Abstract
Introduction:
Bisphenol A is a synthetic compound widely used in the production of polycarbonate plastics and epoxy resins worldwide. As an environmental toxin, it has been reported in plastic equipment and utensils, water bottles and bottle tops, water supply pipes and epoxy resins that coat most of the metal food cans. It is a known endocrine-disrupting chemical and has been progressively replaced by its derivatives including bisphenol S, bisphenol F, bisphenol E, bisphenol AF, bisphenol B and tetramethyl bisphenol F. Bisphenol A and its analogues can bind to estrogen receptors and trigger multiple cellular responses at the organism level.
Methods:
A comprehensive literature review was done utilising electronic databases of PubMed, Google Scholar, Hinari, Connected papers and Science Direct from 1991 onwards. The articles were only included if they reported original relevant research and were limited to articles written in English.
Results:
Animal models, including non-human primates, have been used to study their effects on the endocrine system. Its endocrine disruption activity is reported to be the most studied effect in reproductive biology indicating that it may potentially cause endometriosis in females. Though non-human primates are closely related to humans, limited data exists on their associations between Bisphenol A exposure and its analogues and the pathophysiology of endometriosis.
Conclusion:
Given the current multifaceted knowledge/theory on endometriosis etiology, there is a strong necessity to conduct further biomedical research that utilises non-human primates to study the link between endocrine-disrupting chemicals and its effects on endometriosis.
Introduction
Endometriosis is a benign inflammatory and estrogen-dependent gynecological condition characterised by the presence of endometrial glands and stroma outside the uterine cavity. 1 Global epidemiological studies show that endometriosis affects 10% of reproductive-aged females with annual incidences ranging from 0.12% to 0.72%. 1 The disease’s incidence has risen, however, it is unclear whether this rise is due to disease awareness or environmental contamination. 2
It has an idiopathic etiology accompanied by different theories of pathogenesis. 3 Recent reports indicate that endometriosis is likely attributed to a sundry of endocrine-disrupting chemicals including; bisphenol A (BPA), phthalates, polychlorinated biphenyl (PCBs), atrazine, polybrominated biphenyls (PBBs), 4 dichlorodiphenyltrichloroethane (DDT), 5 dichlorodiphenyldichloroethylene (DDE), dichlorodiphenyldichloroethane (DDD), diethylstilbestrol (DES) and dioxins (TCDD). 6 One of the proposed etiological mechanisms is Sampson’s theory of retrograde menstruation which seems to be the most coherent and explains cases of endometriosis in the pelvis minor. 7 The theory supposes that backflow of endometrial tissues during menstruation into the abdomen results in implantation. 8 Further theories including coelomic metaplasia, lymph vascular metastasis or the embryonic rest theory have been complemented by more recent concepts including lifestyle-related factors, stem cells, hormonal,9,10 immune dysfunction, environmental pollution and/or exposure to bisphenols, 11 and genetic or epigenetic aspects. 12
Despite the presence of multiple theories of endometriosis pathogenesis, the environmental factors that could also pose a threat to and pathogenesis of the disease warrant investigation. To tackle this, experimental investigations including animal models could provide a basis for exploring the proposed association between endocrine-disrupting environmental toxins, such as Bisphenol-A (BPA) exposure and endometriosis development. 13 Numerous efforts have been made to utilise experimental animals, mostly mice 14 and a few non-human primates (NHPs). 15 models in the study of BPA exposure and endometriosis.
The exposure of BPA and other Endocrine Disrupting Chemicals (EDCs) to the reproductive system renders serious abnormalities that may lead to impaired fertility, irregular menstrual patterns, Polycystic Ovary Syndrome (PCOS) and endometriosis. 16 EDCs disrupt normal hormone function by either mimicking or blocking hormones and causing adverse health effects. 17 Long-term exposure to the EDCs can lead to disorders in reproductive-aged females that may be transgenerational. Several studies have reported on the association of BPA and female fecundity, with BPA being detected more frequently in infertile women. 16 Furthermore, BPA experimental research shows that its effects are more critical during prenatal, perinatal and postnatal exposure in the pup’s early life stages and may have transgenerational. 18
Therefore, this review focuses on the effects of BPA and its analogues (Bisphenol S (BPS), Bisphenol F (BPF), Bisphenol E (BPE), Bisphenol F (BPAF) and Bisphenol B (BPB)) on the pathophysiology of endometriosis disease in NHPs as a surrogate for human disease. 19
Methodology
The literature search was done using the electronic databases of Google Scholar, Science Direct (Elsevier), PubMed, Hinari and Connected Papers websites restricted to the period between 1991-2022. The search terms used included; ‘non-human primates’, ‘endometriosis’, ‘bisphenol A,’ ‘bisphenols analogues’ and ‘animal models. The reference lists of all selected articles were reviewed to identify additional papers and the search was limited to articles written in English. Inclusion/Exclusion criteria are listed in Table 1.
