Abstract
Introduction
Fat grafting is widely used to address volume deficiencies and asymmetry in breast augmentation and reconstruction.1,2 Techniques have evolved from simply suctioning and random injection of fat into more advanced procedures that use modern fat preparation techniques, injection of small aliquots, and injection in the correct planes to increase fat graft survival.3,4 More recently, stem cells and growth factors have been used to increase the yield.5,6
Often, large-volume fat grafting is required to address the patient's needs sufficiently. During the early years of large-volume fat grafting, grafting of too much fat, without proper preparation, has been shown to reduce graft survival rates, caused by increased interstitial fluid pressures.7,8 In addition to optimal fat preparation and proper injection techniques, sufficient recipient site preparation is crucial for successful large-volume fat grafting.4,9
Recipient site preparation through pre-expansion promotes an increase in parenchymal space and vascularity. 10 Pre-expansion can be achieved either internally or externally. External mechanical negative pressure induces the generation of 3-dimensional (3D) vascularized scaffolds that are well suited to fat grafting. 11 Based on this concept, an external volume expansion (EVE) device (eg, Brava system, Brava, LLC) can be applied to prepare the recipient site for large-volume fat grafting during breast augmentation and reconstruction. Previous studies have demonstrated its efficacy in increasing overall breast volume and graft survival.12,13
However, conventional EVE devices have limitations, such as requiring a long application duration and complications, including sweating, itching, and rash, which lead to poor patient compliance. 14 Previous studies have demonstrated significant dropout rates related to the long recommended wear-time (11 hours a day) and skin-associated side effects. Reducing wear-time can improve patient compliance by improving daily quality of life and decreasing the side effects. Investigations regarding the amount of expansion according to time and pressure settings are scarce.
This study aimed to compare the efficacy of a newly proposed protocol, cyclic high negative-pressure (CHNP) mode (CHNP EVE, 1 h/day at −55 mm Hg, cycling between a 1-second negative-pressure and a 2-second cooling period) with that of the traditional continuous, moderate-negative-pressure mode (conventional EVE, 8 h/day at −25 mm Hg). If a similar preparation can be achieved with CHNP, the patient can wear the device for 1 hour only before bedtime, which would improve patient compliance.
Materials and Methods
Device
