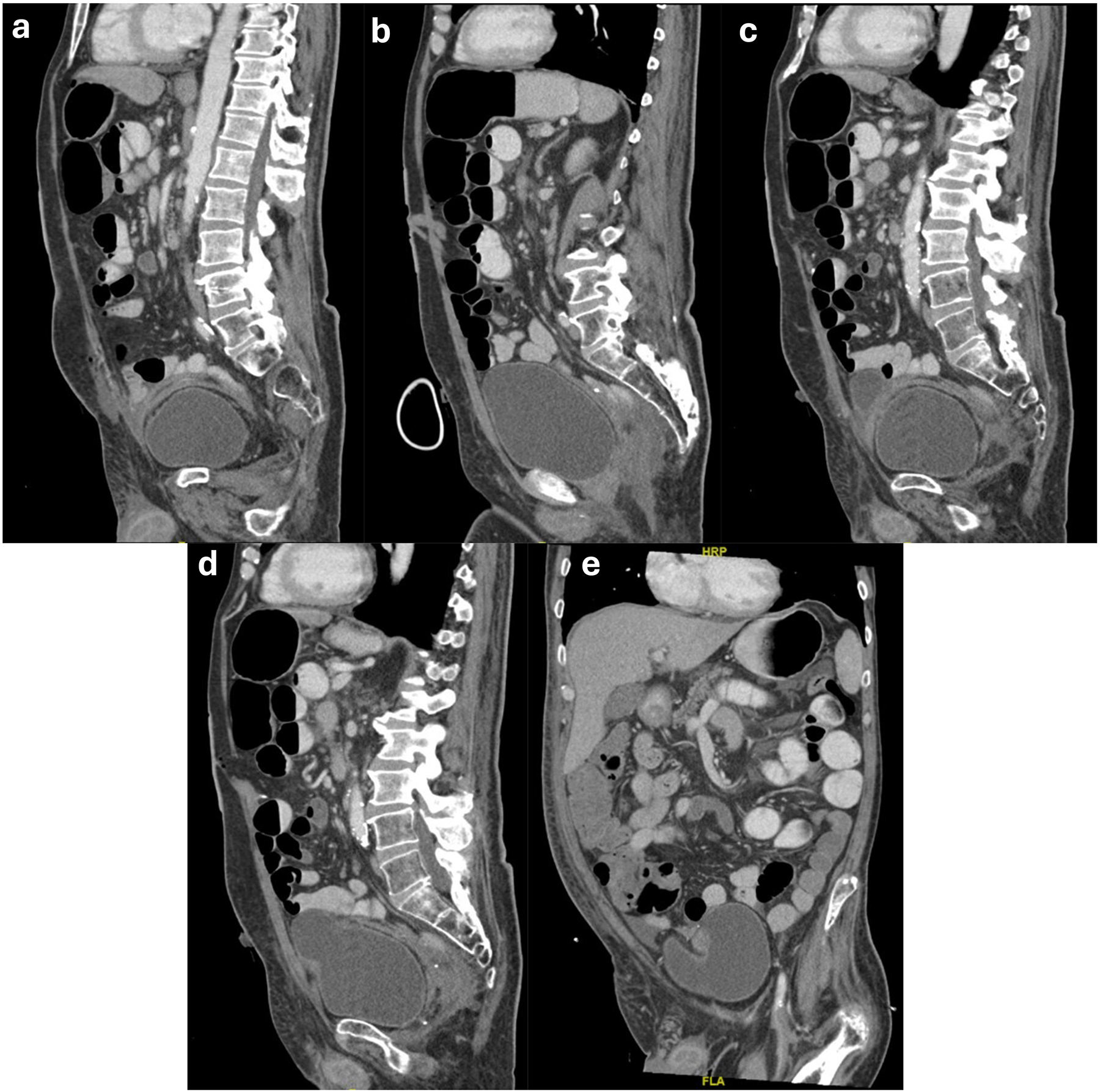
Postoperative computed tomography images demonstrating VRAM flap positioned superior to the bladder. (a) Sagittal view showing flap location relative to bladder. (b) Sagittal view showing marked bladder distention. (c, d) Sagittal images and (e) coronal image demonstrating bladder distortion secondary to VRAM flap.
Case Description #2
A 70-year-old female with squamous cell carcinoma of the anus and vulva underwent robotic-assisted APR with end colostomy, en bloc hysterectomy with bilateral salpingo-oophorectomy, posterior vaginectomy, and VRAM reconstruction (Figure 3(a)). The Foley catheter was removed on POD 3. She subsequently developed acute urinary retention with abdominal pain. During this period, the skin paddle became congested with a violaceous hue and delayed capillary refill (Figure 3(b)). Following Foley reinsertion, flap color, turgor, and capillary refill rapidly improved (Figure 3(c)). Mild epidermolysis was noted without progression. The Foley catheter was maintained for 6 weeks postoperatively. She recovered fully without further flap-related complications.
