Abstract
Selective internal radiation therapy is a form of intra-arterial brachytherapy used to treat primary liver cancer and liver metastases. This article aims to provide an overview of the clinical, economic, organizational legal, social and ethical impact of selective internal radiation therapy using SIR-Spheres Y-90 resin microspheres in the treatment of patients with unresectable, liver-dominant metastatic colorectal cancer who are refractory to or intolerant of chemotherapy. A systematic literature review was performed by querying PubMed, Scopus, EBSCO, CRD and GIN. Two reviewers blindly screened the records retrieved against predefined inclusion/exclusion criteria. The selected studies where summarized following a simplified version of the EuNetHTA Core Model® 2.1. The studies included evaluated selective internal radiation therapy in first-line or further-line treatment and showed a good safety and tolerability profile and significant improvement in efficacy expressed as time to liver progression, progression-free survival and overall survival. Selective internal radiation therapy should be provided in specialized centres and administered by a multidisciplinary team. A hub-and-spoke network could be a viable option to guarantee access to this technology across jurisdictions. The lack of a specific diagnosis-related group tariff accounting for the cost of the device could be seen as the major obstacle to a fair diffusion of this technology. The economic evaluations currently available show the cost-effectiveness of this technology in the population under study. Selective internal radiation therapy using SIR-Spheres Y-90 resin microspheres appears to be a clinically effective and cost-effective option in the treatment of metastatic colorectal cancer patients who are chemotherapy refractory or chemotherapy intolerant.
Keywords
Background
The clinical condition
Colorectal cancer (CRC) is the second most commonly diagnosed cancer in Europe and a leading cause of death both in Europe and worldwide. In 2012, there were 447,00 new cases of CRC in Europe with 215,000 deaths. Worldwide, there were 1.4 million new cases with 694,000 deaths. 1 In Italy, the estimated standardized prevalence rate in 2015 was 355/100,000 individuals aged 0–99 years. The standardized incidence rate, for the same year, was 52/100,000 at the national level. Liver metastases from CRC develop in 50% of patients, but only 25% of those are eligible for surgical resection. The 5-year survival rate after surgery is 20%–40%. 2 After liver resections for colorectal liver metastases (CLM), Abbas et al. 3 have observed a 10-year survival rate of 12%–28%, while Kanas et al. 4 have estimated a median 5-year survival rate of 38% (16%–74%).
Treatment options
According to the most recent ESMO Guidelines over the last decade, the prognosis for patients with metastatic colorectal cancer (mCRC) has improved greatly due not only to an increase in the number of patients being referred for and undergoing surgical resection of their localized metastatic disease, but also to a more strategic approach to the delivery of systemic therapy as well as an expansion in the use of ablative techniques. 5 This reflects the increase in the number of patients who are being managed by a multidisciplinary team and within specialized cancer centres, and the emergence of improved imaging techniques and prognostic and predictive molecular markers.1,5 A simplified treatment algorithm is depicted in Figure 1. Resection remains the mainstay of curative therapy for mCRC, and the treatment strategy should aim to complete resection whenever possible.
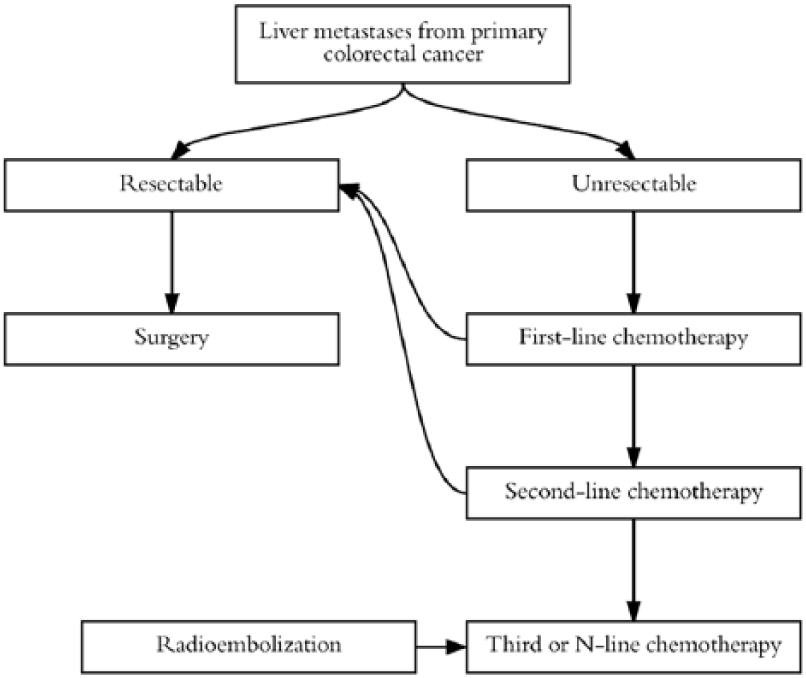
Treatment options for patients with liver metastasis from primary CRC cancer.
Most patients with mCRC present with unresectable disease. 1 For these patients, palliative systemic therapy is the standard of care, with the objectives to prolong overall survival, delay tumour progression and downstage initially unresectable colorectal metastases to resection (conversion therapy). 1 In case of tumour progression after a first-line or ‘induction’ systemic therapy, standard of care is second-line chemotherapy. Standard of care regimens for first- and second-line systemic therapy are including oxaliplatin or irinotecan combined with 5-fluorouracil (5-FU), and sometimes a biological compound (Avastin®, Vectibix® or Erbitux®).2,6
The algorithm in Figure 1 summarizes the potential position for selective internal radiation therapy (SIRT) in the therapeutic strategy, as discussed in this review. This describes SIRT as an option for patients who have failed first- and second-line chemotherapy, and could receive third- or further (‘n-line’) chemotherapy. It should be noted that this figure includes the possibility for patients to receive additional chemotherapy lines before SIRT, in addition to the existing armamentarium of further systemic therapy options (including trifluridine-tipiracil and regorafenib) for the treatment of patients with mCRC beyond first- and second-line treatments. No studies are currently available comparing the outcomes of SIRT and these systemic treatments. Although SIRT could be an alternative for some patients with liver-dominant disease in this setting, SIRT can also be considered as an option for patients failing these treatments.
