Abstract
Credible clinical research is a precondition of evidence-based surgery. If clinical research is not conducted and reported properly, such research can be unreliable, unclear, and misleading. Our journal, Plastic Surgery, aims to improve its quality and thus enhance interest, submissions, and readership. To do so, we must ensure that the articles published in our journal align with these goals. This article guides future clinical research contributors, how to design, conduct and report valuable and reliable research. Readers are informed how to choose a title and keywords that properly reflect the content of the article. The proper organization of a manuscript, and the information that goes into each section is described. Valuable tools like the EQUATOR Network Guidelines, the FINER Criteria and the PICOT Format are described for the reader. These resources help formulate a proper research question and ensure transparency in reporting. Commonly used study designs, and the research questions they answer are presented. This ensures that those engaged in research are choosing the right study design for their research. We outline the statistical information that should be presented in the Methods section and differentiate between the content that should be found in the Results and Discussion sections. As Plastic Surgery strives to publish high-quality, reliable research, it is by the standards presented in this article that we will judge all manuscripts submitted for publication.
Introduction
It is common for both readers and reviewers of clinical research to be befuddled by the message that an article is trying to convey. Oftentimes, this lack of clarity can be caused by incongruous titles (those that do not reflect the substance of the article), inclusion of wrong methods, and lack of transparent reporting. For instance, all of us have been enticed by a catchy title, only to find that the title does not align with the actual content of an article. Furthermore, many of us can sympathize with being confused by an article due to misleading reporting or interpretation of conclusions by the authors. Lastly, many might take notice of common issues within the scientific literature such as the use of an improper research design, selective outcome reporting, a lack of transparency around steps taken to mitigate bias, and lack of admission of areas of uncertainty. Readers and reviewers alike want to see new and relevant research produced using correct and reproducible scientific methods and reported accurately without embellishment or spin. In this article we provide guidelines for authors and reviewers of future articles submitted to our journal, Plastic Surgery. The overall goals are to improve the science in plastic surgery, improve the quality of our Journal and thus iteratively evolve so as to enhance interest, submissions, and readership.
Title and Keywords
The chosen title and keywords of a scientific article should accurately convey the topic of the paper. Many guidelines developed by the EQUATOR (Enhancing QUAlity and Transparency Of health Research) Network 1 require that the title of an article include the study design (eg: case control study, randomized controlled trial, systematic review). Similarly, keywords, should properly capture the content of the article; the use of MeSH 2 terms are often suggested to increase the searchability of an article. Both the title and the keywords should describe the population and/or problem being investigated (eg Breast reconstruction in North American women). The authors encourage readers to refer to some available literature how to properly draft a title.3,4
Proper Content and Order of a Manuscript
Plastic Surgery follows the AMA Manual of Style and recommends formatting articles according to the Uniform Requirements for Manuscripts Submitted to Biomedical Journals. As such, articles should be structured as: Introduction, Methods, Results and Discussion. In addition, the appropriate EQUATOR Network 1 Checklist for the chosen study design (eg: PRISMA Guideline 5 for a Systematic Review) should be used for development and reporting of the study. The appropriate Checklist should be completed (ie: identify on what page of the submitted manuscript each element can be found) and included as an Appendix. It should be stated, in the Methods section, which Checklist was utilized and that it is included. It is also the role of the reviewers to ensure that an author includes a copy of the completed appropriate checklist and that all necessary items of the checklist are addressed within the paper. A list of common study designs and their appropriate Checklists can be found in Table 1. Readers can find all available Checklists at the EQUATOR Network 1 website (www.equator-network.org).
EQUATOR Network 1 Checklists.

*Surgery specific; CONSORT, CONsolidated Standards of Reporting Trials; STROBE, The Strengthening the Reporting of Observational Studies in Epidemiology; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses; SPRIT, Standard Protocol Items: Recommendations for Interventional Trials; CARE, CAse REport; SCARE, Surgical CAse REport; PROCESS, Preferred Reporting Of CasE Series in Surgery; STROCSS, STrengthening the Reporting Of Cohort Studies in Surgery; AGREE, Appraisal of Guidelines, Research and Evaluation; CHEERS, Consolidated Health Economic Evaluation Reporting Standards.