A miniaturized EVE device was made from a 30 mL syringe (Kovax-Syringe®, Korea vaccine) cut into a dome-shaped container, with a base and height of 2.5 cm each. The device was applied to the animal's dorsum after shaving 1 cm from the midline, to avoid pressure on the spine. The device was sealed using Tegaderm® (3M Health Care) and fixed using a Peha-haft bandage (Peha-haft®; Hartmann). Negative pressure was applied using an external suction device (VAC Instill; KCI) (Figure 1).
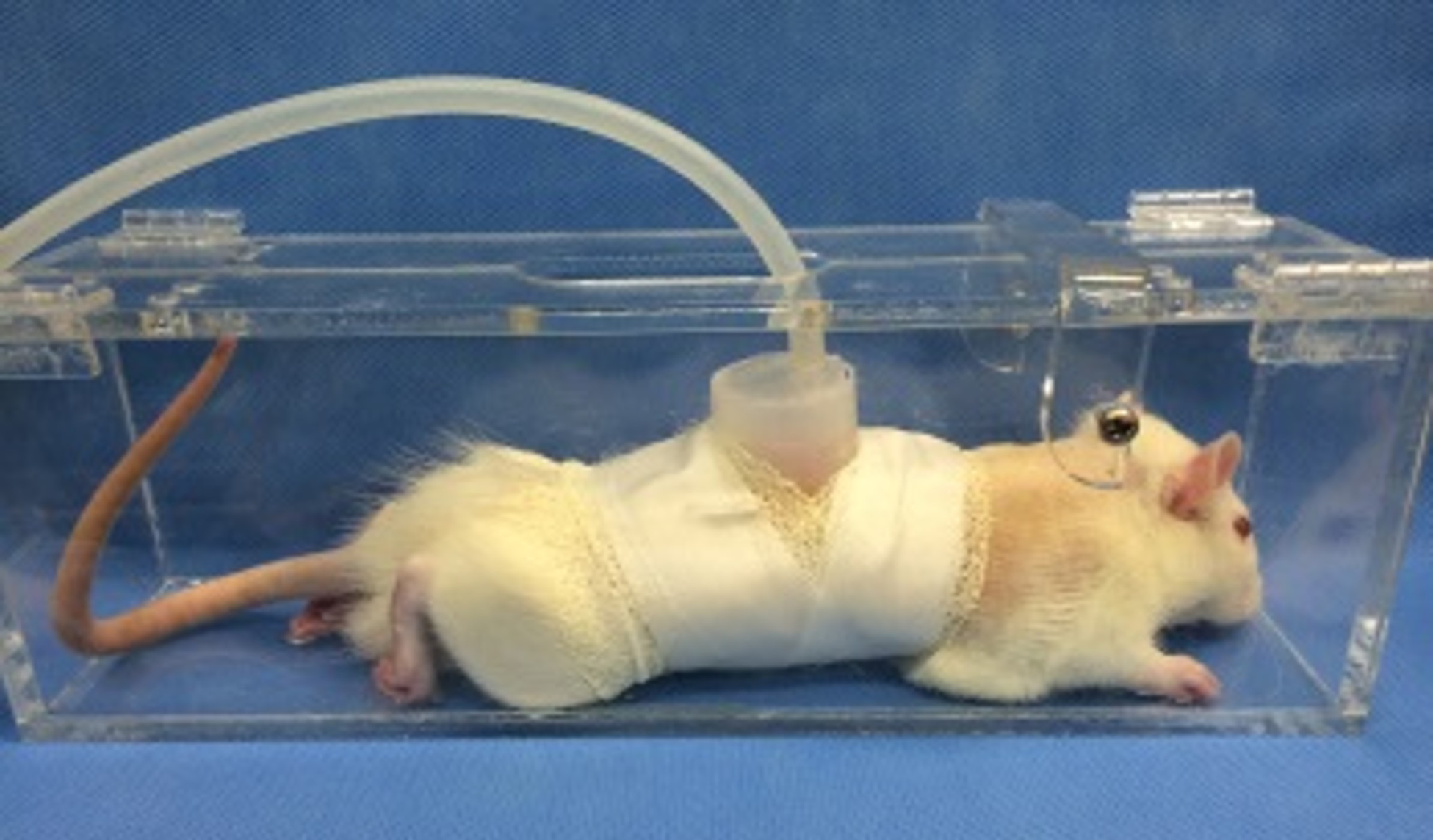
An external volume expansion (EVE) model (a modified 30 mL syringe connected to an external suction device, VAC Instill) applied to the dorsum of a Sprague-Dawley rat and fixed using a Peha-haft compressive bandage.
Animal Model
All animal procedures were conducted strictly according to the guidelines and policy of the National Research Council with approval from the Institutional Animal Care and Use Committee of Seoul National University Bundang Hospital (Approval No. BA1103-079/017-01). Eight-week-old male Sprague-Dawley rats were maintained under light and dark cycles for 12 hours each, in 60% humidity and 22 °C‒24 °C temperature-controlled environments. The rats were allowed access to water and food ad libitum.
The rats were randomly assigned to 3 groups of 6 rats each: the EVE device only, with no pressure settings (control group), the EVE device set at a pressure of −25 mm Hg for 8 h/day (conventional EVE group), and the EVE device set at a pressure of −55 mm Hg for 1 h/day, cycling between a 1-second negative-pressure and a 2-second cooling period (CHNP EVE group). Rats in all 3 groups were placed in a container (Figure 1) for the same amount of time, 8 h/day, then placed in a cage for the remainder of the day. The conventional EVE group received the negative pressure for the entire 8 hours, while the CHNP group received the negative pressure for the first hour only, while no pressure was applied to the control group. The device was applied to the dorsal skin of the mice for 28 days, and all animals were euthanized by CO2 inhalation on day 28.
Full-thickness tissue samples (epidermis to deep fascia) were harvested by a single investigator, fixed with paraformaldehyde (10%) in phosphate-buffered saline (pH 7.2) for 24 hours, and paraffin-embedded. The block was cut into 4 µm thick sections for histological examination.
Expansion Volume Analysis Using Micro-Computed Tomography
On day 28, immediately before euthanization, while the rats were anesthetized using isoflurane (2%-5%) by mask inhalation, expansion volume was analyzed by micro-computed tomography (CT) using a Scanco VivaCT 75 microCT scanner (Scanco Medical). The effective voxel size of the reconstructed images was 40. A global threshold (170) was applied to remove the soft tissue background for 3D image reconstruction. The expanded volume was calculated using the picture archiving and communication system by measuring the tissue above an imaginary plane formed by the base of the apparatus, as shown in Figure 2 (the red line).
