Abstract
Introduction
Stenosing tenosynovitis (trigger finger) is one of the most common presentations seen by hand surgeons.1,2 The pathophysiology of trigger finger involves hypertrophy and inflammation of flexor tendon within its synovial sheath, causing friction and impingement with tendon gliding particularly at the A1 pulley.3–6 Patients with trigger finger present with pain and stiffness of the affected digit(s) and may also experience clicking or locking of their finger(s).3,7
Initial management of trigger finger is nonsurgical, involving rest, splinting, nonsteroidal anti-inflammatory drugs, or corticosteroid injection (CSI).7,8 CSI has a reported resolution rate between 45% and 87% with a single injection and an overall success rate of 47% to 92% after multiple injections.5,9–13
CSI for trigger finger is typically performed through the palm at the level of the A1 pulley of the affected digit.5,12 Various other methods of CSI have been described in the literature, including palmar injections more distally along the affected digit, or injecting through the dorsal aspect of the hand via the webspace. Very few studies have evaluated a dorsal approach to CSI for trigger finger despite a widespread though controversial notion that the dorsal skin of the hand is less sensitive—and thus less painful to inject—than the palmar surface of the hand.14,15 This belief influences regional anesthesia techniques, where a 2-injection dorsal digital block, rather than a single palmar injection, may be preferred by clinicians to minimize injection-associated pain.15–17 Other studies suggest that a single palmar injection may be equally or less painful for patients than a 2-injection dorsal digital block.14–18 Notably, CSI for trigger finger involves only a single injection, regardless of whether it is administered from the palmar or dorsal aspect of the hand.
Given the lack of clarity, this scoping review aims to summarize the literature on dorsal webspace CSIs for patients with trigger finger and describe associated outcomes; specifically, injection-associated pain, distribution of anesthesia, effectiveness, and adverse effects. These findings can inform practice recommendations on CSI techniques for trigger finger and are crucial in evaluating the potential of this technique to improve the needle experience and reduce pain for patients.
Methods
This scoping review adheres to the 6 stages as outlined by Arksey and O’Malley 19 and further expounded on by Levac and colleagues. 20 The Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) Checklist 21 was also used in the development and reporting of this scoping review.
Stage 1: Identifying the Research Question
This scoping review seeks to understand what is known from the existing literature about the effectiveness, pain, and safety of a dorsal approach for CSI in trigger finger management.
Stage 2: Identifying Relevant Studies
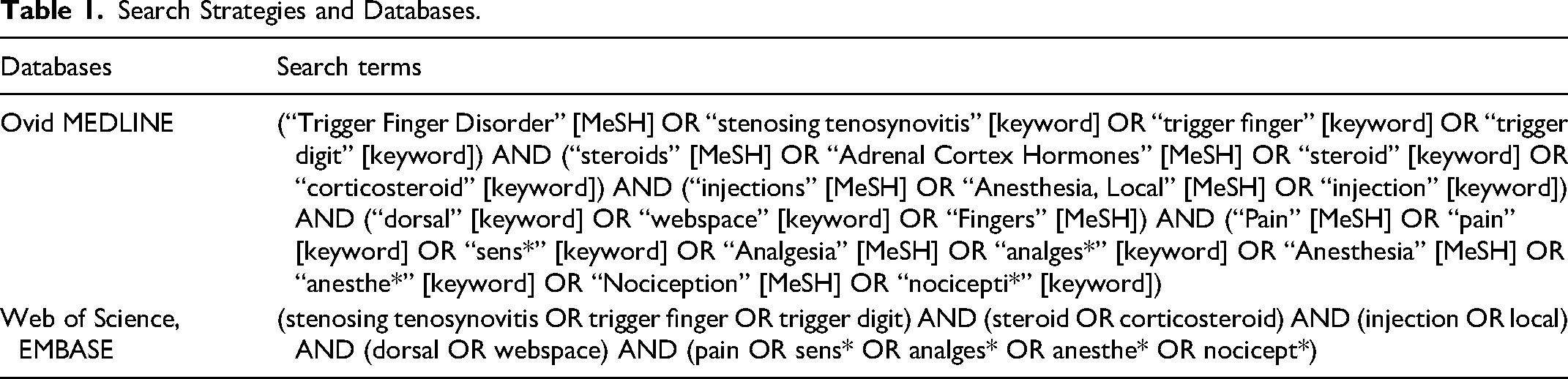
The search strategy focused on terms related to trigger finger, steroids, injections, and pain outcomes (Table 1). The searches were conducted in Ovid MEDLINE, Web of Science, and EMBASE in July 2024.
Search Strategies and Databases.
Literature management software (Covidence) was used for reference management, duplicate removal, title and abstract screening, full-text review, and data extraction.