Details of the inclusion and exclusion criteria.
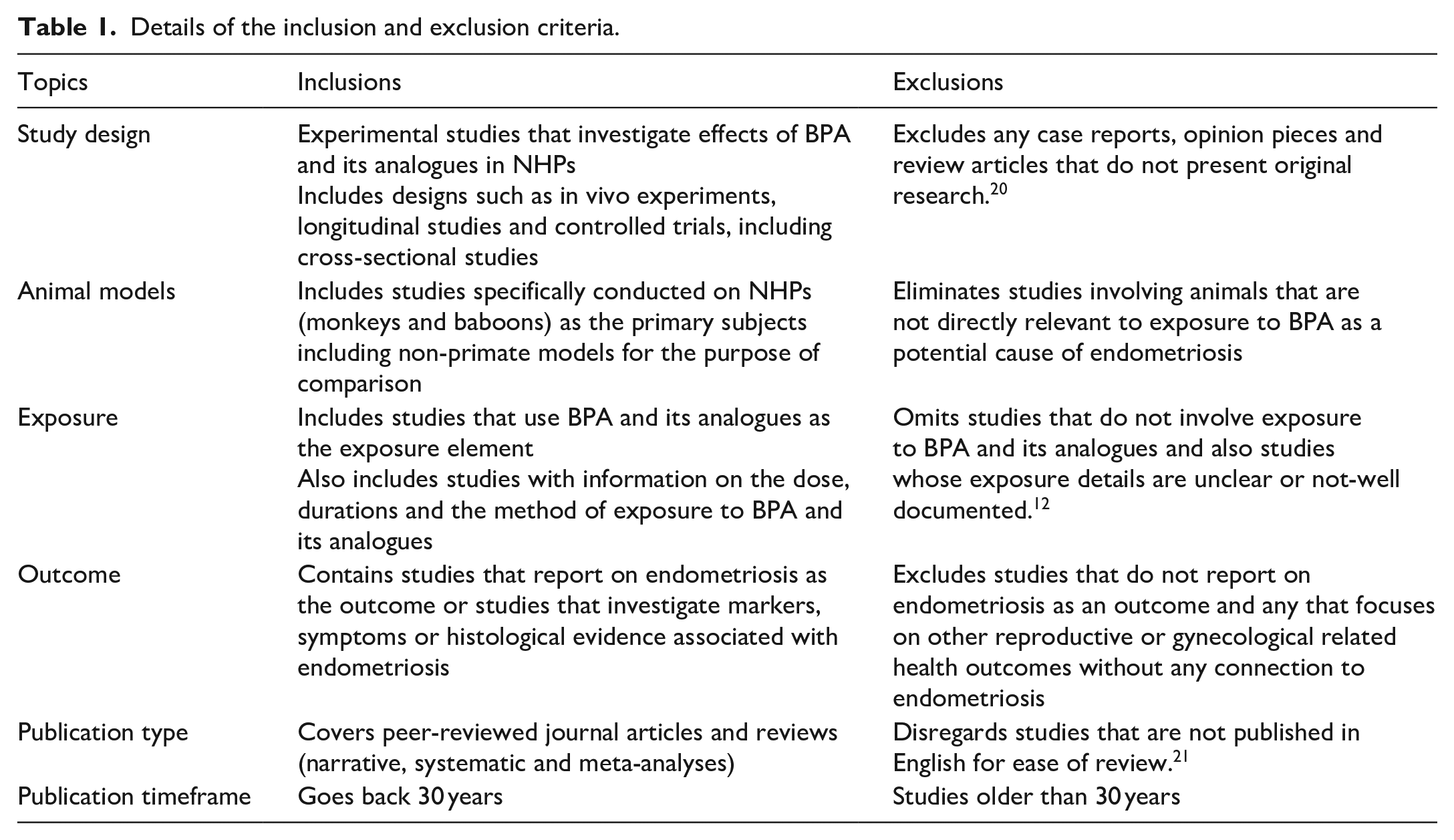
BPA and its analogues as potential cause of endometriosis
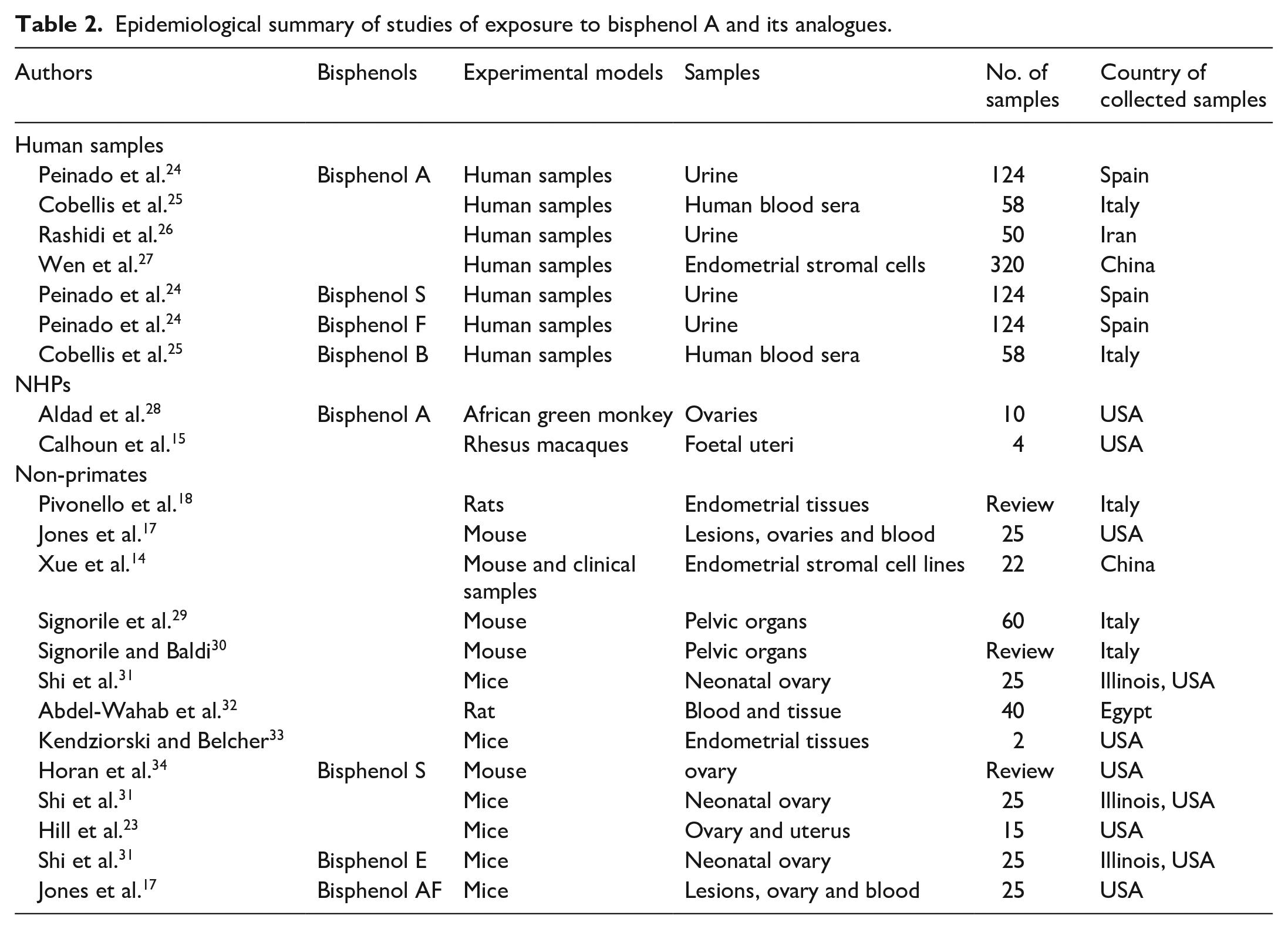
As defined by the World Health Organisation (WHO), EDCs are exogenous substances that alter the function(s) of the endocrine system and cause adverse effects on the organism, its progeny and subpopulations. 22 EDCs are thought to contribute to the pathogenesis of endometriosis, via their ability to mimic the hormone functions of the endocrine system. BPA and its analogues are EDCs that are universally present among common consumer products. 23 Reports indicate that a large amount of natural and man-made chemical toxins are present within our surroundings, hence both animals and humans are exposed to them. 22 Table 2 provides a summary of epidemiological and experimental studies of exposure to BPA and its analogues, whereas Table 3 summarises studies on exposure to BPA and its analogues using human samples.
