Abstract
Introduction
Over the last several decades, paediatric craniofacial centers around the world have reported a dramatic increase in the incidence of children born with metopic craniosynostosis, making it the second most common type of craniosynostosis.1–7
Several epidemiologic studies have suggested that the increase in metopic craniosynostosis cannot be attributed solely to improved diagnosis and awareness of the condition.1,3,7 Some have speculated that the increased incidence and spectrum of severity observed in metopic craniosynostosis may be due to a dose-dependent in utero pharmacological exposure. Population-based studies have suggested that maternal use of thyroid replacement hormone, valproic acid, or folic acid may be associated with metopic craniosynostosis.6,8–10 Unfortunately, rates of use of these agents among the child-bearing age population have remained largely unchanged during the period over which the dramatic rise in metopic craniosynostosis has been observed.
We examined the association between an agent whose increased use has corresponded with the rise in metopic craniosynostosis cases, the fertility drug clomiphene citrate. According to data from the 2002 National Survey of Family Growth, 3.8% of women indicated they used the ovulation drug, a 27% increase from a similar survey conducted in 1995.11,12 The drug is comprised of a 60:40 mix of trans- (enclomiphene) and cis-isomers (zuclomiphene), with the latter having a greater affinity for estrogen receptors. Previous population-based studies have identified an association between craniosynostosis and maternal clomiphene citrate use.13,14 Neither of these studies examined the impact of clomiphene citrate on specific type of craniosynostosis. The fact that clomiphene citrate is taken before conception has been cited as evidence against biologic plausibility; however, several investigators have found that zuclomiphene, the more active isomer in clomiphene citrate preparations, accumulates over consecutive cycles and can be detected in plasma months after administration.15,16 Additionally, female mice injected with clomiphene citrate before ovulation in doses paralleling those in humans had an increased risk of exencephaly. 17
In the current study, we investigated whether a causal link exists between clomiphene citrate or enclomiphene hydrochloride administration and metopic craniosynostosis using a zebrafish model. Zebrafish possess unique advantages over mammalian models, including a short reproductive cycle, large number of progeny, high degree of genetic and developmental conservation with humans, and transparent embryos which permits direct visualization of craniofacial morphogenesis.18,19 Interestingly, however, the zebrafish differs from humans and murine models in that the interfrontal suture, the equivalent of the human metopic suture, remains patent throughout the lifespan of the animal.20,21 We hypothesized that exposure of fertilized zebrafish embryos and larvae to clomiphene citrate and/or enclomiphene hydrochloride will induce fusion of the interfrontal suture akin to metopic craniosynostosis in humans.
Materials and Methods
Animal Model and Maintenance
Wild-type progeny of AB zebrafish (Danio rerio) stocks originally from the Zebrafish International Research Center (ZIRC) were used for all experiments. Animals were raised according to national animal care guidelines and approval for these experiments was received from the Mount Saint Vincent University-Saint Mary's University Joint Animal Care Committee (protocol number 16-03). Larvae were raised to 5.5 mm or 6.5 mm standard length depending on the experimental group. Standard length (SL), defined as the distance from the rostral tip to the caudal peduncle (ie the base of the tail fin), was measured to the nearest decimal with the Nikon NIS Elements image analysis software. These specific SLs were selected as they are points critical to suture development in the zebrafish. 21
Drug Treatment
Once the specified SL was reached, zebrafish larvae were then treated with either 100 nM clomiphene citrate (CC; Sigma-Aldrich, St. Louis, MO; n = 20) or 60 nM enclomiphene hydrochloride (EH; Sigma-Aldrich; n = 20) dissolved in 1% dimethyl sulfoxide (DMSO). The doses of each drug used correspond to human serum levels15,16 and have been shown to be tolerated by zebrafish. 22 Maes et al. have previously shown that low concentrations of DMSO is well-tolerated by zebrafish at various ages (embryos and larvae). 23 Controls included a DMSO solvent control (n = 20) at 5.5 mm SL and a no treatment group (n = 20). The chemicals were added directly to rearing water for five days of continuous exposure and the fish were fed throughout the experimental period. Five days’ duration was selected to ensure as high a dosage as possible that did not result in complete mortality. Water was changed daily and a fresh dose of drug added to the replacement water. After treatment, fish were transferred to new rearing containers containing regular water. Fish were raised for two months to observe the long-term effects on the calvarial development. Two independent trials were conducted. The proportion of fish that survived to the endpoint in each treatment group was recorded. Fish surviving to two months were then euthanized with 0.1% MS222 Ethyl 3-aminobenzoate, methane sulfonic salt (Sigma-Aldrich). Samples were fixed overnight using 10% neutral buffered formalin.
