Abstract
Introduction
Rates of breast augmentation surgery remain high, with over 1.9 million people worldwide having the procedure done in 2023, making it one of the most common cosmetic procedures performed in the world.1,2 In North America, approximately 300,000 procedures were performed in 2023 for implant-based breast augmentation.3,4 This growing use of breast implants highlights the necessary public health measures to assess breast implant safety, which can be achieved with a national breast implant registry (BIR). Despite its recognized importance in the House of Commons Parliament session in November 2023, and societal advocacy dating back decades, Canada remains the only G7 country without an established BIR. 5 BIRs are used globally to study new implant associated risks such as Breast Implant Illness (BII), and Breast Implant Associated Anaplastic Large Cell Lymphoma (BIA-ALCL). 6 The 2010 French Poly Implant Prosthèse (PIP) crisis, in which non-medical grade silicone was used in thousands of implants worldwide, highlighted the consequences of not having a BIR. 7 The absence of a quality registry delayed efforts to control the situation and contact affected patients, resulting in increased costs, time and resource use. 7
BIRs have been instrumental in the investigation of modern implant safety concerns, including BII, BIA-ALCL and implant recalls.6,8–11 BII presents as a constellation of symptoms including fatigue, arthralgia and myalgia after implant insertion, and are alleviated with implant removal. 12 BIA-ALCL is a rare complication associated with the malignant deterioration of the breast implant capsule resulting in 1687 cases and 59 deaths across 51 countries as of 2024.5,13,14 The American PROFILE registry was successfully created to improve data collection and therefore help researchers characterize and understand this rare lymphoma. 13 Given the growing spotlight on BII and BIA-ALCL within recent years, patients’ concerns are mounting on implant safety and the absence of a Canadian BIR. 15 Patients have advocated for a formal implant registry to aid in continual breast implant safety research, improve follow-up care for breast implant patients and facilitate improved communication between patients and surgical teams. 15
The aim of this review was to comprehensively describe the design, development and implementation of existing international breast implant registries. Based on this synthesis, evidence-based recommendations are provided to guide the development and implementation of a Canadian BIR.
Methods
In accordance with the methodological checklist by Prisma, this systematic review was conducted to synthesize the evidence on the strategies used for BIR development and implementation. 16 Databases Medline Ovid, Web of Science and Embase Ovid were searched using the following search terms: (breast or mammo) AND (prosthesis or implant) AND (registry or database) in February 2025 with guidance from an academic librarian. 11 Grey literature sources were also queried with keywords to identify relevant reports from implant registries that are not stored in academic databases.
Articles were included if they were primary journal articles or reports focused on developing or refining breast implant registries. The registries were required to be national, with specific mention of the methods used for registry data collection. Included reports were required to be published in 2000 or later, written in English, and discuss registry data from adults (>18 years old). Articles were excluded if they used non-breast implant related registries or used BIR data to perform research independent from registry development or design.
Quality appraisal and data extraction were completed by two reviewers. Data was collected on patient recruitment, data quality, sustainability, funding, data output reports, international collaboration and patient reported outcome measures (PROMs). Reference lists of published systematic reviews and included articles were reviewed to ensure the quality of our systematic review. Narrative analysis of the data was completed by summarizing the evidence on the structures and strategies used for both the development and sustainability of a functional national BIR.
Results
Study Characteristics
Our search yielded a total of 1577 articles. Once duplicates were removed, 1100 articles were reviewed by title and abstract screening. Sixty-nine articles underwent full text review and a total of 19 articles met inclusion criteria (Figure 1). A total of 19 primary journal articles, and grey literature sources were included (Table 1). Implant registries were identified from the Netherlands (n = 4), Australia (n = 6), United Kingdom (n = 1), Korea (n = 1), America (n = 2), Germany (n = 1) and Denmark (n = 1), or unspecified registry (n = 3). A total of 12 articles discussed how to refine a registry, while others detailed the initial registry development (n = 5) and 2 articles discussed both. Grey literature was also sourced for registries that did not meet inclusion criteria and can be found in Supplementary Tables S1 and S2.