Stage 3: Study Selection
The inclusion and exclusion criteria for selecting studies to include in this review (Table 2) were developed posthoc as we gained familiarity with the existing literature. 19 After removing duplicate studies in Covidence, inclusion and exclusion criteria were applied in a 2-stage screening process. First, 2 independent reviewers screened titles and abstracts to identify relevant literature. Any discrepancies in screening were resolved collaboratively after open discussion. Thereafter, the same 2 authors independently engaged in full-text review and any differences in inclusion were similarly resolved. Specific reasons for exclusion were recorded at the full-text review stage.
Inclusion and Exclusion Criteria.
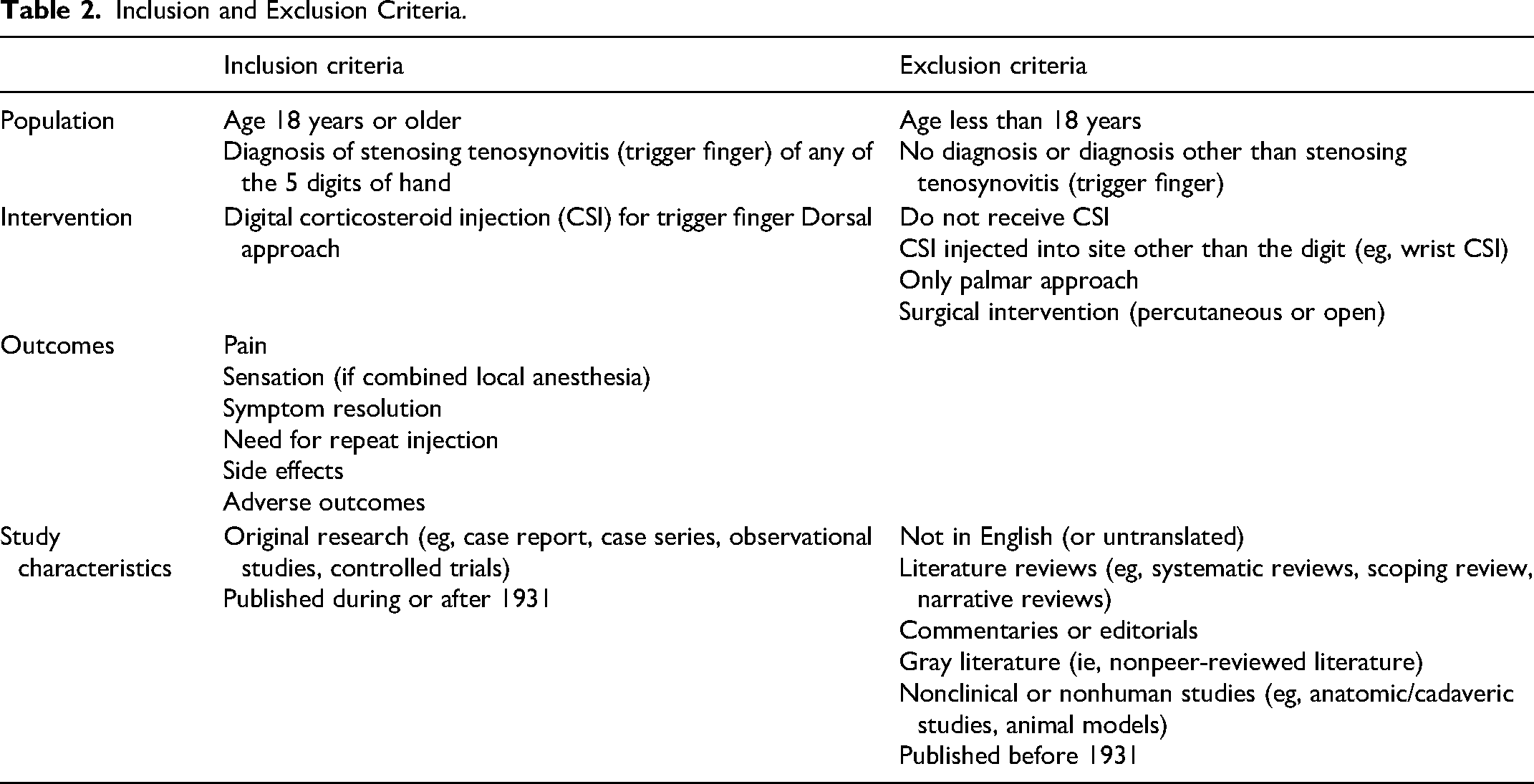
Overall, we included original clinical studies with adult patients diagnosed with trigger finger who received a CSI via a dorsal injection technique. Outcomes included pain, anesthesia (if applicable), efficacy, and adverse outcomes or complications. Studies were excluded if they involved pediatric patients, patients without a diagnosis of trigger finger, patients who received management other than a CSI (eg, A1 pulley release), or if the studies were not in English (or not translated to English).