Whole-Mount Alizarin Red Staining
A standard whole mount acid-free stain 24 was used to visualize the craniofacial skeleton in all samples. Bone was stained using Alizarin red S (Sigma-Aldrich). Briefly, the fish were stained overnight in Alizarin red staining solution. Fish were then rinsed in water and bleached in 3% hydrogen peroxide and 1% potassium hydroxide solution for 20 min. Soft tissues were digested with 1% trypsin (Thermo Fisher Scientific, Inc., Waltham, MA) and 2% sodium tetraborate (Sigma-Aldrich) in distilled water for three nights. All specimens were processed through an ascending series of glycerol in 1% potassium hydroxide solution then transferred to a storage solution of 100% glycerol.
Imaging
All samples were examined either using a Nikon SMZ1000 stereomicroscope or a Nikon Eclipse 50i compound microscope. Samples were photographed using a Nikon DXM120 °C digital camera in all cases. Fish with insufficient staining were omitted from subsequent skull suture morphology assessment performed by an observer blinded to experimental group.
Statistical Analysis
Survival and body length, expressed as mean ± standard error of mean, was compared between groups using a one-way ANOVA with Tukey's multiple comparison test. Proportion of fish surviving or exhibiting cranial abnormalities in each group were compared using a Fisher's exact test and Bonferroni correction for pairwise comparisons. Significance was set at p < .05. Statistical analyses were performed using Prism version 9.2.0 (GraphPad Software, San Diego, CA).
Results
Survival
While fish treated at 6.5 mm SL tended to have poorer survivability (25-30%) compared to fish treated at 5.5 mm SL (>50%) (Figure 1A), only EH treatment at 6.5 mm SL demonstrated a statistically significant reduction in survivability relative to untreated and DMSO controls (p < .02).
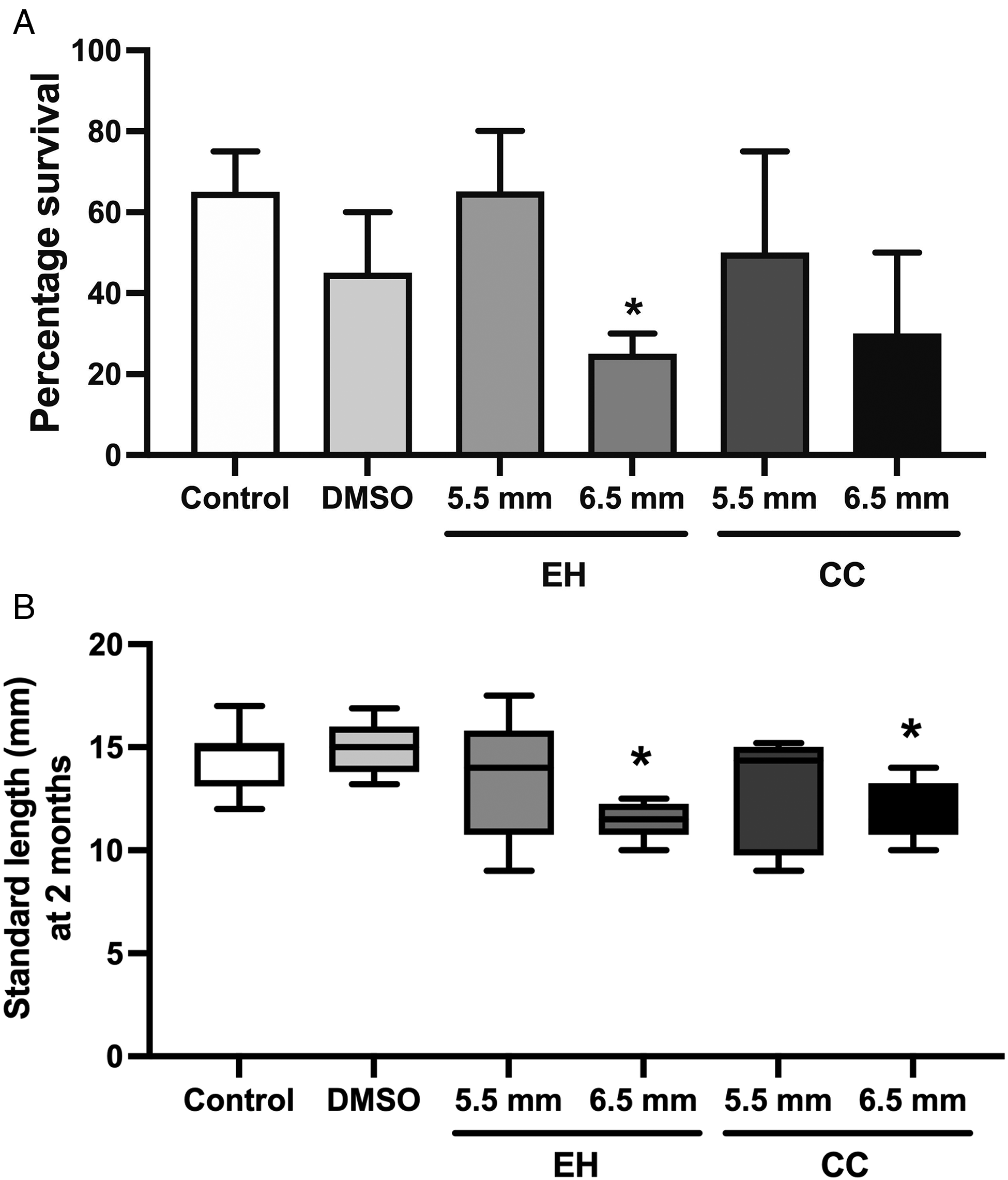