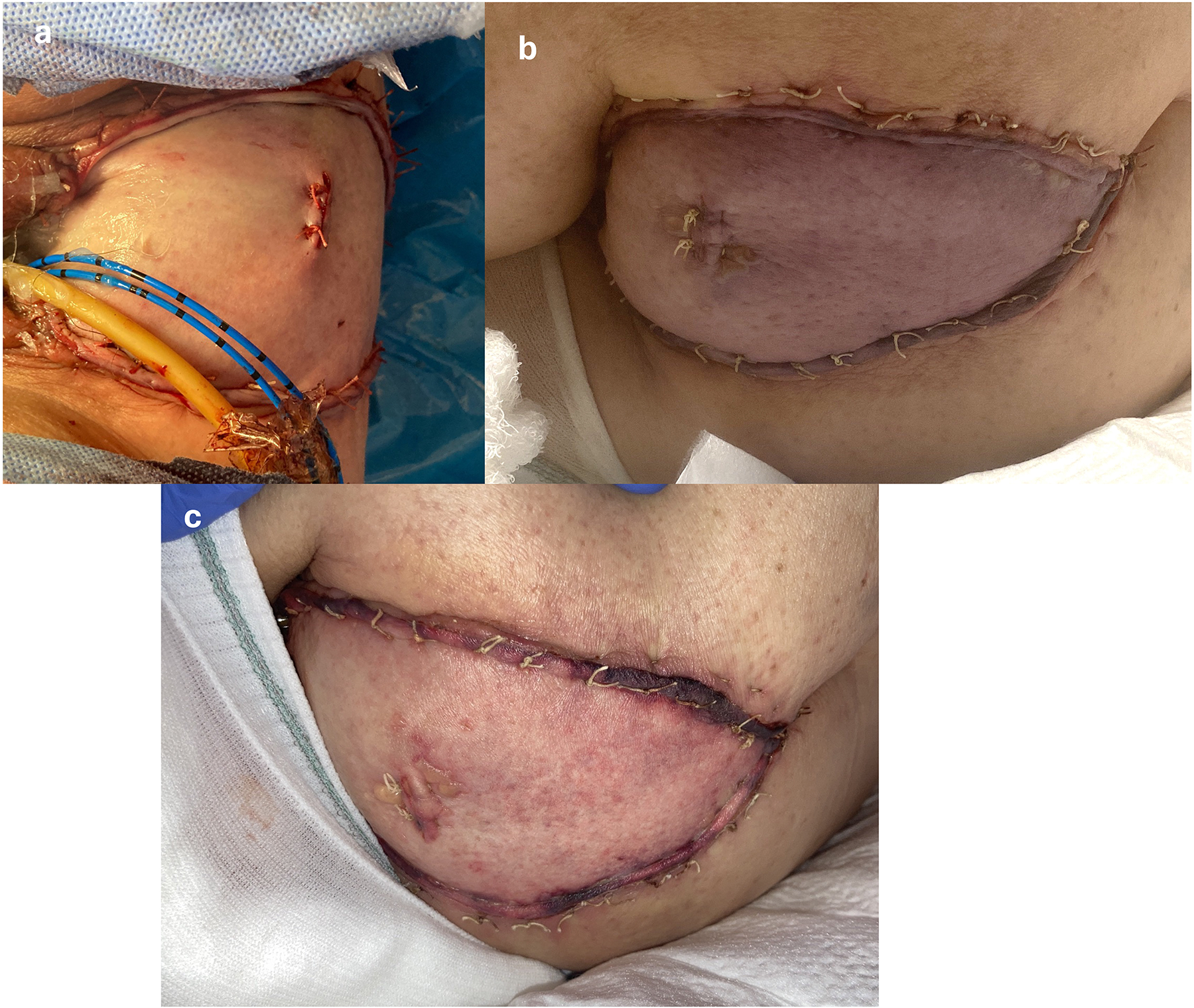
Sequential photographs demonstrating flap congestion after perineal reconstruction with VRAM flap. (a) Immediate postoperative image showing a healthy pink flap after wound closure. (b) After Foley catheter removal on postoperative day (POD) 3, the flap demonstrates marked venous congestion. (c) Following catheter replacement 45 min later, congestion resolves, and the flap returns to its original pink coloration.
Discussion
These cases demonstrate bladder distension as a reversible cause of VRAM flap congestion following APR. In both patients, flap compromise occurred shortly after Foley catheter removal and urinary retention, with radiographic evidence of pedicle compression in one case. Known VRAM complications include infection, dehiscence, and flap necrosis, with reported rates of perineal complications ranging from 10.4% to 22.5%.7,8 To our knowledge, bladder distension has not been previously described as a cause of VRAM pedicle compression.
Bladder decompression resulted in prompt improvement in flap perfusion, suggesting that earlier recognition or prolonged catheterization may prevent avoidable flap compromise. While prolonged Foley use carries a known risk of catheter-associated urinary tract infection, this must be weighed against the consequences of flap ischemia and wound complications. 9 These cases underscore the importance of postoperative urinary monitoring, particularly when VRAM flaps are buried and direct visual assessment is not possible.
Footnotes
Acknowledgments
None.
Author Contributions
NSH was involved in writing original draft; TW, AH, NL in review and editing; NW in supervision, conceptualization, and review and editing. All authors reviewed and approved the final manuscript.
Data Availability Statement
All relevant clinical data are contained within the manuscript. Additional information is available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Institutional review board (IRB) approval was not required for this report of two cases. Written informed consent was obtained from all patients for inclusion in this report and for publication of their clinical details and images.
