Abstract
Introduction
Burn wounds are an ideal culture for microorganisms, and infectious complications are particularly devastating for the admitted burn patient. 1 Burns both compromise the cutaneous barrier of the body and suppress the immune system leading to infection. 2 These infections are a significant cause of morbidity, reintervention and increased overall costs. After initial resuscitation, greater than 50% of the mortality following burn injuries is secondary to infection.3,4
Burn wounds are initially considered to be sterile as the bacterial skin flora is destroyed by the thermal insult. However, surviving bacteria within hair follicles and sebaceous glands, as well as the patient's environment, can lead to rapid colonization of the burn wound. 5 If untreated, microbial colonization can result in infection, delayed healing, and skin graft loss. 5 Notably, McManus et al. 6 reported that 36% of positive burn wound cultures were associated with microbial invasion on histopathologic analysis—considered to be the “gold standard” for the diagnosis of infection in unexcised burn wounds.6,7 Typically, burn wound infections are diagnosed based on patient symptoms and clinical examination; however, microbiological cultures continue to be used in the setting of a clinically suspected infection to identify predominant microorganisms and establish susceptibility. 7
Using regional and national data obtained through the American Burn Association, Heard et al. 8 concluded that older age, male sex, lower extremity involvement, scald injury, and full-thickness burns were consistently associated with burn wound cellulitis in a logistic regression model in a local regional sample of 636 burn wound patients. 8 Despite this research, there exists a paucity of studies evaluating risk factors for burn site colonization in demographic samples, and single-public payer models, representing a current gap in modern burn literature.
Given the association between positive cultures and microbial invasion, particularly in the setting of a clinically suspected infection, efforts to identify wounds at greater risk of colonization can be used to inform clinical management. Ultimately, this knowledge may be used to limit the sequelae of burn wound complications including increased length of stay, additional surgery, and greater health system costs.6,8
Burn units commonly identify and monitor pathogens in their population and environment to both investigate colonization in the clinical unit and guide empirical antibiotics usage in cases of infection. 9 The primary objective of this study was to determine the incidence of microbial pathogens present in burn tissue samples (ie, culture-positive burn wounds on microbiology) and risk factors associated with positive burn wound colonization during primary admission at a single, provincial, tertiary care burn center.
Methods
Study Design
A retrospective observational study (ie, chart review) was performed on all patients admitted to the burn unit (BU) at the Hamilton General Hospital (HGH), a tertiary burn care center with a split provincial population catchment area of approximately 7 million people. Data collection took place in Hamilton, Ontario, Canada, from January 1, 2011, to January 1, 2021. Patient data was collected from electronic medical records in a single network. Institutional research ethics board approval was obtained for this study (Hamilton Integrated Research Ethics Board #13489).
Patient Identification
The medical records of patients admitted the BU with a burn diagnosis were identified by ICD-10 diagnosis codes produced with provincial burn reporting data. Patients were included if (1) they were adults, aged 18 years or older, and (2) they required primary admission for ≥1 day to the burn unit with a diagnosis of a burn (ie, on ICD-10 diagnosis codes). Conversely, patients were excluded if (1) they were readmitted for a burn diagnosis following a primary admission or (2) they were admitted for secondary burn reconstruction. If patients experienced multiple readmissions, only the primary burn admission was analyzed.
Data Collection
Patient data was extracted from electronic medical records. Demographic and clinical information (ie, age, sex, smoking status, diabetes status, total burn surface area [TBSA], location of burn, presence of inhalation injury, full-thickness burn diagnosis, and burn etiology), as well as outcome data (ie, positive burn wound swab or tissue culture) were collected independently and in-duplicate for 20% of participants to ensure adequate agreement using piloted data extraction forms. Disagreements were resolved by consensus between reviewers and the supervising study author. The remaining patient data was extracted independently before being verified by the other reviewer.
