Abstract
Introduction
Peroneal nerve palsy can result from traumatic, iatrogenic, or oncologic insult. Foot drop due to peroneal nerve palsy can drastically impact quality of life, affecting daily occupational and recreational activities.1, 2 Traditional techniques for management of foot drop include ankle-foot orthoses (AFO), direct nerve repair, nerve grafts, or tendon transfers. Unfortunately, these options often provide unsatisfactory results.3–5 Articulated AFOs have a bulky hinge that makes footwear choice challenging and is a weak point, more prone to break down. Nonarticulated AFOs externally immobilize the ankle at 90°, reducing ankle range of motion and preventing normal gait cycle during ambulation. 4 Users of AFOs are often noncompliant due to issues with hygiene, discomfort, and limitations in activities.2–4 Longitudinal nerve traction injuries often result in long segments of nonviable nerve despite physical continuity; thus direct nerve repair and nerve grafting may not be the ideal treatment.5–8 Lastly, the posterior tibial tendon has been repurposed as an internal sling in several variants with reasonable outcomes in recreating a plantigrade, functional foot.9–11 Although the treatment may eliminate the use of AFOs, restored dorsiflexion strength may be weak, and the procedure can lead to complications including hindfoot valgus deformity, arthritis, and flat foot deformity.12, 13
Nerve transfers promote reinnervation by coapting expendable nerve branches or fascicles to injured recipient nerves close to the motor end plates of the target muscle.14, 15 Donor to recipient nerve coaptations can be performed using end-to-end (ETE) or supercharged end-to-side (SETS) technique. The SETS technique uses an expendable distal motor nerve or fascicle to “supercharge” into the side of an injured nerve with maintained continuity.4, 16 SETS nerve transfer may add to the regenerating recipient nerve to more quickly reinnervate the target muscle. 17 Nerve transfer is a well-established reconstructive technique for upper limb peripheral nerve injury.14, 15 Lower extremity nerve transfer (LENT) continues to be an emerging reconstructive method for management of peroneal nerve palsy. Given the novelty of nerve transfers for management of foot drop, there are still a limited number of clinical cases.1, 2, 18–23
A systematic review previously performed at our center illustrated the variability in dorsiflexion strength following nerve transfers in peroneal nerve palsy. 24 The review revealed a bimodal pattern in which there appears to be responders and nonresponders to the intervention. Patient and surgical factors that best predict success or failure of foot drop correction by nerve transfer remain elusive.
The objective of this work is to (1) evaluate the postoperative outcomes after LENT in patients with peroneal nerve palsy and (2) evaluate the patient and surgical factors that best predict successful restoration of ankle dorsiflexion following nerve transfer.
Methods
Study Design and Patient Selection
Following institutional research ethics board approval, a retrospective cohort study of prospectively collected data was performed. The senior author's complete electronic medical records were reviewed for all patients who underwent LENT to correct foot drop from January 2010 to November 2018.
Patients were considered candidates for nerve transfer in cases of peroneal or sciatic nerve injuries causing foot drop that demonstrated 2 or fewer motor units on EMG of peroneal muscles at 3 or more months post injury. At this time point one would expect remyelination to be complete and observed motor units to be representative of the number of intact axons available to reinnervate via collateral spouting. Patients required good residual function of the donor tibial nerve (TN) with British Medical Research Council (MRC) grades 4 or 5. Optimal timing of surgery was assessed on an individualized basis with the goal to move forward with nerve transfer no more than 6 months following nerve injury.
Patients were included in this study if they underwent LENT with a minimum of 6 months of clinical and electrodiagnostic follow-up. Patients with insufficient clinical records were excluded.
Preoperative Assessment
All patients underwent assessment in a multidisciplinary peripheral nerve clinic by both a plastic surgeon and a physiatrist. Patients underwent clinical examination and electrodiagnostic assessment of peroneal nerve continuity. Electrodiagnostic studies utilized tibialis anterior (TA), peroneus longus (PL), and flexor hallucis longus (FHL) to assess motor response of deep peroneal nerve (DPN), superficial peroneal nerve (SPN), and TN, respectively. All patients demonstrated peroneal nerve palsy, with good residual function of the TN. Normal gait is most affected by dorsiflexion at the ankle and TA is the main contributor to this action; thus, we chose this muscle for our analysis. Electrodiagnostic studies were performed by the physiatrist. Patients with residual peroneal nerve function at time of initial consultation were reassessed prior to surgery to confirm no evidence of further recovery.
The decision to perform an ETE versus a SETS coaptation was made on a case-by-case basis. If there was potential for meaningful recovery via the native peroneal nerve, a SETS coaptation was performed. Potential meaningful recovery was determined by preoperative electrodiagnostic findings of motor unit potentials (MUPs) in the absence of clinical signs of reinnervation on physical examination. If MUPs were present, the nerve was not transected and instead a SETS coaptation was performed to preserve any potential recovery.