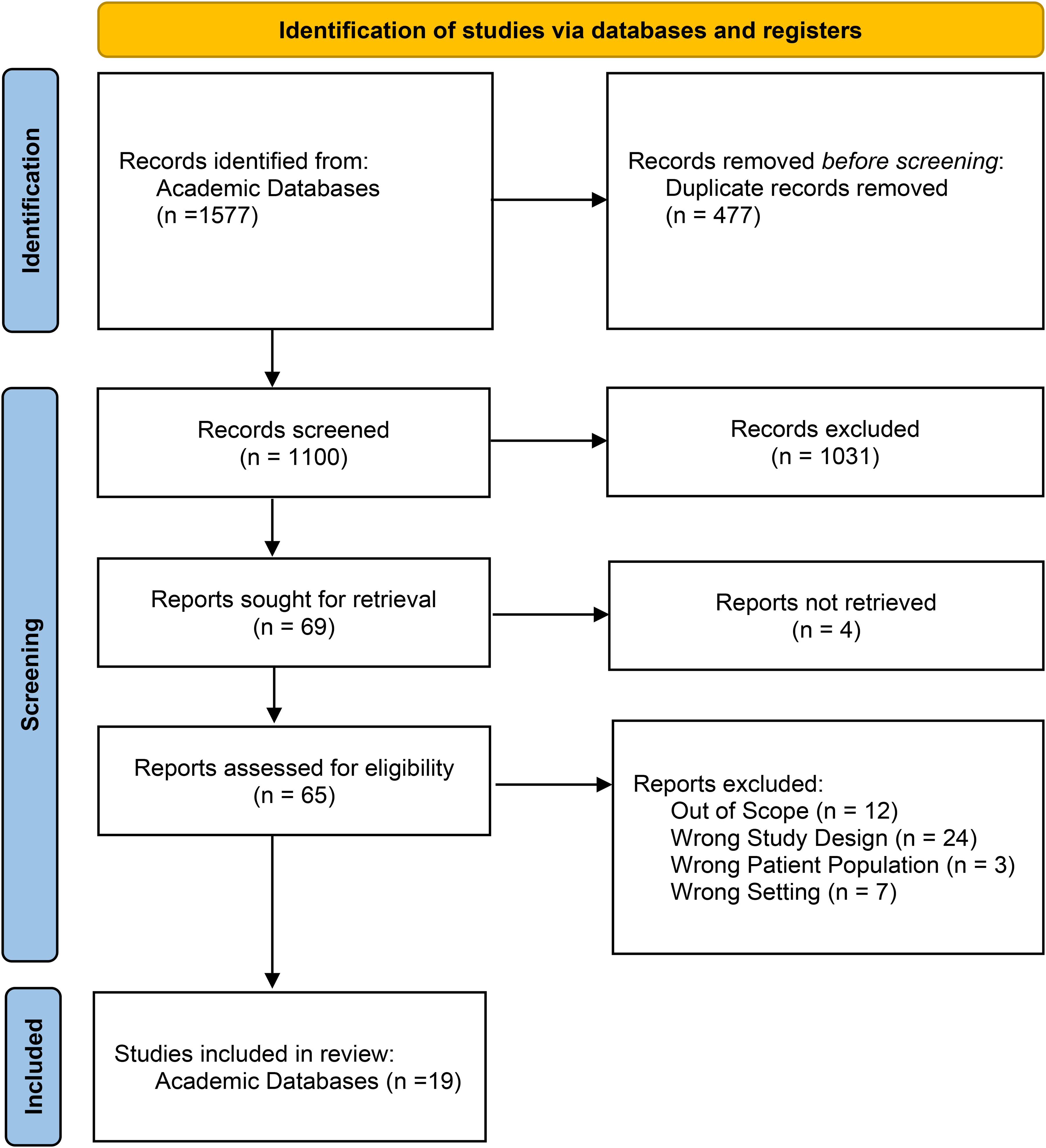
PRISMA diagram created using the PRISMA flow diagram tool. 17
Aims and Scope of Registries.

Characteristics of existing national BIR are summarized and detailed in Supplemental Tables S1 and S2. Evidence-based strengths and weaknesses of each registry are summarized in Table 2. Strengths of a functional BIR include strategies to maximize patient recruitment, optimize quality and completeness of data input, sustainable funding and output reports.
Strengths and Weaknesses of Existing National Breast Implant Registries.

Patient Recruitment
In an opt-in registry, patients must actively consent to participate, whereas in an opt-out registry, patients are included by default unless they choose not to be. Opt-out consent systems are noted in the Dutch, American and Australian registries. A mixed system is used in the United Kingdom, where there is an opt-in system in Ireland and opt-out for patients in England and Scotland. The Korean BIR Pilot study was also an opt-in system. Participation in the German registry is mandatory.