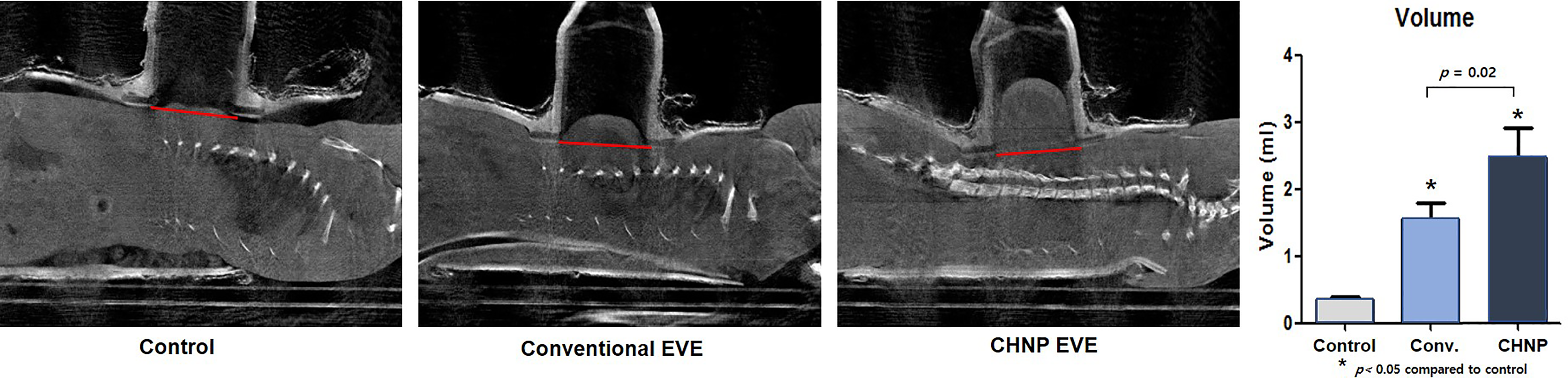
(Left) MicroCT of the rats on day 28 of expansion. The expanded volume was calculated by measuring the tissue above an imaginary plane formed by the base of the apparatus (the red line). (Right) The average calculated volumes were 0.35 ± 0.12, 1.55 ± 0.59, and 2.31 ± 1.20 ml for the control group, conventional EVE group, and the CHNP group, respectively.
Histological Evaluation
Tissue sections of the treated area were stained with hematoxylin and eosin (H&E) and Masson's trichrome (MT) staining. Bright-field images were acquired using a Nikon E200 microscope (Nikon Corp.). Two blinded, independent raters evaluated the prepared slides. Using Image J software (National Institutes of Health), subcutaneous tissue thickness (epidermis to the hypodermis) was measured at 3 random points per sample, and collagen density was calculated by quantification of the blue component at 5× magnification field for each sample.
Immunohistochemistry
For analysis of cell proliferation and angiogenesis, paraffin sections were stained for proliferating cell nuclear antigen (PCNA, 1:200 dilution, Thermo Scientific) and platelet endothelial cell adhesion molecule 1 (PECAM-1, 1:100 dilution, BD Pharmingen) for 1 hour at room temperature. Sections were incubated in the ready-to-use Vecstain ABC complex (Vector Laboratories), followed by diaminobenzidine (Dako North America Inc.). The number of PCNA + adipocytes or new capillaries was counted in 3 random 40× field images by 2 blinded, independent raters.
Statistical Analysis
All values are expressed as the mean ± standard deviation in the figures and text. One-way analysis of variance (IBM SPSS Version 21; IBM Corp.) with Scheffe's posthoc comparisons were used to determine the statistical significance of differences. Differences were considered statistically significant at p < .05.