(A) Survivability of zebrafish at two months. Once animals had reached the defined standard length, treatments were initiated and continued until animals were sacrificed two months later. For the DMSO control group, animals were treated at 5.5 mm standard length. DMSO, dimethyl sulfoxide. EH, enclomiphene hydrochloride. CC, clomiphene citrate. *p = .02 compared to DMSO control, n = 20, experiment performed in duplicate (B) Body length at two months on pooled surviving fish, expressed as Tukey box plot. *p = .04 compared to DMSO control, n ≥ 5.
Growth
Differences in body length were observed after treatment. Based on normative zebrafish growth data, mean SL at 2 months is 14 mm. Among the untreated and DMSO controls, only 15% of fish were less than 14 mm SL at two months of age, whereas 60% of all EH and CC treated fish were less than 14 mm SL. Notably, significant differences in body lengths were observed for fish treated at 6.5 mm SL compared to solvent controls (Figure 1B) indicating an overall stunted growth after treatment with either agent.
Calvarial and Skeletal Development
Whole-mount bone staining the skulls revealed significant effects on calvarial and suture development. In general, the head shapes of treated fish were broader than those of controls, particularly at the level of the occipitoparietal skull. No gross ocular abnormalities were observed. All control fish demonstrated normal skull morphology (Figure 2A), with patent interfrontal sutures (Figure 2B). Of the few DMSO control fish that exhibited cranial vault abnormalities, these were characterized by overlapping sutures (Figure 2C). In contrast, fish treated with EH and CC showed fusion of the interfrontal suture (Figure 2D). Fusion of the interfrontal suture was observed in fish treated with EH and CC at 5.5 mm SL (69% and 50%, respectively; p < .01 compared to controls). Furthermore, compared to controls treatment with CC at 6.5 mm SL caused fusion of the interfrontal suture in 83% of the fish (p < .001). Owing to poor survivability, a significant difference was not observed between fish treated with EH at 6.5 mm SL and controls.
