Abstract
Background
Reduction mammaplasty is defined as the surgical removal of breast tissue and may be done for several indications. This procedure is commonly performed bilaterally to relieve symptoms of macromastia or unilaterally for symmetrization following resection of malignant lesions in the contralateral breast or to correct congenital asymmetry.1–3 A reduction mammaplasty technique can also be used in lumpectomies with a large tumor to breast ratio to facilitate the excision of a large specimen and reshape the breast, minimizing the risk of deformity postradiation, referred to as oncoplastic reduction. 4 According to the American Society of Plastic Surgeons, 97 320 breast reductions were performed by their member surgeons in 2020. 4 There is no recent Canadian equivalent for this data. However, breast reductions for macromastia are covered by all Canadian provincial healthcare insurance plans, and contralateral symmetrizing reduction mammaplasty is frequently included in the management plan of breast cancer patients. 1
Reduction mammaplasty improves quality of life both in patients with macromastia and in patients with contralateral ablative procedures. 5 Globally, mammaplasties have a very high patient satisfaction rate with patients reporting improvements in back and shoulder pain, body image, anxiety, and depression.5–7 Furthermore, bilateral breast reduction for macromastia has been shown to improve patients’ satisfaction with breast appearance, psychosocial well-being, sexual well-being, and physical well-being when assessed with the BREAST-Q scoring scale. 8
Breast cancer is the most common cancer among women worldwide, including in Canada, where it accounts for 1 in 4 of all new cancer diagnoses among women. 9 Breast cancer has a high morbidity and mortality burden—deaths from breast cancer comprise 13% of total cancer deaths among Canadian women each year. 9 Despite regular screening, a small proportion of abnormal pathologies remain radiologically undetected and are found incidentally at the time of breast reduction when resected breast tissue is sent for pathologic examination.5,10
Several American studies have evaluated the incidence of occult breast cancer and other high-risk pathologies in reduction mammaplasty specimens and reported values vary from 0.01% to 1.8% in reduction procedures for macromastia and from 1.1% to 8.0% in mammaplasty following contralateral breast cancer.2,3,5,6,11–13 The last and only Canadian study on this topic was conducted in1999 by Tang et al. This study reported an invasive cancer rate of 0.06%; however, they only examined the presence of invasive cancer and did not report on other high-risk pathologies (atypical ductal hyperplasia (ADH), atypical lobular hyperplasia (ALH), lobular carcinoma in situ (LCIS), ductal carcinoma in situ (DCIS), severe ADH bordering on DCIS). 14 These high-risk pathologies in the literature are described as lesions that have been linked to an increased lifetime risk of developing breast cancer. 15
Furthermore, there is a lack of consensus regarding the importance of risk factors, including personal history of breast cancer, family history of breast cancer, genetic markers, smoking history, patient age and BMI, and weight of the resected specimen. There is consensus about the incidence of high-risk in-situ and malignant lesions increasing with age10,16,17; however, most studies do not account for the fact that women with a personal history of cancer who undergo contralateral breast reduction are often older, compared to women undergoing mammaplasty for macromastia. Prior studies also vary widely in their consideration of family history as a risk factor for breast cancer, which limits generalizability. Similarly, there are conflicting findings on the association between the weight of the resected specimen and the incidence of high-risk or malignant lesions.10,18
The lack of consensus regarding risk factors, the paucity of data on in-situ detection rates, and the low incidence of invasive cancers warrant reinvestigation of postreduction mammaplasty patients in a Canadian context. The aim of this study is to report the incidence of malignant and high-risk breast pathology findings in patients undergoing reduction mammaplasty and contralateral, balancing reduction in cases of unilateral breast conservation therapy.
Methods
Following Research Ethics Board approval, we conducted a retrospective review of electronic medical records for all patients aged 18 and above who underwent reduction mammaplasty at the affiliated academic centers between 2001 and May 2023. This review included both bilateral reductions and unilateral symmetrizing surgeries performed by the 5 plastic surgeons working at the center during this period. Patients were excluded only if data was missing from pathology reports.
