Abstract
Background
Breast reduction mammoplasty (BRM) is one of the most commonly performed procedures by plastic surgeons. The majority of BRM procedures are now performed as outpatient procedures, unless contraindicated due to patient comorbidities. 1 It improves the quality of life for women suffering from back, neck, or shoulder pain, headaches, breast or bra groove pain, and intertrigo as a result of their hypermastia.2,3 Other benefits include improved self-esteem, better sleep, decreased anxiety and depression, and improved fit in clothing in addition to improvement in physical symptoms.3,4 Complications of BRM include infection, delayed wound healing, fat necrosis, areola necrosis, hematoma, and seroma formation5–7. Complication and infection rates have previously been reported in the literature and vary widely based on definitions and antibiotic use. A retrospective study of 512 patients undergoing BRM showed a 30-day complication rate of 32% where complications were defined as infection, delayed wound healing, fat necrosis, and areola necrosis. 6 Previously reported rates of infection in BRM vary widely from 0.8% to 31%2,6–14.
Surgical site infections (SSIs) have been divided into 3 categories by the Centre for Disease Control (CDC): superficial incisional, deep incisional, and organ or space infection. The surveillance period for superficial incisional SSIs is 30 days and 90 days for the other 2 SSI categories for breast surgery. 15 Patient characteristics that have been associated with an increased risk of SSIs include extremes of age, poor nutritional status, obesity, diabetes, smoking, previous site infections or colonization, systemic corticosteroid use, and altered immune response. 16 Risk factors for SSI following breast surgery were identified in a meta-analysis and include age, hypertension, body mass index (BMI), diabetes mellitus, American Society of Anesthesiologists (ASA) class 3 or 4, previous breast biopsy or operation, preoperative chemotherapy, hematoma, seroma, and presence of surgical drain. 17 When postoperative infections occur, they are typically caused by pathogens from normal skin flora such as Staphylococcus species.16,18–20 The classification of breast surgery into one of the CDC's 4 types of surgical wounds is controversial. Breast surgery has historically been defined as “clean” surgery but has also been considered as “clean-contaminated” surgery due to the high incidence of SSIs and presence of bacteria in breast tissue.9,18,21,22
Postoperative prophylactic antibiotics are usually prescribed in addition to preoperative prophylactic antibiotics following this procedure1,9 and continue to be recommended by the American Society of Plastic Surgeons. 23 It is thought that a postoperative course of antibiotics reduces the risk of infection. Unfortunately, there are disadvantages to a prolonged antibiotic course such as the increased risk of antibiotic-related complications including Closteroides difficile colitis, allergic reaction, and the increased risk of developing antibiotic-resistant organisms. 24 There are several studies demonstrating the benefit of preoperative prophylactic to prevent the incidence of SSIs; however, there is a paucity of evidence in the literature regarding the use of postoperative antibiotics in BRM.10,25 Previous studies have been completed in an attempt to answer this question however equipoise remains.2,7,13 This study's purpose is to determine if the addition of prophylactic postoperative antibiotics is more effective in preventing SSIs in comparison to a single dose of preoperative prophylactic antibiotics in BRM. We hypothesize that prophylactic postoperative antibiotics will have no effect on the incidence of SSIs.
Methods
This study was approved by our institution's health research ethics board. A retrospective review of 124 patients undergoing elective BRM between July 2018 and December 2019 by a single senior plastic surgeon was completed. The study's design was adapted from the study protocol for a randomized controlled trial (RCT) published by Garcia et al in 2016. 26 We adapted their protocol to retrospectively compare the effect of postoperative antibiotics between 2 groups, while keeping the remainder of the peri- and postoperative protocol for BRM patients as per the senior author's usual practice. The study groups were created based on the location of surgery at 1 of 2 sites where the senior author routinely performs BRM procedures. Group 1 patients had surgery at a publicly funded hospital, while group 2 patients had surgery at a private surgical center (where publicly funded procedures can take place).