For patients with liver-limited metastases failing the available chemotherapeutic options, radio-embolization with Y-90 resin microspheres has been shown to prolong the time to tumour progression in the liver. 1 Both cetuximab and panitumumab have shown efficacy in the third-line/salvage therapy setting in patients with Ras wild-type tumours and are equally active as single agents. 7 The combination of cetuximab with irinotecan is more active than cetuximab alone, in irinotecan-refractory patients. 8 There is no unequivocal evidence to support administration of the alternative epidermal growth factor receptor (EGFR) antibody, if a patient is refractory to the other; however, there is some evidence that circulating tumour DNA (ctDNA)-guided anti-EGFR re-challenge strategies are effective for patients with acquired resistance to EGFR antibodies.9,10 The multitargeted kinase inhibitor regorafenib has reported activity versus placebo plus best supportive care in two phase III trials.11,12 Regorafenib demonstrated a significant improvement in overall survival (OS) in patients pretreated with cytotoxics and bevacizumab and EGFR antibodies and can be proposed as a standard treatment in this setting. 11 However, concerns over safety have raised some doubts as to whether the labelled dose (160 mg/day 1–21 q4 weeks) is the optimal dose. For this reason, frequent and close monitoring for regorafenib toxicity is recommended. 1
For unresectable metastatic disease, systemic medical therapy (chemotherapy) is the first-choice treatment, but local therapy, such as loco-regional radiotherapy and ablative procedures, may also be utilized to prolong survival or to palliate symptoms (e.g. pain). Potential treatment options available for patients with unresectable, liver-dominant mCRC who are chemotherapy refractory or chemotherapy intolerant are summarized in Table 1.5,13,14
Potential treatment options available for patients with unresectable, liver-dominant mCRC.
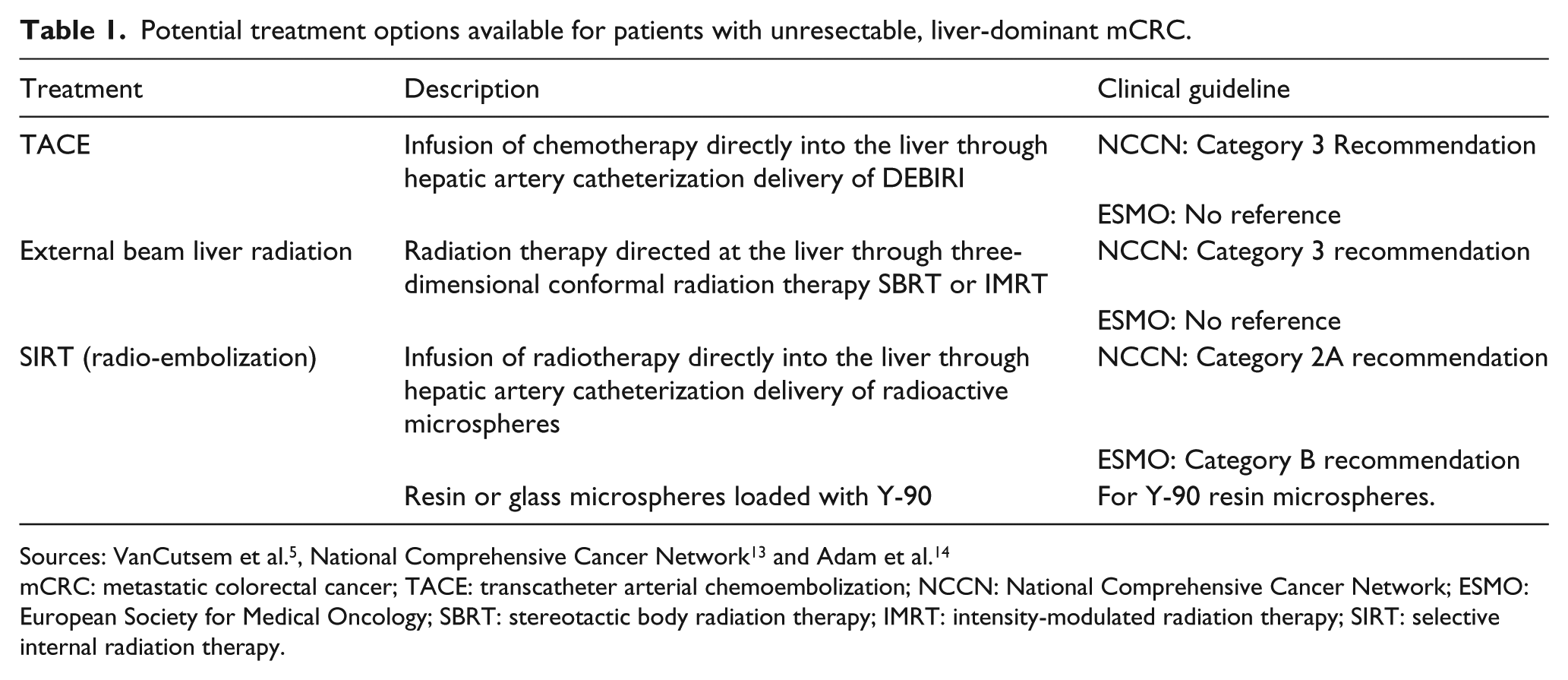
mCRC: metastatic colorectal cancer; TACE: transcatheter arterial chemoembolization; NCCN: National Comprehensive Cancer Network; ESMO: European Society for Medical Oncology; SBRT: stereotactic body radiation therapy; IMRT: intensity-modulated radiation therapy; SIRT: selective internal radiation therapy.
The technology
SIRT, also known as radio-embolization, is a form of intra-arterial brachytherapy used to treat primary liver cancer and liver metastases. SIRT uses resin microspheres including the β-emitter Yttrium-90 (Y-90) 2 and is indicated for patients with unresectable hepatic primary or metastatic cancer, liver-dominant tumour burden and life expectancy of at least 3 months. 15
Y-90 is a pure β emitter, with a physical half-life of 64.1 hours (2.68 days) with a mean tissue penetration of 2.5 mm and a maximum range of 11 mm.16–21 The SIRT procedure is based on the fact that intrahepatic malignancies derive their blood supply almost entirely from the hepatic artery and newly formed arterial vessels inside the cancerous tissue. The microspheres are injected selectively into the appropriate hepatic artery and subsequently become lodged in the microvasculature surrounding the tumour. Very high irradiation doses are delivered to the tumour, whereas the surrounding normal liver parenchyma is less affected by the radiation. 22
SIR-Spheres® Y-90 resin microspheres consist of millions of resin microspheres with an average diameter of about 32 μm (20–60 μm) loaded with Y-90. Typically, about 30–40 million Y-90 resin microspheres (1.0–1.5 GBq) are delivered in a treatment. 23 The microspheres are suspended in sterile water so that they can be delivered by injection, and they have a specific activity of 50 Bq per microsphere at the time of calibration. 24 SIR-Spheres Y-90 resin microspheres have a specific gravity of 1.6 g/mL which means that they become easily suspended in the blood flow when administered, thus allowing optimal distribution in the microvasculature supplying the tumour. This device has a 24-hour shelf life. Before SIRT is undertaken, meticulous coeliac and superior mesenteric angiography is conducted to map the hepatic arterial tree and to detect and occlude, using microcoil embolization, every collateral vessel that arises from the hepatic artery that could lead to extrahepatic deposition of microspheres. At a second hepatic arterial catheterization conducted separately after the therapy-planning arteriography, Y-90 resin microspheres suspended in sterile water are injected under intermittent fluoroscopic visualization, alternating with 5% glucose and contrast medium, to assess for preserved antegrade hepatic arterial flow.16,25,26
Contraindications for SIRT include (a) pretreatment angiogram indications of flow to the gastrointestinal tract – such as those visualized by the pretreatment Tc-MAA scan – which cannot be corrected by catheter embolization techniques, (b) an excessive shunting to the lungs as quantified by the Tc-MAA scan that would result in 30 Gy lung dose on a single administration, (c) excessive tumour burden with limited hepatic reserve or biochemical evidence of reduced liver function as potentially indicated by elevated levels of bilirubin (widely suggested cut-off: 2 mg/dL), (d) markedly abnormal synthetic and excretory liver function test (LFT) and (e) significantly altered international normalized ratio or partial thromboplastin time, or reduced serum albumin.