Data extraction fell under 3 categories: demographics, risk factors identified during preoperative consultation, and pathology findings. Demographic data included age at the time of surgery and sex. Data extracted from preoperative consultation notes included personal history of breast cancer, family history (first or second-degree relatives) of breast cancer, and BMI. Personal history and family history of breast cancer were recorded as present or absent. Specimen weight and pathologic findings were collected from pathology reports. Patients were categorized as having malignant lesions, high-risk lesions, low-risk/benign lesions, or no abnormal pathology findings. Malignant lesions included invasive lobular carcinoma, or DCIS, and invasive ductal carcinoma. High-risk lesions included ADH, ALH, LCIS, and severe ADH bordering on DCIS. Low-risk findings included fibrocystic changes, fibroadenoma, duct ectasia, intraductal papilloma or sclerosing adenosis. If more than one finding was reported in a single specimen, it was classified according to the lesion of highest severity.
Patients were divided into 2 groups. Group A consisted of patients who underwent bilateral reduction for symptomatic macromastia. Group B consisted of patients who underwent balancing breast reduction for symmetrization purposes post unilateral breast-conserving surgery for cancer. The patient demographics were analyzed by patient whereas the unit of measurement for analysis of pathology findings was by breast.
Statistical analyses were performed using IBM SPSS version 26.0 (Macintosh. VA, NY). Independent t-tests and Pearson's Chi-squared test with Yates’ continuity corrections were used to compare patient demographics with continuous variables reported as mean and standard deviation. Categorical variables were calculated as frequency and percentage. Multivariate regression analysis was used to identify independent predictors for high-risk and malignant lesions with ORs and 95% Confidence Intervals (CI). All tests were 2-sided, and significance was set at P < .05. Furthermore, a subgroup analysis was carried out to calculate an age-adjusted incidence of malignant and high-risk pathology findings between patients with and without a personal history of breast cancer.
Results
Patient Characteristics
This retrospective study included a total of 1383 breasts from 872 patients who underwent reduction mammoplasties at the affiliated academic centers between January 2001 and May 2023. Group A consisted of 1022 breasts from 511 patients who had bilateral mammaplasties for macromastia. Group B consisted of 361 breasts from patients who underwent a unilateral reduction postbreast-conserving surgery. Table 1 shows demographic data for the 2 groups. Group B was significantly older (56.9 ± 9.3 vs 41.1 ± 13.6 years, P = .003) while Group A had a higher mean BMI (33.1 ± 8.4 vs 30.1 ± 5.8, P < .0001) and higher reduction weights (680.5 ± 374.0 and 629.7 ± 321.1 g, P = .015) (Table 1). BMI and reduction weight were significantly correlated for Group A (r = 0.52, P < .05) and Group B (r = 0.55, P < .05). Of the 872 patients, preoperative consult notes were identified for 512 patients; 211 for Group A and 301 for Group B. Fifteen (1.5%) of the patients with identified consult notes had a documented personal history of breast cancer. Despite patients in Group B undergoing balancing reduction, only 8 (2.7%) of these patients had a documented conversation about a previous breast cancer diagnosis with their plastic surgeon. A history of breast cancer diagnosis was discussed and documented for 7 (3.3%) of patients in Group A. One hundred eight (21.1%) patients reported a family history of breast cancer; 35 of these patients were in Group A and 73 were in Group B.
Patient Perioperative Demographics.
