Abstract
Introduction
Throughout the 20th and 21st centuries, the number of patients seeking cosmetic procedures has dramatically increased in popularity. In 2018, cosmetic procedures accounted for 14.1% of surgical procedures and 18.7% of nonsurgical procedures in the United States. 1 Liposuction is one of the most popular methods currently used to correct deep and superficial fat accumulation and was the most popular body contouring procedure in 2014.2,3 However, liposuction has several risks, including asymmetry, hyperpigmentation, seromas, hematomas, edema, venous thromboembolism, abdominal and bowel perforations, necrotizing fasciitis, and sepsis. 2
As an alternative to liposuction, there have been recent developments in nonsurgical fat reduction methods, which led to the creation of cryolipolysis. Cryolipolysis involves the placement of a cooling applicator on an area where the patient wishes to reduce their fat for 60 min at 10 °C. 4 One leader in this field is CoolSculpting® (ZELTIQ Aesthetics, California) which was the first cryolipolysis device to receive Food and Drug Administration (FDA) approval and has been available for over 10 years. 5 CoolSculpting® devices have been associated with high levels of satisfaction when used on the abdomen, back, flanks, thighs, arms, and the submental area. 5
In the United States, in 2012, over $130 billion was spent on medical and health research. 6 In order to increase transparency, the Physician Payments Sunshine Act was passed. This included the creation of the Open Payments Registry, which sought to report any financial interactions between industry and US-based physicians. 7 Financial conflicts of interest have recently been a topic of discussion in plastic surgery as they may affect clinical judgment and may influence patient care, clinical practice, and research. 8 Under-reporting relevant financial conflicts of interest has also raised concerns around full industry transparency. 9 As such, studies that are funded by a manufacturer are more likely to report positive findings with regard to the device provided by the funder. 8
This study aims to determine whether physicians who received funding from Allergan aesthetics, the parent company of ZELTIQ Aesthetics, declared a financial conflict of interests or stated that funding was received in the reporting of published positions on cryolipolysis devices. Furthermore, we examined if receiving industry payments is more likely to have a positive or neutral association in the literature.
Methods
To determine the association between author conflicts of interest and published positions on the CoolSculpting® cryolipolysis device and the extent to which authors disclose financial conflicts of interest, we employed a study design used previously to explore these associations in the fields of tumour treatment, hypoglossal nerve stimulation and treatment of lower urinary tract symptoms.9-11 As this study did not involve healthcare records, it was deemed exempt from institutional ethics review.
Search Strategy and Article Selection
We performed a Google Scholar (Google LLC, California) search on January 17, 2022, to identify all the articles that cite the pivotal trial in the 2016 Lasers in Surgery and Medicine article, “Safety and Efficacy of Cryolipolysis for Non-Invasive Reduction of Submental Fat.” To access these articles, the “Cited by” feature was used to access all articles that cited the FDA article which are accessible on Google Scholar. This article was chosen due to its contributions to the FDA approval of ZELTIQ aesthetics’ CoolSculpting® system. The identified articles were screened for inclusion using titles, abstracts, and full-body texts. Articles were included if they were editorials, guidelines, review articles, commentaries, randomized control trials (RCT)s, or prospective or retrospective studies focused on the CoolSculpting® cryolipolysis device. It was not possible to include articles published after 2020 as the data from the Centers for Medicare & Medicaid Services (CMS) Open Payments Registry was only available up until 2020 at the time this study was conducted. Articles containing no US-based authors were also excluded, as the CMS Open Payment Registry is limited to US-based physicians. This was a limitation in our study that the United States was the only English speaking country with an accessible registry of payments.
Article Coding
Full-text articles published by US authors were selected based on inclusion criteria and were then read independently. Two separate investigators systematically coded all articles as “favourable” if they expressed views supportive of the cryolipolysis devices or as “neutral” if the articles outlined any concerns or limitations of the intervention. Disagreements were resolved by correspondence to ensure an appropriate rating was given to each article.
Conflict of Interest Data
A separate investigator blinded to article coding collected data regarding financial conflict of interest. Author-reported conflicts of interest were recorded if explicitly declared in the published article and compared to the CMS Open Payments Registry. We downloaded the Open Payments Registry data for each physician author from 2014 to 2020, the most recent data available. 12 We screened for direct or in-kind payments from Allergan, the parent company of ZELTIQ aesthetics, which manufactures the CoolSculpting® cryolipolysis system. All payments that were specified for unrelated products or conditions were excluded from this study. We recorded the total number of payments, cumulative payment totals, and nature of payments. Authors with no documented payments from Allergan were coded as having no financial conflict of interest. As the cryolipolysis device has been owned by multiple companies, some payments may not have been registered on the CMS Open Payment Registry as a result of the transitions.