The target population
According to the Italian National Institute of Public Health, 52,000 new cases of colorectal cancer were diagnosed in Italy in 2015. Of these, about 30% exhibit liver metastatic disease. These patients are eligible for first-line chemotherapy, but approximately 15% of them will achieve surgically operable disease. The remaining 85% will receive second-line chemotherapy. Again, 85% of patients will not become eligible for surgery and, thus undergo third-line chemotherapy. About 30% of the third-line patients are chemo-refractory and about 90% of them will be eligible for SIRT procedure as a third-line option (3380), while the remaining 10% will be considered not eligible for SIRT due to physical or technical contraindications. Moreover, after third-line treatment, at any further treatment line the target population decreases by 15% due to the availability of other options.
A survey performed by the Italian Agency of National Health Services (Age.Na.S) and involving 19 Italian centres performing SIRT revealed that SIRT was used as the first line in only 21.2%, meaning that that chemotherapy still represents the best option as first-line treatment. The majority of hospitals (54.5%) declared providing SIRT in one session, while only two hospitals did so in two sessions and the remaining three of them in one or two sessions. All these centres employ SIR-Spheres Y-90 resin microspheres to perform SIRT. 2 The most recent ESMO guidelines recommend SIRT as a treatment option for patients with liver-limited disease failing the available chemotherapeutic options. Moreover, radio-embolization with Y-90 resin microspheres (and chemoembolization) of colorectal liver metastases in earlier treatment lines is considered as an interesting strategy as ‘consolidation treatment’ but should be limited to clinical trials. 1
Objectives
The aim of this study is to provide an overview of the clinical, economic, organizational legal, social and ethical impact of SIRT using SIR-Spheres Y-90 resin microspheres in the third-line treatment of patients with unresectable, liver-dominant metastatic colorectal cancer who are refractory to or intolerant of chemotherapy.
Methods
A literature review and a field research were performed.
Definition of the research question
The research question was made explicit by using the PICO model. Patients with unresectable, liver-dominant metastatic colorectal cancer who are chemotherapy refractory or chemotherapy intolerant represented the population under study (P); SIR-Spheres Y-90 resin microspheres were the intervention being assessed (I), whereas the best supportive care was the comparator (C). Outcomes of interest (O) were overall survival (OS), progression-free survival (PFS), HR-QoL and resection rate (see the ‘Efficacy’ section); adverse events (AEs) and side effects (see the ‘Safety’ section); costs and cost-effectiveness (see the ‘Economic impact’ section); and investments, training, patients flow, workflow (see the ‘Organizational impact’ section).
Search strategy
Five databases were queried to gather evidence needed to conduct the assessment, namely PubMed, Scopus, EBSCO, including Cochrane Central Register of Controlled Trials, Cochrane Database of Systematic Reviews, Health Technology Assessments, NHS Economic Evaluation Database, CRD Database and GIN (Guidelines International Network) database. The search string launched on PubMed and Scopus as well as the key words utilized in the other databases are reported in Supplemental Table S1. No temporal limits where imposed to our search strategy, but only pieces of evidence published in Italian or English language and reporting the key words in the title and/or abstract were included. Further evidence was identified though manual search. Moreover, data concerning the technical features of the technology were provided by the manufacturer. Finally, to get a deeper understanding of the use of this technology within the Italian setting, experts’ opinion was asked during the assessment.
Inclusion/exclusion criteria
Records retrieved though the search strategy were considered eligible unless they met one or more of the following exclusion criteria:
Not relevant to the technology under study;
Not relevant to the condition under study;
Neither English nor Italian language;
Type of study not relevant (case report, editorial, preclinical study);
Not sufficient information on any of the aspects under study.
Study selection process
Records retrieved were formatted into an MS Excel worksheet containing the following for each record: an ID number, the database in which it was found, indication of whether it was a duplicate or not, first author, year of publication, title, reference citation, link to the abstract and name of the reviewer who selected it. A drop-down menu indicated whether it was to be included or excluded, another drop-down menu showed the reasons for exclusion, and some more drop-down menus listed the HTA domains in which the article could be considered relevant.
The first screening, based on title and abstract, was done by two senior researchers blindly, while two junior researchers extracted data from the selected studies.
Reporting of results
Results have been discussed narratively. Evidence has been organized based on selected items of the EuNetHTA Core Model® 2.1. 27 The full list of items considered in the current analysis is reported in Supplemental Table S2. In this article, only the most relevant pieces of evidence have been summarized. The full analysis of the studies included in the safety and efficacy section is reported in Supplemental Tables S3 to S6, respectively.
Results
Safety
Kennedy et al. 28 conducted a single-arm retrospective study which was meant to enrol approximately 1000 patients from December 2012 to June 2013 (MORE study). Six hundred and six patients were eventually enrolled in 11 US centres; of these, 160 elderly (⩾70 years) and 446 younger (<70 years) received SIRT using Y-90 resin microspheres. A further analysis was conducted in 98 very elderly patients (⩾75 years). SIRT appeared to be equally well tolerated by elderly and younger patients, with no statistically significant differences across age groups were reported in terms of AEs of any grade (p = 0.433) or grade 3 events (p = 0.615), although gastrointestinal events (any grade) were less likely to be reported in the elderly patients than in the younger patients. Common grade 3+ events reported in the elderly and the younger patients were abdominal pain (3.1% vs 6.1%), gastritis or duodenitis (1.3% vs 0.2%), nausea (0.6% vs 1.3%), vomiting (1.3% vs 1.3%), fatigue (5.6% vs 4.5%), ascites (1.3% vs 2.0%), hyperbilirubinemia (3.8% vs 2.7%), and RE-induced liver disease (REILD) (1.3% vs 0.2%) respectively. Similar findings for grade 3 events were also observed in the analyses of the very elderly with the exception of grade 3+ abdominal pain, which was less likely to be reported in elderly (1.0% vs 6.1%; p = 0.029). The most common event was mild to moderate fatigue, which tended to be more frequent in the elderly patients than in the younger patients (41.3% vs 34.5%), but between-group differences were not statistically significant. Overall, the reporting of liver–function-related AEs was low and not significantly different from the younger cohorts in the analyses of either the elderly or the very elderly.