Female patients with breast hypertrophy were included. Two study groups of 62 patients were formed based on the location of surgery at 1 of 2 surgical centers. Patients were not formally assigned to either group; operating room availability and patient comorbidities determined the location of surgery. Sample size was determined with a 2-sample non-inferiority calculation to achieve a power of 80% and significance level of 5%. This was compared to proportions described in previous studies.2,26 Each group followed 1 of 2 antibiotic regimens: the first regimen consisted of a single preoperative intravenous cefazolin dose (group 1), while the second regimen consisted of a single preoperative intravenous cefazolin dose followed by a 5-day course of oral antibiotics (group 2). In group 2, patients received 5 days of oral cefalexin unless they had an allergy to penicillin or cephalosporins, in which case they received oral clindamycin. All patients were offered postoperative follow-up with the senior author. The usual follow-up schedule consisted of a follow-up visit on postoperative day 1, at 2 weeks, and at 6 to 8 weeks postoperatively. For patients living outside of Winnipeg, they were typically seen by the senior author on postoperative day 1 and then followed by their family physician closer to home. Primary outcome was the incidence of SSIs. Secondary outcome measures included the incidence of complications such as SSIs, delayed wound healing, dehiscence, and/or antibiotic-related complications.
Data collection included type of BRM and pedicle, the pathology results from the resected breast tissue specimen (benign or malignant), location of surgery, use of suction-assisted liposuction (SAL), combination with other procedures, use of breast drains, weight of resected breast tissue specimen, time to follow-up, operative time, complications, and presence of SSI. Demographic information collected included patient age, BMI, antibiotic allergies, and past medical history including smoking history, diabetes, obstructive sleep apnea (OSA). The criteria for SSIs (superficial incisional, deep incisional, and organ/space SSIs) outlined by the CDC were used to identify SSIs during our retrospective review of patient records. 15
Data were analyzed by calculating percentages, mean, median, and standard deviation. The Mann–Whitney test was used to compare age, BMI, total breast resection weight, operative time, and time to follow-up. Other nominal demographic data was analyzed using either the Fisher’s exact test or chi-squared test if one or more of the 2 × 2 table cells had a count of more than 5. The Fisher’s exact test was used to compare the incidence of SSIs and chi-squared test was used to compare the incidence of complications between groups 1 and 2, respectively. P value of .05 was considered statistically significant. The data analysis was completed using SPSS statistical software.
Results
One hundred and twenty-four patients undergoing BRM were analyzed, 62 in group 1 and 62 in group 2. Demographic information, patient characteristics, and procedure details are summarized in Table 1. Mean patient age was 44 years old and patient age ranged from 16 to 79 years old. Mean patient BMI was 31.4 kg/m2, BMI ranged from 20 to 52 kg/m2. Mean total breast resection weight was 1152 g, with a range of 112 to 5196 g. Ninety-nine percent (123/124) of BRM procedures were Wise-pattern reductions and 85% (106/124) were superomedial pedicles. Mean patient follow-up between all patients was 107 days. Mean operative time was 108 min in group 1 and 113 min in group 2. When the cases with other procedures were excluded, the mean operative time for BRM only was 100 min in group 1 and 107 min in group 2.
Patient Characteristics and Procedure Details for Groups 1 (Preoperative Antibiotics only) and 2 (pre- and Postoperative Antibiotics).