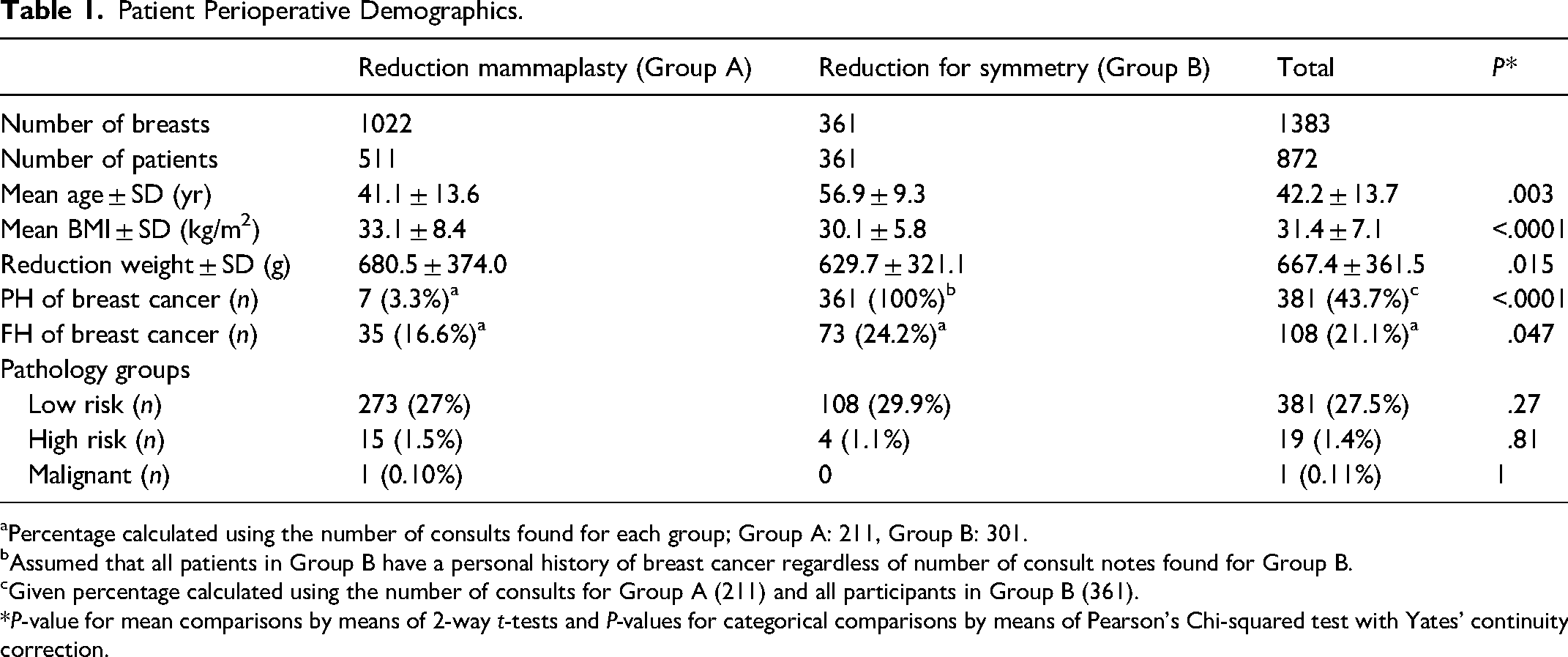
Percentage calculated using the number of consults found for each group; Group A: 211, Group B: 301.
Assumed that all patients in Group B have a personal history of breast cancer regardless of number of consult notes found for Group B.
Given percentage calculated using the number of consults for Group A (211) and all participants in Group B (361).
*P-value for mean comparisons by means of 2-way t-tests and P-values for categorical comparisons by means of Pearson's Chi-squared test with Yates’ continuity correction.
High-risk and malignant pathology incidence was 1.4% overall. The sole malignancy detected was in a patient who had a bilateral reduction (Group A) without a prior breast cancer history. Multivariate analysis revealed BMI as a significant predictor for high-risk and malignant pathologies (OR 1.134, 95% CI [1.012-1.271]). There was no statistically significant difference in the incidence of low-risk findings between the 2 groups (27.0% in Group A vs 29.9% in Group B, P = .27). The most common pathological finding was fibrocystic changes (Table 2).
Incidence of Low-Risk, High Risk, and Malignant Lesions.