A prospective, randomized, multicentre international trial was conducted by van Hazel et al. 29 to assess the safety of SIRT using Y-90 resin microspheres in addition to standard fluorouracil-, leucovorin- and oxaliplatin-based chemotherapy (SIRFLOX) in patients with previously untreated mCRC. The study started in August 2006 and ended in November 2016. Five hundred and thirty-two patients were enrolled. In the study, 73.4% and 85.4% of patients experienced grade 3+ AEs in control and SIRT arm respectively. Hematologic toxicities were reported at a higher rate in SIRT compared with control (p < 0.05). Also, 1.9% of control group and 3.7% of SIRT patients reported grade 5 AEs of any causality. Four treatment-related grade 5 AEs were attributed to chemotherapy (two cardiac-related events in control and one respiratory failure and one febrile neutropenia in SIRT), two were attributed to SIRT (hepatic failure and radiation hepatitis) and one was attributed to both chemotherapy and SIRT (hepatic failure in SIRT). Serious AEs were reported less frequently in control patients (41.6%) than in SIRT patients (54.1%; p = 0.005). The authors showed that five patients experienced SIRT-related hepatotoxicity (radiation hepatitis or hepatic failure). Both cases of radiation hepatitis, one of which was fatal, occurred 2–3 months after SIRT and were treated with low-molecular-weight heparin, diuretics and corticosteroids. Two patients experienced fatal hepatic failure – one case occurring 5 days after SIRT and the other case >2 years after SIRT.
In a retrospective study, Tohme et al. 30 evaluated the tolerability outcomes among elderly (⩾70 years) and younger patients (<70 years) with liver-dominant mCRC who received SIRT using Y-90 resin microspheres as salvage therapy. From 2002 to 2012, 107 consecutive patients with unresectable mCRC treated with SIRT after failing first- and second-line chemotherapy have been included in a study, out of which 44 were elderly and 63 were younger patients. The study was performed at the University of Pittsburgh Medical Center. SIRT was equally well tolerated in both groups and common procedure-related AEs (fatigue, nausea and/or vomiting, abdominal pain, fever and increased bilirubin) were predominately of mild to moderate intensity and of short duration. Post-treatment hepatic toxicity was assessed between the elderly and younger patients and found to be relatively mild in both groups. No patient had fulminant hepatic failure after treatment. One patient in each group developed a late (later than 30 days) grade 4 toxicity that was related to a biliary stricture and resolved with endoscopic retrograde cholangiopancreatography and stenting. No patients developed post-treatment gastric or duodenal ulceration.
van Hazel et al. 31 conducted a randomized trial to compare the response rate, time to progressive disease, and toxicity of a regimen of systemic fluorouracil/leucovorin chemotherapy versus the same chemotherapy plus a single administration of SIR-Spheres Y-90 resin microspheres in patients with advanced colorectal liver metastases. Twenty-one patients with previously untreated advanced colorectal liver metastases, with or without extrahepatic metastases, were randomized into the study: 5-FU 425 mg/m2/day plus leucovorin 20 mg/m2/day for five consecutive days and repeated at four weekly intervals versus the same chemotherapy plus a single administration of Y-90 resin microspheres that was administered on the third or fourth day of the second cycle of chemotherapy. There were more grades 3 and 4 toxicity events in patients receiving the combination treatment. One patient in the combination arm died from chemotherapy-induced neutropenic sepsis after the fourth chemotherapy cycle. Four patients developed transient abdominal pain at the time of injection of the Y-90 resin microspheres that resolved with narcotic analgesia. One patient treated with SIRT plus chemotherapy developed a liver abscess in the site of a necrotic tumour mass following treatment and recovered quickly after drainage of the abscess. One patient developed radiation induced liver cirrhosis. As this patient weighed 43 kg, treatment with 2.5 GBq of Y-90 activity was considered excessive.
The main features and results of the studies discussed here are listed in Table 2.
Summary of evidence on safety.
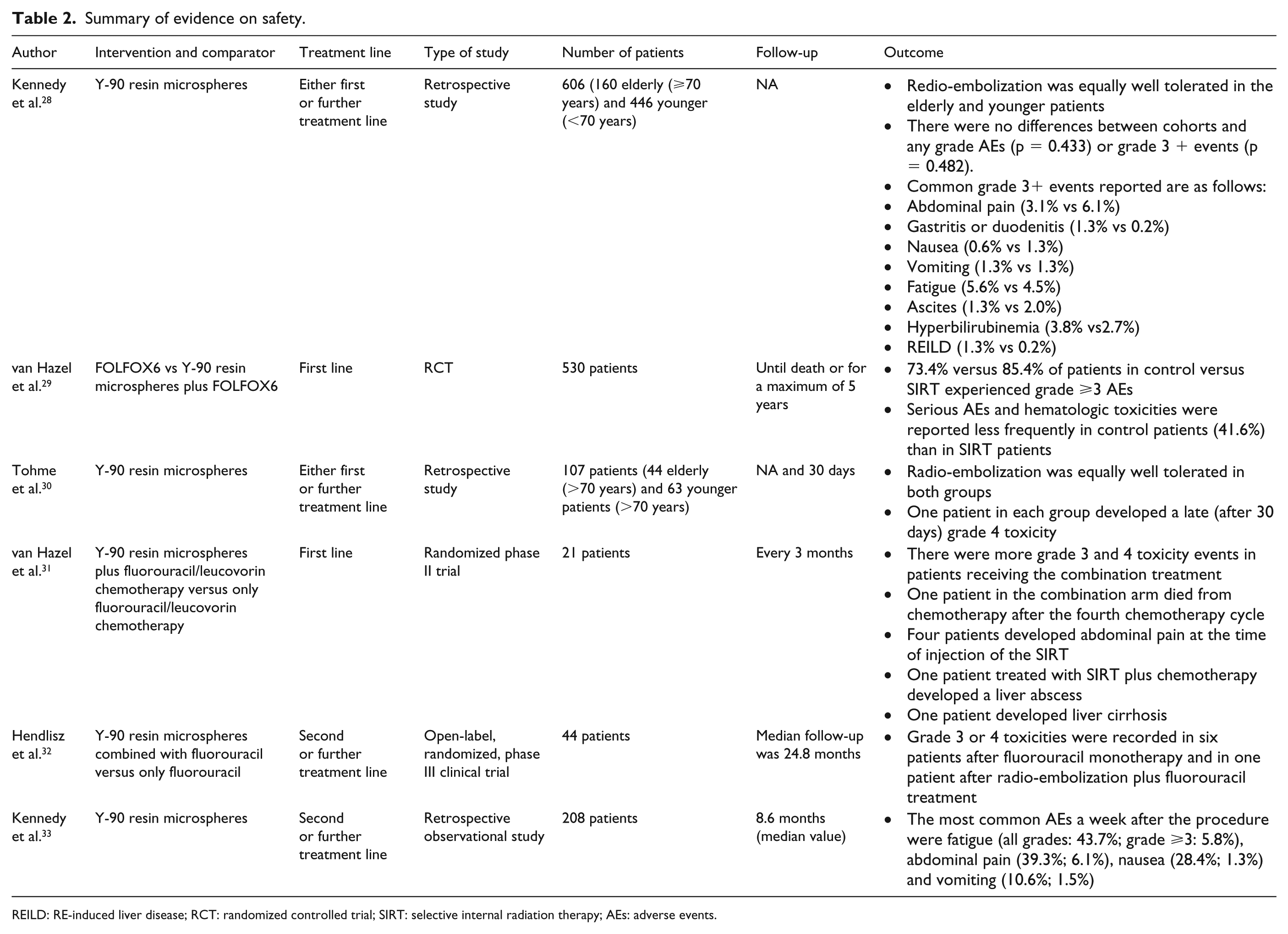
REILD: RE-induced liver disease; RCT: randomized controlled trial; SIRT: selective internal radiation therapy; AEs: adverse events.