Results
Volume Calculation
MicroCT imaging was performed to study tissue swelling at the end of the expansion cycle on day 28. In the conventional EVE group and CHNP EVE group, macroscopic local swelling of the soft tissue was observed (Figure 2). The average volumes for the control, conventional EVE, and CHNP EVE groups were 0.35 ± 0.12, 1.55 ± 0.59, and 2.31 ± 1.20 ml, respectively (Figure 2). EVE groups showed an increase in volume by 4.4 ± 1.7- and 6.6 ± 3.1-fold, respectively, as compared to the control group. Furthermore, the average volume in the CHNP EVE group (2.31 ± 1.20 ml) was significantly higher than that in the conventional EVE group (1.55 ± 0.59 ml, p < .05).
H&E stained sections were analyzed for subcutaneous thickness at 5× magnification (Figure 3). The average thickness of subcutaneous tissue was 1478.3 ± 76.3, 2499.6 ± 206.7, and 2418.2 ± 210.1 μm, for the control, conventional EVE, and CHNP EVE groups, respectively (Figure 3). The difference in subcutaneous tissue thickness between the control and EVE groups was statistically significant. However, there was no statistically significant difference between the 2 EVE groups.
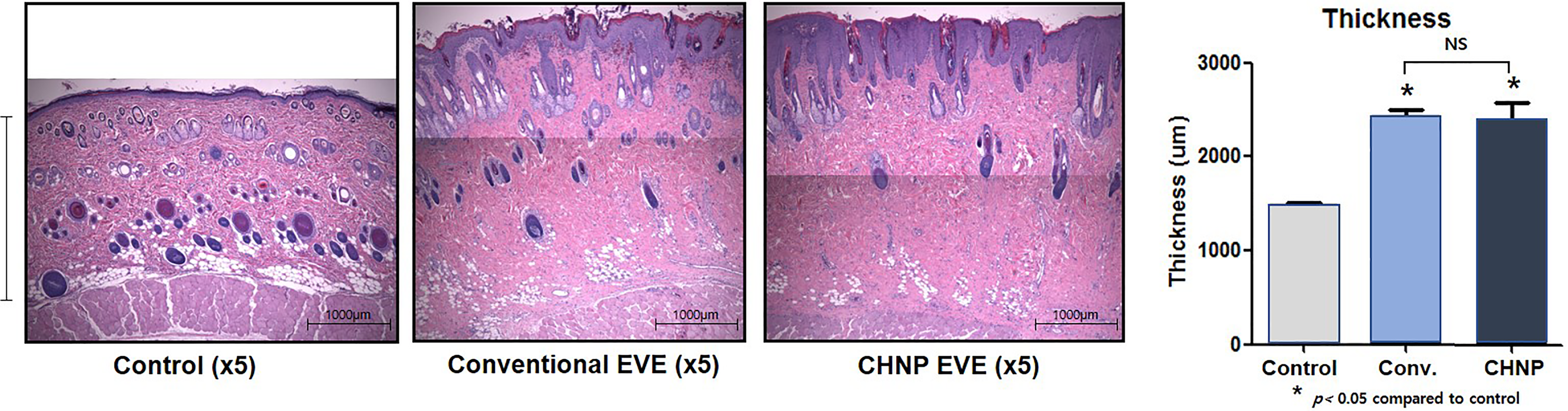
(Left) H&E staining. Subcutaneous tissue thickness (epidermis to the hypodermis; an example shown as the vertical bar on the far left) was measured at 3 random points per sample, analyzed under 5× magnification. (Right) The average thicknesses of subcutaneous tissue were 1478.23 ± 76.3, 2499.6 ± 206.7, and 2418.2 ± 210.1 μm, for the control, conventional EVE, and CHNP EVE groups, respectively. Both EVE groups had significantly thicker subcutaneous tissue than the control group, but there was no statistical difference between the 2 groups.
Collagen Density and the Number of Adipocytes
The level of collagen deposition was analyzed by MT staining. Compared to the control group, the levels of collagen deposition were significantly higher in both EVE groups (Figure 4). There was no statistically significant difference in collagen density between the 2 groups.