Epidemiological summary of studies of exposure to bisphenol A and its analogues.
Summary of studies on exposure to BPA and its analogues using human samples.

The female reproductive system is very sensitive to hormonal imbalances and modifications of the endocrine system’s functions. 35 Generally, BPA is a well-studied EDC and can interfere with human hormonal balance even at long-lasting low-dose exposure. 36 Upon exposure, as a selective Estrogen Receptor (ER) modulator, BPA has been shown to bind to estrogen, thyroid, glucocorticoid, peroxisome proliferator-activated receptors and steroid enzymes among other molecular targets. 37 It then interferes with the gene expression of several estrogen (ER) receptor subtypes (ERα, ERγ and ERβ) and can subsequently cause pleiotropic effects on the reproductive system, behaviour and metabolism. 38 BPA interrupts the foetal pulsatile secretion of Gonadotrophin-Releasing Hormone (GnRH) which negatively affects the function of the hypothalamic-ovarian axis. 36 As a result, continuous pre-and postnatal exposure induces future structural changes to the female reproductive system (ovary and uterus), leading to abnormalities and possibly diseases like endometriosis. Additionally, several BPA alternatives (BPS, BPB, BPF and BPE) have also been implicated in having similar effects to BPA on the endocrine system, thereby negating their safety. Among the analogues, 39 tetramethyl bisphenol F (TMBPF) is reported to be the most recently produced. 40
Use of animal models for BPA exposure
NHP models for BPA exposure
Studies on the effects of BPA exposure using NHPs have been done using both baboons (Old World Monkeys) and macaques (New World Monkeys). However, the exposure of BPA analogues has not been reported using NHPs but only in mouse models.
17
Studies on BPA have been shown to impact the midbrain dopamine neurons and hippocampal spine synapses in NHPs.
41
The results showed that exposure to BPA induced abnormalities in the foetal ventral mesencephalon and hippocampus in two months pregnant rhesus monkeys. Consequently, the same study showed a reduction in spine hippocampal synapses among juvenile vervet monkeys upon BPA exposure. Various research has revealed that consistent developmental exposure to EDCs (including BPA) interferes with the response of animal models to hormonal challenges later in life. Upon BPA and its analogues, Kathryn
Summary of studies on exposure to BPA and its analogues using NHP models.
