Abstract
Introduction
Dupuytren's disease (DD) is a progressive fibroproliferative disorder characterized by thickening and shortening of the palmar fascia, causing contracture of the digits. 1 Though relatively common, the incidence of DD is highest in patients of northern European origin, with prevalence ranging from 2% to 42% in diverse Caucasian populations. There are several risk factors associated with the development of DD, including male sex, diabetes mellitus, hypertension, dyslipidemia, alcoholism, and smoking.2,3
DD is more common in males than females. 4 Traditionally, estimates suggest an overall male-to-female ratio ranging from 3:1 to 9.5:1. 4 There is evidence that the male-to-female ratio is not as discrepant as previously thought. 4 Sex variation in DD, however, is not limited to incidence but persists in age of onset, bilateral versus unilateral hand involvement, age at the first operation, and recurrence of the disease. 4 Recent studies report earlier age of onset in men, which has been associated with more aggressive disease.5,6 There are described sex differences in disease severity, 7 reoperations, and postoperative complications.4,8 Although the etiology of DD remains unknown, strong evidence of sex variation throughout the clinical course of the disease may suggest different underlying pathophysiological mechanisms, and it warrants studying sex differences in robust methodological designs, such as clinical trials.
There are various surgical and conservative treatment options for DD, and some observational studies have investigated the safety as well as tolerability of each approach. Peimer et al 9 identified a lower incidence of adverse events in Clostridium histiolyticum collagenase treatment compared to fasciectomy, including nerve injury (0% vs 3.8%), neurapraxia (4.4% vs 9.4%), and complex regional pain syndrome (0.1% vs 4.5%). Another study concluded that fasciectomy produces a greater mean magnitude of correction for digital contractures compared to collagenase. 10 However, sex-specific efficacy and complications and would need to be investigated in clinical trials to account for known confounders.
The objective of this systematic review was to determine the proportion of female participants’ enrollment into clinical trials examining treatments for DD and to assess sex differences in tolerability and complications for DD treatment.
Methods
This systematic review and meta-analysis was conducted and reported in accordance with Preferred Reporting Items for Systematic Reviews and Meta-Analyses for Equity (PRISMA-Equity) guidelines 11 and registered on the PROSPERO International prospective register of systematic reviews (CRD42020204525).
Literature Search Strategy
An electronic search was conducted in Ovid Medline (1946—November 2020), Ovid Embase (1947—November 2020), and EBSCO CINAHL (1952—November 2020) from database inception to November 2020. The search strategy utilized a combination of subject headings, text words, and author-supplied keywords. A combination of the following subject heading terms was included for Ovid MEDLINE database search: “Dupuytren Contracture,” and any of the following terms “Radiation,” “Radiotherarapy,” “Radiofrequency Therapy,” “Collagenases,” “Injections,” “Fasciotomy,” “Needles,” “Surgery,” “Sex,” “Gender Identity,” “Gender role,” “Bias,” and “Randomized Controlled Trial.” Similar search strategies were used for the other databases.
Study Selection Criteria
We included controlled trials of adult (≥ 18 years old) male and female patients with isolated DD that evaluated conservative or non-conservative treatment, including injections, percutaneous needle aponeurotomy (PNA), radiation, and surgery. All included studies were published in the English language. Details on randomization were not required in eligible clinical trials. Exclusion criteria were studies that did not indicate the sex distribution of study participants, did not include female participants, articles not published in English, articles missing an abstract or full text, and observational studies (eg, case reports or series, protocols, guidelines, literature reviews, and editorials). Full text was considered missing if there is only a published abstract or the article was not accessible through the authors’ academic institution.
Selection of Eligible Studies
Four reviewers independently assessed the titles, abstracts, and full text for inclusion using Covidence systematic review management. Each article was assessed by at least 2 independent reviewers. The screening was completed in 2 stages: first, articles were screened by title and abstract and second, eligible abstracts underwent full-text review. Any discrepancies were resolved by consensus and by consulting the senior author (HB).
Data Extraction and Study Quality Assessment
Data extraction of included articles was also performed by 2 independent reviewers, and any discrepancies were resolved by consensus and by the senior author. The following data were extracted from included articles: bibliographic data, primary and secondary objectives, study design, study duration, inclusion and exclusion criteria, the total number of participants, number of male and female participants, age of participants, treatment methods or arms, and number of male and female participants in each treatment arm. Furthermore, we extracted data on the following if applicable: any exploration of sex differences in clinical outcomes, subgroup analyses on sex differences, assessment of sex differences in tolerability of treatment, and discussion of potential differences in outcomes or complications attributed to sex differences. No formal quality assessment was completed as the proportion of female enrollment was the primary estimate for the meta-analysis, and it was assumed to be independent of the study quality.
Statistical Analysis
Categorical variables were reported as frequencies and percentages, and the overall sex distribution was analyzed. Heterogeneity between the studies was investigated by calculating I2 and Tau2. We performed a meta-analysis on the proportion of female participants in eligible studies using the random-effects model to calculate the proportion of female participants. A Funnel plot was visualized, and Egger's regression test was conducted to assess for the presence of publication bias. Additionally, we performed a subgroup analysis for studies including pharmacological therapeutics in DD, such as collagenase injections, to estimate the overall proportion of female participants. Although we used the random-effects model in the interpretation of the results, we also reported the fixed-effects model estimates. All the analyses were completed using R statistical software.