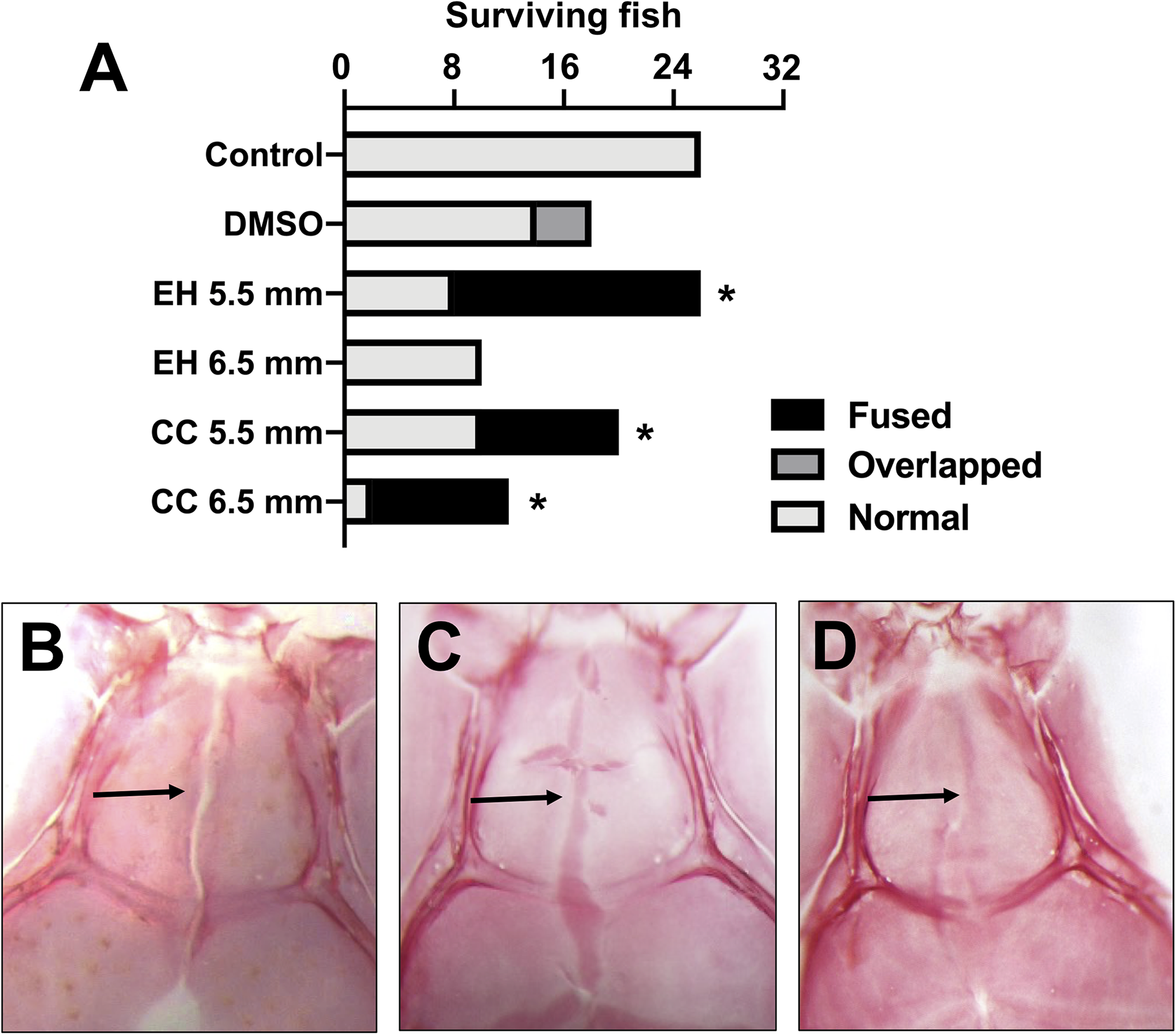
(A) Interfrontal suture morphology in pooled surviving fish. *p < .01, n ≥ 5. (B) Representative whole mount Alizarin red stain of untreated control fish indicating patent interfrontal suture (arrow). (B) Representative stain of control fish showing patent interfrontal suture. (C) Representative stain of a fish showing overlapping interfrontal suture (arrow). (D) Representative stain of a fish showing fused/absent interfrontal suture (arrow).
As calvarial abnormalities have been shown to be associated with rib anomalies and vertebral fusion, 25 we also examined the remainder of the zebrafish skeleton to assess if these contributed to the shorter body length observed in treated animals. As demonstrated in Figure 3, no such anomalies were observed.