Efficacy
Two comparative retrospective studies demonstrated significant OS improvements associated with Y-90 resin microspheres in the chemotherapy-refractory or intolerant setting.
First, Bester et al. 34 evaluated the efficacy of Y-90 resin microspheres in 339 patients with chemotherapy-refractory liver metastases. Patients were recruited between 2006 and 2011. Another 51 patients were instead referred back to their treating physician for conservative treatment or best supportive care. OS was determined for the whole treated cohort and the standard-care cohort, as well as for the treated CRC group and the treated non-CRC group. For the whole treated cohort, the median OS after the first treatment with Y-90 microsphere radio-embolization was 12.0 months (95% confidence interval (CI): (10.7, 14.5); p < 0.001), and there was a significant improvement in OS in those patients treated with Y-90 resin microspheres compared with the standard-care cohort. 34
Second, Seidensticker et al. 35 performed a matched-pair comparison of patients who received radio-embolization with Y-90 resin microspheres plus best supportive care (BSC) or only BSC for extensive liver disease. Twenty-nine patients who received radio-embolization were retrospectively matched with a contemporary cohort of 500 patients who received BSC from three centres in Germany. Patients in both groups had a similar performance status (Karnofsky index, median 80% (60%–100%)). Compared with only BSC, radio-embolization with Y-90 resin microspheres prolonged survival (median, 8.3 vs 3.5 months; p < 0.001) with a hazard ratio of 0.3 (95% CI: (0.16–0.55); p < 0.001) in a multivariate Cox proportional hazard model. 35
Kennedy et al. 33 evaluated 208 patients in the United States treated with Y-90 resin microspheres in liver brachytherapy. Median survival is 10.5 months for responders but only 4.5 months in non-responders. Radioactive microspheres produced an encouraging median survival. Partial responses were found in 74 of 208 patients (35.5%), stable disease or minor response was found in 114 of 208 patients (55%), and progressive disease was found in 21 of 208 patients (10%). Positron Emission Tomography scans showed response in 176 of 208 patients (85%) and no response or progression in 31 of 208 patients (15%). Radioactive microspheres produced a significant objective response rate.
Tohme et al. 30 found no significant difference with regard to overall median survival between younger (8.4 months; 95% CI: (6.2, 10.6)) or elderly patients (8.2 months; 95% CI: (5.9, 10.5); p = 0.667). The presence of extrahepatic disease was associated with a significantly worse median survival in both groups. The authors have concluded that radio-embolization with Y-90 resin microspheres appears to be as effective for the elderly as it is for younger patients with mCRC.
Hendlisz et al. 32 conducted a prospective, multicentre, randomized phase III trial in patients with unresectable, chemotherapy-refractory or chemotherapy-intolerant, liver-limited mCRC comparing arm A (FU protracted intravenous infusion 300 mg/m2 days 1 through 14 every 3 weeks) and arm B (SIRT plus intravenous FU 225 mg/m2 days 1 through 14 then 300 mg/m2 days 1 through 14 every 3 weeks) until hepatic progression (NCT00199173). Overall response rates in arms A and B were 0% and 9.5%, respectively, and disease control rates (partial response and stable disease) were 35% and 86%, respectively. The study met its primary endpoint with Y-90 resin microspheres plus FU significantly improving time to local progression (TTLP) and time to tumour progression (TTP) compared with only FU. There was no significant difference in median OS between the treatment arms: 7.3 months for arm A (two patients alive at the time of analysis) and 10.0 months for patients in arm B. The OS result was confounded by crossover as 10 out of 23 patients in arm A received SIRT on progression. This procedure was a valid therapeutic option for chemotherapy-refractory, liver-limited mCRC.
In the study by van Hazel et al., 31 patients treated with the combination of SIR-Spheres plus chemotherapy had a median survival of 29.4 months compared to 12.8 months for patients treated with chemotherapy alone (p = 0.025). Moreover, in this study, the response rate was evaluated using RECIST criteria. Although several patients in the chemotherapy arm showed some diminution in tumour size with treatment, no patient qualified for a response. No complete responses were recorded in either group. The response rate for 11 patients receiving the combination treatment was significantly greater than for 10 patients receiving only chemotherapy (First Integrated Response; 10 PR, 1 SD vs 0 PR, 6 SD, 4 PD, p < 0.001 and Best Confirmed Response; 8 PR, 3 SD vs 0 PR, 6 SD, 4 PD, p < 0.001). The time to progressive disease was significantly longer for patients treated with the combination of SIRT plus chemotherapy (18.6 vs 3.6 months). This trial demonstrated that the addition of a single administration of SIR-Spheres to a regimen of systemic fluorouracil/leucovorin chemotherapy significantly increased treatment-related response, time to progressive disease and survival.
The main features and results of the studies discussed here are listed in Table 3.
Summary of evidence on efficacy.