Results
A total of 4759 references were identified through the database search. After the screening was completed, a full-text screening was completed for 59 references. We excluded 31 references based on the eligibility criteria. The most common reason for exclusion was a wrong study design (n = 12), followed by a reply, protocol or commentary on an RCT (n = 5) and a follow-up or a secondary analysis of a previous trial (n = 5). One trial was excluded for including only male participants. 12 Figure 1 illustrates the flowchart of the study with the reasons for exclusion.
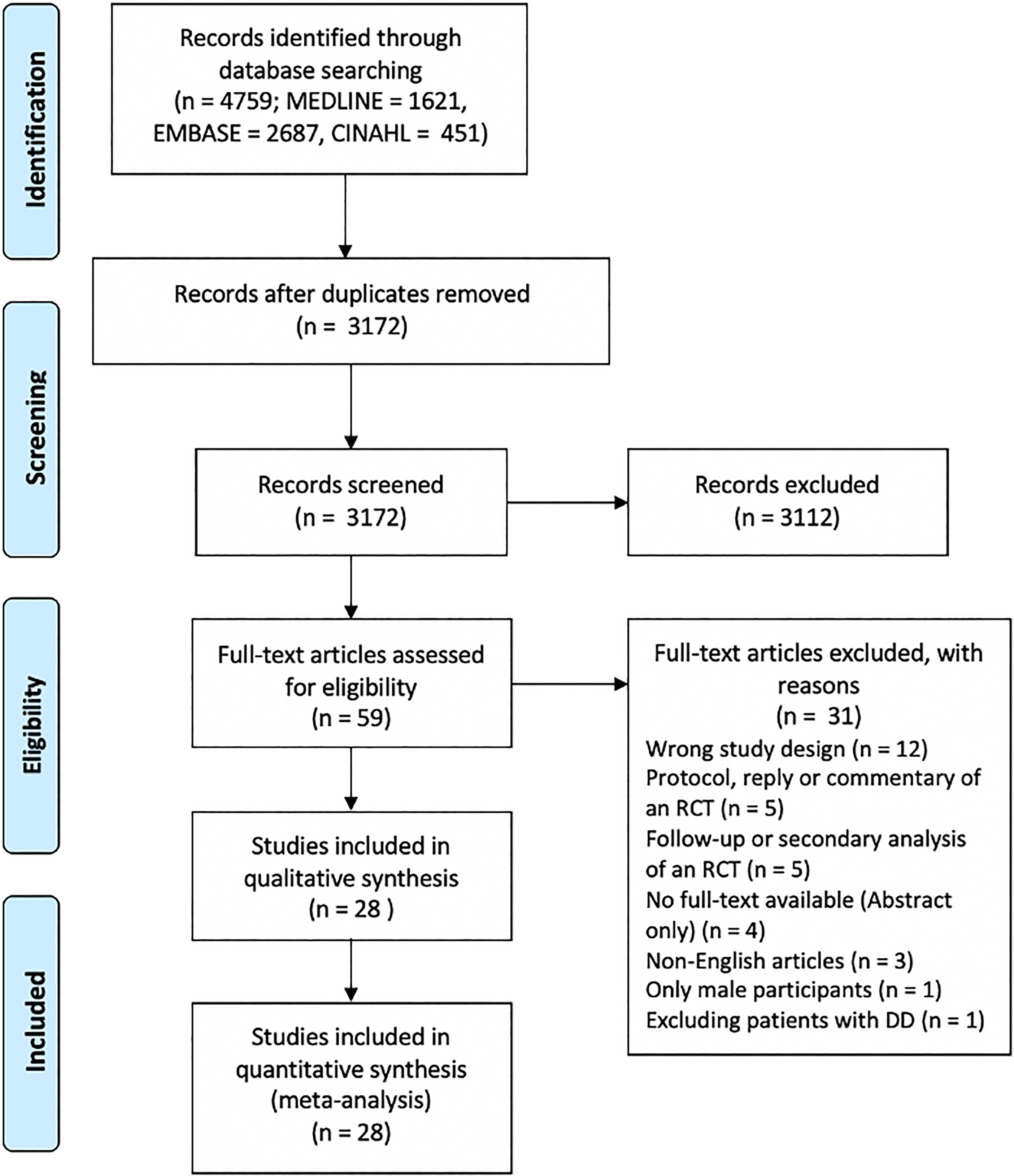
The flowchart of the systematic review. The PRISMA flow illustrates that a total of 3172 abstracts were screened, and 59 full-text articles were reviewed. There were 31 studies excluded following full-text review, and the study reviewed 28 studies.
A total of 28 studies were eligible for the final review. The total sample size was 2853, and the overall number of female participants was 545 (19.1%).13–40 Not all the studies specified the sex differences of the study participants per treatment arm. As such, we did not report the overall number of participants within each arm of eligible trials. No studies assessed sex differences in the primary outcome or performed a subgroup analysis on male and female participants. Only 2 studies indicated a discussion of differences in complications between male and female participants.28,33 Only one study discussed tolerability related to sex differences after administering tamoxifen. 33 There were 13 studies (46.4%) that used pharmacological therapeutics in their treatment arms, and 5 studies (17.9%) had a placebo arm in their trial. Table 1 summarizes the characteristics of eligible studies.