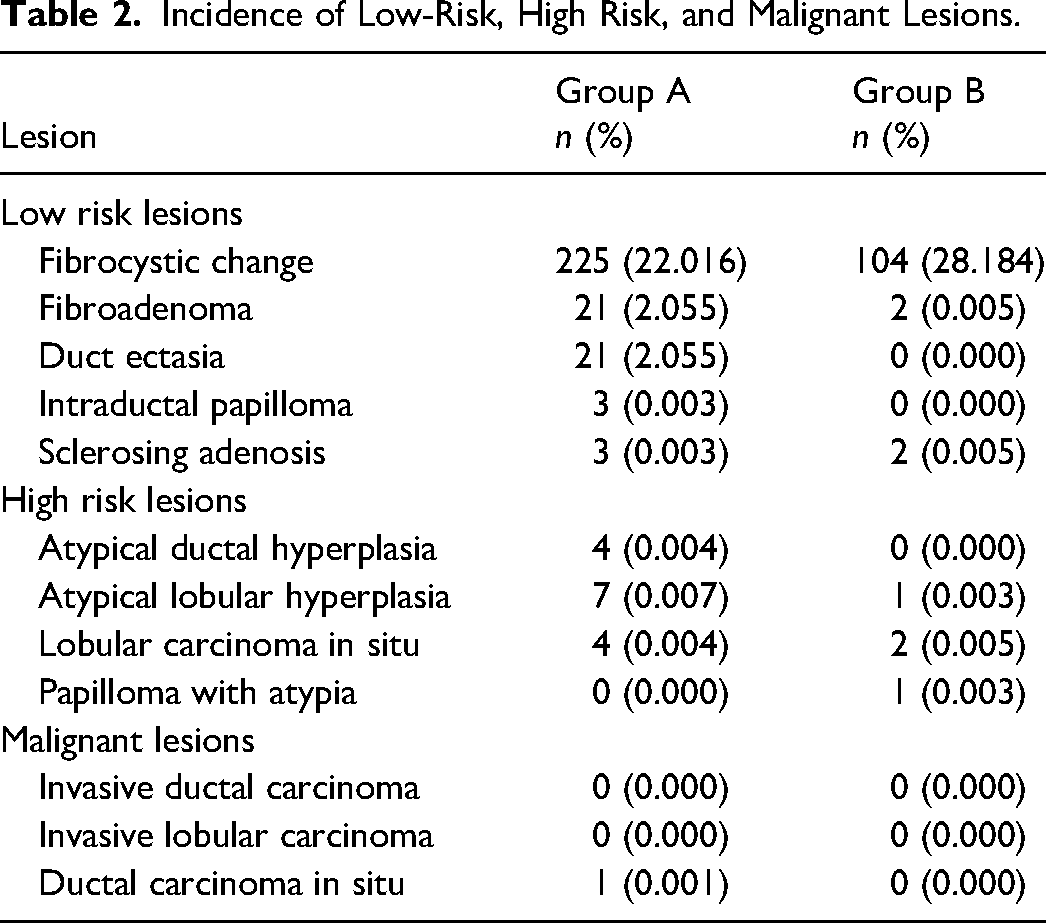
On multivariate regression analyzing impact of family history, BMI, bilateral/unilateral reduction, reduction weight, and height; BMI was the only factor found to have a significant association with the finding of a high-risk pathology (OR of 1.134, 95% CI [1.012-1.271]).
Discussion
In this study, we found the incidence of high-risk or malignant findings in reduction mammaplasty specimen at 1.4%, consistent with previously reported breast cancer incidence rates discovered at the time of breast reduction.2,3,5,6,11–13 We identified one patient with a malignant pathology finding at the time of reduction mammaplasty. This patient was undergoing bilateral reduction mammaplasty for benign symptomatic macromastia and did not have a personal history of cancer. They did, however, report a family history of breast cancer and were over the age of 50 at the time of surgery.
Our findings suggest that malignancies are uncommon in patients undergoing reduction mammaplasty, potentially reflecting the effectiveness of Canada's breast cancer screening programs. Patients who reported a personal history of breast cancer were on average older than patients without a history of cancer emphasizing that increasing age is a risk factor for breast cancer. Historically, in Ontario, patients were only recommended to undergo breast cancer screening between the ages of 50 and 75 years old unless they fall into a high-risk group. 19
This has recently been changed to begin screening women at age 40 starting in the fall of 2024 in an attempt to detect more cancers at earlier stages. 20 The results of our retrospective review are consistent with the body of literature suggesting individuals younger than 50 are unlikely to have incidental breast findings on pathologic examination in the absence of preoperative examination findings. This highlights the potential for these changes in the screening program to result in an increase in false positive results, or delay the screening of individuals in high risk groups.
