Abstract
Introduction
Dupuytren's Disease (DD) is a benign fibroproliferative condition that gradually causing flexion contractures to the digits leading to issues with hand function. Although multiple etiologies have been implicated in this condition, it is thought to be mostly transmitted through an autosomal dominant gene with variable penetrance primarily affecting patients with Northern European ancestry. 1 The prevalence of DD in Western countries varies from 0.6% to 31.6% and increases steadily with increasing age. 2 Many surgical approaches exist to manage DD, including percutaneous needle aponeurotomy, open fasciotomy, and most commonly, limited open palmar fasciectomy.3,4 Non-surgical management options such as injections with steroids or verapamil have been previously proven ineffective and rejected clinically. 5 After its approval by the Food and Drug Administration (FDA) in 2010, 6 collagenase injection (CI) by Xiaflex® became a common and legitimate treatment for DD.
Several studies have compared cost and clinical effectiveness of CI to earlier treatment options. Research comparing the cost of these procedures has resulted in conflicting results. In 2013, a decision analysis study concluded that CI was more cost-effective as compared to fasciectomy and needle aponeurotomy. 4 One year later, a systematic review and economic evaluation 7 concluded that limited palmar fasciectomy (LPF) was the most cost-effective choice for moderate to severe contractures. The results from these studies must be interpreted while considering their limitations. For instance, the decision analysis model 4 relied on secondary data and in the systematic review, 7 the Markov model that the authors used was fraught with considerable uncertainty about the appropriateness of many assumptions and parameters used in the model. More recently, a propensity-matched analysis 8 and a multi-center non-randomized controlled observation study 9 stated that procedure-related costs were lower for CI as compared to fasciectomy at 1-year and 26-weeks post procedure, respectively. While these more recent studies (2020 and 2022, respectively) are of better methodological quality, a large, long-term, randomized controlled trial (RCT) with cost-effectiveness analyses using patient-level data is still required to make a true comparison between these two techniques.4,7–9
In terms of clinical effectiveness, results also vary. Two multi-center double-blind randomized placebo-controlled trials demonstrated the clinical safety, efficacy, effectiveness, and tolerability of CI.10,11 The long-term efficacy and safety of CI was confirmed in 3-year 12 and 5-year 13 non-treatment follow-up studies. In both studies, CI had a recurrence rate comparable to surgical approaches with no long-term adverse effects.12,13 A retrospective case-control study 14 concluded that, at 1-year post procedure, significantly more joints treated with fasciectomy met the criteria for “resolution of joint contracture” as compared to CI joints. A 12-week propensity score matched study 15 found that CI was not significantly different from LPF in reducing metacarpophalangeal and proximal interphalangeal joint contractures. However, treatment using CI, provided a more rapid recovery of hand function than LPF and was associated with fewer serious adverse events. 15 Lastly, a non-randomized prospective observational study 9 found that, at 26-weeks post-procedure, there was no significant difference in quality of life or disability in fasciectomy and CI patients; however, the CI group showed a significantly larger extension deficit.
While the above-mentioned studies are informative, they present limitations. The propensity matched 14 and the retrospective 15 studies have limitations built into their study designs and both recommend an RCT for head-to-head comparison of CI and LPF. The presented RCTs10,11 are methodologically more robust; however, are limited as they compared CI to placebo. Many trial methodologists would find concern in using a placebo as the control limb when existing surgical techniques exist, such as the commonly performed palmar fasciectomy. As such, the need for an RCT comparing CI and LPF is still required for a true comparison of these two common procedures. The purpose of this study was to conduct an RCT comparing the clinical effectiveness of CI and LPF in DD patients 12-months post-procedure.
Materials and Methods
This prospective RCT was performed from February 2018 to November 2021. All study procedures were approved by the local Research Ethics Board and all participants provided written informed consent. This study was registered with ClinicalTrials.gov on April 1, 2016 (NCT02725528).
Patient Screening and Recruitment
Patients were screened for eligibility by their treating surgeon during their consultation using a study screening form outlining the inclusion and exclusion criteria (Table 1). If eligible, patients were briefly introduced to the study by the surgeon; a research assistant then provided further information and obtained consent.
Inclusion and Exclusion Criteria.