Summary of the Characteristics of Eligible Studies.
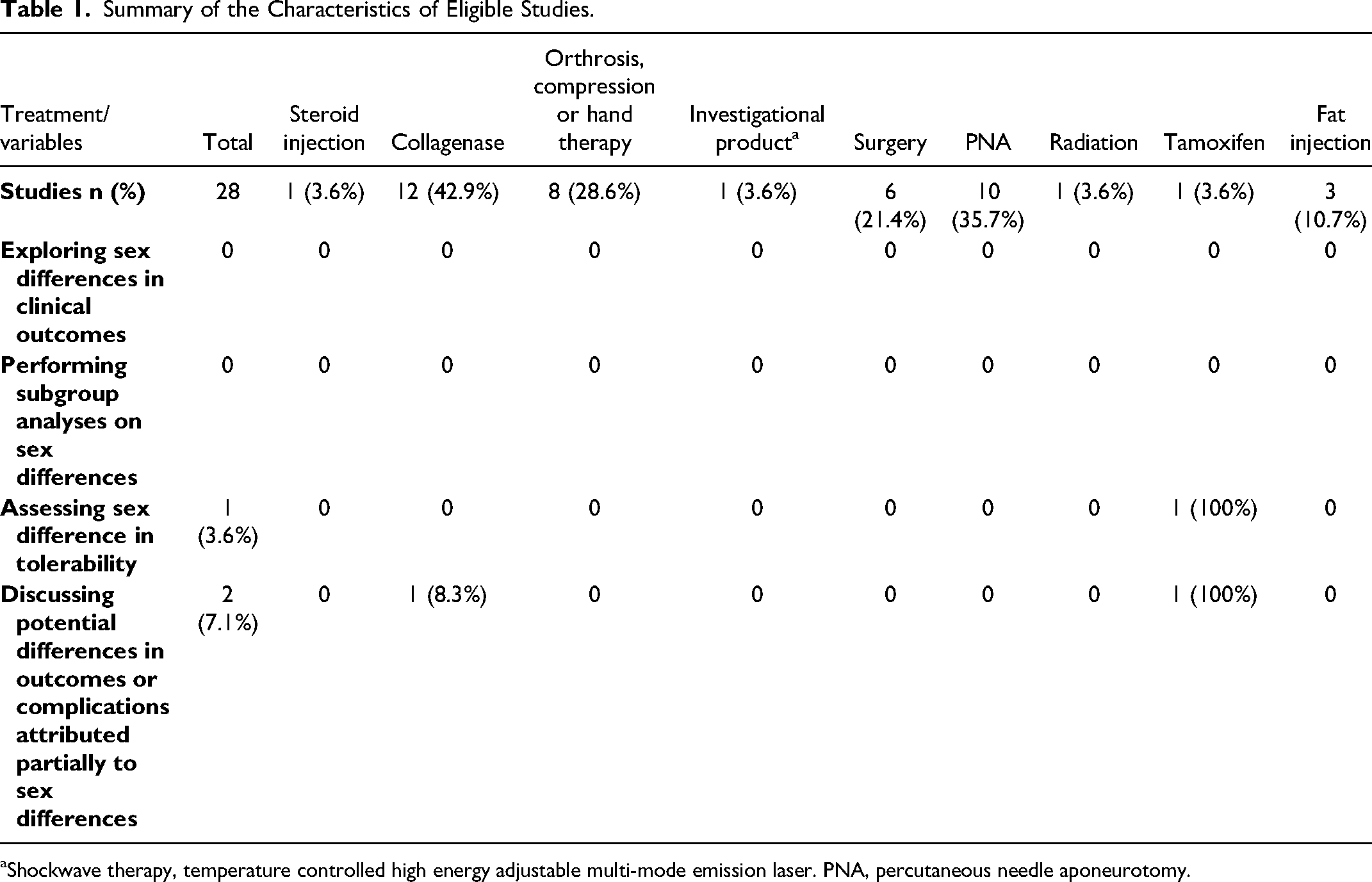
Shockwave therapy, temperature controlled high energy adjustable multi-mode emission laser. PNA, percutaneous needle aponeurotomy.
There was a significant heterogeneity between the studies (I2 = 77.9% [95% CI: 68.4-84.5%]; Cochrane Q P < .0001, Tau2 = 0.01). As such, we performed a meta-analysis using the random-effects model to calculate the proportion of female participants in eligible studies. Cumulatively, the percentage of female participants enrolled in the studies was 19.5% [95% CI: 16.1-23.0%]. The estimate from the fixed effect model was 18.3% [95% CI: 16.9-19.8%]. Figure 2 illustrates the Forest plot from the analysis. A funnel plot was used to assess for the presence of a publication bias (Figure 3). There was no evidence of bias on visual assessment or with the use of statistical methods (Egger's test P = .26).