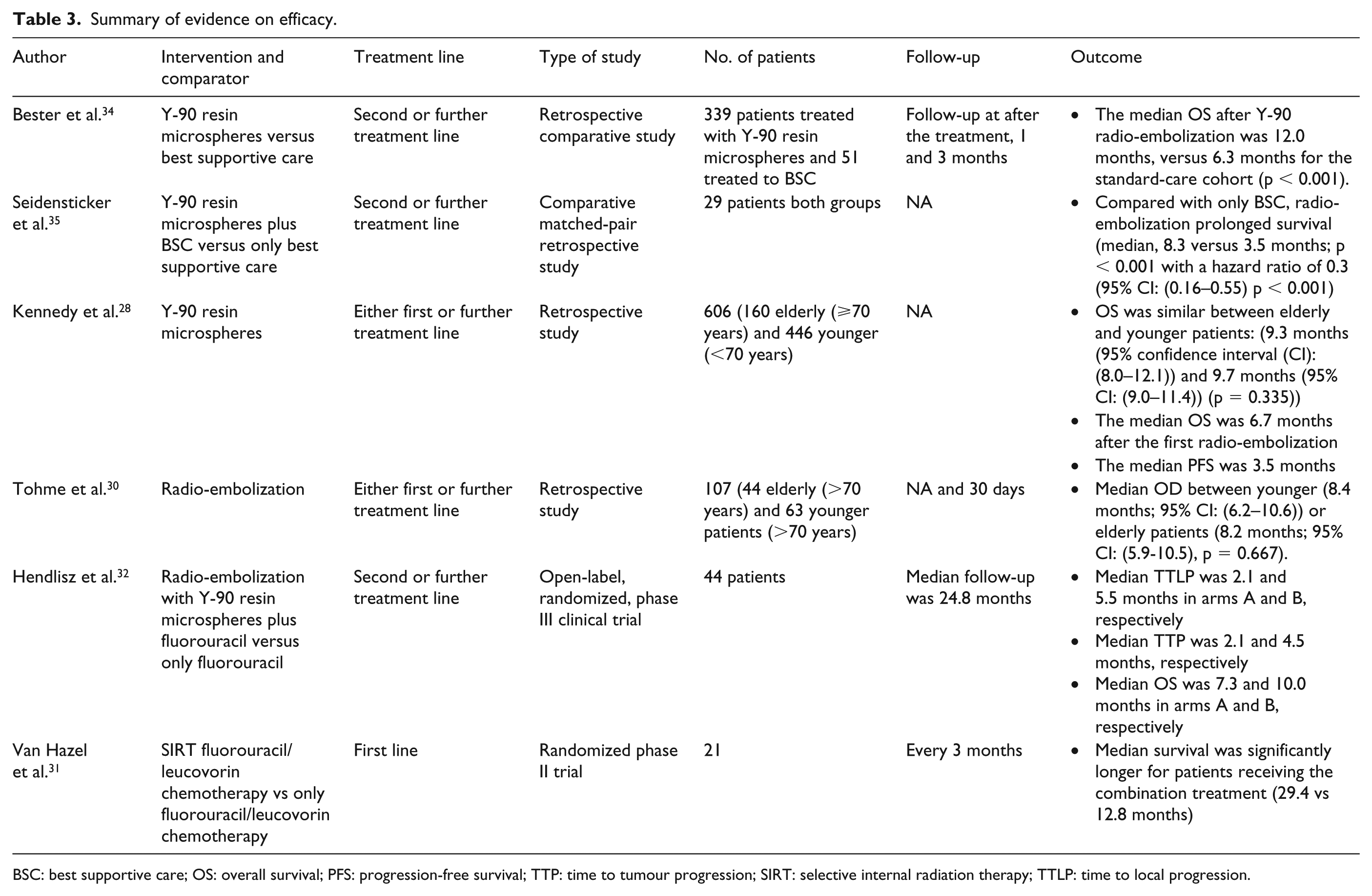
BSC: best supportive care; OS: overall survival; PFS: progression-free survival; TTP: time to tumour progression; SIRT: selective internal radiation therapy; TTLP: time to local progression.
Organizational aspects
A patient being considered for SIRT will be admitted for a work-up procedure before the treatment.23,36 The pretreatment work-up consists of (a) angiography to identify the hepatic vasculature feeding the tumour(s) to ensure that the blood supply to the tumour(s) is suitable for highly selective injection and to identify hepatic vascular connections to the gastro intestinal (GI) tract to ensure that these are sufficiently small to avoid radiation pneumonitis or radiation gastritis; (b) injection of macro-aggregated albumin labelled with Technetium-99m (99mTc-MAA). This is a diagnostic injection predicting the distribution of microspheres demonstrating the degree of hepato-pulmonary shunting and the connections between liver vascularization and GI tract that could result in delivery of radiation to non-target tissue; (c) single single-photon emission computed tomography (SPECT) scan using a gamma camera to demonstrate the degree of lung shunting, confirm avoidance of GI tract shunting, map the deposition of MAA in target lesions and confirm the degree to which healthy tissue will be spared by radiation; and (d) review of SPECT scan to calculate the lung shunt and confirm that it is below the acceptable threshold, determine if SIRT is the appropriate treatment option and establish the appropriate dose for the patient. The work-up can be performed in an outpatient setting.
If SIRT is confirmed as the appropriate treatment option, the treatment date is defined. Under local anaesthesia, patients are injected with radioactive microspheres usually via a trans-femoral catheter into the branches of the hepatic artery. The administration of SIRT using Y-90 resin microspheres is predominately performed in a single session.
SIRT procedures must be performed by an expert radiologist, under aseptic conditions in an interventional radiology/ theatre setting. 37 According to Age.Na.S, centres performing SIRT should be equipped with a tumour board, a nuclear medicine unit with licence to store and dispose of Yttrium-90 and a hot lab to prepare the activity, a planar gamma camera and/or SPECT-CT to perform the 99mTc-labelled MAA scan after the pretreatment work-up procedure and the Bremsstrahlung scan after SIRT administration, an interventional radiology suite, equipped with an angiography and licenced to use Yttrium-90 within interventional procedures, a licence for authorized users to administer Yttrium-90 microspheres and a medical physics unit with facility to perform Yttrium-90 dosimetric calculations and manage radiation protection before, during and after the procedure. 2 However, these are, generally, already owned by hospitals for performing other procedures; thus they do not represent a specific investment to perform SIRT.