Primary Outcome Definition
The primary outcome of this analysis was the incidence (ie, new cases of an outcome over the study time horizon) of any culture-positive burn wound (ie, obtained from debrided tissue sample or wound swab), identified by isolated microbial culture and sensitivity, performed during the patient's primary burn admission. At our institution, burn wound cultures are performed for clinically suspected infection; criteria for culture include conversion of a superficial partial-thickness to deep partial-thickness injury, change in wound/skin appearance, eschar separation, and delayed healing/graft issues. Given the subjective nature of a burn wound infection/cellulitis diagnosis, we only included patients with clinical signs of infection or those requiring debridement in our regression if they had a concordant positive burn wound culture.
Data Analysis
To address the primary objective, descriptive statistics (ie, percentages, means, standard deviations) were reported to determine the incidence of BU patients with culture-positive wounds on microbiology during their admission course. To evaluate clinical and demographic patient variables associated with culture-positive burn wounds, a univariate logistic regression analysis was performed. Independent variables were selected a priori based on their hypothesized association with the outcome of interest (ie, culture-positive burn wounds) based on review of the literature.4,5,8 The variables identified were age (continuous, years at time of BU admission), sex (male/female), smoking status (categorical, smoker/nonsmoker), TBSA (continuous, %), diabetes status (categorical, yes/no), burn site (categorical, head and neck/upper limb/lower limb/trunk, yes/no), presence of full-thickness burn (categorical, yes/no), inhalation injury status (categorical, yes/no), and burn etiology (categorical, scald/flame/chemical/electrical/contact). A Wald test or generalized likelihood ratio test was used to evaluate the statistical significance of each independent variable (P < .05).
Statistically significant variables from the univariate logistic regression analyses were included in a multivariable logistic regression model. Goodness-of-fit of the multivariable model was evaluated using the Hosmer–Lemeshow test, where P ≥ .05 implies the model is appropriate. Multicollinearity was evaluated using the standard errors (SE) of independent variables within the logistic regression analysis, where a SE of >2 was used to denote multicollinear independent variables. Given a rule of thumb of 10 events (N = 173 culture-positive burn wounds) per independent variable, the sample was determined to be sufficiently powered.10,11 Listwise deletion (ie, complete case analysis) was used to address missing data within regression analyses. Statistical significance was set at P ≤ .05. All analyses were performed using SPSS 26.0.
Results
The sample consisted of N = 634 patients. Most patients were male (72.1%), with a flame burn injury (62%), and had a mean age of 47.6 (±18.0) years and a TBSA of 13.5% (±14.8). See Tables 1 and 2 for complete patient clinical and demographic characteristics.
Demographic and Clinical Characteristics (N = 634).

N denotes the frequency of individual participants. N% represents the percent of the total sample.
Distribution of Participant Variables.
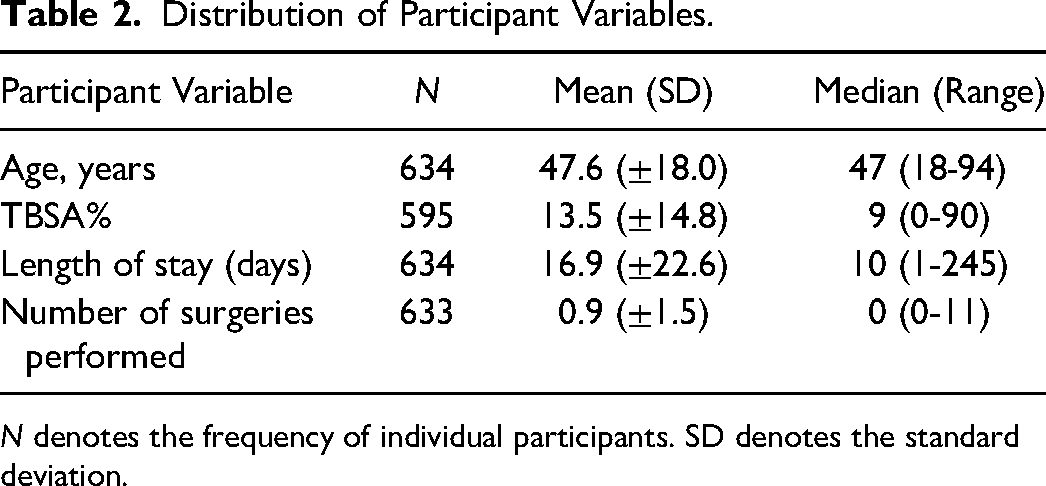
N denotes the frequency of individual participants. SD denotes the standard deviation.