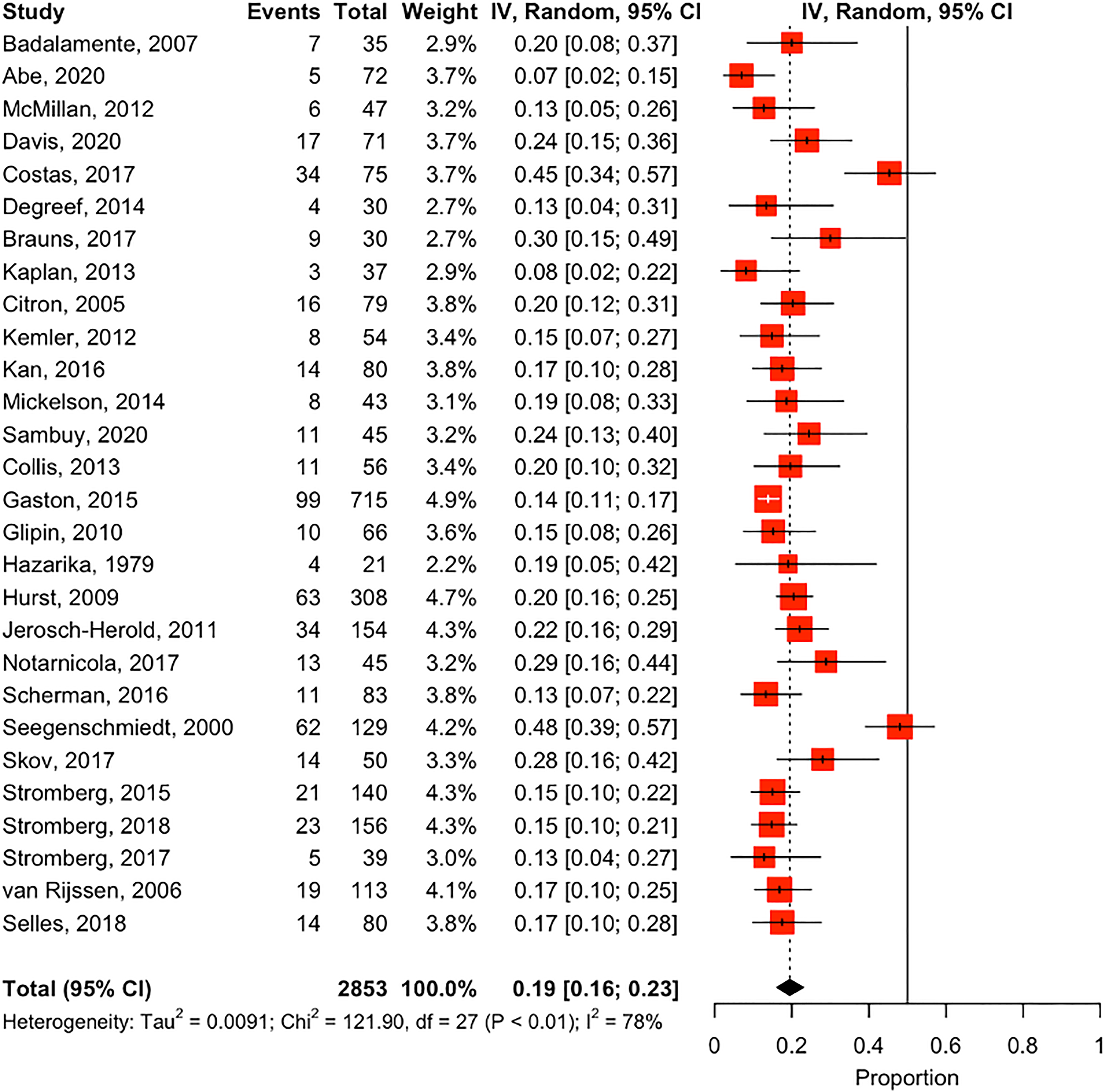
A forest plot estimating the proportion of female participants in eligible studies. The straight line in the forest plot indicates the null value (50%). Values closer to one favor more female enrollment, and values closer to 0 favor more male enrollments. Most eligible studies had more male participants compared to female participants. The random effect estimate was 19.5% [95% CI: 16.1-23.0%].