Stage 4: Charting the Data
A data extraction form was developed, tested, and revised by 2 members of the research team who also completed extraction. After independent extraction was complete, discrepancies in extracted data were collectively reviewed and finalized based on consensus.
Effectiveness of dorsal CSI was defined as either complete symptom resolution or the proportion of patients not requiring a repeat injection for trigger finger symptoms. Pain was defined as measured using a validated pain score, such as the visual analogue scale (VAS) or numerical rating scale. Specifically, the VAS for pain is typically scored out of 10, with higher scores indicating more pain. 22
Assessment of Risk of Bias
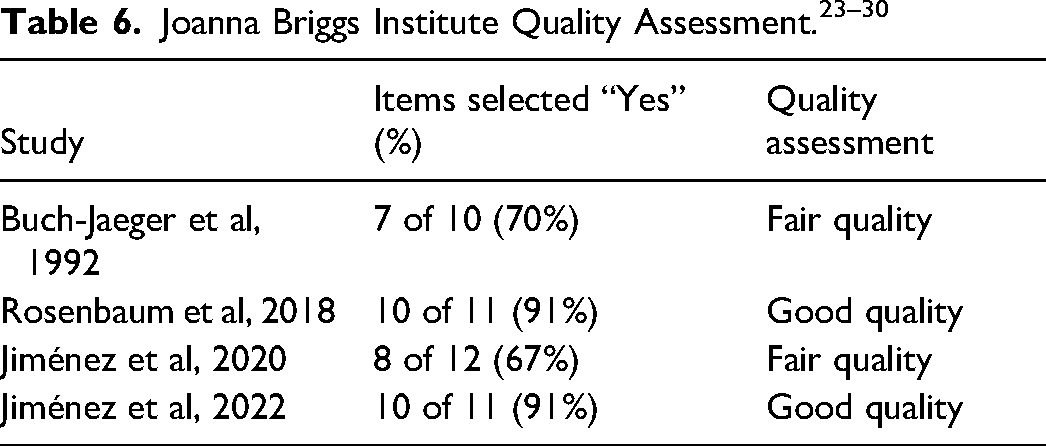
Based on recommendations by Levac and colleagues, 20 we also conducted a methodological quality assessment using the Joanna Briggs Institute (JBI) Critical Appraisal Tools for case series, cohort studies, and randomized controlled trials (RCTs).23–25 Two reviewers discussed the JBI Critical Appraisal Tools to establish shared understanding before independently critically appraising each included article using the appropriate JBI tool and then reviewing appraisals collectively for agreement.
In order to summarize the risk of bias for the included studies, we classified the studies based on the proportion of JBI checklist items answered as “Yes.” Specifically, we followed the rating score system as described by Kanda and colleagues 26 : very poor quality (0%-25% “Yes”); poor quality (26%-50% “Yes”); fair quality (51%-75% “Yes”); good quality (76%-100% “Yes”).
Stage 5: Collate, Summarize, and Report the Results
The following data were extracted and reported from included studies: study characteristics, setting, study population, intervention characteristics, outcomes (specifically measures of pain, sensation, effectiveness, and complications), and main findings.
Stage 6: Consultation Exercise
As recommended by Arksey and O’Malley 19 and re-emphasized by Levac and colleagues 20 we engaged in consultation with an academic hand surgeon and junior plastic surgery resident familiar with trigger finger management, specifically dorsal CSI techniques. After completing Stage 5 of the scoping review, both clinicians critiqued the findings of the review, provided content expertise to contextualize the meaning of the results, and highlighted areas for potential further investigation.
Results
Searches of the databases were completed in June 2024 and yielded 66 articles. After the removal of duplicates, title and abstract screening, and full-text review, 4 articles were included (Figure 1).