Clinical decisions upon the treatment of mCRC should be discussed within a multidisciplinary team that should include both a colorectal surgeon and a hepatobiliary surgery specialist and/or a liver surgeon as necessary, with the compulsory inclusion of a pathologist and a diagnostic radiologist, as well as radiation and medical oncologists. An interventional radiologist/nuclear physician may also be included as the role of ablative treatments is gaining increasing importance.1,38–40
Costs and economic evaluation
The societal cost of mCRC
mCRC is characterized by a significant economic burden. A study conducted in the United States on 598 patients revealed that the mean total cost per patient (medical plus pharmaceutical costs) was US$252,200; outpatient hospital visits (excluding radiation and surgery) contributed most to the total cost, at a mean cost of US$71,334. Hospitalization costs, with or without surgery (mean, US$56,862), accounted for 33% of the US$176,135 unadjusted mean cost for medical services (ambulatory visits, emergency department visits, laboratory/radiology services and inpatient admission). Chemotherapy and biologics were also costly (mean, US$31,112 and US$38,276 respectively). Moreover, CRC stage at diagnosis and the number of lines of treatment after metastasis had a statistically significant association with cost (p < 0.001). 41 A similar pattern was observed by Paramore et al. 42 who found an additional annual cost for patients with mCRC of US$97,031 more for mCRC cases compared to non-mCRC controls. The main cost drivers for mCRC were hospitalizations (US$37,369) and specialist visits (US$34,582), which included chemotherapy administration. 42
A large Finnish cost-of-illness study reported that the total cost for the cross-sectional six-month period were €22,200 in the primary treatment state, €2,106 in the rehabilitation state, €2,812 in the remission state, €20,540 in the metastatic state and €21,146 in the palliative state. Most of the costs were direct medical costs. The informal care cost was highest per patient in the palliative care state, amounting to 33% of the total costs. The productivity costs varied between disease states, constituting 19%–40% of the total costs, and were highest in the primary treatment state. 43
Terminal metastatic CRC patients are those consuming the highest amount of healthcare resources. Mittmann et al. 44 estimated that patients approaching death received an average of eight home care visits at CAD$800 within a 30-day time horizon. Overall, patients with advanced CRC who were approaching death were found to require a moderate level of home care support, resulting in costs of about CAD$5,000 over the six-month time horizon.
Using regression methods, Hanly et al. 45 focused on costs for patients and their families in the Irish setting. In the diagnosis and treatment phase, weekly informal care costs per person were as follows: hospital-based costs, incurred by 99% of carers (mean = €393), domestic-based time costs, incurred by 85% (mean = €609), and domestic-based out-of-pocket (OOP) costs, incurred by 68%, (mean = €69). Ongoing costs included domestic-based time costs incurred by 66% (mean = €66) and domestic-based OOP costs incurred by 52% (mean = €52).
The cost-effectiveness of SIRT
Evidence on cost-effectiveness of SIRT comes from a unique cost-effectiveness model, adapted to different jurisdictions.
Pennington et al. 46 compared Y-90 resin microspheres to best supportive care (BSC) in patients with inoperable liver-dominant, chemotherapy-refractory mCRC from a UK perspective. Survival data from a comparative retrospective cohort study was analysed and used in a state-transition cost-effectiveness model, using quality-adjusted life years (QALYs) gained as the measure of effectiveness. The model incorporated costs for the SIRT procedure, monitoring, further treatment, AEs and death. Utility values, reflecting patient quality-of-life, were taken from a published source. SIRT using Y-90 resin microspheres compared to BSC improved OS by a mean of 1.12 life years and resulted in a cost per QALY gained of £28,216. In sensitivity analysis, this varied between £25,015 and £28,817.
Using the same model structure, Taieb et al. 47 assessed the cost-effectiveness of SIRT with SIR-Spheres Y-90 resin microspheres compared to BSC from the perspective of the national health service in France. A wide range of sensitivity analyses were performed on utility, OS and costs. The cost per QALY gained with SIRT was €30,610 ranging from €25,015 to €31,817 in the sensitivity analysis. The results demonstrated that SIRT was a cost-effective option in the treatment of patients with chemotherapy-refractory, liver-dominant mCRC in France.
Cosimelli et al. 48 adapted the same model to the perspective of the Italian National Health Service. The model included costs for treatment acquisition, pretreatment work-up and delivery of microspheres, and chemotherapy received in addition to, instead of or after, SIRT. In addition, costs of managing AEs and the cost of death were included. Costs were obtained from the University Hospital in Bologna, the Italian Agency of Medicines (Agenzia Italiana del Farmaco (AIFA)) and the literature. Utility data was not available from the study; so it was taken from a recent economic evaluation run by the National Institute of Clinical Excellence for the UK on the same target population.
SIRT increased survival resulting in a life-year gain of 1.35 (2.12 vs 0.98) life years and a QALY gain of 0.83 (1.52 vs 0.70). The costs of SIRT, monitoring and further treatment were greater in the SIRT arm with partial cost offset through a reduction in AEs. Overall, SIRT led to an increase in costs of €24,626 (€39,973 vs €15,347), resulting in a cost/QALY of €29,850. Probabilistic sensitivity analysis showed a 97% chance of SIRT being cost-effective at a threshold of €50,000/QALY. The authors concluded that SIRT using Y-90 resin microspheres could potentially be a cost-effective option in the treatment of patients with chemotherapy-refractory liver metastases resulting from CRC (Table 4).
Summary of evidence on cost-effectiveness.