In the Introduction, we expect the author(s) to clearly state and justify the problem they are investigating. To do so they need to introduce the subject, preferably with primary references and reviews, to outline current gaps in the literature or areas in need of further research. One should also consider the FINER Criteria as a tool to justify the relevance of a study. The FINER Criteria, developed by Hurley and colleagues 15 is briefly explained in Table 2. After justifying the reason for the investigation, one should clearly state the research question. We recommend structuring this research question using the PICOT Format, developed by Richardson and colleages 16 (Table 3) to ensure that the research question is well defined. A nebulous question often leads to uncertain conclusions and this can be considered wasteful research.
The FINER Criteria. 15
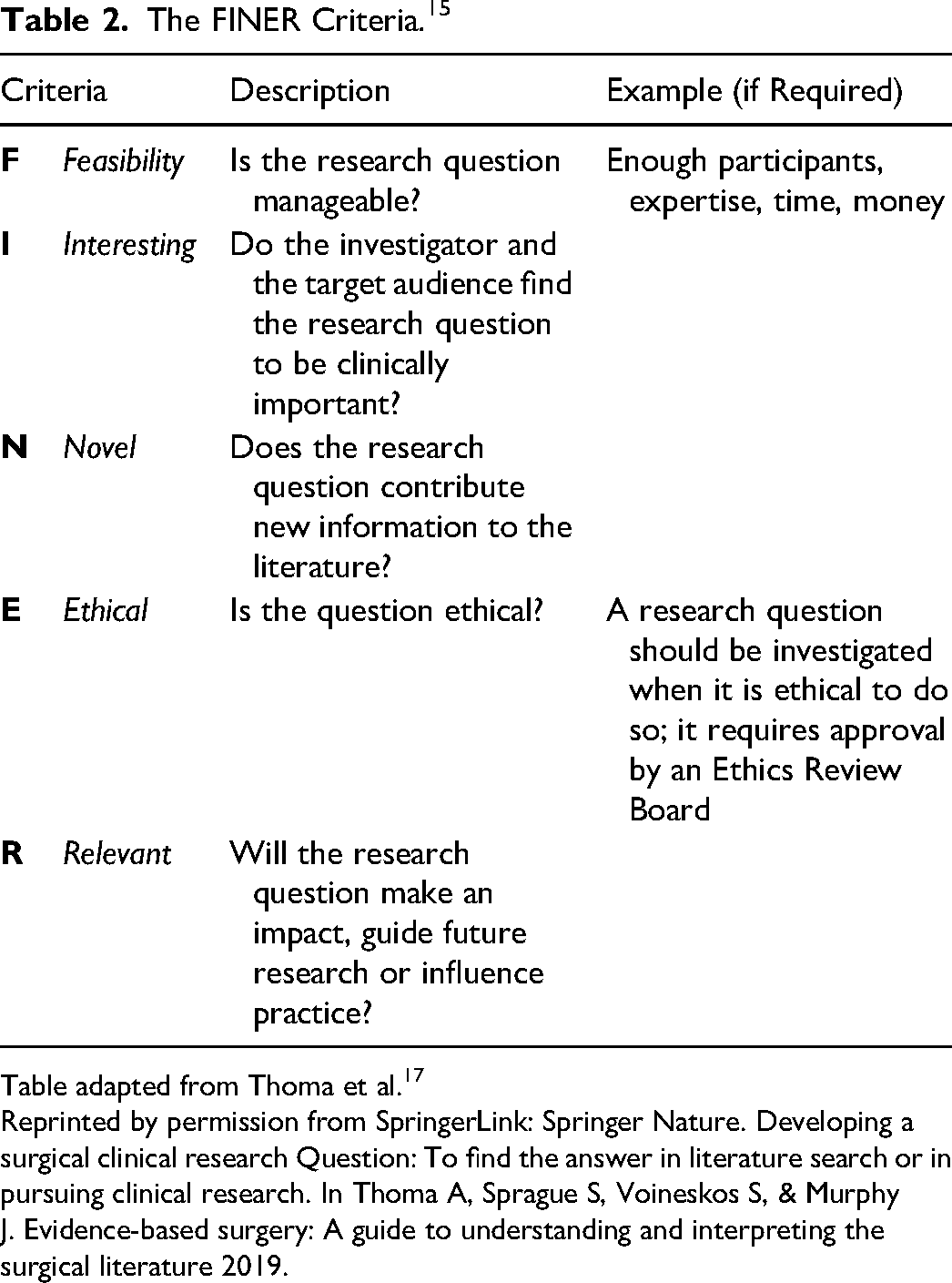
Table adapted from Thoma et al. 17
Reprinted by permission from SpringerLink: Springer Nature. Developing a surgical clinical research Question: To find the answer in literature search or in pursuing clinical research. In Thoma A, Sprague S, Voineskos S, & Murphy J. Evidence-based surgery: A guide to understanding and interpreting the surgical literature 2019.