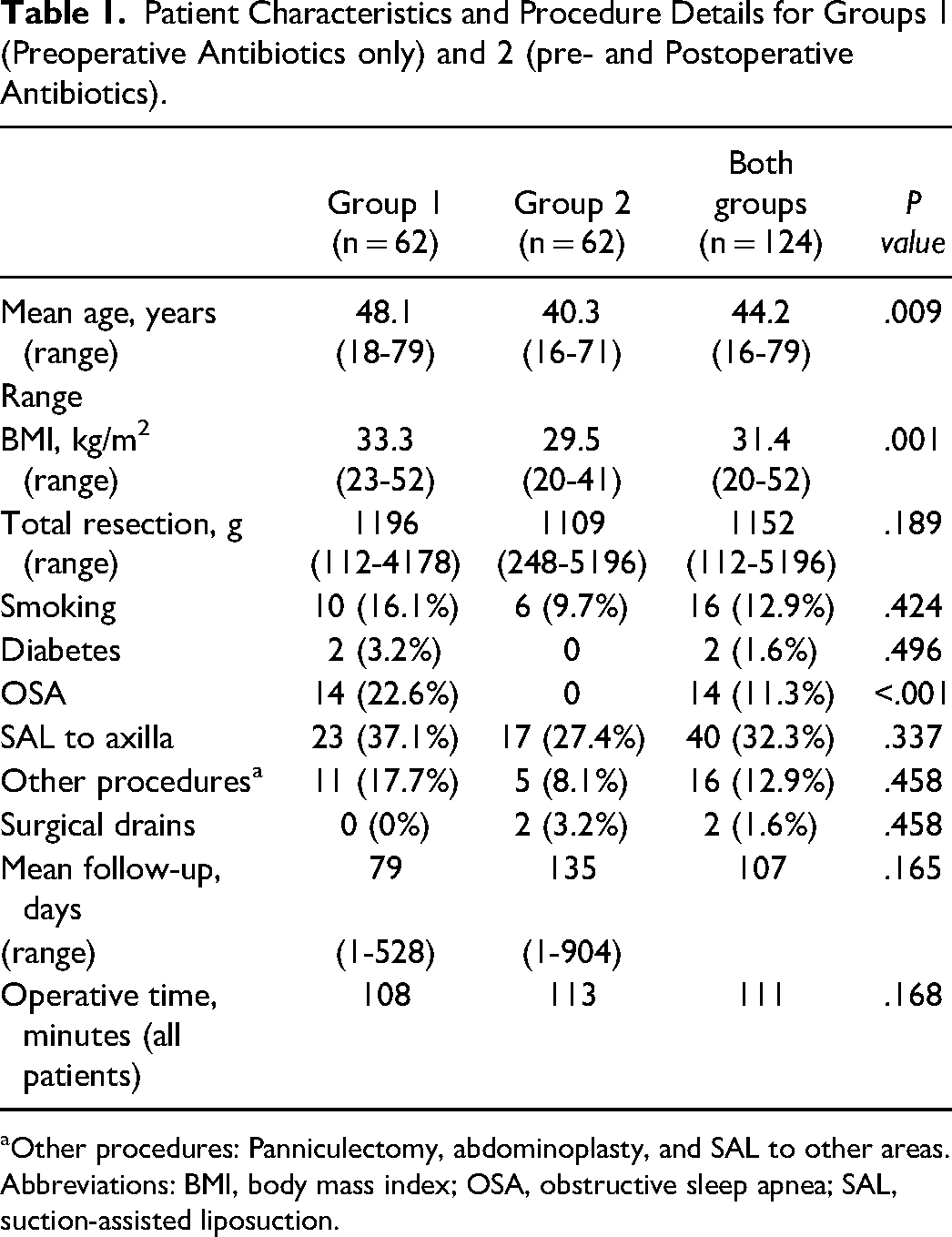
Other procedures: Panniculectomy, abdominoplasty, and SAL to other areas.
Abbreviations: BMI, body mass index; OSA, obstructive sleep apnea; SAL, suction-assisted liposuction.
The incidence of SSI in both study groups is illustrated in Table 2. Overall SSI rate was 5.6% (7/124). Infection rate in group 1 was 8.1% (5/62) in comparison to 3.2% (2/62) for group 2. This result was not statistically significant, with a P value of .44.
Incidence of Surgical Site Infections (SSIs) in Groups 1 and 2.