Abbreviations: MCP, metacarpophalangeal; PIP, proximal interphalangeal;
The inclusion criteria was originally a 90% coverage of Xiaflex®, which was changed in October of 2018 (1 year after the start of recruitment). Health insurance coverage was included as an inclusion criterion to eliminate ethical issues and bias of financial security of patients as each vial of Xiaflex® costs $1200.00 CDN.
Randomization
Immediately following consent to participate in the study, patients were randomized to either the intervention group (CI) or control group (LPF). Parallel randomization was done in a 1:1 ratio using REDCap with block randomization; the randomization log was created and entered into REDCap by a biostatistician not involved in data collection or analysis. Due to the nature of the procedures, physicians and patients could not be blinded. Details about the two procedures can be found in Supplemental Material A.
Data Collection
At baseline, patients completed a demographics form and received an envelope containing the 1-week pre-procedure questionnaires, and the 1-day pre-procedure questionnaires. Patients were reminded when the questionnaires needed to be completed and were told to return their questionnaires at their one-month follow-up with their surgeon. At the one-month follow-up, baseline questionnaires were collected, and one-month questionnaires were given to patients. If possible, patients completed these questionnaires in clinic; alternatively, the patients were asked to complete the questionnaires at home and mail them back to the clinic using a provided self-addressed, stamped envelope. The same procedure was followed at 3-, 6-, and 12-month follow-ups. As data collection for this study took place during the COVID-19 pandemic, patient appointments were impacted. To avoid missing follow-up days, patients had their questionnaires mailed to them if appointments were canceled or the patient did not show. At all in-person appointments, range of motion (ROM) measures were taken using a goniometer. Prior to the COVID-19 pandemic, ROM measurements were taken by a research assistant. Due to safety restrictions during the COVID-19 pandemic, ROM measures were done by the treating surgeon. Data on ROM is presented with digital loss of extension as the unit of measurement. Approximately 1-year after the last patient completed the EXTEND study, we received additional ethical clearance to review the medical files of consenting participants to determine if the rate of additional intervention to the same digit was different between the two groups up to 4 years later. We were able to contact 15 of the patients; all 15 granted us permission to review their medical file.
Outcome Measures
The primary outcome was health state measured by the Michigan Hand Questionnaire (MHQ). 16 Secondary outcomes were health status (The Health Utility Index-3 (HUI-3)), 17 function (The Unité Rhumatologique des Affections de la Main (URAM) 18 and The Southampton Dupuytren's Scoring Scheme (SDSS)), 19 and ROM of treated digits. Measurements for all outcomes were taken at baseline (1-day and 1-week pre-procedure), and 1-, 3-, 6-, and 12-months post-procedure.
The MHQ is a hand-specific questionnaire that measures function, activities of daily living, pain, and satisfaction. 16 In the current study, the MHQ score represents the hand of the digit treated for DD (ie, if the treated digit was on the right hand, the MHQ score represents the right hand). The HUI-3 is a generic questionnaire that measures health status 17 ; it can be used to calculate Quality Adjusted Life Years (QALY), an important component in cost-utility analysis. The URAM is a DD-specific health-related quality of life measure 18 ; the total score is postulated to be related to disability associated with DD. Lastly, the SDSS is a disease-specific questionnaire measuring the amount of disability attributed to DD. 19 More detailed information for each outcome measure, including scoring, interpretation, and psychometric properties can be found in Supplemental Material B.
Data Analysis
Descriptive statistics were used to evaluate demographic, clinical characteristics, quality of life outcome scores, and performance tests. Baseline scores were calculated by taking the average score of the 1-day and 1-week baseline measure for each outcome measure. QALY was calculated by multiplying the difference between quality of life, as measured by the HUI-3 score before and after the intervention by the remaining years of life of the average patient. 20 Missing data points on questionnaires were replaced using mean imputation, this was required for a total of three questions, all considered “missing at random.”