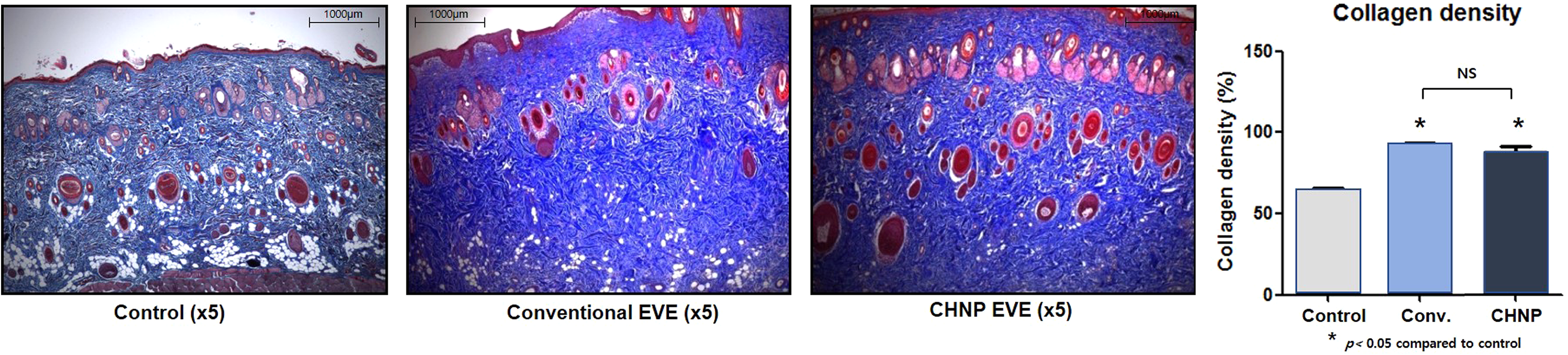
(Left) MT staining. (Right) Collagen deposition was significantly higher for both EVE groups.
PCNA immunohistochemistry showed an increased density of PCNA + subdermal adipocytes in Groups I and II. The average number was measured by counting PCNA + adipocytes at 3 random 40× fields. The average number of PCNA + adipocytes was 11.3 ± 0.9, 22.2 ± 5.2, and 18.7 ± 3.0 for the control, conventional EVE, and CHNP EVE groups, respectively. Compared to the control group, the adipocyte proliferation rate was 1.9 and 1.7 times greater in the EVE groups (Figure 5, p < .05). There was no statistically significant difference in the proliferation rate of adipocytes between the 2 EVE groups (Figure 5).
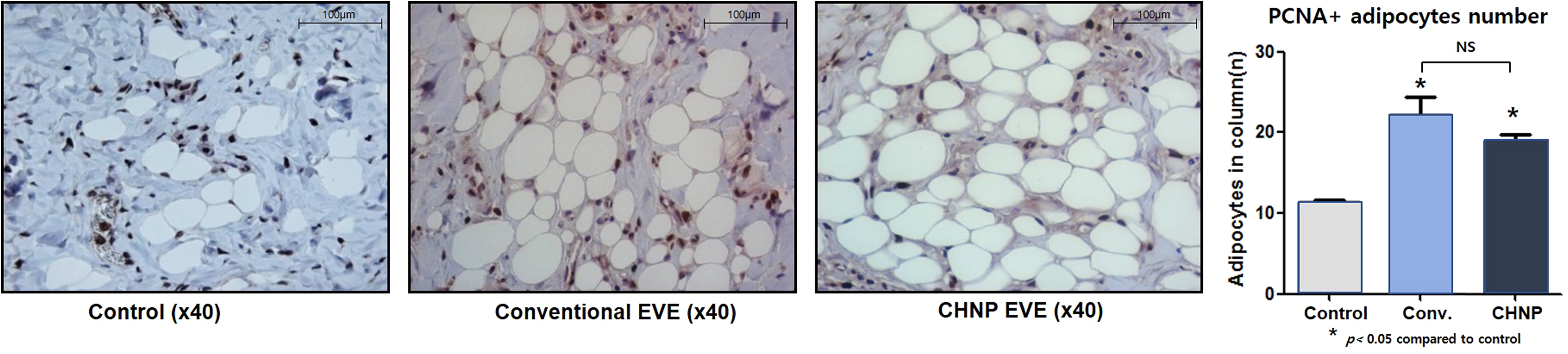
(Left) PCNA immunohistochemistry. Increased density of subdermal PCNA + adipocytes is evident. (Right) The average number of PCNA + adipocytes was 11.3 ± 0.9, 22.2 ± 5.2, and 18.7 ± 3.0 for the control, the conventional EVE, and the CHNP EVE groups, respectively, which reflect adipocyte proliferation rates 1.9 and 1.7 times greater for the EVE groups compared to the control (p < .05).