Surgical Technique
Surgical procedures were performed by a plastic surgeon with fellowship training in nerve surgery. A tourniquet was applied to the upper thigh as a precaution but was not inflated. The peroneal nerve was initially decompressed and explored to inspect the level and extent of injury. The DPN, SPN, and branches to TA and PL were isolated, inspected under the operating microscope, and stimulated intraoperatively at least 20 min following decompression of the nerve. Once intraoperatively confirming peroneal nerve palsy, the recipient nerves were selected based upon intraoperative findings including level of injury, the diameter of the recipient, the branching nerve anatomy, the location and length of the donor nerves, and intraoperative nerve stimulation. Redundant TN fascicles to FHL and flexor digitorum longus (FDL), or a more proximal branch of mixed fibers to FHL/FDL were identified using a nerve stimulator and selected for transfer. Recipient nerves such as the deep peroneal component of the common peroneal nerve (CPN), the DPN, and the SPN were identified based on anatomy or intraneural dissection; branches to TA and PL were identified by tracing nerve branches from the end organ, proximally.
The decision to perform 1 versus 2 nerve transfers was made on a case-by-case basis, dependent on intraoperative anatomical findings of the donor and recipient nerves. Two nerve transfers were performed whenever possible. Nerve transfers were made to DPN or directly to branches to TA to optimize dorsiflexion recovery whenever possible. When 2 transfers were possible the preference was to perform coaptations to TA and PL to recover dorsiflexion and provide ankle stability. Instances where only 1 nerve transfer could be performed were due to scarring and unhealthy appearing recipient nerve fascicles or challenging donor nerve anatomy with risk of downgrading TN function.
In the setting of ETE coaptation, the selected donor and recipient nerves were divided distally, and proximally, respectively. In the setting of SETS coaptation, an epineurial window was created in the recipient nerve. All coaptations were performed using 8-0 nylon suture with a range of 2 to 4 sutures and fibrin glue under an operating microscope.
Postoperative Management
Following a routine wound check at 2 weeks, patients were examined for clinical and electrodiagnostic signs of motor neuron recovery at 5 months postoperatively, and serially thereafter. Early postoperative assessments involved evaluating motor units in TA while patients were asked to curl their toes. The presence of motor units in TA (recipient) with isolated engagement FHL and/or FDL (donor) was interpreted as reinnervation by nerve transfer.
Study Measures and Data Collection
For all patients who met the inclusion criteria, data was collected regarding age, sex, body mass index (BMI), mechanism of injury, side of injury, date of injury, date of surgery, comorbidities, preoperative and postoperative dorsiflexion and ankle eversion MRC strength and MUPs, and length of follow-up. Two independent reviewers completed data extraction. In cases of disagreement a consensus method was used.
Primary outcome measures were (i) postoperative ankle dorsiflexion and ankle eversion strength evaluated by the MRC grade strength scale, and (ii) the presence or absence of motor units in TA or PL muscle groups on postoperative EMG examination. Time to clinical recovery was defined as first noted contraction of the TA and/or PL muscle on physical examination. Time to EMG recovery was defined by first noted TA and/or PL nascent MUPs. A good clinical outcome was defined as a postoperative ankle dorsiflexion of MRC 4 or greater. Secondary outcome measures were (i) postoperative use of AFO, and (ii) patient and surgical factors.
Statistical Analysis
Statistical analysis was performed using descriptive statistics and nonparametric tests. Descriptive statistics were used to illustrate preoperative, perioperative, and postoperative data. The Wilcoxon signed rank sum test was utilized to compare pre and postoperative dorsiflexion and ankle eversion MRC grade strengths. The Mann–Whitney U test was used to evaluate differences in patient and surgical factors between the good and poor outcomes groups. Differences with P-value less than .05 were considered statistically significant. All nonparametric tests were performed using Statistical Package for the Social Sciences (SPSS) software version 21.0 (IBM Corporation).
Results
Patient Demographics and Injury Patterns
Nine patients underwent nerve transfer for peroneal nerve palsy. Most patients were male (n = 5), with a mean age of 38.3 years old (range, 18-57 years) (Table 1). Five patients suffered high-energy traumatic knee dislocations and 1 patient sustained a high-energy traumatic hip dislocation. One patient suffered a low-energy knee dislocation from ambulation, 2 patients experienced iatrogenic injuries secondary to arthroplasty hip surgeries, and 1 patient sustained an injury secondary to nerve tumor resection (Table 1).
Patient Demographics, Injury Site, and Mechanism.