Subsequently, all authors’ first and last names with a middle initial, if available, were searched on a Google Search (Google LLC, California) to determine if they were a physician (MD or DO), as well as searching for any partnerships with ZELTIQ aesthetics. If available, authors were searched on LinkedIn (LinkedIn, California) to further investigate if any authors were direct employees of ZELTIQ aesthetics.
Results
Article Coding
Ninety-one articles were identified referencing the pivotal FDA-approving article on Google Scholar. Of these, 19 articles were based on authors from the United States. Seven articles were labelled neutral, and 12 were labelled favourable.
Distribution of Payment Status Among Authors
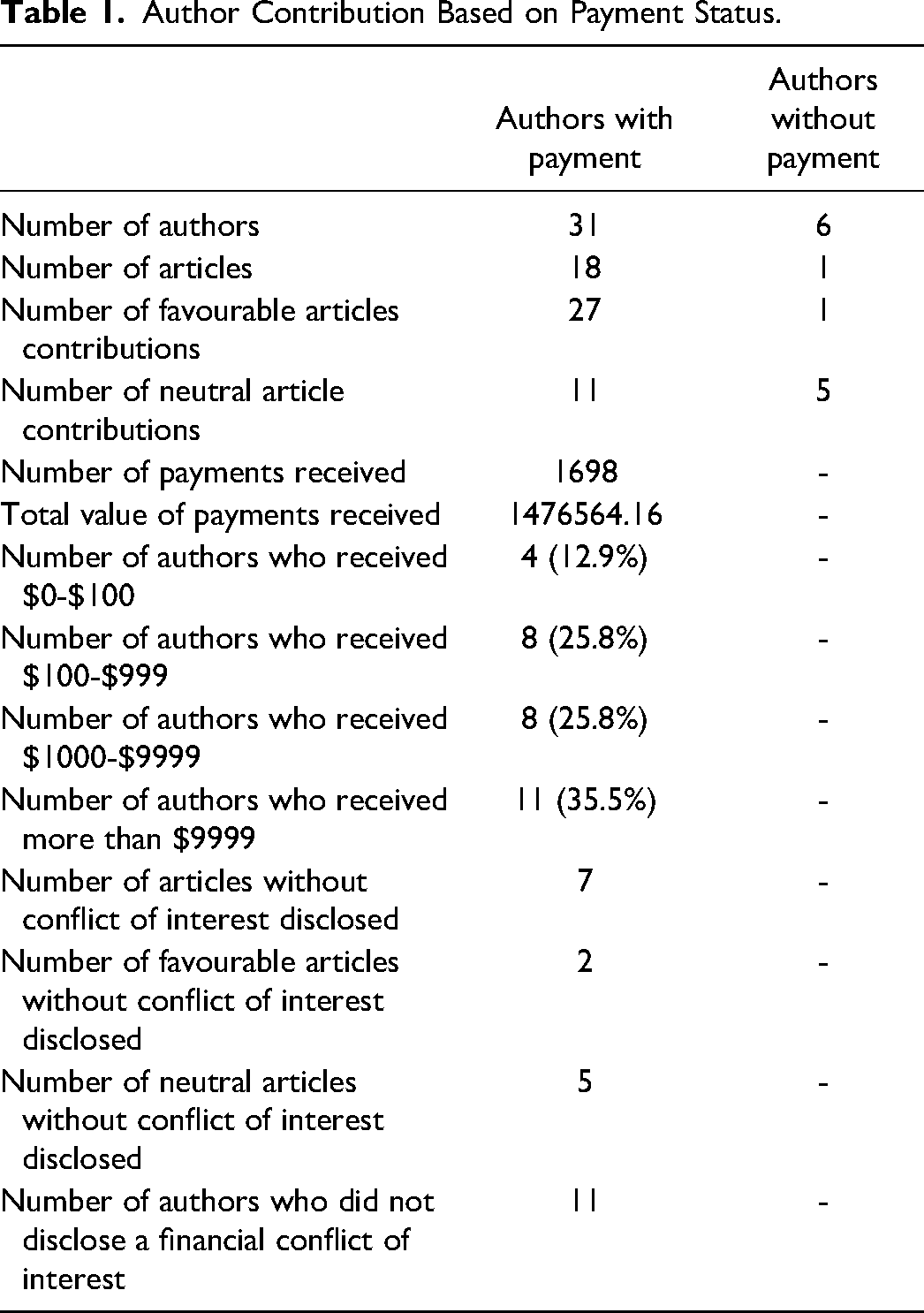
Of the 37 authors, all of whom are physicians from the United States, from the articles that met the inclusion criteria, 31 (83.8%) had accepted at least one payment from a manufacturer. Since 2014, collectively, the sum of money received by these authors from the manufacturer equated to $1,476,564.16 for a total of 1698 payments. The majority of these payments to physicians based in the United States were for consulting fees (66.4%), followed by compensation for services other than consulting (23.4%), and to a lesser extent, travel and lodging (6.5%), food and beverage (3.5%), gifts (0.2%), and education (0.06%). Of the authors who received payment, 11 (35.5%) received more than $10,000. The number of articles written was not correlated to the amount of money received.