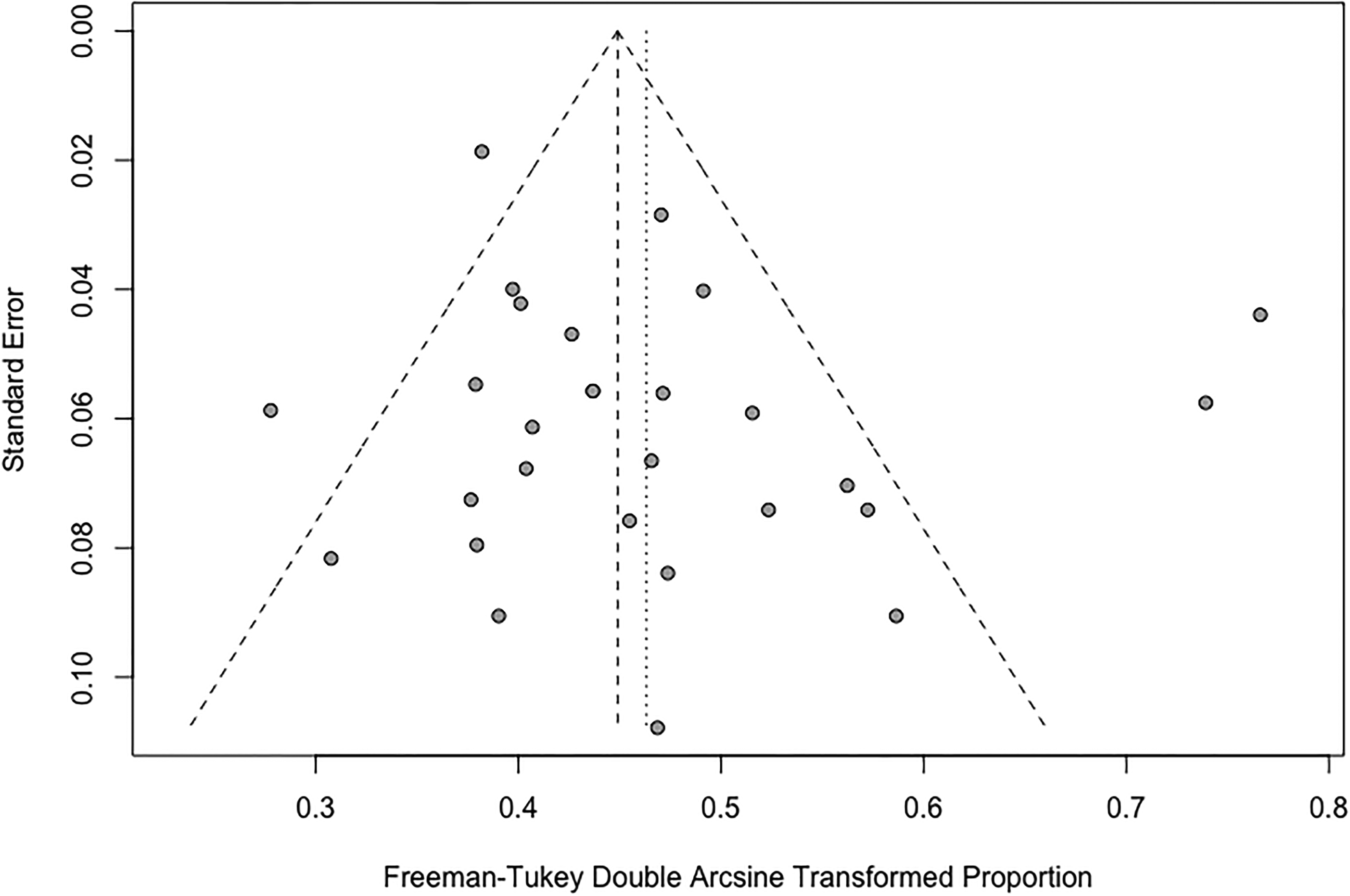
A funnel plot for eligible studies assessing the risk of publication bias. There were some outliers in both directions of the plot. Visual assessment of the funnel plot indicated no strong evidence of asymmetry (ie, no strong evidence of publication bias).
We further performed a subgroup meta-analysis on the 13 studies that used pharmacological therapeutics to estimate the proportion of female participants. The heterogeneity between the studies was significant (I2 = 76.1% [95% CI: 59.2-86.0%]; Cochrane Q P < .0001, Tau2 = 0.006). The meta-analysis indicated that the proportion of female participants enrolled in studies using pharmacological therapeutics was 17.1% [95% CI: 13.1-21.4%] (Figure 4). The estimate from the fixed effect model was 16.0% [95% CI: 14.3-17.7%]. There was no evidence of bias on visual assessment or with the use of statistical methods (Egger's test P = .47, Figure 5).