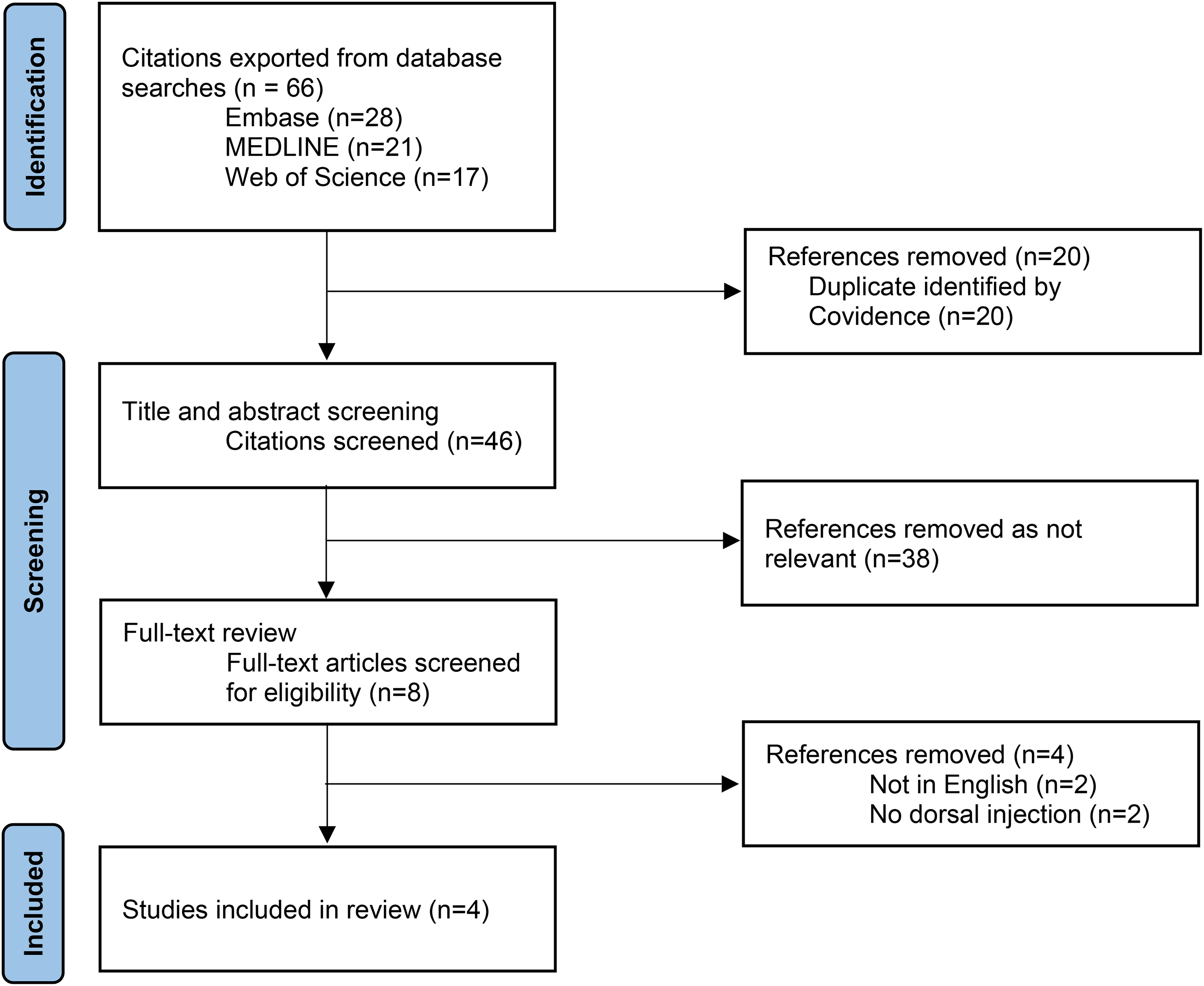
PRISMA flow chart of literature inclusion and exclusion process.
Study Characteristics and Setting
Included studies consisted of a case series (n = 1), cohort studies (n = 2), and an RCT (n = 1). Geographically, studies took place in Spain (n = 2), France (n = 1), and the United States (n = 1). All participants were recruited in surgeons’ clinics.