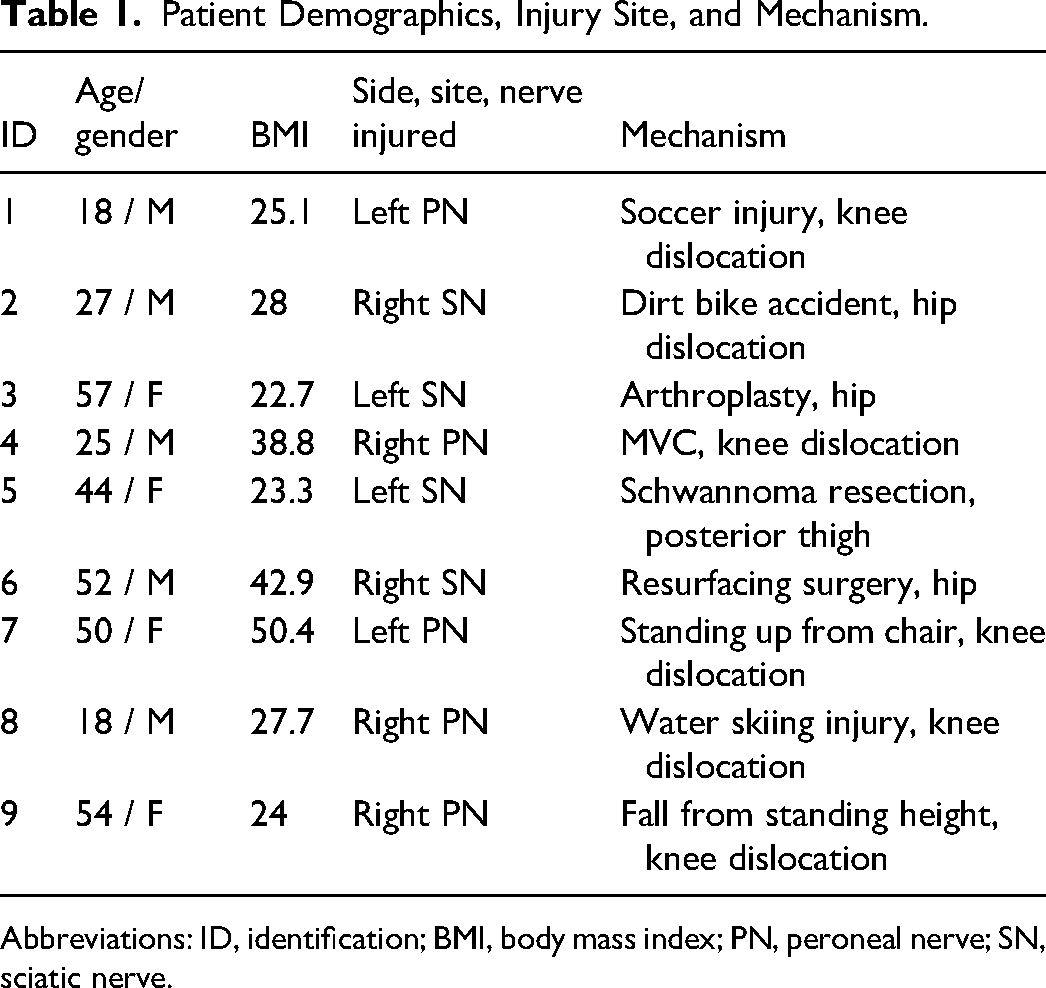
Abbreviations: ID, identification; BMI, body mass index; PN, peroneal nerve; SN, sciatic nerve.
Patient Factors
All but 1 patient (78%) demonstrated MRC 0 ankle dorsiflexion and eversion strength at initial assessment. Notably, Patient 5 presented after left sciatic nerve schwannoma resection. She was using an AFO at time of consultation. Though she regained some dorsiflexion and ankle eversion strength (MRC 3 and 4 respectively), electrodiagnostic studies 3 months post injury demonstrated a paucity of motor units. One would expect recovery due to remyelination to be complete at this time point. She was dissatisfied with her level of function. The patient wished to pursue nerve transfer after thorough counseling on surgical and nonsurgical options. Given the patient's goals, the time lapse since injury, the poverty of motor units for collateral sprouting, and the distance of axonal regrowth required prior to irreversible fibrosis, we felt the benefit of a SETS nerve transfer outweighed the risk.
Patients had normal or near normal function of the TN with preoperative plantar flexion strength MRC 4 or 5 (Table 2).