Life expectancy was set at 79 years for males, and 84 years for females. 21 Between groups analysis for PROMs, QALYs and ROM at 12-months were determined using one-way analysis of covariance (ANCOVA). Group was entered as the independent variable, the 12-month outcome of interest was entered as the dependent variable and the baseline value for the corresponding outcome measure was entered as the covariate. Statistical significance was considered at p < .05; all analyses were performed using SPSS® version 26.0. 22
Results
The required sample size to achieve power for the proposed study was 128 patients, 64 per group (See Supplemental Material C). Unfortunately, due to several factors, adequate sample size was not reached, and therefore, power was not achieved. A total of 122 patients were screened for eligibility over the course of the study; 100 of those were excluded according to the eligibility criteria listed in Table 1. The original inclusion criterion of 90% insurance coverage for Xiaflex® resulted in a high percentage of patients being deemed ineligible. Changing this criterion to 80% coverage resulted in an improved recruitment rate; however, recruitment was still slow due to patient preference for the non-invasive nature of CI over LPF. Ultimately, the coup de grace of this study was early termination due to the discontinuation of Xiaflex® in Canada, announced in May 2020.
A total of 22 patients were eligible to participate in this study; all consented to participate and were randomized. Following randomization, one patient withdrew from the study citing dissatisfaction with randomly assigned procedural group as the reason for withdrawing. In other words, after being randomized to the patient's assigned procedural group (surgery) they elected to withdraw consent to participate in the study. All other patients received the treatment they were randomized to. Questionnaire completion varied by questionnaire and by follow-up date. Figure 1 illustrates the flow of patients from screening through to study completion. Demographic information is included for the 21 patients who continued with the study following randomization. Patients were mostly (85.7%; n = 18) male with an average age of 65.3 (SD = 9.2). Fifteen patients (68.2%) were treated on their right hand and the majority (45.5%) had their little finger treated (Table 2).
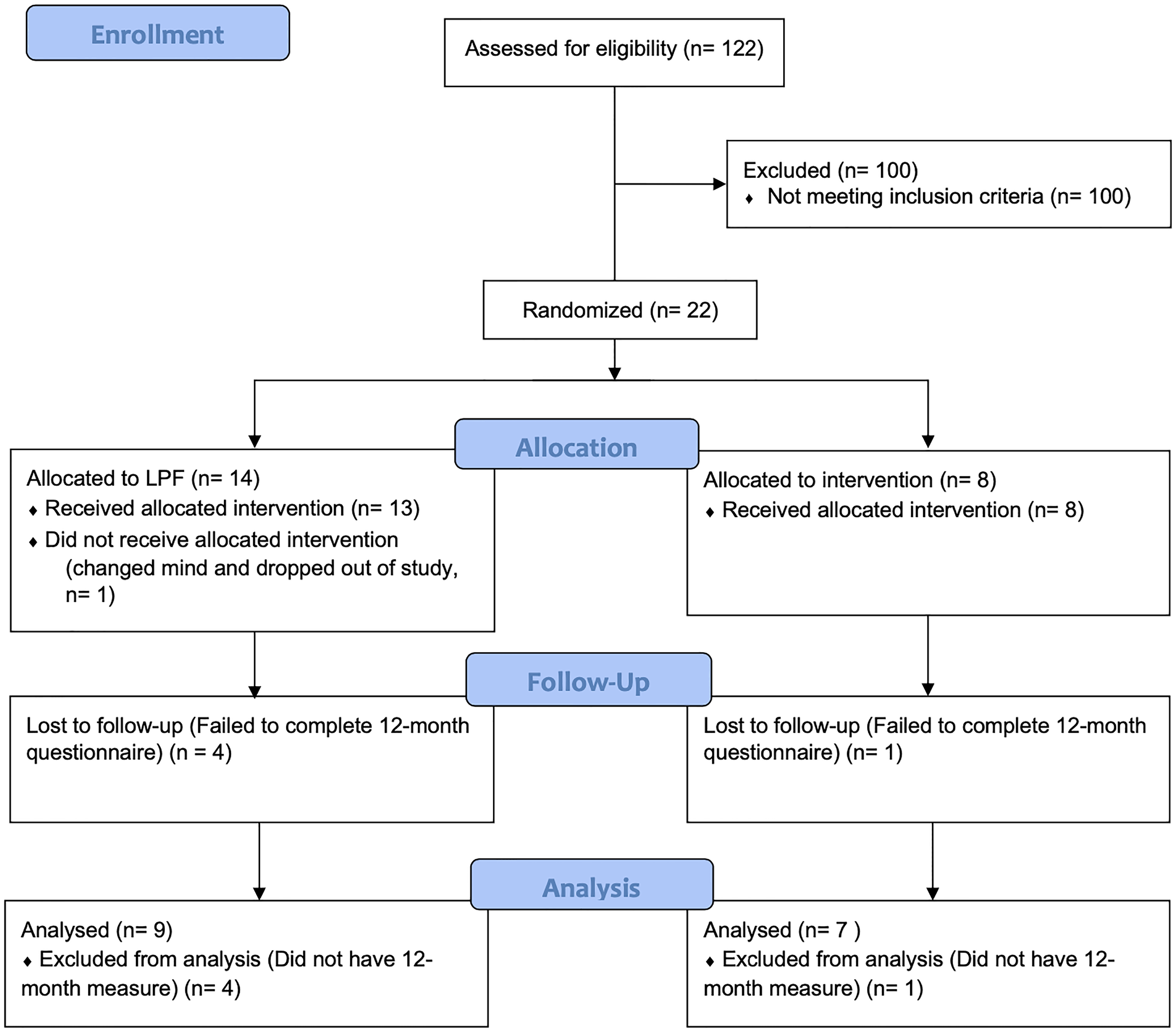
A CONSORT flow diagram that illustrates the flow of patients from screening through to study completion. *Note: the CONSORT Flow Diagram is representative of the primary outcome (Michigan Hand Questionnaire).