Study Population
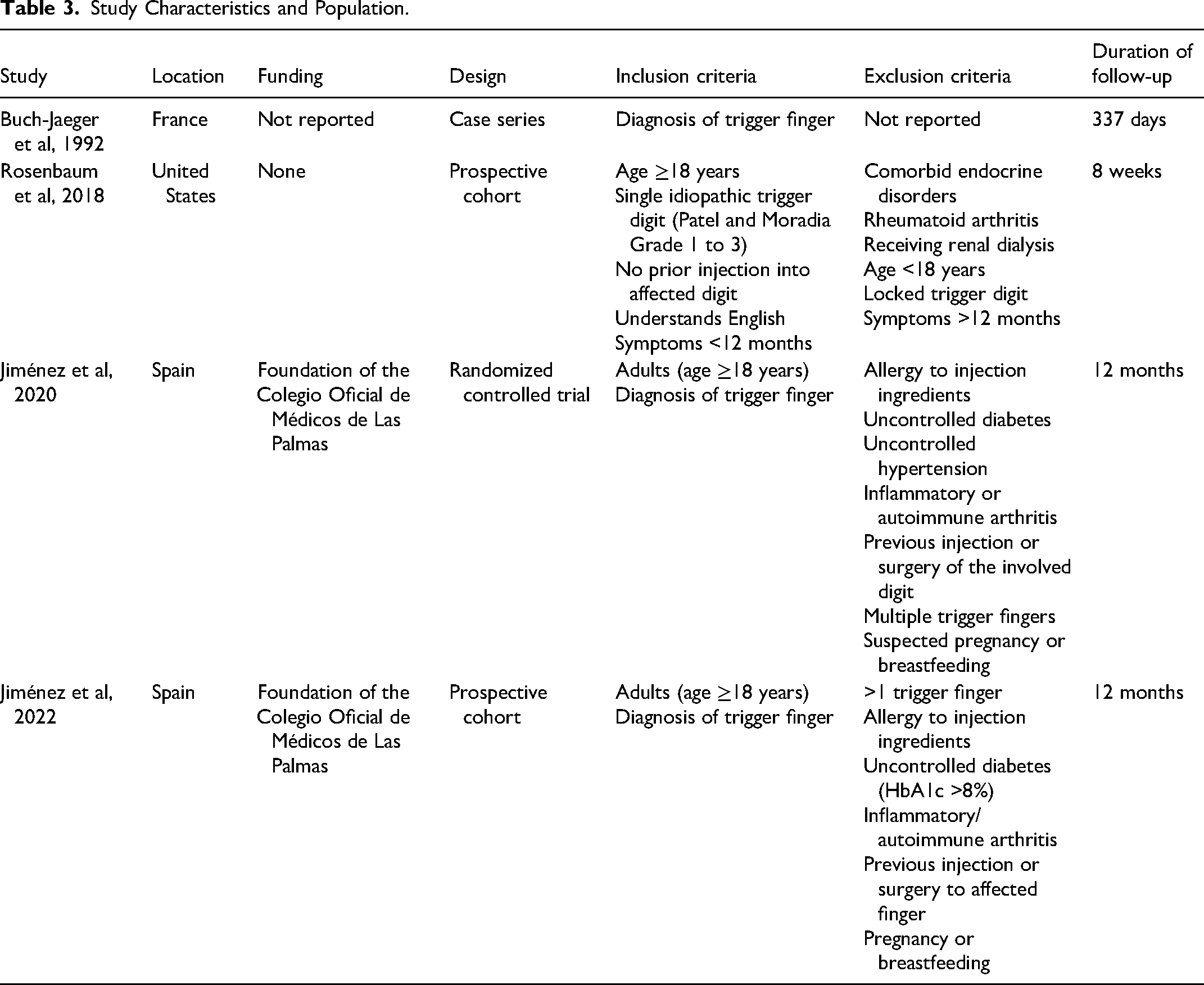
All studies included adult patients with a diagnosis of trigger finger. Exclusion criteria were reported by 3 studies which included, omitting patients with arthritis, prior CSI, and/or surgical treatment of the affected digit(s).27–29 Rosenbaum and colleagues also excluded all patients with diabetes mellitus (Tables 3 and 4). 29
Study Characteristics and Population.
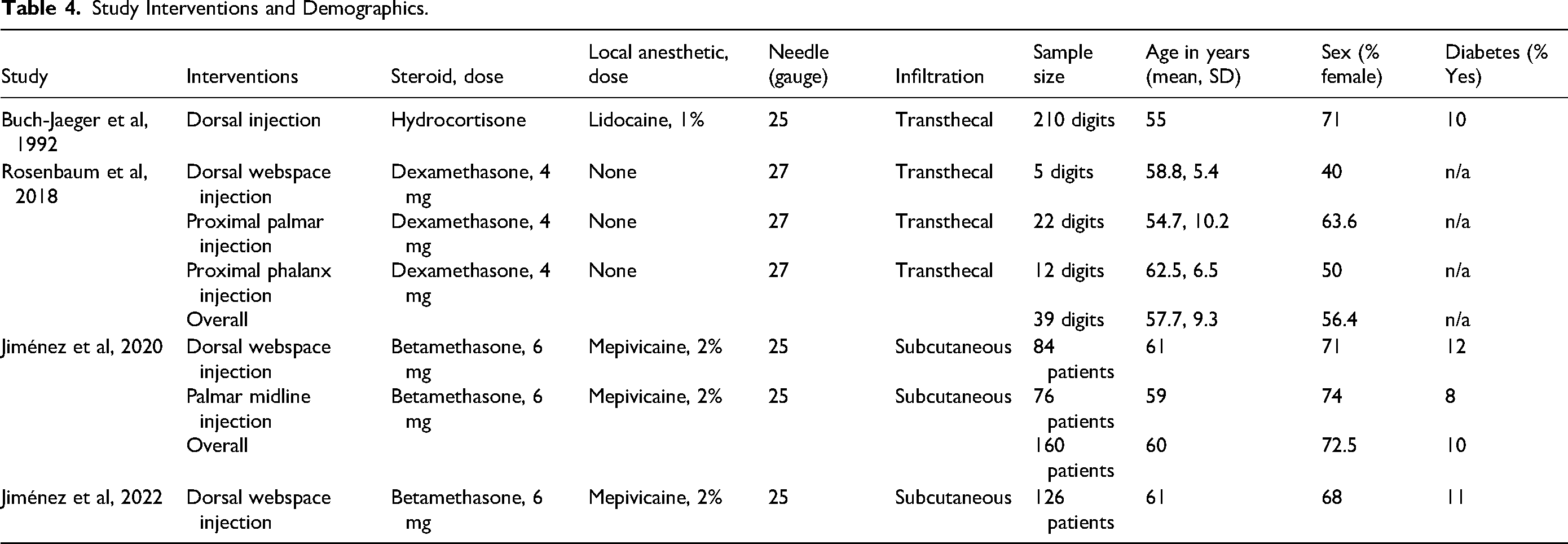
Study Interventions and Demographics.