Surgical Factors Including Time to Surgery, Nerve Transfers, Postoperative Outcomes, and Follow-up Duration
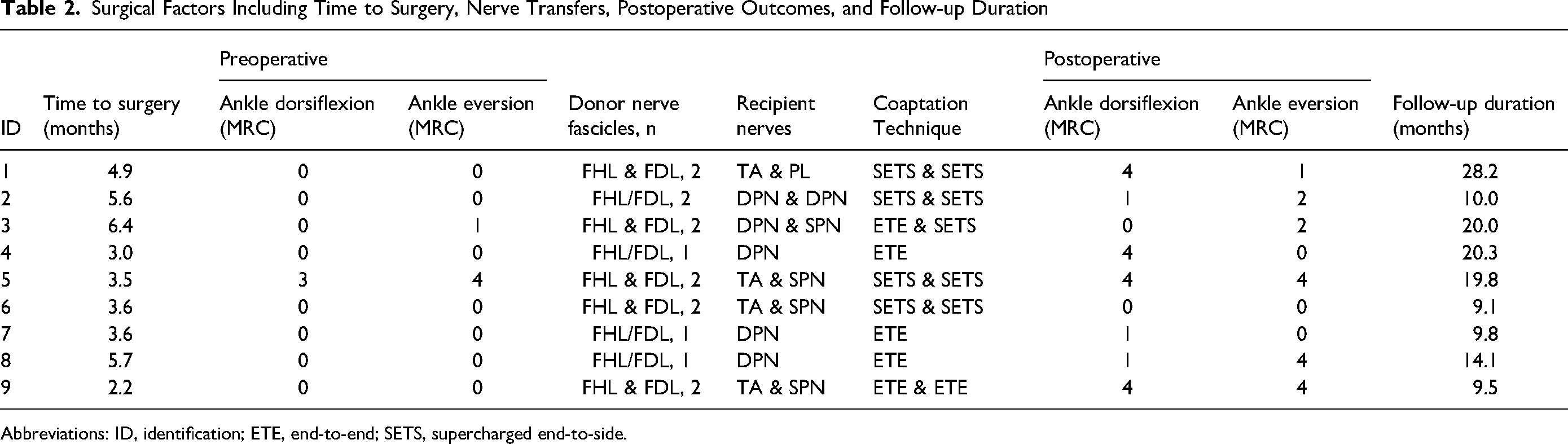
Abbreviations: ID, identification; ETE, end-to-end; SETS, supercharged end-to-side.
Surgical Factors
Patients underwent nerve transfer surgery a mean of 4.3 months following injury (range, 2.2-6.4 months). There were no complications or donor site morbidities resulting from the nerve transfer surgery.
The peroneal nerve was decompressed in all patients. Decompression occurred at the time of nerve transfer in 8 patients. In one case the primary surgeon was consulted intraoperatively by the Orthopaedic Surgery service for assessment of a transected peroneal nerve. Given the mechanism and signs of traction injury, the nerve was first decompressed and tagged by the primary surgeon for later nerve transfer surgery. Patient 8 presented in a delayed fashion, 5 months after knee dislocation with complete foot drop; therefore, knee reconstruction by the Orthopaedic Surgery service was performed prior to nerve transfer, during the same operation.
TN fascicles to FDL and FHL were utilized as donors in all 9 patients (Table 2). The branch to TA, the branch to PL, the DPN, the SPN, and in one instance the deep peroneal component of the CPN were utilized as recipients. Most patients received 2 nerve transfers (67%). A SETS transfer was performed in 5 patients (56%) with the remainder performed in an ETE fashion (44%).
Postoperative Results
Mean follow-up was 15.6 months, with a range of 9.1 to 28.2 months (Table 2). After nerve transfer surgery 4 patients (44%) achieved MRC 4 motor recovery of ankle dorsiflexion, 3 patients (33%) obtained MRC 1 motor recovery, and 2 patients (22%) did not recover any motor function (Table 2).
Both ankle dorsiflexion (P = .015) and ankle eversion (P = .041) increased significantly after nerve transfer surgery. The mean ankle dorsiflexion strength increased from MRC 0.3 preoperatively to MRC 2.1 postoperatively (Table 3).
Preoperative and Postoperative DF and AE Strength (MRC) N = 9.

Abbreviations: DF, dorsiflexion; AE, ankle eversion; MRC, Medical Research Council.
Mean ± standard deviation.
*Wilcoxon signed rank sum test.
In 3 of 4 patients that achieved meaningful recovery (MRC 4 or greater), the mean ankle dorsiflexion strength increased from MRC 1 preoperatively to MRC 4 postoperatively. One patient had a preoperative dorsiflexion strength of MRC 3, which increased to MRC 4. Electrodiagnostic recovery was noted at a mean of 7.7 months (range 5.1-11.8 months), while clinical recovery was noted shortly after at a mean of 9.3 months (range 5.1-20.0 months) (Table 4).
Postoperative Recovery (n = 8).