Angiogenesis and Vascular Remodeling
Immunohistochemical staining for PECAM-1 was performed to quantify angiogenesis. EVE groups showed 2.5 and 2.7 times higher vessel density than the control group (Figure 6, p < .05). However, there was no statistically significant difference in PECAM-1 + blood vessel density between the 2 EVE groups (Figure 6). Furthermore, the EVE groups showed an increase in vascular networks and thickened endothelial walls of blood vessels.
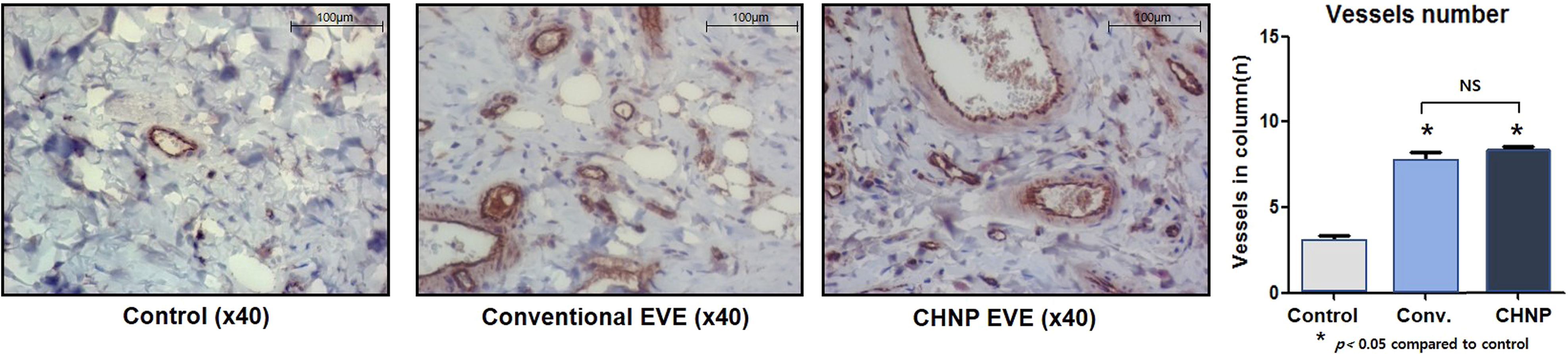
(Left) PECAM-1 immunohistochemistry. Increased vascular networks and thickened endothelial walls of the vessels. (Right). Compared to the control group, the conventional EVE and the CHNP Eve groups had 2.5 and 2.7 times higher vessel density (p < .05).
Discussion
Autologous fat grafting has become an indispensable tool for both esthetic and reconstruction surgeons. Initially, the fat graft was used for a small-volume transfer, to alleviate small volume deficiencies, but recently, a large-volume transfer has become successful due to advances in the field. Small-volume fat grafting is predictable, but large-volume fat grafting is less so, with a high variation in fat graft survival. 15
Proper recipient site preparation is crucial for increasing the survival of adipocytes in large-volume grafts. In his review article, Nahabedian emphasized 4 important principles of recipient site preparation for large-volume fat grafting: the graft-to-capacity ratio, ideal plane, stromal vascular fraction (SVF), and external soft tissue expansion. 16 The interstitial fluid pressure should not be too high after injection, to increase the graft-to-capacity ratio. 4 Furthermore, the injection must be performed in the ideal plane to allow the graft to be in close proximity to the host tissue to survive through plasmatic imbibition until adequate neovascularization has taken place. SVF has been shown to increase fat survival after grafting. 17
External soft tissue expansion and the graft-to-capacity ratio are intertwined because the main purpose of an EVE is to increase the graft-to-capacity ratio. External expansion increases the volume of the subcutaneous tissue while increasing the interstitial volume and promoting angiogenesis, 4 providing a scaffold for large-volume fat grafts with an increased likelihood of fat survival.
Previous reports have shown the effectiveness of preoperative use of EVE (eg, the BRAVA system) with the conventional protocol of 10 h/day for 4 weeks, at −15 to −25 mm Hg pressure. However, some studies have also reported complications associated with a long wear-time, which caused poor patient compliance. Wearing the device for 10 hours can interfere with the patient's daily activity and may even cause insomnia in some patients. 18 The device can cause skin irritation, erythema, and bullae formation. Some reports have reported skin irritation, in as many as 57.8% of patients. 19
If the wear-time can be reduced while maintaining similar efficacy compared to the traditional protocol, patient compliance and effectiveness can be improved. Several animal model studies have been conducted to study different settings for EVE. Giatsidis et al 20 tested intermittent stimulation in the hope of reducing the overall wear-time. Their study showed that moderate-intensity intermittent stimulation (0.5 hour per session, 6 times/day for 5 days at −25 mm Hg) had better angiogenesis compared to high-intensity intermittent stimulation (same setting as moderate intensity but for 1.5 hours per session for 5 times/day). 20 The shorter session time of the moderate-intensity group allowed the tissue to recover after subcritical ischemia.