Intervention Characteristics
All studies (n = 4) involved CSI for trigger finger delivered through the dorsal aspect of the hand via the webspace. Two studies compared a dorsal webspace CSI to 1 or more palmar CSI techniques: proximally over the metacarpal head at the A1 pulley (n = 2)27,29 or distally in the palmar surface of the first phalanx of the affected digit (n = 1). 29 Half the studies (n = 2) employed subcutaneous injection27,28 and the other half (n = 2) used a transthecal injection technique.29,30
Three different steroids were used in the CSI across the 4 studies; specifically, betamethasone,27,28 dexamethasone, 29 or hydrocortisone. 30 Most studies (n = 3) used a combined local anesthetic: mepivacaine 2%27,28 or lidocaine 1%. 30
Outcomes
Effectiveness
All studies included reported on CSI effectiveness, either as symptom resolution and/or reinjection rates for symptomatic persistence or recurrence.
Buch-Jaeger and colleagues 30 reported a 73.5% (128 of 174) symptom resolution rate 1 month after a single CSI. Of those with complete symptom resolution at 1 month, 82% (98 of 119) had no symptom recurrence at 6 months post-CSI. At 12 months, 21% (25 of 119) of all patients followed required a second CSI.
Rosenbaum and colleagues 29 found that 40% (2 of 5) of patients receiving a dorsal webspace CSI required repeat injection at 4 or 8 weeks, compared to 66.7% (8 of 12) in both the proximal palmar and proximal phalanx groups. Overall, however, the authors found no statistically significant difference in repeat injection rates across the 3 different injection sites.
In their RCT, Jiménez and colleagues 27 found that the initial CSI was successful in 51% (37 of 72) of patients in the dorsal webspace arm compared to 47% (31 of 66) in the palmar arm; after repeat injections, the overall symptom resolution rates at 12 months were 67% (48 of 72) for the dorsal technique and 56% (37 of 66) in the palmar technique. At 3 months postinitial CSI, there were an equal number of recurrences (n = 4) in each group. None of the abovementioned results achieved statistical significance.
Jiménez and colleagues 28 reported a 54% (62 of 114) symptom resolution rate at 12 months for a single dorsal injection and 66% (16 of 24) for a second CSI. The overall success rate was 68% (78 of 114). There were 4 symptomatic recurrences at 12 months of follow-up.
Sensation
None of the studies that used a combined local anesthetic in the injection (n = 3) assessed or reported sensation or anesthetic distribution for dorsal CSI.27,28,30
Pain
Three of the 4 studies included in this review reported injection-associated pain using the VAS.
Rosenbaum et al 29 found that dorsal CSI were associated with a 6.8 (1.8) mean (SD) VAS score, compared to 6.6 (2.6) for proximal palmar injection and 6.0 (2.8) for proximal phalanx injection, but no statistical significance (P = .754) in pain scores overall.
Jiménez and colleagues 27 reported a statistically significant difference in mean (SD) VAS pain scores; 3.6 (2.4) for dorsal injection and 5.4 (2.7) for palmar midline injection (P < .001).
Jiménez et al 28 reported an overall mean (SD) VAS score of 3.8 (2.3) for dorsal CSI.
Complications
None of the 4 included studies reported any adverse effects or complications associated with dorsal CSI for trigger finger. No complications were reported for any palmar injection techniques also evaluated in the included studies (Table 5).27–30
Study Results by Intervention.