Financial Conflicts in Articles
In total, 85.7% (6 of 7) of neutral articles had at least one author who received payments. And 100% (12 of 12) of articles published by authors based in the US labelled as favourable had at least one author who received payments. Seventy-five percent (9 of 12) of the favourable articles assessed had a first author who received a payment. Furthermore, 91.7% (11 of 12) of articles deemed favourable had all contributing authors receive a payment. Meanwhile, only one author who had not received payment contributed to a favourable article (Table 1).
Author Contribution Based on Payment Status.
Disclosures of Conflicts of Interest
In total, 29.7% (11 of 37) of authors from the United States who received payments clearly stated in their manuscript that they did not declare a financial conflict of interest. Three of these instances occurred in favourable articles, whereas 8 of these instances occurred in neutral articles. And 56.8% (21 of 37) of authors from the United States received payment from Allergan within 1 year of publishing their article. Of the authors, who were evaluated in this study, who wrote favourable articles and received payment, 69.6% (16 of 23) of authors received payment anytime between 2014 and the date of publication.
Discussion
With the rapid evolution in technology, this has led to the creation of many new medical devices. However, before widespread dissemination of these products, they should be well studied before making claims and recommendations for patients. In the process of reporting on these products, many physicians often have a certain relationship with the companies that manufacture their devices. These relationships can be disclosed, but are sometimes under-reported in the literature.
This study demonstrates that, in the United States, in almost all cases where cryolipolysis devices were provided a positive view in the literature, the authors received industry payments. We found that the majority of authors from the United States contributing to the cryolipolysis literature in any capacity received some nature of the payment. Furthermore, we identified 11 instances in which funding was received but not disclosed by the authors.
Interestingly, we found more articles written in the United States that were coded as neutral failed to disclose financial conflicts of interest compared to articles coded as favourable. This may be a result of several factors. For articles coded as neutral, many of the primary authors did not receive payments and may be unaware of all conflicts of interest held by the authorship group. Furthermore, authors who attend conferences or events sponsored by the manufacturing company may be receiving in-kind payments that they may not know are associated with the device manufacturers. Several of these authors who were assessed received less than $100 of in-kind payments over a 6-year period. This topic is nuanced, however, as any payment awarded by a device manufacturer has the ability to bias an author's opinion regardless of a specific dollar amount. Future research could look at how payments labelled as consulting fees are associated with favourable articles in the literature compared to historically smaller payments such as in-kind food and beverage.
CoolsculptingⓇ (ZELTIQ Aesthetics, Allergan, California) and other cryolipolysis devices have become a popular alternative to liposuction since FDA approval in 2010. 13 The popularity of cryolipolysis for nonsurgical fat loss is not only due to its accessibility but also the medical and scientific literature that supports its efficacy and safety on various body parts. However, while such validation studies help guide practices, critical assessment of studies, including funding sources and authors’ financial conflicts of interest, should be factored into the interpretation of results. Despite disclosure requirements in most major journals, of the assessed articles, there were 11 instances where a financial conflict was identified by our team but not disclosed in the article. Inconsistent disclosure has the potential to significantly bias the interpretation of research, jeopardizing the integrity of the validation process for new technologies. Commercialization of medical science offers both benefits and harms. A more obvious benefit includes acceleration of new discoveries to the bedside, but this must be balanced with the inherent bias and loss of objectivity associated with commercialization. 14 Thus, it is imperative that conflicts of interest are disclosed in the literature. Maintaining rigorous disclosures increases the integrity of the literature and protects investigators, clinicians, and patients from the potential harms of commercialization. 15
With more than 4 million treatments completed worldwide, cryolipolysis is one of the most widely performed cosmetic procedures. 16 In the literature, patient satisfaction rates following cryolipolysis are quite high, with a 2022 study finding that 89.6% of participants were satisfied or very satisfied with the results. 17 However, potential side effects exist, and the same study found that 21.4% reported treatment-emergent adverse events. Adverse events range from minor symptoms such as site erythema, numbness and oedema to more serious complications such as persistent pain, skin hyperpigmentation, motor neuropathy, and paradoxical adipose hyperplasia. 13 Interestingly, one of the treatments for paradoxical adipose hyperplasia is liposuction, the more invasive procedure that cryolipolysis attempts to replace and as such, patients should be aware of these more severe complications. As shown in our study, all but 1 author based in the United States who wrote a favourable article on cryolipolysis received industry payments. This conflict of interest may lead to downplaying of adverse events and, therefore, may be problematic from a patient safety perspective.