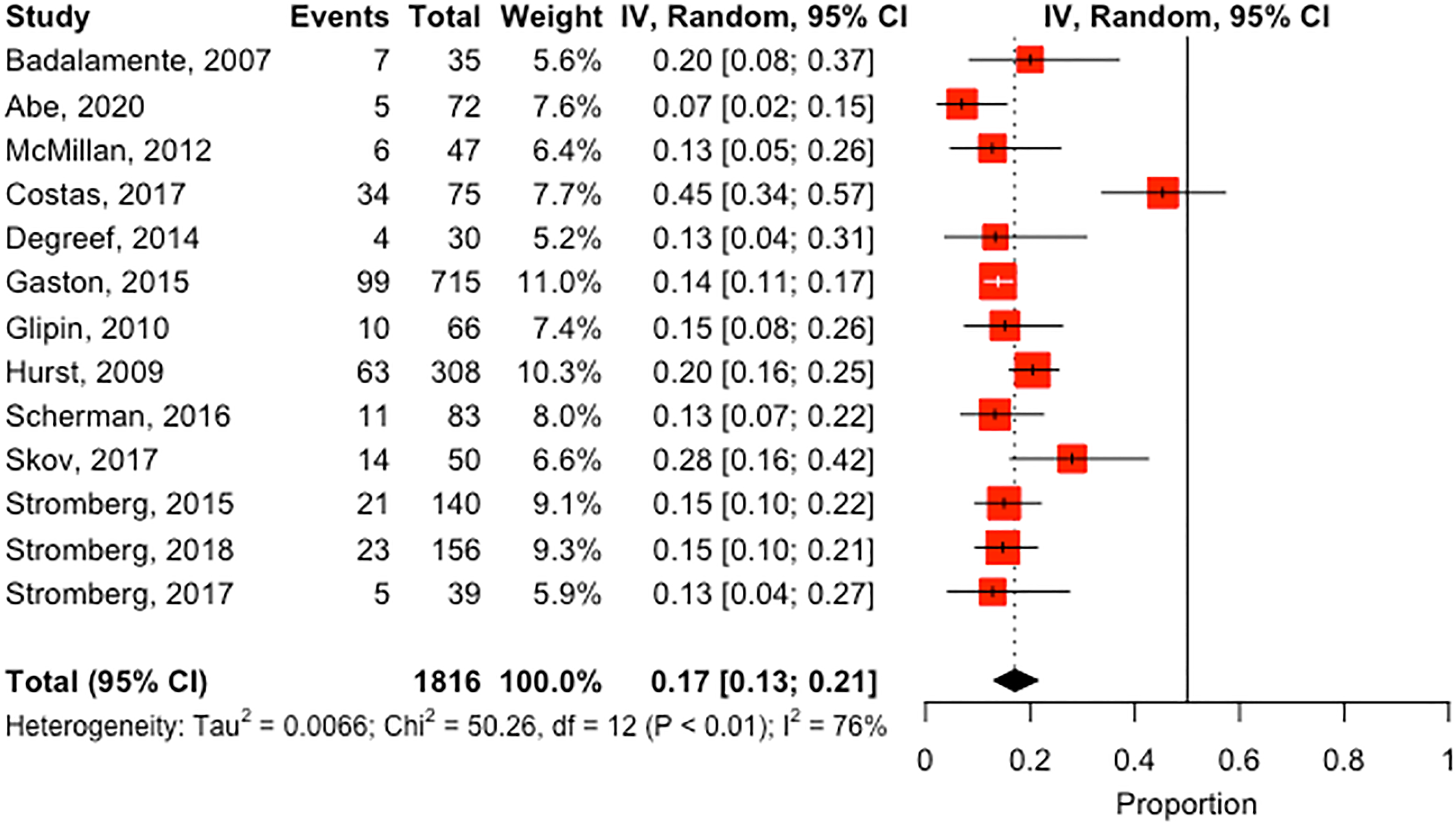
A forest plot estimating the proportion of female participants in eligible pharmacological therapeutic studies. The straight line in the forest plot indicates the null value (50%). Values closer to one favor more female enrollment, and values closer to 0 favor more male enrollments. Most eligible studies had more male participants compared to female participants. The random effect estimate was 16.0% [95% CI: 14.3-17.7%].
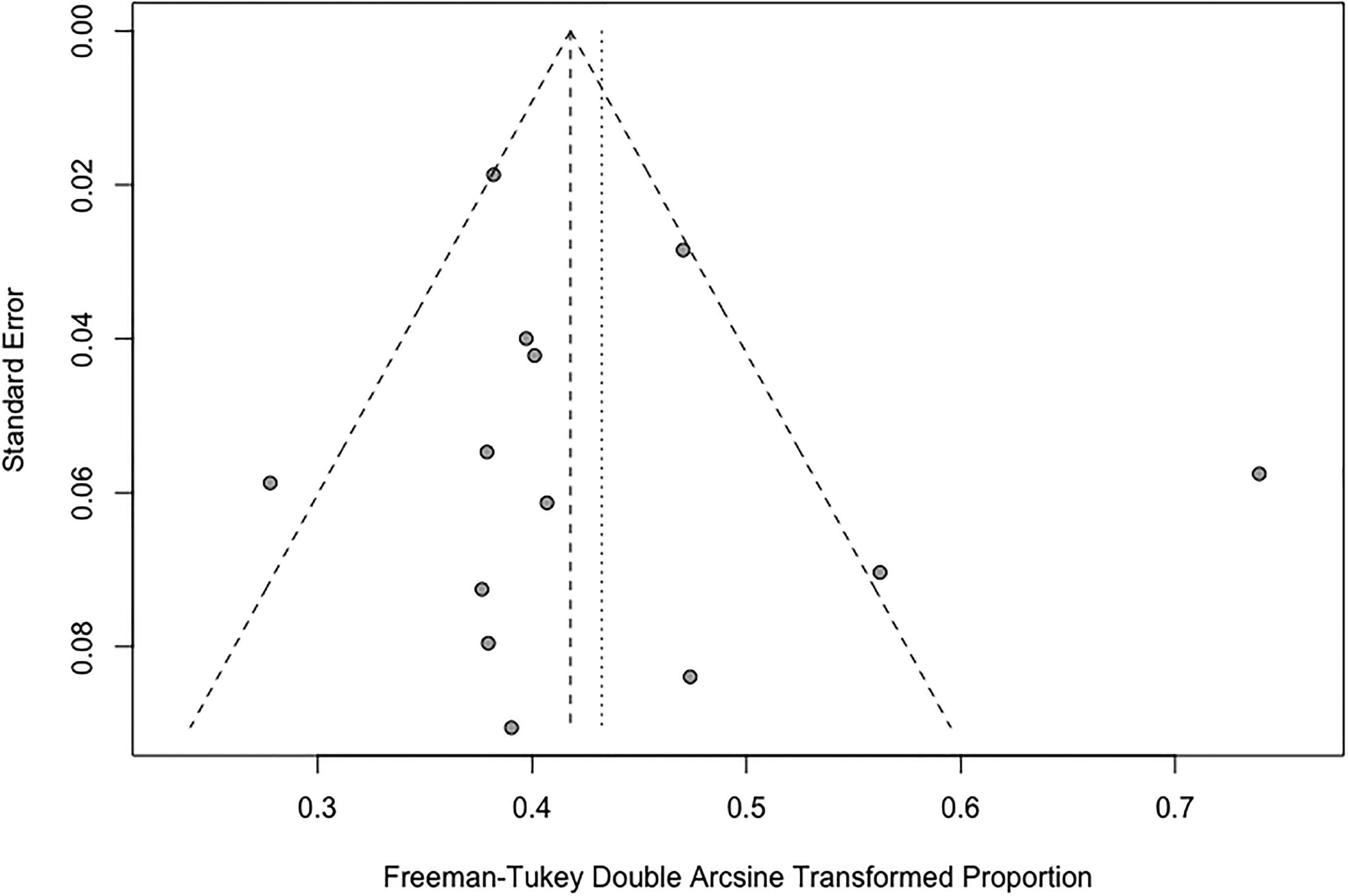
A funnel plot for eligible pharmacological therapeutic studies assessing the risk of publication bias. There was one obvious outlier of the funnel plot. Visual assessment of the funnel plot indicated no strong evidence of asymmetry (ie, no strong evidence of publication bias).
Discussion
This systematic review and meta-analysis identified low emphasis on examining sex differences in clinical trials for DD. Females were less likely to be enrolled compared to males in most of the clinical trials. The overall female enrollment in clinical trials was less than 20%, with a very limited number of studies accounting for tolerability or complications. No clinical trial in this systematic review explored sex differences in the treatment effect of DD.
The prevalence of DD and the male-to-female ratio varies in the literature. Traditionally, the sex differences in the prevalence of DD were thought to be as high as 9.5:1. However, modern United States (US) data suggest a lower difference in prevalence. Anthony et al 4 reported the difference in prevalence as 1.7:1. Interestingly, the difference in ratio approached 1:1 with increasing age, suggesting that females have a later onset of DD. Lanting et al 41 also reported similar results with a male-to-female ratio of 1.2:1, and the authors suggested a difference in the course of DD between males and females. It is also possible that DD is underdiagnosed in females. Degreef and De Semet 42 randomly examined hands in Belgium and found a more negligible difference in