Our study design aimed to reduce the total wear-time by increasing the negative pressure and utilizing cyclic pressure. In our preliminary study using 8-week-old male Sprague-Dawley rats, we applied different negative pressures ranging from −25 to −80 mm Hg in increments of 5 mm Hg to test for pressure tolerance. At pressures lower than −60 mm Hg, the rats were agitated and struggled to remove the device. In addition, EVE at −55 mm Hg for 8 hours did not cause severe erythema or skin necrosis in the rats. Therefore, we designed our experiment at −55 a cyclic pressure as the experimental group.
To evaluate the effect of CHNP, the CHNP EVE group (−55 mm Hg for 1 h/day) was compared to the conventional EVE group (−25 mm Hg for 8 h/day) and the control group (the device only). On day 28, EVE groups had higher volumes than the control group. Histological evaluation demonstrated a similar increase in the thickness of the subcutaneous tissue between the 2 EVE groups. PCNA staining also showed a similar increase in subdermal adipocyte density.
Previous animal studies have demonstrated that the increase in the volume and thickness of the subcutaneous tissue is due to transient tissue edema.12,21 One benefit of using EVE before fat grafting is to reduce the compression on the graft and reduce the interstitial fluid pressure to improve graft survival. 22 Therefore, large-volume fat grafting should be performed immediately after the completion of EVE treatment.
Recent studies have emphasized the importance of vascular bed formation through angiogenesis to increase graft survival.22,23 Immunohistochemistry against PECAM-1 demonstrated significantly higher vessel density in EVE groups, as well as thickened endothelial walls. Conventional and CHNP group differences were not statistically significant, showing a similar effect on angiogenesis in the recipient bed. Similar to previous studies based on murine models, EVE groups demonstrated an increase in the density of subdermal adipocytes in PCNA immunohistochemistry. The proliferation rate of adipocytes was 1.9-fold greater in the conventional EVE group than in the control group. However, this increase was not statistically significant as compared to that in the CHNP EVE group.
Our findings demonstrated that in murine models, CHNP can achieve similar results in angiogenesis, subcutaneous tissue thickening, and adipocyte proliferation as compared to the conventional setting, while markedly reducing the daily device wear-time from 8 hours to 1 hour. The comparable efficacy noted in the experimental group arises from 2 main factors: higher pressure and the cyclic mode. A higher negative pressure causes ischemia and hypoxia, thereby promoting angiogenesis. The high negative pressure causes direct occlusion of the vessels and an increase in the distance between blood vessels and cells, creating hypoxic conditions. A cyclical or intermittent application of negative pressure causes more angiogenesis than a static application of a device in skin expansion and wound-healing models.24–26 However, an intermittent application can cause discomfort to the patient in a clinical setting, decreasing patient compliance.24,27
This study had several limitations. First, murine models differ significantly from humans. Further clinical studies are needed to validate the efficacy of CHNP settings in patients with fat grafts to find the optimal treatment setting. In addition, while the rats did not exhibit short-term complications caused by high negative pressure, patients may experience more discomfort and higher rates of pressure-associated complications, such as erythema and skin discoloration. Second, long-term follow-up data on complications, angiogenesis, and subcutaneous thickening were not available, as the rats were sacrificed and biopsied on the last day of treatment. Despite these limitations, the findings of this study can guide us in finding the optimal setting for EVE, to maximize fat graft survival while reducing patient discomfort and associated complications.
Conclusion
CHNP produced comparable results in recipient site preparation (as demonstrated by subcutaneous tissue thickening and angiogenesis) as the conventional protocol, while markedly reducing the daily wear-time from 8 hours to 1 hour. Although further clinical data must be acquired, our new setting seems promising and provides a much more patient-friendly pre-expansion environment.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Seoul National University (grant number 1425088861).