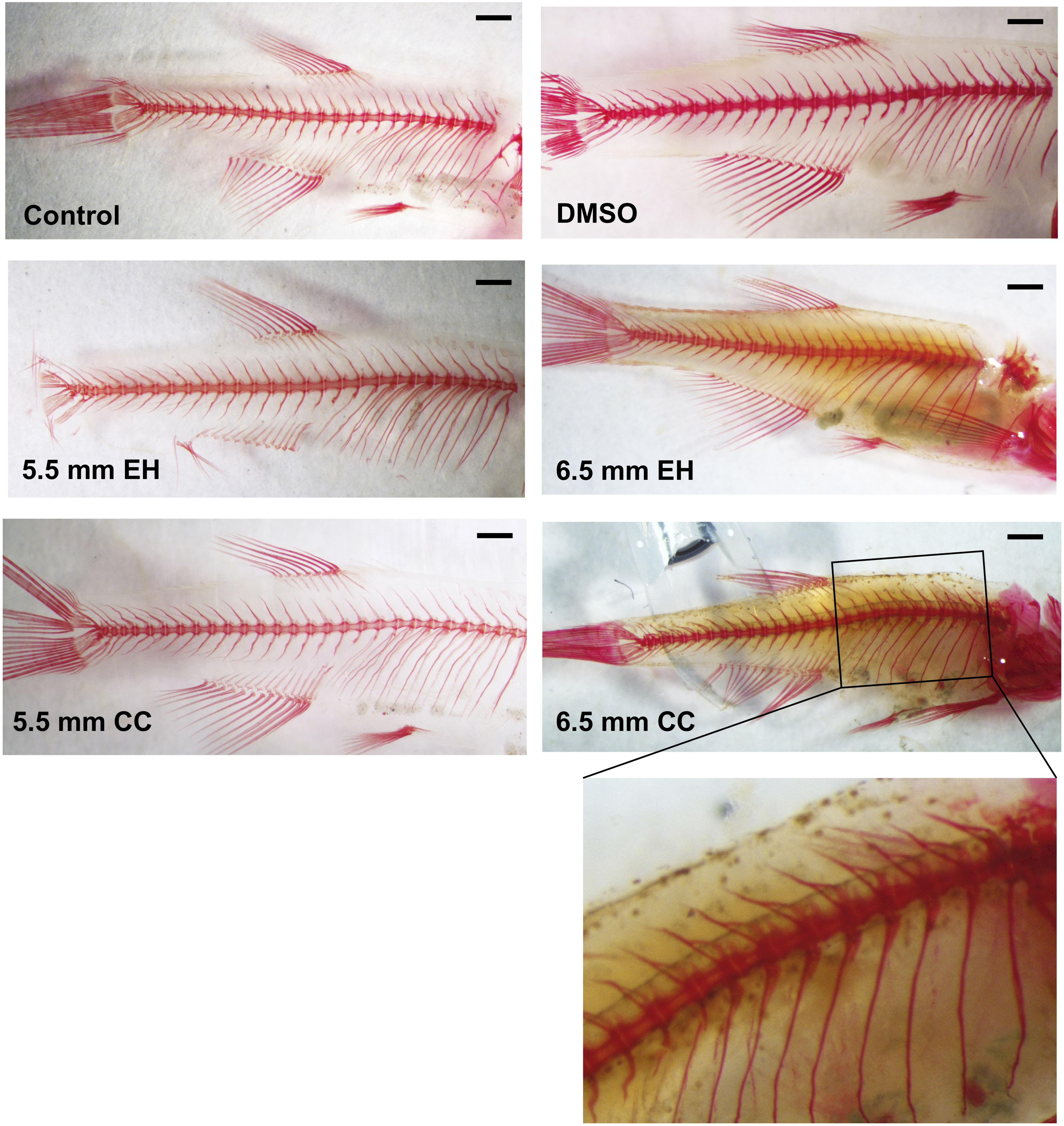
Representative Alizarin red stain of vertebral and rib morphology in pooled surviving fish from each treatment group. At increased magnification, no vertebral fusions are observed (inset). Bar = 1 mm.
Discussion
Our results provide direct evidence, for the first time, that the active compounds in common fertility drugs, clomiphene citrate and enclomiphene hydrochloride, can affect calvarial development, specifically at the level of the interfrontal suture. Clomiphene citrate is a non-steroidal ovulation-inducing drug that selectively binds estrogen receptors. Although its fertility activity is due principally to its inhibition of negative feedback of gonadotropin release from the hypothalamic-pituitary-gonadal axis, it also has direct effects on the uterus; its effects on other tissues, including the craniofacial skeleton, have not been investigated. 26 Our data suggest that CC may have a greater effect on calvarial development than EH. This may be due to the greater biological activity of the zuclomiphene isomer in CC. Another possibility is that those animals treated with EH at 6.5 mm SL developed more severe calvarial abnormalities and died prior to two months. Further research is needed to explore this effect in depth.