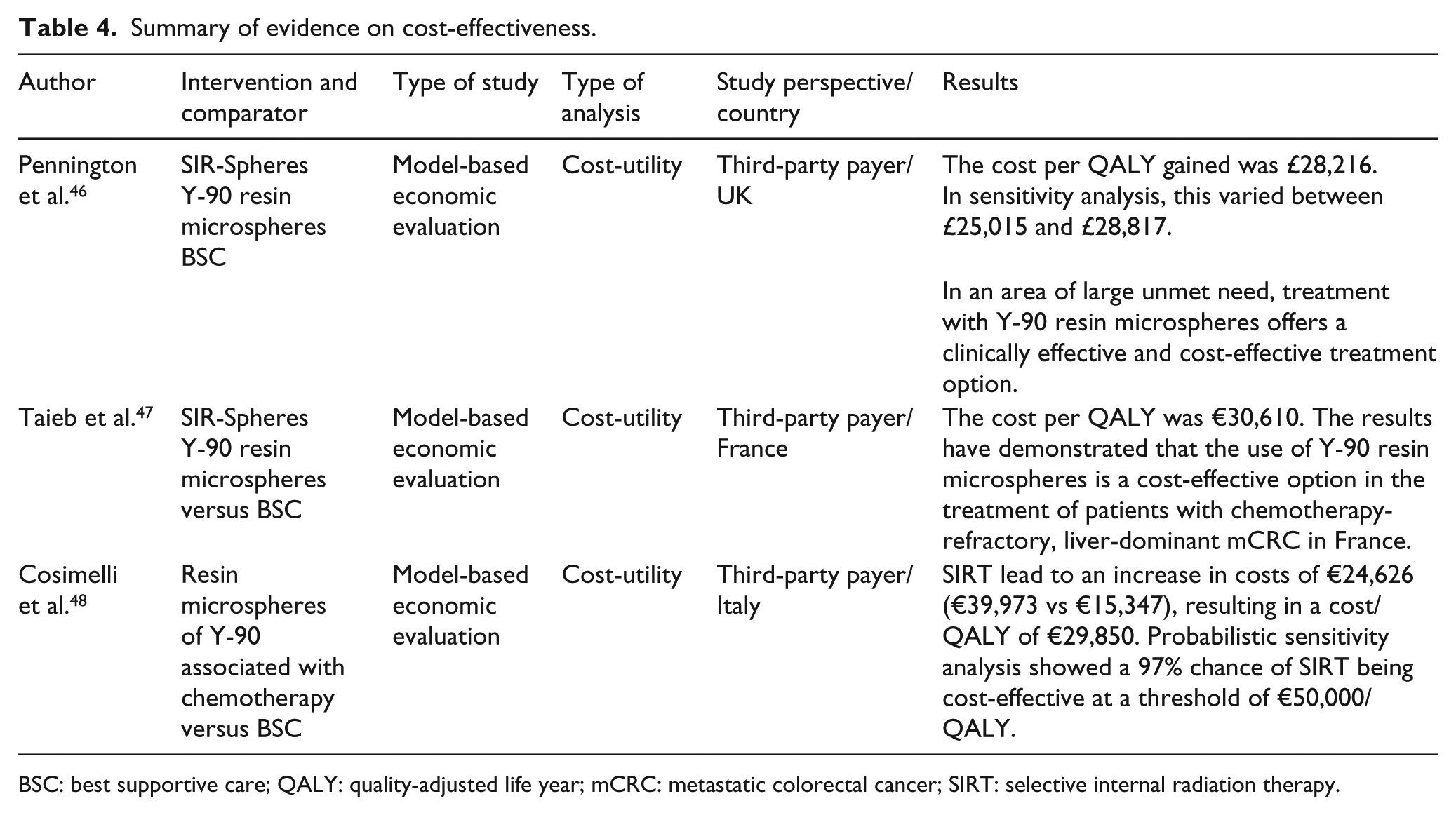
BSC: best supportive care; QALY: quality-adjusted life year; mCRC: metastatic colorectal cancer; SIRT: selective internal radiation therapy.
Ethical, legal and social issues
The new ESMO guidelines, recommend SIRT using Y-90 resin microspheres for patients with liver-limited metastases of colorectal cancer failing the available chemotherapeutic options, that is those who are refractory or intolerant to chemotherapy. 1 For patients being able to undergo SIRT means having a further chance of surviving cancer. Cosimelli et al. 49 in their study measured patients QoL before SIRT and 6 weeks after treatment. They administrated cancer and site-specific questionnaires to 14 patients (out of 50 patients included in the study). The interpersonal and technical skills of healthcare operators and the information they provided on treatment were judged good by patients (European Organization for Research and Treatment of Cancer questionnaire on satisfaction of cancer inpatients, EORTC IN–PATSAT 32). Compliance was good (mean 8 out of a 10-point scale, where 10 is the maximum score). Anxiety and depression levels before treatment were borderline, but anxiety decreased significantly after 6 weeks, while the depression score did not change. According to the authors, the results show good overall QoL with SIRT.
Treatment options for mCRC patients are limited and for this reason, these patients can be considered particularly vulnerable; this population is defined by Pennington et al. 46 as having a large unmet need. For this reason, a realistic explanation of the ‘strengths’ and ‘weaknesses’ of the technology is necessary to allow the patient to express a conscious consent to the therapy. As underlined by Chiarolla et al. 2 this point is relevant from the ethical point of view with particular reference to patients’ expectations and hopes. Anyway, the attitude towards this treatment is usually positive and its side effects are regarded as tolerable.
The relatively high cost of the technology along with the lack of a specific reimbursement within the Italian NHS, could be perceived as an obstacle to patients’ equal access to care. Once economical sustainability is ascertained, also organizational requirements (and sustainability) for an effective delivery of the technology in Italian regional context is required.
Discussion and conclusion
So far, SIRT has been considered as a third-or-over line of treatment. In this study, SIRT proved to have a positive safety and efficacy profile and a favourable cost-effectiveness ratio in this population of patients. However, the treatment pattern for patients with liver metastases from primary CRC has been changing dramatically over time and the proportion of patients treated with biological agents is now much greater than in the past. Moreover, regorafenib and trifluridine-tipiracil have been validated in randomized phase III clinical trials, and approved for reimbursement by AIFA as a third-line treatment in mCRC. These drugs are also available in other European countries, although regorafenib is not recommended in this setting in Germany and in the UK. In this evolving context, future research is required to estimate the cost-effectiveness of SIRT compared to these drugs in third-line mCRC.
This technology may also be beneficial in earlier lines of treatment. Although phase III randomized trials have not demonstrated a benefit of SIRT in combination with standard-of-care chemotherapy versus only chemotherapy in terms of OS, 50 a recent meta-analysis of two of these trials has demonstrated a statistically significant extension of OS for patients with a right-sided primary colorectal cancer receiving SIRT (median: 22.0 vs 17.1 months; HR = 0.641; p = 0.008). 51 It should be noted that evidence in this setting cannot be applied to patients with chemotherapy-refractory or chemotherapy-intolerant disease.
In conclusion, SIRT using SIR-Spheres Y-90 resin microspheres appears to be a clinically effective and cost-effective option for the treatment of well-selected mCRC patients who are chemotherapy refractory or chemotherapy intolerant. The implementation of this technology is recommended in hospital centres with adequate technological infrastructure and appropriate personnel training. Compliance with the treatment protocol is important for the success of the procedure. Further research is needed to recommend the use of SIRT in earlier treatment lines.
Supplemental Material
Supplemental_Material – Supplemental material for SIR-Spheres® Y-90 resin microspheres in chemotherapy refractory or intolerant patients with metastatic colorectal cancer
Supplemental material, Supplemental_Material for SIR-Spheres® Y-90 resin microspheres in chemotherapy refractory or intolerant patients with metastatic colorectal cancer by Silvia Coretti, Filippo Rumi, Dario Sacchini and Americo Cicchetti in Global & Regional Health Technology Assessment
Footnotes
Acknowledgements
This study was made possible by an unconditional grant provided by Sirtex Medical Europe GmbH. Publication of the study results was not contingent on sponsor’s approval, and views expressed here are those of the authors and not necessarily those of the funders.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