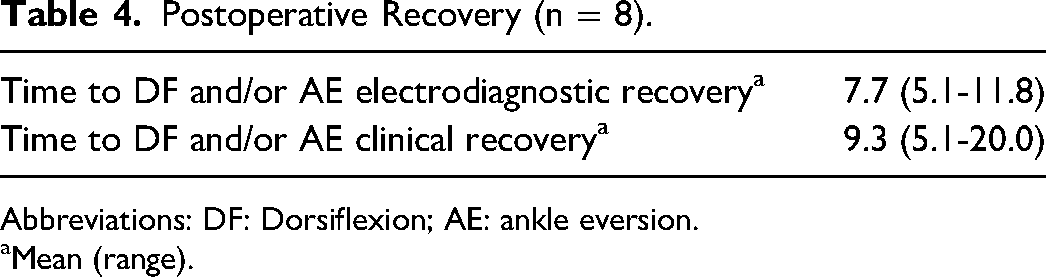
Abbreviations: DF: Dorsiflexion; AE: ankle eversion.
Mean (range).
Postoperative use of AFO
Of the 4 patients that achieved a dorsiflexion strength MRC 4, 3 (75%) used an AFO as needed for strenuous activities postoperatively and 1 patient (25%) discontinued use of an AFO altogether (Table 5).
Postoperative Use of AFO (n = 8).
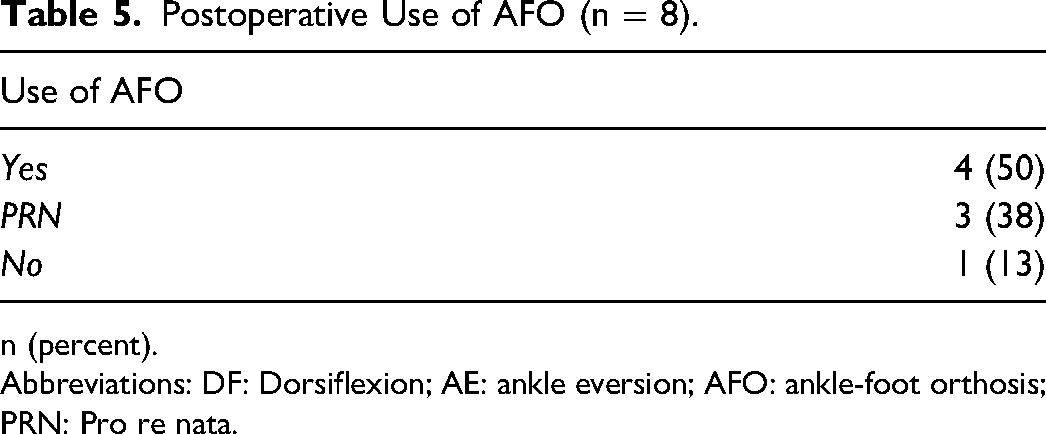
n (percent).
Abbreviations: DF: Dorsiflexion; AE: ankle eversion; AFO: ankle-foot orthosis; PRN: Pro re nata.
Impact of Patient and Surgical Factors
When comparing prognostic factors between the good and poor outcome groups, a shorter time to surgery (3.4 months vs 5.0 months) was significantly associated with better outcomes (P = .049). While not statistically significant, the good outcome group tended to be younger, had lower BMIs, and shorter time to EMG changes and clinical strength recovery. While not statistically significant, patients in the good outcome group received at least 1 nerve transfer directly to the TA nerve branch directly as opposed to the main trunk of the DPN more often than the poor outcome group (75% vs 20%) (P = .099) (Table 6).
Comparison of Patients With Good and Poor Outcomes.