The incidence of complications in both study groups is presented in Table 3. Overall incidence of complications was 29.0% (36/124); 38.7% (24/62) in group 1 and 19.4% (12/62) in group 2. This result was statistically significant, P value .03. Complications consisted of delayed wound healing, wound dehiscence, SSIs, and 2 hematomas requiring evacuation.
Incidence of Complications in Groups 1 and 2.

Discussion
The study groups were different in terms of surgery location, patient characteristics, and resident participation. As mentioned above, one location is a tertiary health care facility and the other is a private surgical facility. Publicly funded procedures such as BRM and additional cosmetic procedures such as SAL occur at both facilities. Patients requiring overnight admission following BRM due to medical comorbidities, such as OSA, fell into group 1 (publicly funded). This difference between our study groups was clearly illustrated in our results: patients who had surgery in the hospital setting were higher-risk patients. Another key difference between groups is trainee participation. Residents typically only participate in procedures occurring in hospital settings at our institution, and therefore would only have been involved in BRM cases occurring in the hospital setting (group 1). At the private surgical center, an experienced surgical assist was present during BRM cases to help with suturing. The senior author uncommonly places surgical drains in BRM and if drains are placed for any procedure they are removed on postoperative day 1. Use of surgical drains was not statistically different between groups. Drains were only used in 2 patients in group 2. This difference between groups is unlikely to have affected results as they were rarely used and only prolonged usage of drainage tubes has been previously identified as a risk factor for SSIs. 17 Otherwise, the patients in both study groups were treated the same in terms of intraoperative risk factors and follow-up. Sterile technique was followed, and the senior author was present during all cases as the main surgeon. Outpatient follow-up was arranged at similar intervals for both groups and we showed no statistical difference for mean follow–up between groups.
The incidence of infection in our study was comparable to previous studies. There was a statistical difference in some demographic variables between study groups (age, BMI, and OSA) placing group 1 at higher risk for surgical complications. Despite this difference, group 1 (higher risk group) did not receive postoperative prophylactic antibiotics and we were still able to show no evidence of a significantly lower infection rate between groups. Our results are a true representation of the patient population seen at our institution as we did not exclude patients based on smoking, comorbidities, BMI, age, or resection weight—all known risk factors for SSI after breast surgery.6,17,27 This is a real-world patient population outside of the clinical trial setting.
Interestingly, we were able to show a statistical significance between the rate of complications in the 2 study groups. As mentioned above, complications were defined as SSIs, delayed wound healing, dehiscence, and/or antibiotic-related complications. This difference between groups is likely multifactorial and due to increased risk factors for SSIs, resident participation, lack of prophylactic antibiotics, and possible failure to identify minor SSIs. Group 1 patients consisted of higher-risk patients (greater age, higher BMI, and greater number with OSA). The rate of infection was higher at 8.1% (but not statistically significant) and the complication rate was much higher at almost 39% (P = .03). Resident participation in plastic surgery has been studied and is deemed overall safe28–31. We found that operative time was not affected by resident participation. In fact, the mean operative time was shorter in group 1 where residents were present. It has been shown, however, that resident involvement is a risk factor for minor surgical complications such as SSIs and wound dehiscence.5,32 This confounding factor may explain the higher rate of SSIs and complications in the group having surgery in the hospital setting (group 1). Ahmadi et al in 2005 showed a significant decrease in delayed wound healing among patients receiving preoperative antibiotics only. 7 In all the complications recorded in our patient population, all but 2 cases (hematomas requiring evacuation) could be attributed to infection. A course of postoperative antibiotics likely would have improved this, although we cannot conclude this due to the heterogeneity of our study groups. Finally, there is a possibility that some minor SSIs were misdiagnosed as a complication due to the retrospective nature of the study. The CDC SSI criteria is broad and can lead to ambiguity and underreporting of infection, especially while applying the criteria in the setting of retrospectively collected data.