The PICOT Format. 16
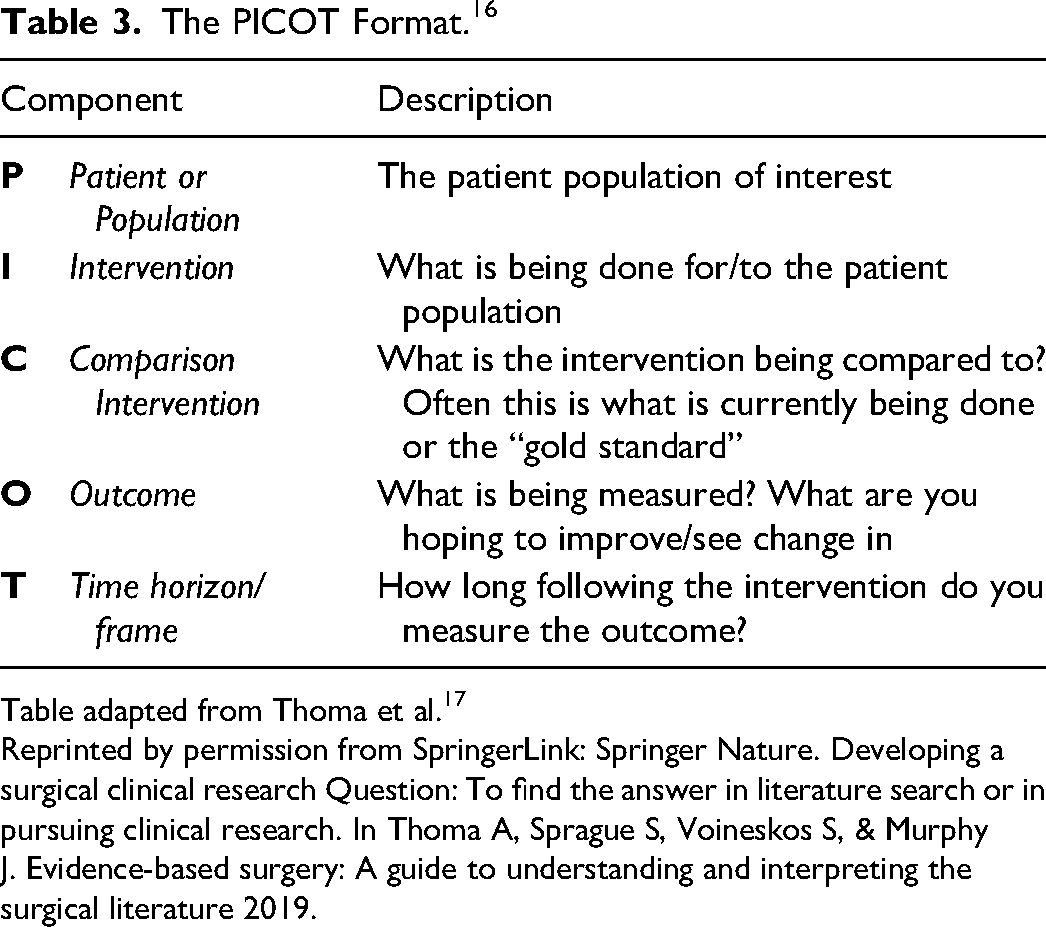
Table adapted from Thoma et al. 17
Reprinted by permission from SpringerLink: Springer Nature. Developing a surgical clinical research Question: To find the answer in literature search or in pursuing clinical research. In Thoma A, Sprague S, Voineskos S, & Murphy J. Evidence-based surgery: A guide to understanding and interpreting the surgical literature 2019.
The Methods section is one of the most important sections of a paper as it shows the readers to what extent your findings can be trusted. Your methods section should clearly describe elements of your study including information on the patient population (if applicable), your study outcomes, the tools used to measure these outcomes, the time horizon of your study, and how you analyzed your data. The chosen study design must be appropriate, in that, it can properly answer your research question (See Table 4). For example, in a research study where you want to determine if procedure A is more harmful than Procedure B, the RCT design cannot be used as it is unethical to proceed with such a study that submits patients in one of the groups to a harmful approach. However, a Case-Control study design is definitely an appropriate choice. Study outcomes should be selected based on evidence from previous literature, not out of convenience. Think for a moment, and ask yourself, are the outcomes I am using in this study critically important or trivial? You can justify your outcome choice with key references, such as Core Outcome Sets, 18 previous primary studies, or systematic reviews. If you do not provide evidence-based justification for your outcomes, reviewers will assume that you have not done your homework on the subject.