Data Quality
All registries studied involve physician or surgeon data input into the registry. The Dutch and American (NBIR) registries use a barcode system for data input. The UK BIR has an interactive dashboard online. The Korean BIR is currently creating a mobile app program for physicians and patients. It is unclear how data input is managed in the German registry. International Collaboration of Breast Registry Activities (ICOBRA) minimum data set requirements are used by the following registries: Dutch, United Kingdom, Australian, Germany and Korea. The Australian BDR was the only registry that used PROMs; however, this is being adopted by the American NBIR.
Sustainability
Private funding is used by the Dutch BIR via a €25 patient surcharge obtained through the patient or insurance. Government funding is used by United Kingdom, Australian and Korean registries. Ministry of Health Government funding was used in the German registry for initial development, but a future financial plan is unknown. The funding sources of the American registries are also unclear.
Output Reports
Annual public output reports based off registry findings are released by the Dutch, United Kingdom, Australian and American (NBIR) registries.
Discussion
Recruitment and Opt-Out System
An opt-out system is one where all patients who have received breast implants are automatically enrolled in the implant registry, and can be observed in the American NBIR, Dutch, United Kingdom and Australian registries. 18 The German registry is the first registry with mandatory participation, although this registry is not yet functional. 19 In contrast, the Korean registry used an opt-in system due to study design, but recommended the use of an opt-out system. 1 To highlight the importance of an opt-out system we can compare capture rates between the Canadian Joint Replacement Registry (CJRR) and Australian Joint Registries. The Canadian registry had an implant capture rate of 68.2% from 2020 to 2021; a lower capture rate than the opt-out Australian National Joint Replacement Registry of 97%.7,20,21 In general, countries that use an opt-in system have significantly lower capture rates, leading to the premature closure of previous registries; for this reason, we recommend an opt-out system for a Canadian registry.7,22
Administrative Burden
High administrative burden for plastic surgeons has limited registry participation and data accuracy in existing registries, prompting proposed solutions like barcode data entry systems and financial incentives.23–25 Though it is widely recognized that registry participation includes increased administrative burden for participating surgeons, a survey for the Dutch BIR users reported that participation was of value despite this burden. Recommendations for increased surgeon participation and data accuracy are discussed below.
While the registries included in this review do not offer financial incentives to participating physicians or residents, such incentives are recognized as one of the most important motivators for physician participation.25,26 The use of a barcode system can be seen in the Dutch BIR where the barcode automatically adds the unique device number, global trade number, expiration date, lot number and serial number into the registry form.23,26,27 Notably, the American National Breast Implant Registry endorses multiple pathways of secure data input into their registry including: manual data entry, a barcode scanning app and a device tracking app. The device tracking app is a new feature which allows physicians to fulfil the federal requirements for breast implant device tracking which is simultaneously forwarded to implant manufacturers. 28 As registry design evolves to reduce administrative burden, equal attention must be given to ensure data security. In the Report of the Standing Committee on Health by the House of Commons Canada, published November 2023, the Canadian Institute for Health Information (CIHI) suggests that greater government infrastructure and legislation are needed to ensure adequate registry privacy and confidentiality.5,25
Data Quality: ICOBRA, PROMS, FAIR
The use of minimum international datasets through ICOBRA are also highly recommended to facilitate better data linking between countries; a strategy that is already used by the CJRR.7,21,25 This includes collecting data on patient demographics, patient history, patient outcomes, surgical procedure and intraoperative techniques such as glove changing.7,10,29,30 Though not included in ICOBRA, to account for factors influencing quality of care, surgical and institutional details could be recorded. Overall, it is important to ensure that the development of a Canadian BIR uses internationally acceptable datasets, to prepare for future international collaborations.