Our study is the first to compare a single dose of IV antibiotics to that and a course of oral antibiotics. At our institution, BRM is typically performed as an outpatient procedure unless overnight admission is required due to OSA or other comorbidities. For that reason, it is common practice to only administer a single IV dose of antibiotics at the induction of anesthesia. There is currently no evidence in the literature supporting the optimal use of perioperative prophylactic IV antibiotics, whether administered as a single dose or over the course of 24 h. Veiga et al published their study protocol for a randomized trial but the results are not yet available. 33 Our study results suggest that a single dose of IV antibiotics is sufficient to prevent SSIs in a high-risk patient population.
We found 3 similar studies to ours from a review of the literature. Most recently in 2020 Garcia et al published the results of a RCT comparing the use of postoperative prophylactic antibiotics (7-day course of cephalexin) versus placebo. 13 Their results showed an infection rate of 0.8% overall and no statistical difference between groups in terms of incidence of infection. The low infection rate is due to their carefully selected patient population, which consisted of young healthy females with a normal BMI. They reported a mean resection weight of 790g and 695g in the control and study groups, respectively, much lower than the mean resection weight of 1152g we reported in our study. All patients in this trial received IV antibiotics preoperatively and for the first 24 h postoperatively, which may not have been necessary in such a low-risk patient population. A second study published by Veiga-Filho et al in 2010 prospectively investigated the difference between a group of patients receiving IV antibiotics from induction of anesthesia until discharge from hospital on postoperative day 1 and 6 days of oral antibiotics versus no antibiotics at all. 2 They showed a significantly lower rate of SSI in the group that received antibiotics (14% SSI vs 2% SSI) but were not able to differentiate between the benefits of pre- versus postoperative prophylactic antibiotics. Finally, the last study was published in 2005 by Ahmadi et al compared 3 groups: no antibiotics, preoperative antibiotics only, and both pre- and postoperative antibiotics. 7 They found no significant difference in the incidence of infection between their study groups and the average incidence of infection was 24%. Two meta-analyses showed that the administration of prophylactic postoperative antibiotics remains controversial.10,12
Limitations to this study include its retrospective nature and designation of study groups, which resulted in heterogeneous study group demographics and biased results. The formation of study groups based on location resulted in the formation of a higher risk patient population in group 1. Despite this difference and the fact that this group did not receive prophylactic postoperative oral antibiotics, we found no statistical difference in the infection rate. Another large source of bias results from the fact that this study is based on a single senior author's practice. Data collection was limited to the medical records of the senior author and did not include visits to ambulatory clinics or emergency departments, unless this was reported to the surgeon and noted in the patient's record. There is a possibility that some cases of infection have been missed due to patients presenting elsewhere and failing to notify the senior author's office. The identification of infection cases was also dependent on diagnosis in the medical record by the senior author or information in the chart meeting the definitions of infection by the CDC as mentioned above. A multivariate analysis may have been able to control for some of the differences between our study groups, but due to our study's sample size, this type of analysis was not feasible. Another limitation is the fact that our study design was based on an RCT non-inferiority trial despite being retrospective in nature. There is a risk that our study is underpowered due to this. A RCT would be useful as a follow-up study to confirm our results and eliminate biases associated with our study groups, data collection, and identification of SSIs.
Conclusion
Study results suggest that there is no significant difference between giving postoperative prophylactic antibiotics and not giving postoperative prophylactic antibiotics with respect to the rate of SSIs for BRM. A RCT, with a larger sample size to allow for a multivariate analysis, would be a useful follow-up study to confirm our results.
Footnotes
Consent
Informed consent was deemed unnecessary due to the retrospective nature of the study. No personal identifying information was collected, and aggregate data was analyzed. Study participants were given a unique study number.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Helsinki Statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Previous Conference Presentations
American Society of Plastic Surgeons: Virtual Plastic Surgery Meeting; Virtual Meeting; October 16 to 18, 2020
Canadian Society of Plastic Surgeons Annual Meeting 2020; Virtual Meeting; June 2020
REB Approval
This study was approved by the University of Manitoba Research Ethics Board (Bannatyne), HS23144, H2019:335.