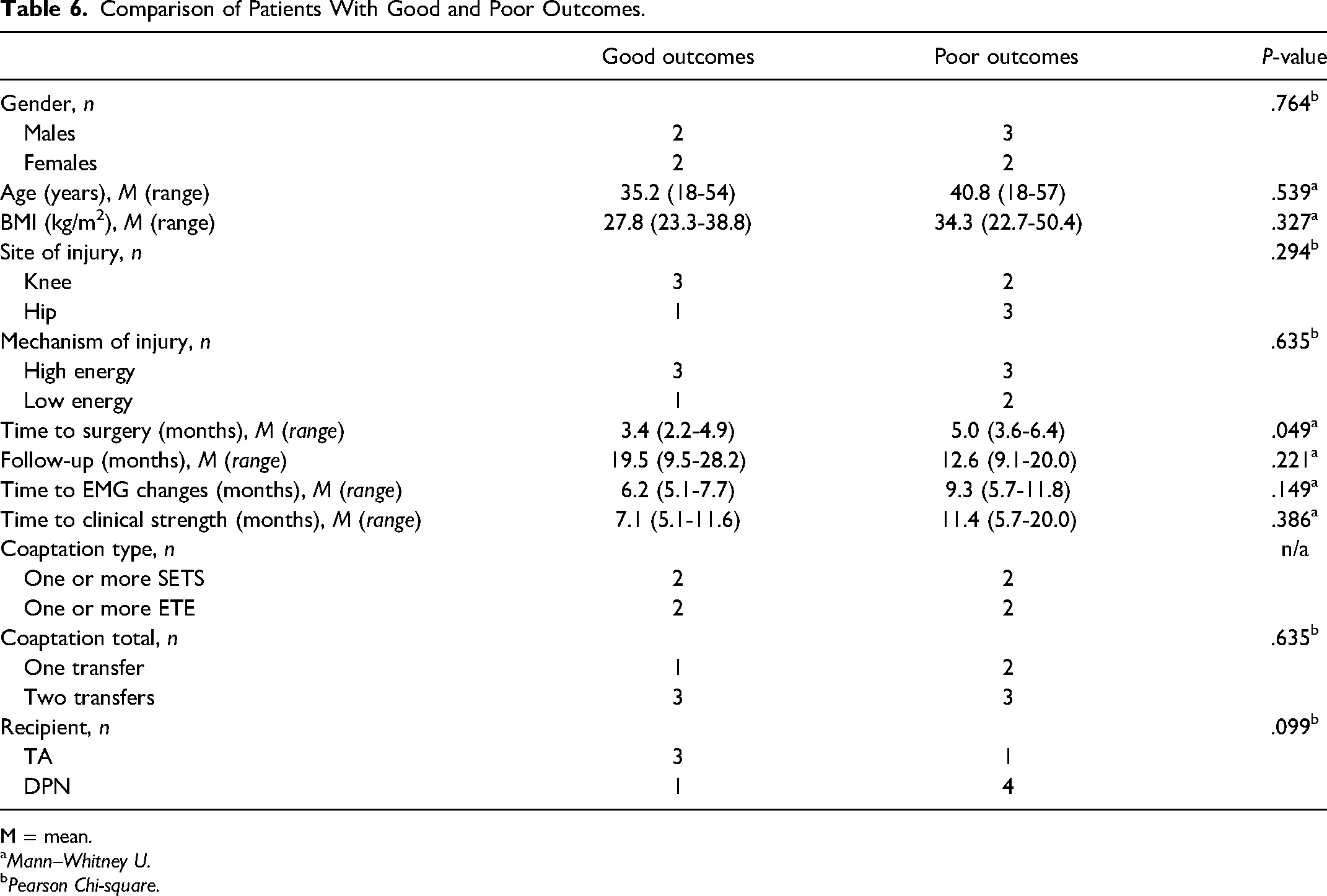
M = mean.
Mann–Whitney U.
Pearson Chi-square.
When assessing surgical factors such as nerve donors, recipients, number of transfers, and coaptation technique, no meaningful differences could be identified between the 2 outcome groups (Table 6).
Discussion
Nine patients were included in this retrospective cohort study with a mean follow-up time of 15.6 months. Patients underwent nerve transfer surgery a mean of 4.3 months following injury. Ankle dorsiflexion strength increased significantly from a mean MRC 0.3 preoperatively to 2.1 postoperatively, and 4 patients demonstrated postoperative dorsiflexion strength of MRC 4 or greater. A shorter time to surgery was the only prognostic factor significantly associated with better outcomes.
Donor and Recipient Nerves
There is no consensus on the optimal donor nerve in LENT for peroneal nerve palsy. Nath et al revealed promising results after transfer of either the SPN or partial TN to DPN, with 10 of 14 patients achieving postoperative ankle dorsiflexion strength of MRC 4 or greater. 1 Flores et al utilized the branch to soleus as donor to DPN with only 1 of 10 patients achieving postoperative ankle dorsiflexion strength of MRC 4 or greater. 20 Giuffre et al, Leclère et al, and Ferris et al reported on partial TN transfers directly to the TA.2, 21, 23 Giuffre et al saw good ankle dorsiflexion recovery in only 1 of 11 patients, Leclère reported good outcomes in 3 of 6 patients, and Ferris et al reported ankle dorsiflexion strength of MRC 4 or greater in 7 of 9 patients.2, 21, 23 In this study, TN fascicles to FDL and FHL were utilized as donors in all 9 patients. Donor fascicles from FHL and FDL are advantageous as toe flexion is synergistic to ankle dorsiflexion. This is in comparison to other posterior compartment donor options such as nerves to the gastrocnemius or soleus muscles, which are antagonistic to dorsiflexion.