Alignment of Research Question with Study Design.

Information from Del Fabbro; 21 MA/SR, Meta-analyses and systematic review; RCT, Randomized Controlled Trial; CSM, Computer Simulated Model; QALY, Quality-Adjusted Life Years.
Next, authors are expected to provide justification for using specific outcome measurement tools (ie: instruments, scale, Patient Reported Outcomes [PROs]) to measure their chosen outcomes. Justification for outcome measurement tools can be provided through access information from previous literature, or database like the one created by The COSMIN Initiative. 20 When justifying the use of a specific questionnaire, PRO or scale, you need to include their clinimetric properties (eg: validity, reliability and responsiveness to change) and provide supporting references. A recent article expands on the preconditions of improving the science in plastic surgery and we encourage future authors to review this resource. 21
In addition to the above content, the Methods section should also include a detailed statistical analysis description. This should explain, clearly, to the readers how your collected data, cleaned, analyzed, and reported your data, as well as what program(s) were used to do so. We recommend that your statistical plan is developed, carried out, and reported with the help of a biostatistician. The authors also need to outline the steps they have taken to avoid the introduction of bias in their study. For example, if an RCT study is submitted, we would expect the authors to tell how they randomized their patients (ie: randomization software, random number generator). Furthermore, were the patients, researchers or outcome assessors blinded? This type of information is important when determining the bias, and therefore the credibility, of a study. 22
The Results section goes into detail about what was found, and how this finding relates to the research question and any stated hypotheses. This is not a space for data analysis or drawing conclusions. As opposed to simply listing one's data in paragraph format, tables and figures should be efficiently constructed to support a brief description of germane results that were found. At present some journals require authors to make their raw data available so other authors are able to validate, replicate, reproduce, reinterpret, and archive the primary study's findings. 23 Specifically, data availability statements describing a submitting author's compliance with a specific journal's data policy and specific methods of data sharing such as in repositories, supporting information files, etc represent essential components of submissions in an effort to enable greater data transparency. In the future, this will likely become a requirement for all journals.
The Discussion section briefly reiterates important background information, the justification of the investigation and the research question. Authors should then discuss how their results compare to earlier literature and provide directions for future research. The Discussion section should also discuss limitations and uncertainties. Despite our best efforts, a clinical research study is not like a chemistry or physics experiment. Most clinical research is pragmatic (one performed under real life circumstances) and there is always some uncertainty on the findings and conclusions. It is important that, in the Discussion section, the authors recognize the limitations and areas of uncertainty. These limitations are important as they inform future investigators what else needs to be done to address this uncertainty in future research. A final tip for the discussion section is to create a cogent, organized, and structured message to be conveyed to the reader as opposed to one giant collage of sentences that jump from topic-to-topic and create a difficult translation of content for reviewers and ultimately readers to follow.
Finally, we take this opportunity to comment on systematic reviews, a commonly published study design. Let's face it, systematic reviews are commonly published because they are easier to perform than well executed primary studies. In our view, if methodologically robust primary studies are performed, there would not be a need for systematic reviews! Frequently the rationale for submitting a systematic review is not strongly supported. Authors need to familiarize themselves why we use this study design. 24 The reviewers will be critical if SRs do not provide a strong rationale for the need for a systematic review or do not follow the PRISMA reporting guidelines. Furthermore, if a systematic review compares two interventions or approaches and is based on less than ten studies or less than three studies examine a particular outcome, such SRs will not be accepted for publication.
Summary
Surgeons, physicians, and researchers often turn to clinical research to help guide decision making and improve patient quality of care. As such, it is critical that published research is properly conducted, interpreted, and reported. The above article outlines the expectations to which all manuscripts submitted for publication in Plastic Surgery will be held. Although the adoption of the practices suggested in this article may add steps to the research and reporting process, they help to ensure that the research is rigorous, credible, and transparent. The authors of this article, as well as the editorial team at Plastic Surgery encourage researchers and knowledge users to consider the suggestions in this article and utilize the reference list as a way to further educate oneself on the requirements for high quality research.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr. Achilles Thoma may receive royalty payments from any sales of his book “Evidence-Based Medicine: A Guide to Understanding and Interpreting the Surgical Literature”
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics Approval
Not required
Informed Consent
Not required
Supplemental material
Supplemental material for this article is available online.