The introduction of PROMs in registries has been a quality improvement initiative adopted by ICOBRA, Australian and American (NBIR) registries, and we recommend this in a Canadian registry.28,31 PROMs are classified as outcome measures reported in a registry directly from the patient without any interpretation from the clinician.32,33 PROMs in the Australian registry allow patients to comment on their satisfaction level as it pertains to implant look, feel, rippling, pain and tightness after surgery and can be reported on a patient mobile dashboard.31–34 This captures important feedback from patients as it pertains to implant satisfaction and performance.10,32
To ensure registry data is usable and accessible FAIR principles must be considered during the data collection phase. This ensures data is findable, accessible, interoperable and reusable. This would allow physicians to evaluate their own patient outcomes as they can easily view and evaluate their own provision of surgical care, which in turn may encourage registry participation. 26
Funding and Governance
The importance of continual long-term funding must be emphasized, as registries in the past have halted activity due to funding constraints, as seen in Austria. 7 Registries can be funded by the government, patients, or grants. Government funding is used in the United Kingdom, Australian, German and the Korean Pilot BIRs; although, a noted weakness of government funding is that it is dependent on the political climate. Government involvement can also be useful in the development of a registry through their ability to mandate hospital, clinic and surgeon participation in the BIR which would lead to increased participation rates. 22 Alternatively, consistent funding can be maintained from patient contribution, as showcased by the Dutch BIR where there is a €25 charge per implant; however, a patient surcharge could cause the patient to refuse participation in the registry. 22 Finally, funding can also be accessed through grants, as seen in the Dutch BIR where a €130,000 non-government grant was used for the initial development of the registry. 27
Health Canada's Medical Devices Directorate stated that the financial cost of such registry is unknown until a registry scope was determined. 35 To gain perspective on potential financial implications of a Canadian BIR, we can adopt the funding model used by the CJRR which receives funding from Health Canada and provincial/territorial governments.20,36 Although the financial needs of a registry may seem costly at first, the development can reduce total public health spending. 7
Proper registry governance is another essential component of a successful registry, as it can aid in superior development and maintenance of a BIR. 25 The Australian and American registries are good examples as they have continual stakeholder support which includes a steering committee, university partners, data publications committee and a regulatory board.33,37 For the governance of a Canadian BIR, we can look to the CJRR for guidance. The CJRR is governed by an advisory board comprised of orthopedic surgeons, and representatives from the Ministry of Health, Arthritis Society, Canadian Orthopedic Association and the Canadian Orthopedic Nurses Association. 36 Similar stakeholders for a Canadian BIR could include: plastic surgeons, patients, CIHI, The Breast Implant Safety Alliance, Canadian Institute of Health Research, Canadian Society of Plastic Surgeons, Canadian Society for Aesthetic Surgery, Public Health and University partners.
Output Reports
It is recommended that Canada includes annual output reports of registry findings. The use of output reports has been noted in many successful implant registries such as the Dutch, United Kingdom, American and Australian breast implant registries. The American NBIR has a committee including an epidemiologist and statistician responsible for data reporting, research and publications to guarantee prompt and accurate output reports. 28 These reports can be made publicly available for clinicians, implant manufacturers and patients to promote research dissemination.
Limitations
This study was limited to the inclusion criteria specified in our database research. As a result, we did not assess some successful breast implant registries such as those in: Austria, Italy, France, Sweden, Spain or Russia. To account for this, we have added information in Supplementary Table S2. The results and recommendations for a Canadian BIR, may benefit from the perspectives of these registries. Further, some included studies were published in the early 2000s and the registry design may have been updated since. To address this bias, we reviewed grey literature sources to include the most updated information on existing registries.
Conclusion
We recommend the development of a registry that has low administrative burden, incentives for participating physicians, continual funding partners, governance with relevant stakeholders, an opt-out system and the inclusion of PROMs and ICOBRA minimum dataset.
Supplemental Material
sj-docx-1-psg-10.1177_22925503251355977 - Supplemental material for Recommendations for a Canadian Breast Implant Registry
Supplemental material, sj-docx-1-psg-10.1177_22925503251355977 for Recommendations for a Canadian Breast Implant Registry by Victoria M.S. Rea, Raveena Gowda, Emma Nicholson and Kathryn V. Isaac in Plastic Surgery
Footnotes
Acknowledgments
Dr Kathryn Isaac is holder of the Dr Patricia Clugston Chair in Breast Reconstruction Surgery at the University of British Columbia and would like to acknowledge the support of VGH & UBC Hospital Foundation's donors and partners who made this Chair possible.
Author Contributions
Victoria M.S. Rea did data collection, data analysis, manuscript writing, review and editing manuscript.
Raveena Gowda did data collection, data analysis, manuscript writing, review and editing manuscript.
Emma Nicholson did project conception of protocol and data review.
Kathryn V. Isaac did project administration, supervision, conceptualization, manuscript writing, review and editing manuscript.
All authors have read and approved the final manuscript.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Considerations
This article does not contain any studies with human or animal subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement on Related Presentations
This abstract was presented at the 2023 Canadian Society of Plastic Surgeons Annual Meeting.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