Low-risk findings were identified in 27.5% of specimens, the most common finding being fibrocystic change. There was no significant difference in the incidence of low-risk findings between patients undergoing bilateral versus unilateral reduction. This is consistent with previous studies of breast reduction patients indicating that up to a third of women between the ages of 20 and 45 will have some histologic evidence of fibrocystic changes.21,22 The mean BMI for Group A was larger than that of Group B, which is consistent with their differing surgical indications. Additionally, we identified a correlation between higher BMI and the presence of high-risk and malignant breast pathologies. The reason for this occurrence is likely multifactorial, with the known risk of increased estrogens and an increased inflammatory state which are associated with an increased BMI.23,24 These findings highlight the importance of ensuring that all patients, especially the patients with higher BMIs undergo preoperative breast examination by their surgeon. Patients should be counselled, that, even though it is small, there is a risk of finding a malignant lesion during their surgery and the impact of such a finding with respect to further management should be discussed.
The importance of gathering accurate and adequate information during the preoperative consultation of a patient seeking breast reduction irrespective of the reason cannot be overemphasized. Unfortunately, a large number of our patients, mostly in group A, were missing preoperative consults and the ones available were further missing important information on personal and family history of breast disease. This limits the analysis that can be carried out in such retrospective reviews. The adoption of a standardized questionnaire for patients who choose to undergo reduction mammaplasty that includes important information including BMI, detailed past medical and surgical history, radiation history, family history of breast disease is a key step in the ability to conduct meaningful research that can further assist in appropriate counselling of our patients. Even if the risk of an incidental abnormal finding is low at about 1%, postoperative management of these patients can be complex and includes surgical, medical, and radiation oncology consultations, possible further surgical resection including complete mastectomy to reduce the risk of local recurrence.3,11 Furthermore, the possibility of reexcision to obtain clear margins without the need for complete mastectomy highlights the importance of specimen orientation and en-bloc resection, where possible.2,25 In-depth preoperative discussions are essential for enabling patients, particularly those with elevated BMI, to make well-informed decisions before proceeding with surgery and be prepared for the possibility of a pathologic finding. Future studies should be done prospectively, using a standardized preoperative questionnaire and exam to accurately measure the risk of abnormal and specifically malignant, lesions in this patient population.
Limitations
A key limitation of our study is its retrospective design, which restricted our ability to gather thorough, uniform data on patients’ risk factors as indicated by the lack of detailed, preoperative consultations. Factors including timing of cancer detection, institutional changes in reconstruction practices, and differences in cancer type are important considerations that could impact outcomes such as patient age at surgery and the risk of atypia or malignancy. These can be addressed in a future study with a larger population and ideally at a site with comprehensive electronic medical records. Additionally, the small sample size likely diminished the statistical significance of our findings. Despite these, our results are consistent with previous literature on the incidence of malignant pathology findings in mammaplasty specimens. Implementing a standardized preoperative questionnaire for those contemplating reduction mammaplasty could greatly enhance the accuracy and comprehensiveness of data, benefit patient counselling, management of patients with abnormal findings and help design future research. Prospective studies with improved electronic medical records and larger cohorts are recommended to facilitate data collection and provide more conclusive evidence on the risks of incidental high-risk or malignant of pathology findings at the time of breast reduction surgery.
Conclusions
Our findings present updated Canadian data, aligned with previously reported incidence rates of pathological findings in mammaplasty specimens and highlight the correlation between BMI and pathology risk. These results are significant for clinicians who perform and counsel patients about breast reduction surgery, whether they perform breast reconstruction or not. Acknowledging the shortcomings of the data presented above highlights the importance of developing a standardized approach to collecting pre- and postoperative data across multiple Canadian institutions to provide a comprehensive overview of the Canadian context. Moreover, as breast cancer screening guidelines broaden to include younger individuals, we may observe increased rates of oncoplastic and balancing reductions. Collecting accurate preoperative data through this period of transition will be essential for our healthcare system.
Footnotes
Author contributions
BH: developed study design, data collection, data analysis, and manuscript preparation; KJ: assisted with study design, data collection, data analysis, and manuscript preparation; SS: assisted with study design, data collection, data analysis, and manuscript preparation; LK: assisted with data analysis, presentation, manuscript preparation, and submission formatting; RP: assisted with ethics application and study design; DM: assisted with manuscript preparation and editing; GM: oversaw and supervised project, advised on study design, data analysis, and manuscript preparation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Research ethics statement
Ethics approval was obtained from the Queen's University Health Sciences and Affiliated Teaching Hospitals Research Ethics Board. Internal ethics approval number: SURG-578-22. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