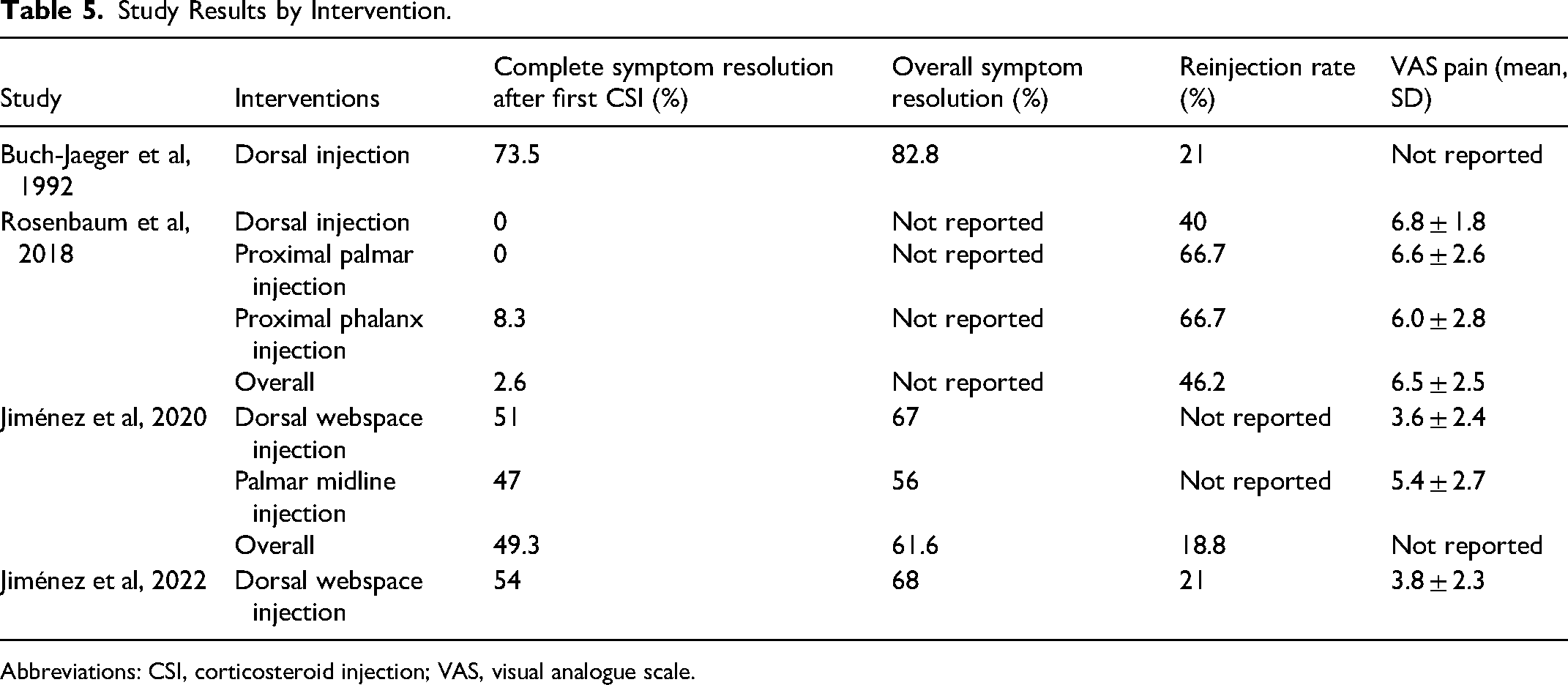
Abbreviations: CSI, corticosteroid injection; VAS, visual analogue scale.
Quality Assessment
Of the 4 included studies, 2 were of good quality and 2 of fair quality. The case series 30 lacked clear inclusion/exclusion criteria and did not describe how they identified or measured trigger finger in participants. The RCT did not conceal allocation and no blinding was used. 27 Both observational studies did not describe strategies to address incomplete follow-up (Table 6).28,29
Discussion
This scoping review examined the available evidence on the use of dorsal CSI for managing trigger finger. Despite the prevalence of trigger finger and the frequent use of CSI as a nonoperative treatment, there is limited evidence evaluating a dorsal approach for CSI. Furthermore, only 2 of the 4 studies compared dorsal and palmar injection techniques for trigger finger management, and we identified only 1 RCT comparing dorsal and palmar injections, highlighting the need for more high-quality studies on this topic.
Effectiveness
Symptom resolution with dorsal CSI was similar to rates reported in the literature. Both studies which compared dorsal and palmar injection techniques found similar or slightly better symptom resolution and reinjection rates for dorsal CSI but no statistically significant differences. Overall, this review suggests that a dorsal approach for CSI is noninferior to palmar injection for managing trigger finger.