Factors for recipient nerve selection included mechanism of injury, variability in anatomy, branch size matches, and length required to ensure tensionless transfers. While not statistically significant, patients in the good outcome group received at least 1 nerve transfer to the TA nerve branch more often than the poor outcome group (75% versus 20%) (P = .099). Coapting directly to the motor branch to TA decreases the distance to the motor end plates of the most powerful dorsiflexor muscle. 24 More proximal coaptation to DPN increases the distance required for axonal regrowth and distributes the regrowth across TA as well as other anterior compartment musculature that provides less powerful dorsiflexion. 24
Clinical Outcomes
Our group previously performed a systematic review including a total of 41 patients who underwent nerve transfer for foot drop. 24 None of the patients included in the review were from our group. The review demonstrated variety in donor nerve and recipient nerve selection, and inconsistency in overall clinical outcomes. Of the 41 patients included, the average postoperative dorsiflexion strength was MRC 2.1. The outcomes were clustered in 2 groups at the upper and lower limits of the MRC scale, revealing that there are responders and nonresponders after nerve transfer for peroneal nerve palsy. The current study similarly determined a postoperative ankle dorsiflexion mean MRC of 2.1, with bimodal distribution of postoperative outcomes (Figure 1). Four patients achieved a good outcome (MRC 4) whereas 5 patients did not (MRC 0 or 1).
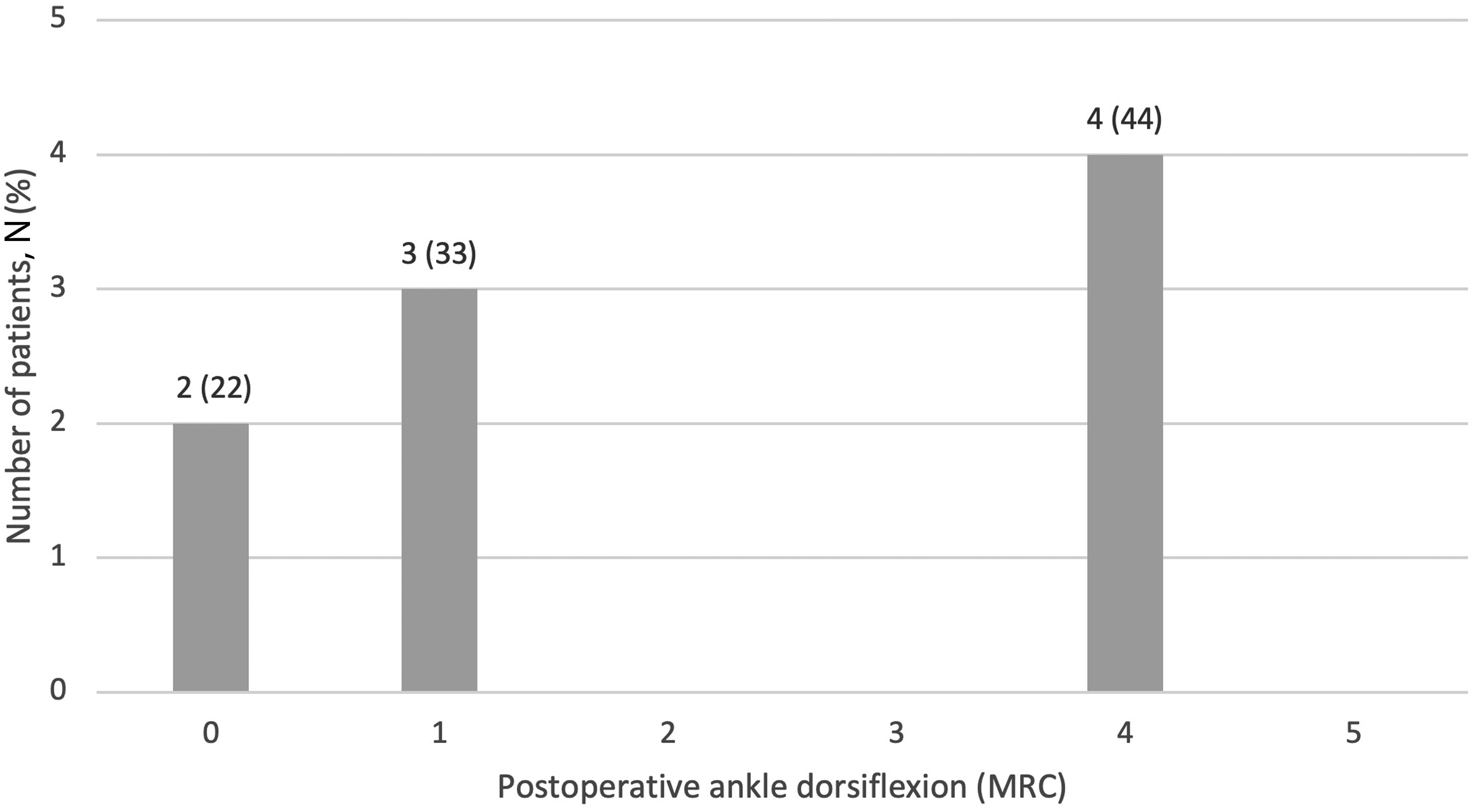
Distribution of postoperative ankle dorsiflexion of all patients (N = 9).