Descriptive Analysis
Of the 634 patients included in this study, 239 were found to have undergone either wound or tissue culture (37.7%). One hundred sixty-five of these patients underwent wound swabs, 37 underwent tissue cultures, and an additional 37 underwent both. Of the 202 total wound swabs performed in patients with clinical suspicion of infection, 141 wound swabs were positive (69.8%). Likewise, of the 74 total tissue cultures performed, 60 were found to be positive (81.1%). In total, 173 unique patients (27.3% of the total sample) demonstrated a positive burn wound culture. This data is presented in Table 3. Distribution of the burn wound culture frequency by calendar year is illustrated in Figure 1. The frequency of common pathogens isolated from burn wound cultures is presented in Table 4. A complete list of isolated pathogens is reported in
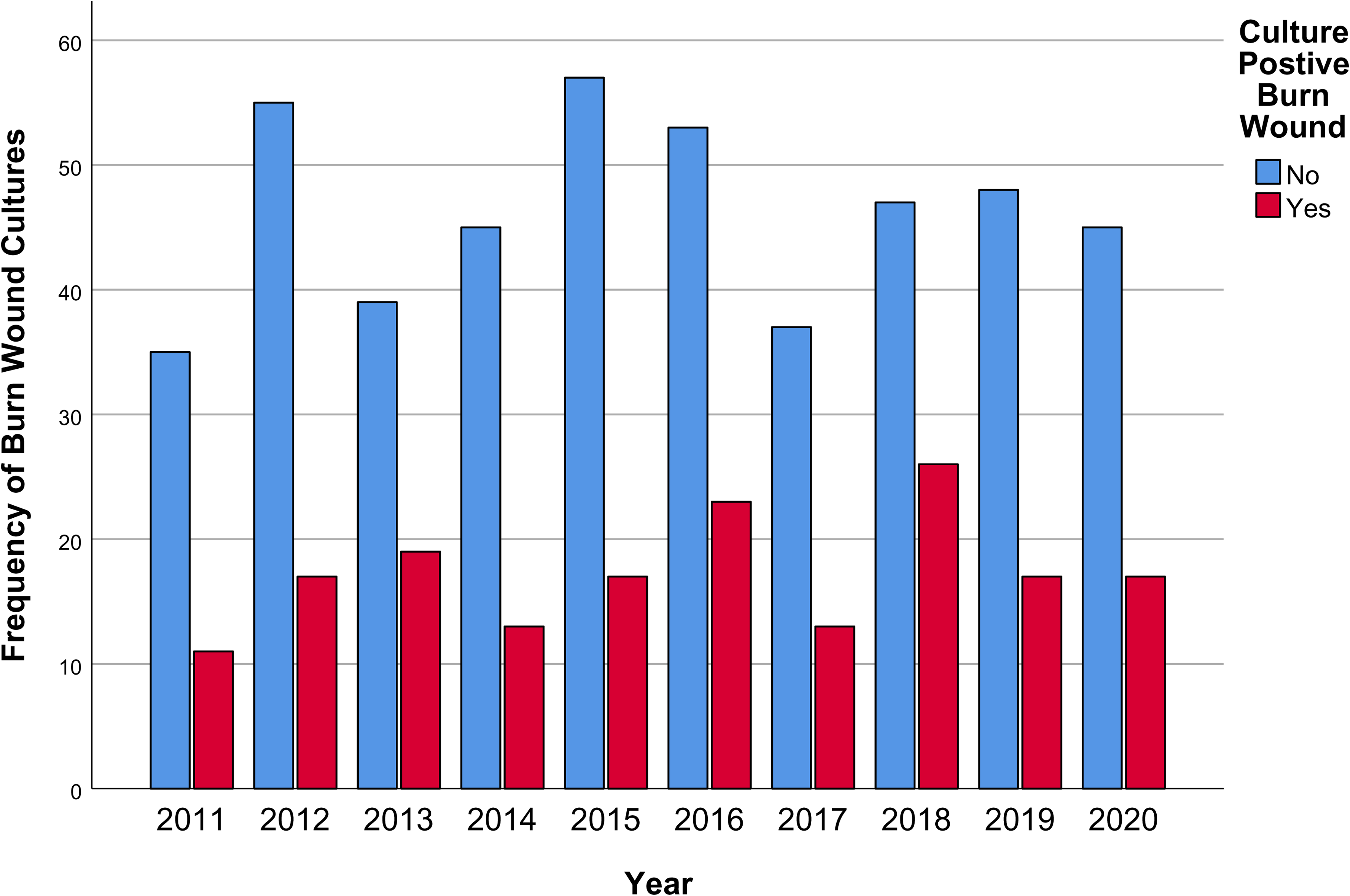
Frequency of burn wound cultures per calendar year.