Demographics and Baseline Measures.
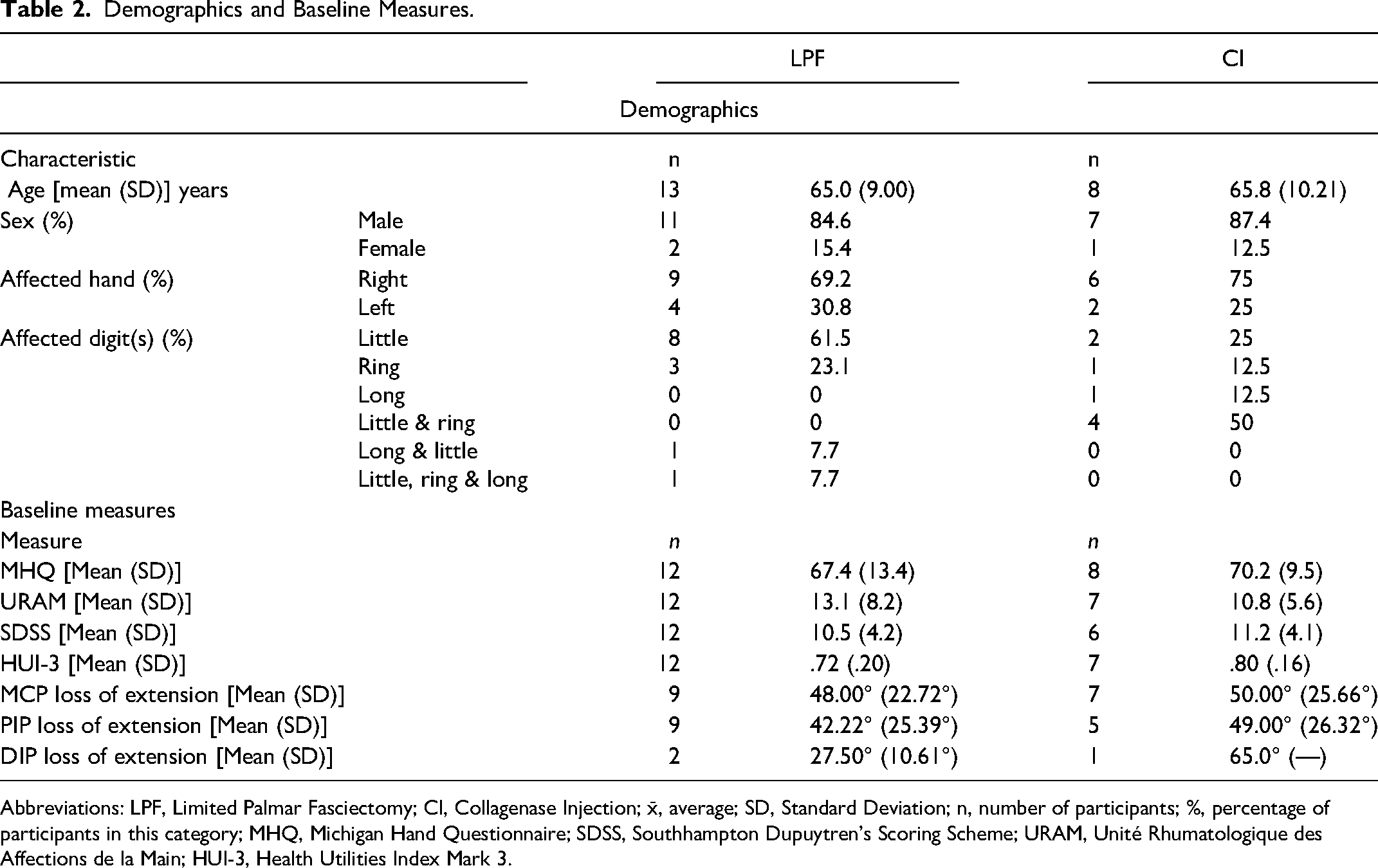
Abbreviations: LPF, Limited Palmar Fasciectomy; CI, Collagenase Injection; x̄, average; SD, Standard Deviation; n, number of participants; %, percentage of participants in this category; MHQ, Michigan Hand Questionnaire; SDSS, Southhampton Dupuytren's Scoring Scheme; URAM, Unité Rhumatologique des Affections de la Main; HUI-3, Health Utilities Index Mark 3.