Sensation
None of the studies which used a combined anesthetic agent assessed the distribution of anesthesia or changes in sensation postinjection. Future research should address this gap and compare the anesthetic distribution in both palmar and dorsal CSI as this has the potential to influence postinjection pain experiences for patients with trigger finger. 31
Pain
Pain associated with CSI for trigger finger is not insignificant for patients.31,32 This review found that VAS scores for pain associated with dorsal CSI were consistent with or lower than scores reported in the literature for palmar CSI. 31 When compared to palmar injection techniques, VAS pain scores for dorsal CSI were similar 29 or significantly lower, 27 suggesting there may be a potential advantage to a dorsal approach in reducing patient discomfort during injection. It is important to note that VAS pain scores were comparatively higher for all 3 injection techniques evaluated by Rosenbaum and colleagues 29 which may be due to the lack of a combined local anesthetic, impacting the pain scores reported soon after injection.
Complications
None of the studies in this review reported any adverse events or complications from dorsal CSI, suggesting it may be a similarly safe alternative to palmar CSI in trigger finger management.
Quality Assessment
In the 4 studies included in this review, we determined 2 studies were of good quality, while the remaining 2 were of fair quality. Notably, there was an absence of allocation concealment and blinding in the RCT 27 ; however, one must consider the practical difficulty in blinding for a study comparing injection locations and techniques, especially where a main outcome is pain associated with the intervention itself. Nevertheless, this review highlights the need for more rigorously designed and reported studies to better evaluate the effectiveness and safety of dorsal CSI for trigger finger.
Limitations
This review has some limitations. Most studies included (n = 3) were case series or observational studies, and only half of them (n = 2) compared dorsal CSI to palmar techniques. We recommend researchers conduct more high-quality trials directly comparing outcomes for palmar and dorsal CSI in trigger finger management. Additionally, the plane of CSI differed among studies, however, this is unlikely to substantially influence the results as transthecal and subcutaneous CSI have been found to have generally similar outcomes in trigger finger management. 33 Our review may also be impacted by publication bias; specifically, studies that may have found dorsal CSI to have worse or equivocal outcomes in trigger finger treatment may not have been submitted or accepted for publication, skewing the available evidence. 34
Implications for Practice
When choosing an approach for CSI in trigger finger, clinicians might consider medical evidence, patient-specific factors, and personal familiarity with injection techniques. This review revealed that, while current evidence does not definitively favor a dorsal approach over a palmar approach for CSI in trigger finger management, the potential for reduced injection-associated pain with the dorsal approach in combination with evidence of similar effectiveness and safety warrants further consideration.
Future Directions and Recommendations
Given the sparse evidence on dorsal CSI in the context of trigger finger, particularly the limited number of controlled studies, future research should focus on well-designed RCTs with larger sample sizes and standardized outcome measures in order to provide more robust evidence as to the optimal CSI technique for trigger finger. These studies should aim to directly compare dorsal and palmar CSI techniques, including assessments of pain, anesthetic distribution (where applicable), symptom resolution, and adverse effects. Additionally, researchers should consider qualitative or mixed-method studies characterizing clinicians’ awareness, familiarity, and comfort with dorsal CSI techniques for managing trigger finger.
Conclusion
This scoping review found that a dorsal CSI for trigger finger may offer comparable effectiveness and safety with potentially lower injection-associated pain compared to a palmar approach; however, the current literature is insufficient to make conclusive clinical recommendations. Future research should address existing gaps and methodological limitations to better guide clinical decision-making and improve patient experiences when performing CSI for managing trigger finger.
Supplemental Material
sj-docx-1-psg-10.1177_22925503251379892 - Supplemental material for Dorsal Approach for Corticosteroid Injection in Trigger Finger Management: A Scoping Review
Supplemental material, sj-docx-1-psg-10.1177_22925503251379892 for Dorsal Approach for Corticosteroid Injection in Trigger Finger Management: A Scoping Review by Muhammadhasan Nasser, Kaitlyn Soro, Natasha Barone and Kevin J. Zuo in Plastic Surgery
Footnotes
Acknowledgments
The authors would like to acknowledge the contributions of Daniel Antflek for providing critical feedback and refinement of the manuscript.
Author Contributions
MN and KJZ contributed to the conception and design of the review. MN and KS were responsible for screening and data extraction. MN synthesized and analyzed the data, with additional analytical input from KS, KJZ, and NB. MN drafted the manuscript, which was critically revised by KJZ, NB, and KS. KJZ supervised all aspects of the study. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
This article does not contain any studies with human or animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Muhammadhasan Nasser was supported by the Comprehensive Research Experience for Medical Students Program through the Temerty Faculty of Medicine and Division of Plastic Surgery, University Health Network. These agencies had no role in the design, collection, analyses, or interpretation of the results of this study or in the preparation, review, or approval of the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.