Results of Patients who Underwent Wound Swab and/or Tissue Culture.

Types and Frequency of Common Pathogens Isolated From Burn Wound Cultures.

N denotes the total number of positive burn wound cultures sent to microbiology lab. N% represents the percent of total positive burn wound cultures sent.
Logistic Regression Analysis
The results of the univariable logistic regression analyses are reported in Table 5. Participants who were older in age (OR 1.02 [1.01-1.03], P < .01), diabetic (OR 1.91 [1.13-3.23], P = .02), had a larger burn TBSA % (OR 1.05 [1.04-1.07], P < .01), full-thickness burn (OR 3.79 [2.62-5.49], P < .01), and inhalation injury (OR 2.47 [1.67-3.65], P < .01), with involvement of the lower limb (OR 1.59 [1.12-2.25], P < .01) and abdomen/trunk (OR 2.40 [1.67-3.43], P < .01) demonstrated a statistically significant increased odds of a positive burn wound culture in our sample.
Univariable Logistic Regression Analysis.
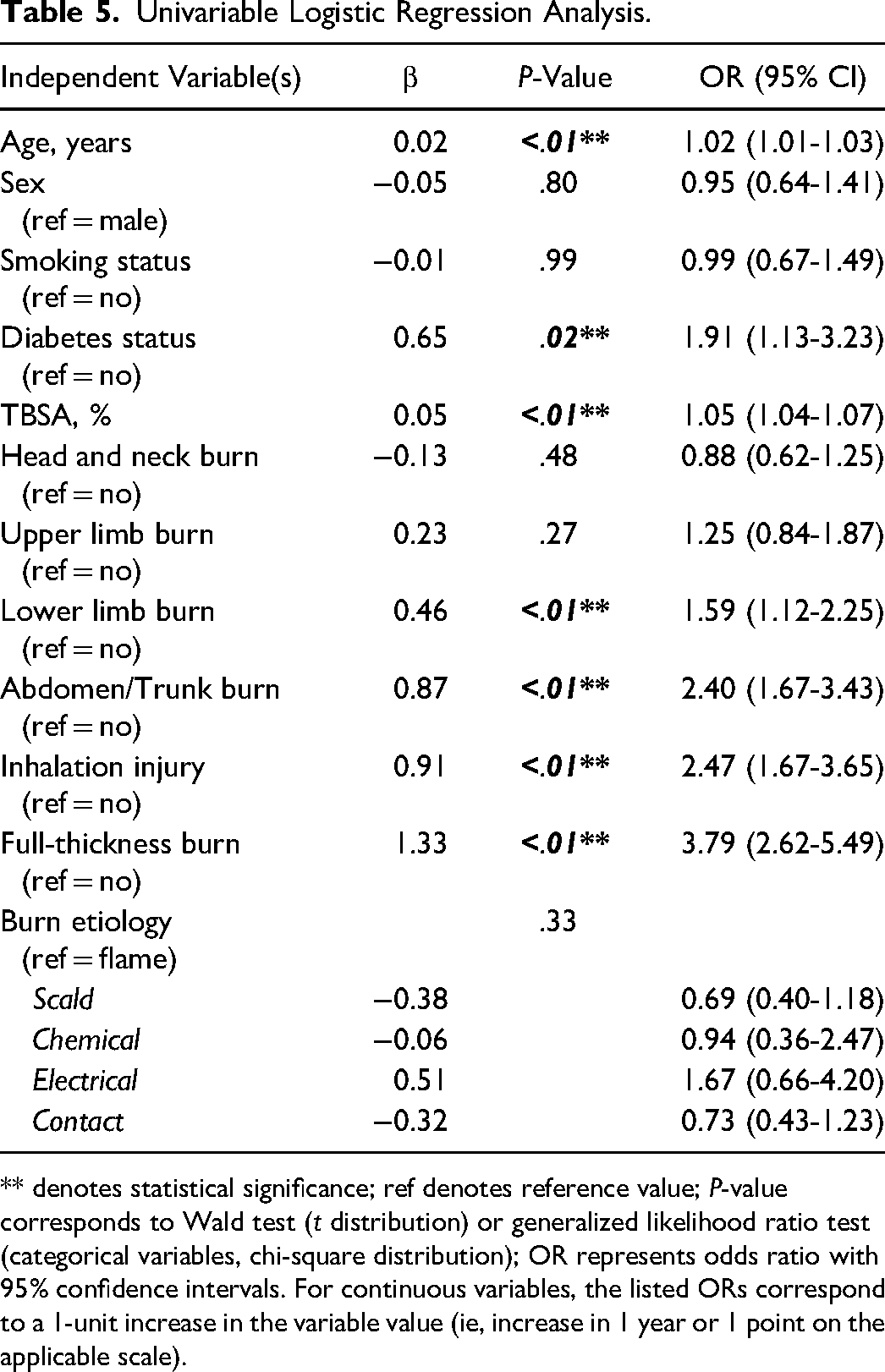
** denotes statistical significance; ref denotes reference value; P-value corresponds to Wald test (t distribution) or generalized likelihood ratio test (categorical variables, chi-square distribution); OR represents odds ratio with 95% confidence intervals. For continuous variables, the listed ORs correspond to a 1-unit increase in the variable value (ie, increase in 1 year or 1 point on the applicable scale).
When these statistically significant variables were included in a multivariable logistic regression model, only participant age (P = .05), diabetes status (P = .03), TBSA (P < .01), and presence of a full-thickness burn (P < .01) remained statistically significant after adjusting for all other variables (Table 6). Goodness-of-fit of the model was determined to be sufficient (Hosmer–Lemeshow test, P = .31).
Multivariable Logistic Regression Analysis of Statistically Significant Variables

** denotes statistical significance; ref denotes reference value; SE refers to standard error; P-value corresponds to Wald test (t distribution) or generalized likelihood ratio test (categorical variables, chi-square distribution); OR represents odds ratio with 95% confidence intervals. Goodness-of-fit of the multivariable model was evaluated using the Hosmer–Lemeshow test. For continuous variables, the listed ORs correspond to a 1-unit increase in the variable value (ie, increase in 1 year or 1 point on the applicable scale).
Discussion
This is the largest study to evaluate the incidence of burn wound colonization in a regional demographic sample within a single-public payer context. From our N = 634 sample, we identified 173 patients with culture-positive burn wounds during their primary admission, corresponding to an incidence of 27.3%.
In addition, through univariable logistic regression analyses, we note that increasing participant age, diabetes status, greater burn TBSA, presence of full-thickness burns, inhalation injury, and lower limb and trunk involvement were associated with a statistically significant increased odds of a positive burn wound culture. Of note, age, diabetes status, TBSA, and diagnosis of full-thickness burn remained statistically significant (P ≤ .05) after adjusting for all statistically significant factors in a multivariable logistic regression model.