Available data for all outcome measures at all follow-ups are reported in Table 3. No statistically significant difference was detected between groups for any of the PROMs or for ROM at baseline (p > .05). There was no statistically significant difference between groups at 12 months after controlling for baseline scores for MHQ p = .15, 95% CI [−26.4, 6.8], SDSS p = .70, 95% CI [−4.0, 5.8], URAM p = .89, 95% CI [−14.4, 12.6], HUI-3 p = .72, 95% CI [−.2, .2]. Similarly, there was no statistically significant difference in extension gained between groups at 12 months after controlling for baseline in the MCP p = .07, 95% CI [−9.5, 15.1] or PIP p = .76, 95% CI [7.3, 32.1]. Differences in loss of extension from baseline to 12 months could not be calculated for DIP due to lack of data. The mean (SD) 12-month QALY for the LPF group and CI group was calculated as .93 (2.8) and .83 (1.7), respectively. The 12-month QALY did not statistically differ between groups (p > .05). The additional review of patient files revealed that, as of August 15, 2022, three patients from the CI group required palmar fasciectomy on the same digit. One patient in the LPF group required repeat surgery.
Patient-Reported Data for LPF and CI Groups by Follow-Up.
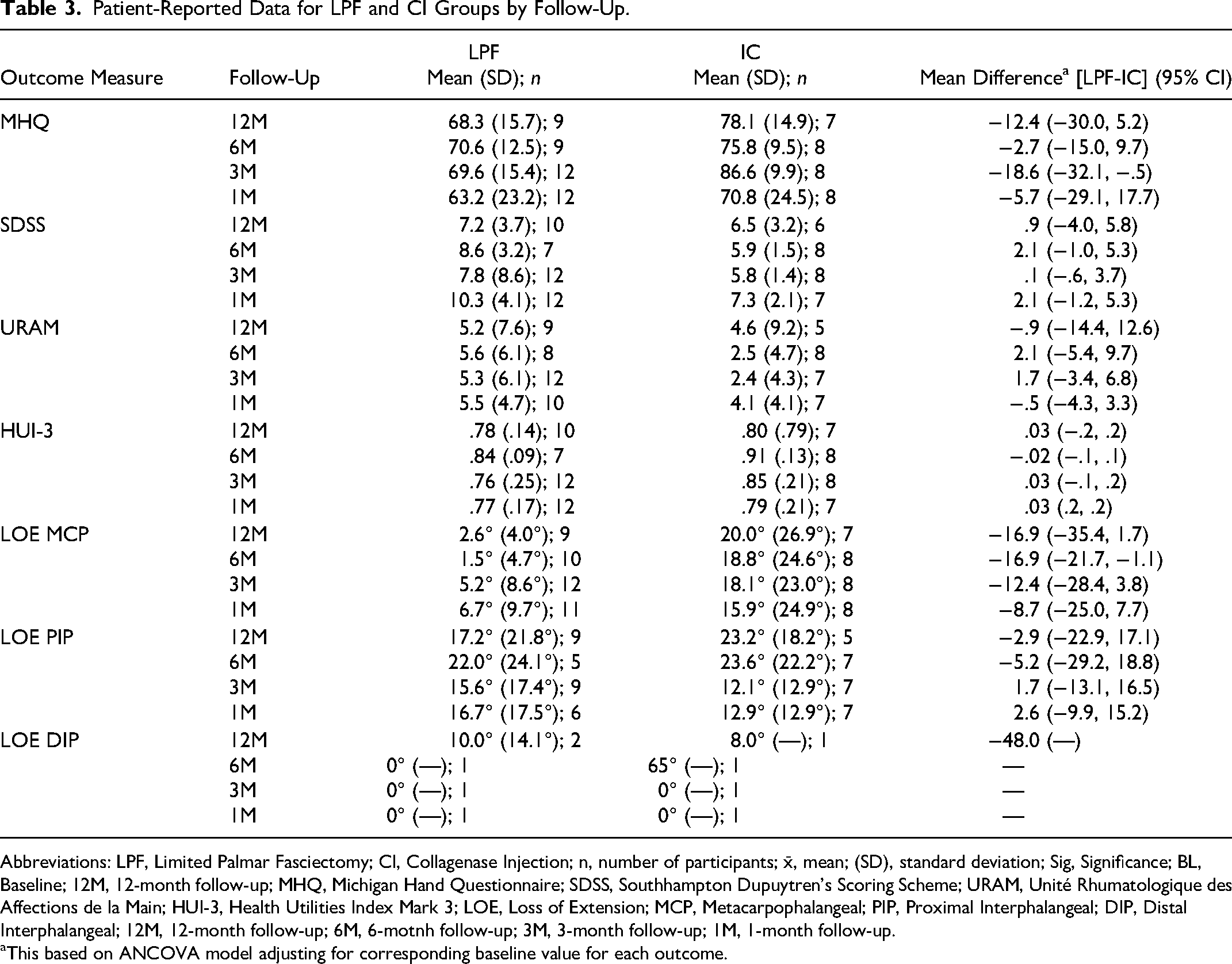
Abbreviations: LPF, Limited Palmar Fasciectomy; CI, Collagenase Injection; n, number of participants; x̄, mean; (SD), standard deviation; Sig, Significance; BL, Baseline; 12M, 12-month follow-up; MHQ, Michigan Hand Questionnaire; SDSS, Southhampton Dupuytren's Scoring Scheme; URAM, Unité Rhumatologique