Prognostic Factors
No specific positive or negative prognostic factors have been established in LENT. Giuffre et al hypothesized that low energy mechanisms leading to isolated peroneal nerve injuries, younger age, and earlier time to surgery may be positive prognostic factors. 2 There are fundamental differences in high energy traction-avulsion and crush injuries to the peroneal nerve versus low energy insults of iatrogenic or oncologic mechanisms. Giuffre et al previously postulated that high energy injury to the common or DPN may cause a zone of injury affecting the nerve proper and motor end plates. 25 Based on histologic examination Prasad et al hypothesized high energy traction injuries extend to the myoneural junction, creating scar that cannot be penetrated at the level of the end organ. 26 This may explain why Giuffre et al had poorer outcomes when examining high-energy, traumatic nerve injuries at the level of the knee, compared to other studies with mixed mechanism of injuries. The mechanisms of injury in this study were varied, including traumatic, iatrogenic, and oncogenic occurring at the level of the knee or hip. The prognostic value of mechanism of injury is still unclear at this stage.
SETS coaptation was selected in patients when there was potential for native peroneal nerve reinnervation. When preoperative assessment demonstrated dorsiflexion strength of MRC 0, but EMG studies displayed few present motor units, SETS coaptation was utilized due to some evidence of axonal continuity. This was seen in patients with high sciatic nerve injury with minimal axonal continuity of peroneal nerve present at initial assessment, as evidenced by only 1 or 2 mature motor units. The morphology of present motor units reflected residual units from a near complete axonal injury, not recent reinnervation. Little native recovery was anticipated given both lack of axons to contribute to collateral sprouting, and substantial distance regenerating axons must travel to the target motor endplate before permanent muscle atrophy and fibrosis occur.
It is difficult to verify if patients with good outcomes achieved MRC 4 or greater because of nerve transfer, nerve decompression, or native reinnervation in the setting of SETS coaptation. In their retrospective study of 20 patients with CPN palsy from various mechanisms, Thoma et al described improvement of dorsiflexion from peroneal nerve decompression alone. 27 The authors reported an improvement from MRC grade 0 to MRC grade 3-5 in 10 patients. Postoperative electrodiagnostic examinations involved evaluating motor units in TA (recipient) while patients were asked to curl only their toes, thus engaging FHL and/or FDL (donor). The presence of motor units in TA when one is engaging the donor nerve may be suggestive of innervation by nerve transfer.
When assessing patient factors, only a shorter time to surgery was significantly associated with better outcomes. This is not surprising as it is well accepted that denervation atrophy is time dependent. 23 While not significant, patients in the good outcomes group tended to be younger, had lower BMIs, and had shorter time to surgery. Perhaps some of these factors are important prognostically but were not statistically significant given the limited power of this small cohort.
Limitations
Limitations in this study include a small cohort size, the heterogeneity of the patient population, and follow-up duration. The small cohort size and heterogeneity limited the power of statistical tests in identifying prognostic factors. That said, the outcomes demonstrated in this work are consistent with systematic review findings in both the mean postoperative MRC and bimodal distribution. Therefore, we feel that our cohort is appropriately representative of this patient population. The follow-up electrodiagnostic studies were initiated at 5 months postoperatively and the overall follow-duration was short in some cases with a range of 9.1 to 28.2 months. It is possible that patients had signs of recovery prior to the first postoperative assessment. In those with follow-ups less than 12 months, patients may have further evolution of clinical and electrodiagnostic reinnervation not captured in the data. This may underestimate the extent to which patients recovered.
Conclusion
This series of 9 consecutive patients by a single surgeon adds to the growing body of literature examining the merit of TN transfers in the management of peroneal nerve palsy. The study demonstrates a significant increase in postoperative dorsiflexion strength with TN transfers. Our results reinforce the findings of the systematic review previously performed by our center demonstrating that there are responders and nonresponders to LENT. While lesser time to surgery appeared to predispose patients to a successful nerve transfer other patient and surgical factors that prognosticate successful restoration of ankle dorsiflexion strength remain elusive.
Footnotes
Author Contributions
Julia B. Lichtenstein: Preparation of ethics application, data extraction, data analysis, manuscript preparation, presentation preparation, manuscript submission. Linden K. Head: Preparation of ethics application, data extraction, data analysis, manuscript preparation and editing, presentation editing. Gerald Wolff: Ethics application editing, manuscript editing, presentation editing. Kirsty Boyd: Principal investigator, overseeing of research project, ethics application editing, manuscript editing, presentation editing.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Statements
This article does not contain any studies with human or animal subjects.
Institutional Review Board (IRB) Approval
This study was approved by the Ottawa Health Science Network Research Ethics Board (Identification Code: 20180884-01H). All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2013.