Interestingly, we observed that the timing of drug administration had a significant impact on survivability, with later exposure more likely to be lethal to the developing larvae. This may be due to effects on other organ systems at later critical stages. For example, between 6.2 to 6.6 mm SL, the fin rays and swimbladder (an organ critical to controlling buoyancy) are developing. 27 We speculate that disruption of these critical structures by CC and EH could have led to poor feeding, growth, and, ultimately, survival. Necropsies and additional dose-response experiments should shine further light on the mechanisms at play.
In addition to effects on survivability, both CC and EH had impacts on body growth. This is in keeping with previous human and mouse studies demonstrating decreased fetal growth with CC exposure.17,28 The observed intrauterine growth restriction in animals and humans with CC exposure has been attributed to anti-estrogenic effects on the endometrium causing reduced blood flow or directly on embryogenesis. We speculate that the anti-estrogenic activity of CC has similar effects in zebrafish, as estrogen signaling has been shown to be critical to early zebrafish cardiac and brain development.29,30
The most striking observation of our study is that CC or EH exposure is associated with fusion of the interfrontal suture, a structure that typically remains patent throughout the life of the zebrafish. Aberrations in signaling pathways known to induce craniosynostosis in humans also result in suture fusion in zebrafish, rendering them excellent for modeling of craniosynostosis. 31 While previous clinical registry studies have examined an association between CC and all types of isolated craniosynostosis,13,14 our animal study suggests that the interfrontal suture is more susceptible to disruption by CC than other sutures. We speculate that this is due to signaling pathways unique to interfrontal suture formation compared to other sutures, a hypothesis supported by recent transcriptome analysis of the interfrontal suture in mice. 25 As in zebrafish, under normal circumstances the anterior interfrontal suture never fuses in mice. 32 Previous work in mice has pointed to a role of estrogen receptor signaling in posterofrontal suture fusion. James et al. showed that estrogen receptor-α is upregulated at the time of suture fusion and that application of the estrogen receptor antagonist fulvestrant can maintain suture patency in postnatal mice. 33 The apparent discrepancy between this latter observation and our own finding that an estrogen receptor antagonist induces suture fusion may be attributable to species- and stage-specific differences in estrogen activity.
While zebrafish have increasingly been used to study craniofacial development over the last 30 years owing to their sequence homology with the human genome, rapidity of embryogenesis, and transparency allowing for visualization of embryogenesis, there are limitations to this animal model.34,35 Unlike mice and humans, zebrafish cranial development occurs at an earlier stage post-fertilization. As in rodents, but unlike humans, zebrafish sutures remain patent throughout life. Finally, zebrafish bones flanking the coronal sutures derive from mesoderm, whereas in mammals coronal sutures mark the boundary between anterior neural crest bones and posterior mesoderm bones. Such species differences may limit the relevance of our findings to humans.
Despite the limitations of small sample size, this preliminary study points to novel avenues of investigation. Future work will focus on dose-response experiments and necropsies to identify the cause of early lethality noted in the 6.5 mm SL experimental group. While this preliminary study focused on whole-mount staining to specifically examine suture development, we plan to use three-dimensional microcomputed tomography to assess craniofacial morphology in greater depth. Additionally, while we tested the effect an estrogen receptor antagonist on suture patency, we did not examine estrogen receptor expression, either through immunostaining or in situ hybridization, during zebrafish cranial suture development. Finally, we will examine whether exogenous 17-β estradiol can maintain interfrontal suture patency in fish treated with CC or EH. By further characterizing the expression of estrogen receptors and ligands in the cranial skeleton of zebrafish, we hope to provide essential insights into the signaling pathways that govern the development of metopic craniosynostosis.
Footnotes
Ethics Approval
Approval for this work was received from the Mount Saint Vincent University-Saint Mary's University Joint Animal Care Committee (protocol number 16-03). Informed consent was not applicable.
Statement of Financial Interest
The authors have no financial interests or conflicts-of-interest to disclose
Availability of Data and Materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Authors' Contributions
TF-O performed the animal treatments, collected survival data, and performed histological examinations. MB developed the concept, analyzed and interpreted the data and wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nova Scotia Health Research Foundation (grant number Development Grant (#2014-15).).