des Affections de la Main; HUI-3, Health Utilities Index Mark 3; LOE, Loss of Extension; MCP, Metacarpophalangeal; PIP, Proximal Interphalangeal; DIP, Distal Interphalangeal; 12M, 12-month follow-up; 6M, 6-motnh follow-up; 3M, 3-month follow-up; 1M, 1-month follow-up.
This based on ANCOVA model adjusting for corresponding baseline value for each outcome.
Discussion
Limited palmar fasciectomy and CI are two common management techniques for DD in North America. Previous RCTs comparing CI to placebo have shown the efficacy and safety of CI10–15; however, this is a clear issue as existing standard treatment approaches exist. At the time of this writing, there is no research directly comparing LPF and CI. As such, the aim of this study was to provide a head-to-head comparison of these techniques to determine which approach is more effective in the management of DD. Unfortunately, several barriers prevented adequate recruitment and, concomitantly, statistical power was not achieved. Given the small sample size, it would be incorrect for readers to conclude that LPF and CI are equivalent based on the presented data. However, this information is still valuable due to the paucity of Level I evidence in the current literature. Data from this study can be pooled with other data in systematic reviews and meta-analyses to help provide the definitive answer our study was unable to provide. In addition, it is the responsibility of clinical researchers to make data publicly available to prevent wasted research resources 23 and to respect the time and effort of patients.