the proportion between males and females with early DD. It is unclear, however, whether males are more likely to seek care or if females are less likely to be evaluated and diagnosed with DD. Stahl and Calif 43 reported a delay in female presentations following the onset of DD by approximately 1 year compared to males. DiBenedetti et al 44 surveyed 23 103 participants from the US general population and identified a higher prevalence of self-reported symptoms suggestive of DD with a ratio of approximately 1:1. However, more males received a DD diagnosis or treatment compared to females (63% vs 37%). Therefore, the traditional male-to-female ratio may not be accurate in the general population.
In addition to the sex differences in the prevalence, there are known sex differences in the onset and disease progression of DD. Females appear to have a later onset of DD and a higher age at the first surgery compared to males. 6 Although the sex difference between males and females approached 1:1 in one study, the ratio for receiving fasciectomies was 3:1. 4 Other studies identified a higher sex difference in fasciectomy with a ratio of 5.9:1. Females are also less likely to have reoperations. 6 Some authors suggest females tend to have a milder disease than males.6,43 However, the literature encompasses conflicting results, with the lack of high-quality comparative studies. Ferry et al 45 reported a higher incidence of complex regional pain syndrome in females who underwent surgery for DD. Females were also less likely to achieve a complete range of motion at the proximal interphalangeal joint. The disease severity was more profound in females compared to males, measured with the Disabilities of the Arm, Shoulder and Hand (DASH) questionnaire. Females scored 12.8 points higher in the DASH questionnaire, on average, compared to males. Genetic factors leading to a worse DD in females have also been described. 46 Therefore, there are known sex differences in DD that are worth accounting for in clinical trials.