The risk factors associated with burn wound colonization identified in our study are similar to the risk factors identified by Heard et al. 8 Similarly, the authors concluded that increasing age, male sex, full-thickness burns, and lower extremity involvement were significantly associated with burn wound cellulitis. In contrast to Heard et al. 8 our sample was unable to identify a statistically significant increased odds associated with burn etiology (ie, scald burn) and patient ethnicity. Furthermore, our study demonstrates that diabetes significantly increases the odds of burn wound colonization by approximately 1.9 times (from 1.1 to 3.2 times increased odds), relative to patients without a diabetes diagnosis. The association between diabetes and infectious burn complications is also well supported in the literature. 5 Specifically, Barsun et al. 12 reported a 44% incidence of infection in a sample of 68 diabetic patients presenting with lower extremity burns. 12 The similar risk factors identified within this study, in the setting of the existing literature, support the association between positive burn wound cultures and progression to cellulitis/infection.
Notably, positive burn wound cultures are used within this study as an objective adjunct for helping to identify burn site infection. At our institution, burn wound cultures are utilized in the setting of a suspected clinical infection to identify predominant microorganisms and establish susceptibility. 13 Given that the best approach to burn wound infection management is primary prevention, the authors believe that patients with diabetes, full-thickness burns, higher TBSA involvement, and older age should be monitored very closely for progression to infection. 5 Knowing these risk factors may increase your clinical suspicion for infection in the setting of equivocal physical findings and reduce the threshold for initiating antimicrobial or operative treatment. While not directly analyzed in this patient sample, efforts to prevent infection are likely to limit its known sequelae including increased patient morbidity, mortality, length of stay, procedures, and healthcare system costs.3,4,6,8
This study has several limitations. First, given the retrospective nature of this observational study, the authors could not be blinded; therefore, extraction of potential exposures/outcomes may be biased. Second, authors were limited to the existing documentation of ICD-10 codes and exposures reported in the patient's electronic medical record; as such, missing data and incorrect clinical reporting may have biased the results of our analysis. Finally, the primary outcome of this analysis (ie, positive burn wound cultures) serves as a risk factor for more patient-important outcomes, such as clinical infection and cellulitis necessitating antibiotic administration or sepsis secondary to burn site infection. As such, the authors acknowledge that burn microbial colonization may not universally lead to clinically significant adverse patient outcomes, which may bias these study results. 13 It should be emphasized that wound contamination is not the same as wound infection. Given this, wound cultures should never be used as a substitute for thorough physical examination, and treatment decisions should never be initiated based on positive wound cultures alone. At our institution, burn sites are typically swabbed/debrided and sent for microbial culture and sensitivity when clinical infection is suspected. In this way, burn wound cultures are a useful adjunct to—but not a replacement for—clinical assessment when determining if a wound is infected.
The strength of this study stems from its relatively large demographic sample that is likely representative of the provincial population given that our institution serves as a burn referral center in a publicly insured healthcare setting. Additional strengths stem from the use of an objective outcome assessment, as determined by microbial pathogen culture, in the setting of suspected clinical infection.
Conclusion
In conclusion, the authors identified a 27.3% incidence of positive burn wound colonization in a 10-year sample of patients who were admitted to a single, tertiary burn center. Risk factors associated with this outcome include increasing participant age, diabetic status, larger burn TBSA, presence of full-thickness burns, inhalation injury, and lower limb and trunk involvement. In the clinical practice setting, the authors believe that patients with these clinical and demographic risk factors should be monitored closely to prevent and manage progression to clinical infection. Going forward, future research should seek to further establish the association between positive wound cultures, in the setting of clinical suspicion of infection, and microbial invasion on histopathological analysis to strengthen its use as an objective, adjunctive measure of infection moving forward.
Supplemental Material
sj-docx-1-psg-10.1177_22925503231214760 - Supplemental material for Incidence and Risk Factors Associated with Microbial Colonization of Burn Wounds: An Observational Study
Supplemental material, sj-docx-1-psg-10.1177_22925503231214760 for Incidence and Risk Factors Associated with Microbial Colonization of Burn Wounds: An Observational Study by Lucas Gallo, MD, MSc, PhD(c), Patrick Kim, MD, Matteo Gallo, MD, Oluwatobi Olaiya, MD, MSc, Cheryl Main, MD, Ronen Avram, MD, MSc, James Bain, MD, MSc, Achilles Thoma, MD, MSc, Sophocles H. Voineskos, MD, MSc, and Christopher Coroneos, MD, MSc in Plastic Surgery
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