Physicians are a leading drivers of healthcare spending, and most research on interactions between their interactions with industry have focused on pharmaceuticals. Several studies have shown associations between pharmaceutical industry payments and clinical research, guidelines, and recommendations.18-20 Recently, research has investigated if this association exists within the device manufacturing industry. Our results were consistent with literature across a number of medical specialties demonstrating that projects funded by device manufacturers are more likely to have a positive stance on the product and more likely to contribute to the literature.9-11,21,22 As such, careful examination of industry influence on such publications should be an essential consideration for clinicians and clinics adopting technology such as cryolipolysis devices.
Inherently, a level of codependence must exist between physicians and industry. To advance healthcare and improve patient care requires physicians and industry to collaborate in order to implement products in healthcare settings. Collaboration with industry can offer many benefits as they can provide funding for research and new technology. However, this implementation needs to be done ethically, with patient safety as the primary goal. As a result, physicians who publish on new devices need to be mindful of their conflicts of interest and how their implicit and explicit biases may affect the results and set goals to produce accurate and truthful results. On the contrary, it also requires consumers to be mindful that industry has a role in the literature, and those who publish may have implicit biases that can influence the results before implementing new technology into their practice. Doing so helps to maintain public trust and protect the safety of patients while also advancing healthcare.
Strengths and Limitations
This study is the first of its kind to examine an association between financial conflicts of interest and articles’ positions on cryolipolysis devices. We reviewed the conflicts of interest of 37 authors, providing a large sample size for assessing the disclosure of conflicts. Our study was limited by the information available in the CMS Open Payments Registry to identify funding sources. Thus, our study does not account for conflicts of interest that were not disclosed or completed within this database. It is important to note these results are most specific to articles published in the United States as, culturally, financial conflicts of interest may have more or less of an effect on an author depending on the recipient's relationship with industry, finances, and several other factors. Further, our study only accounts for financial conflicts of interest and, therefore, may underestimate the impact of interpersonal or other types of business relationships with industry as well as conflicts of interest formed after the publication of the article. Furthermore, consulting companies often hire physicians as third-party employees, those relationships would not be identifiable in our search and could have resulted in a conflict of interest. Although we outline our criteria for article coding, no formal guideline is available for evaluating an article's tone or position. We did not examine the scientific validity of the articles to assess whether conflicts of interest biased the actual results of the study; however, this is a potential future avenue of study. Finally, we recognize that this data is specific to the United States, and may not be generalizable to Canada or other regions. Similar datasets are not available in Canada to our knowledge, but may improve understanding of this issue on a regional level.
Conclusion
Our study has shown that most published opinions from authors from the United States on cryolipolysis devices for nonsurgical fat reduction were written by physicians who received payments from the device manufacturers. Furthermore, it identifies a gap in the authors’ disclosure of financial conflicts of interest. When being consumers of the literature, it is always important to be mindful that industry may be biasing the messaging surrounding their products. Our article, in alignment with a number of similar articles across different fields, emphasizes the importance of transparency among authors in disclosing their financial conflicts. Additionally, our study reinforces the need for medical professionals to exercise a high degree of caution and scrutiny when basing clinical decision making on supposedly objective evidence published in the literature.
Statement on any previous study related presentations and publications, including published abstracts:
- Poster presentation: “An analysis of industry-related payments towards physicians – Cryolipolysis” Medical Student Research Showcase. Kingston, ON. Nov 16, 2022. - Oral presentation: “An analysis of industry-related payments towards physicians – Cryolipolysis” Plastic Surgery The Meeting. Boston, MA. Oct 28, 2022.
Footnotes
Author Contributions
Idea was developed by KA, AK, and AS. All authors contributed to the development and revision process of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