Though the etiology of DD remains unknown, the epidemiological and clinical research to date draws attention to the strong influence of sex on the onset and progression of the disease. Despite the prolific body of research on DD, sex differences received little attention in clinical trials for DD. Randomized clinical trials represent one of the most robust ways for researchers to showcase the effectiveness and illustrate the clinical impact of an intervention. A critical component is recruiting a well-balanced study population and potentially powering the studies to explore sex differences. 47 Nonetheless, it has long been recognized that sex-based biases, particularly female under-enrollment, are present in participant selection for these trials.48,49 Previous studies have established that there is substantial female under-representation in clinical studies on chronic kidney disease, cardiovascular disease, stroke, non-sex-specific cancers, and HIV/AIDS.49–52 A previous review of all manuscripts published in 2011 and 2012 in Annals of Surgery, American Journal of Surgery, JAMA Surgery, Journal of Surgical Research, and Surgery found that less than one-third of human surgical clinical trials performed data analysis by sex, and that there was a wide variation in the inclusion and the matching based on the sex variable. 53
Sex differences are not known for pharmacological therapeutics in Dupuytren's. For example, some pharmacological therapeutics had different efficacies between males and females in stroke 54 and glioblastomas. 55 In Alzheimer's disease, there are known sex differences in pharmacological treatment that were not described in previous clinical trials. 56 Our study had similar findings in DD. None of the clinical trials reported secondary analyses for sex-specific efficacy in DD, with exceptionally limited data on sex differences. In fact, there was a trial excluded from the analysis that did not include any female participants. 12 Future clinical trials should collect data on sex differences and thoroughly report their results.
It is critical to understand sex differences in DD related to therapeutics. Pagnotta et al 57 reported a higher expression of androgen receptors in the palmar fascia compared to patients with carpal tunnel syndrome. Some authors described the development of an animal model for DD that was described exclusively in male rats. 58 Although this model is promising for exploring the sex differences in DD, excluding female rats will prevent the evaluation of sex differences. Nevertheless, clinical data provide a higher level of evidence for sex differences, and clinical trials can arrive to more accurate conclusions. It is biologically plausible that sex differences might interact with some of the therapeutics. In a randomized trial for high-dose tamoxifen for DD, only 4 females were enrolled into the trial, and only one female participant received tamoxifen. 33 The small proportion of female participants limits the generalizability of the findings to females with DD. The effect of other pharmacological therapeutics is not known due to a critical deficiency in the literature that was identified in this study.
There are multiple approaches to explore sex differences in DD. Clinical trials can stratify randomization by sex to ensure equal enrollment. Ad hoc and secondary analyses can also be performed for each sex as an exploratory analysis to determine the effect of treatment on each sex. Complications should also be reported for each sex and relevant variables. Observational studies should account for sex in their statistical models. Stratifying the results of a trial by sex is essential to identify sex differences in response, tolerability and complications. Funding agencies can consider sex differences in their applications or have specialized funding streams for sex differences. The Canadian Institutes of Health Research (CIHR) recognized the importance of incorporating sex differences in research and issued a mandate for universal inclusion when appropriate. 59 Furthermore, some funding opportunities by CIHR require mandatory training on sex and gender differences and the inclusion of a research plan that reflects the implementation of this policy. 60 In this study, although the proportion of female participants enrolled in clinical trials might be close to the traditional epidemiology of DD, eligible clinical trials were underpowered to examine sex differences. Sex differences are known to impact DD onset and progression and were not explored as a secondary analysis in any of the trials. Thus, it is essential to consider some of the stated approaches in future clinical trials by investigators and funding agencies.
There are several strengths to the study. We identified a critical gap in clinical trials for DD. All trials did not report analyses on sex differences for their primary outcome, with a limited number of trials exploring complications and tolerability. Our results favour exploring sex differences in future studies. We also performed an additional analysis on pharmacological therapeutics where the literature further supports exploring sex differences. We conducted a sensitive search strategy to identify eligible studies. The study outlines an important issue in DD clinical trials and identifies potential solutions for future studies.
The study had some limitations. There is heterogeneity in the investigated treatments used in clinical trials with potentially varying degrees of importance in specifying sex differences. We attempted to overcome this limitation by performing another analysis on trials that included pharmacological therapeutics in DD, and we arrived at the same conclusion. We did not formally assess the quality of the included trials, and we were also unable to perform additional analyses on female participants within each arm due to the lack of universal reporting. The current reporting guidelines encourage authors to report the baseline characteristics for each treatment. We also limited the study to English literature. Some of the sex differences in enrollment might be related to regional variations of the disease. However, designing the trials with enough statistical power to explore sex differences would require a higher proportion of female participants. Finally, some trials only reported the number of female hands, and they did not clearly report the number of female participants. We calculated the number from tables and may not have accurately estimated the correct proportion. It is critical for clinical trials to have transparent reporting and not to violate statistical assumptions, such as statistical independence. Nevertheless, we provided close estimates in the limited number of trials that had this limitation, and we drew conclusions that are unlikely to be impacted by the precision of the estimate.
In conclusion, sex differences are not well defined in clinical trials for DD. Only a very limited number of studies explored sex differences in complications and tolerability, with no studies reporting analyses on the differences in treatment response. Clinical trials are underpowered to examine sex differences in DD, given recent epidemiological studies. Future clinical trials and observational studies should consider sex stratification, ad hoc analyses, reporting sex differences in the primary outcome, tolerability and complications.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
IRB Approval Statement
The study was exempt from IRB approval as it was a systematic review of the existing literature.
Informed Consent Statement
The study did not require consent as it was a systematic review.
Ethical Statement
The study was conforming to the Declaration of Helsinki.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