The limited available data from the current study suggests that both techniques are acceptable for the management of Dupuytren's contracture. A definitive improvement in PROMs from baseline to 12 months was noted in both groups and no statistical difference was detected. At 12 months, the difference between groups for the primary outcome, the MHQ, was 9.77, which falls within the MCID range of 8–13. 24 Lastly, we found that 1–4 years post-procedure, three CI patients required palmar fasciectomy and one LPF patient required a second surgery on the same digit. These findings suggest an increased rate of recurrence in the CI group, this conflicts with earlier studies.12,13
Readers should consider the following limitations when interpreting this study. First is the small sample size, which prevents statistical conclusions of superiority. Readers are discouraged from concluding equivalence between CI and LPF due to limited power. Several factors prevented adequate recruitment including health insurance coverage, treatment preference for the minimally invasive CI, and the discontinuation of Xiaflex® in Canada. Second, is the short time-horizon (12-months); recurrence in Dupuytren's Disease is often noted several years following intervention. Although we did a brief follow-up to determine if additional surgery was needed 1-4 years later, we did not collect functionality, satisfaction, quality of life, or range of motion at this point. It is also important to note that this study did not compare or go into detail regarding the type of DD that was treated. Investigators planning to conduct future RCTs comparing procedures of differing invasiveness should first perform a feasibility study to determine interest in patient populations. Studies should also plan for a longer follow-up and prepare for the difficulties of doing so (eg, older patient population, decreasing patient commitment for data collection, asking patients to return for non-medically needed follow-up). Lastly, investigators should consider differences between types of DD (ie, pretendinous involving the MCP vs PIP joints) in their analysis. Despite the limitations of this study, it is the first published RCT comparing CI to LPF using a combination of generic and condition-specific scales; future trials should consider doing the same.
Conclusions
Based on available 12-month data, CI and LPF are reasonable for the management of DD. Results should be interpreted with caution given the small sample size; the authors strongly discourage readers from interpreting these results as equivalence between CI and LPF. Future research should consider a pre-trial feasibility study, less restrictive inclusion criteria, additional analyses based on DD type, and a longer follow-up.
Supplemental Material
sj-docx-1-psg-10.1177_22925503231161066 - Supplemental material for Randomized Controlled Trial Comparing the Clinical Effectiveness of Collagenase Injection (Xiaflex®) and Palmar Fasciectomy in the Management of Dupuytren's Contracture
Supplemental material, sj-docx-1-psg-10.1177_22925503231161066 for Randomized Controlled Trial Comparing the Clinical Effectiveness of Collagenase Injection (Xiaflex®) and Palmar Fasciectomy in the Management of Dupuytren's Contracture by Achilles Thoma, Jessica Murphy and Lucas Gallo, Bimpe Ayeni, Lehana Thabane in Plastic Surgery
Supplemental Material
sj-docx-2-psg-10.1177_22925503231161066 - Supplemental material for Randomized Controlled Trial Comparing the Clinical Effectiveness of Collagenase Injection (Xiaflex®) and Palmar Fasciectomy in the Management of Dupuytren's Contracture
Supplemental material, sj-docx-2-psg-10.1177_22925503231161066 for Randomized Controlled Trial Comparing the Clinical Effectiveness of Collagenase Injection (Xiaflex®) and Palmar Fasciectomy in the Management of Dupuytren's Contracture by Achilles Thoma, Jessica Murphy and Lucas Gallo, Bimpe Ayeni, Lehana Thabane in Plastic Surgery
Supplemental Material
sj-docx-3-psg-10.1177_22925503231161066 - Supplemental material for Randomized Controlled Trial Comparing the Clinical Effectiveness of Collagenase Injection (Xiaflex®) and Palmar Fasciectomy in the Management of Dupuytren's Contracture
Supplemental material, sj-docx-3-psg-10.1177_22925503231161066 for Randomized Controlled Trial Comparing the Clinical Effectiveness of Collagenase Injection (Xiaflex®) and Palmar Fasciectomy in the Management of Dupuytren's Contracture by Achilles Thoma, Jessica Murphy and Lucas Gallo, Bimpe Ayeni, Lehana Thabane in Plastic Surgery
Footnotes
Author Contributions
AT: conceived and directed the study, contributed patients to study. AT, JM, BA, LT: contributed to the study design and interpretation of the results. JM, LG: analyzed the data. JM: coordinated the study from inception and data collection. LT: contributed to the statistical analysis and reporting of the data. AT, JM, BA, LG, and LT: contributed to the manuscript preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Statements
All study procedures were approved by the Hamilton Integrated Research Ethics Board (#2521). All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. Informed consent was obtained from all patients for being included in the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Physician Services Incorporated, (grant number PSI Foundation Grant #16-48).
Registration
This study was registered with ClinicalTrials.gov (NCT02725528).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