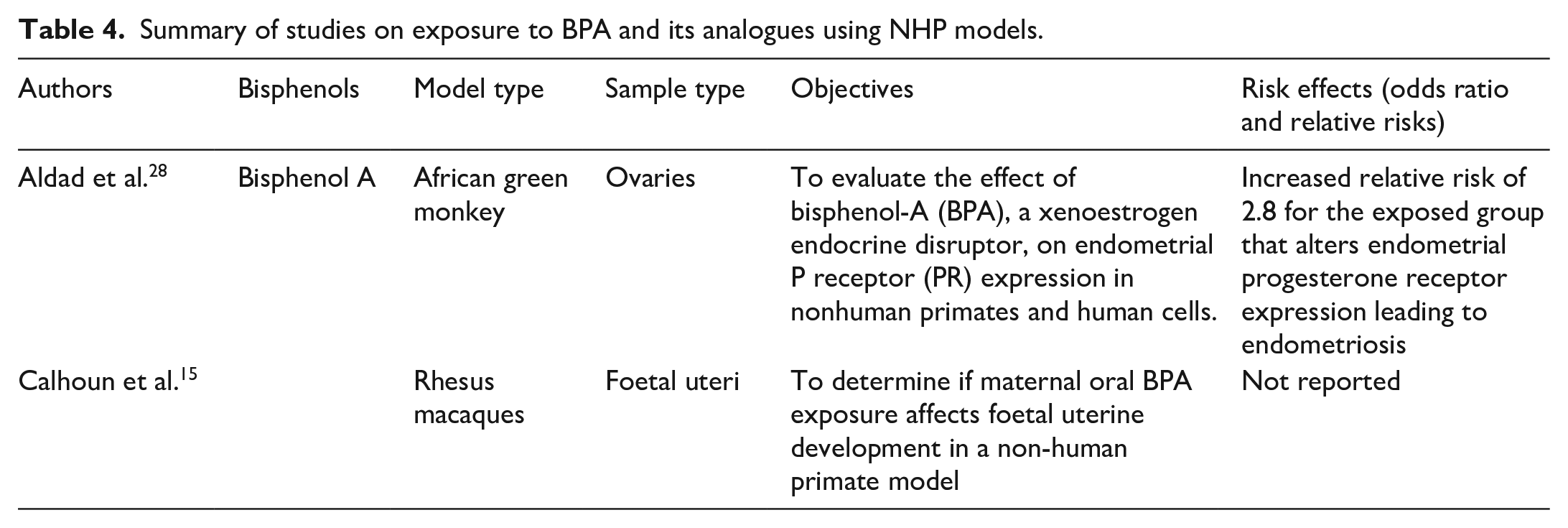
NHPs for BPA exposure as a potential cause of endometriosis
Early studies on the effects of 2,3,7,8-tetrachlorodibenzo-
Discussion
The review performed a literature search of experimental animal studies which examined the EDC of BPA and its analogues. While several studies monitored non-primate animals, the association between these endocrine disrupters’ exposure and the female reproductive function focusing on endometriosis has not been explored in NHPs. 46 Here, we have reviewed the current literature on BPA with its analogues and summarised the current state of knowledge.
Due to increased industrialisation, thousands of man-made chemicals have been developed with few undergoing rigorous safety assessments before commercial use. Ubiquitous exposure to these compounds, many of which act as EDCs, including BPA, has been suggested to contribute to the increasing incidence of numerous diseases, including endometriosis. 5 BPA exposure has been associated with an increase in the body’s inflammatory reactions. 47 Inflammation plays a crucial role in the development of lesions, adhesions and discomfort in endometriosis. Additionally, BPA exposure has been linked to epigenetic changes, which can affect gene expression without altering the underlying DNA sequence. Endometriosis progression and growth may be influenced by epigenetic modifications that lead to the dysregulation of genes involved in hormones and immunological function. 48
Many EDCs including BPA have been confirmed to dysregulate the immune system. 47 This potentially impairs the body’s ability to recognise and destroy aberrant endometrial tissue. The development and function of the female reproductive tract depend on hormone concentrations and balance. Endocrine disruptors interfere with the synthesis, metabolism and action of hormones, leading to the dysregulation of normal physiological processes and potentially promoting the development of diseases. Endometriosis is intimately associated with steroid metabolism and associated pathways, corresponding to the dominant roles estrogen receptors (ESRs) and progesterone receptors (PGRs) play in uterine biology. 12 Therefore, environmental toxicants that either mimic estrogen or enhance estrogenic exposure in the endometrium are thought to increase the risk of endometriosis. BPA as an endocrine disrupter has been reported to exert estrogenic activity which may interfere with the female reproductive system. 16
Animal studies show the association between exposure to BPA and the development of endometriosis. The action of BPA is reported to be similar to the action of estrogens in animals. Additional epidemiological studies have indicated that BPA exposure may potentially be associated with alterations in hormone levels and impairment of ovary and uterine functions. 35 Despite several studies on endometriosis using NHPs, 49 few studies have assessed the relationship between endometriosis and BPA in these animals. 28 BPA as a known endocrine disruptor has been shown to be a reproductive toxicant in animal models, its structural analogues are increasingly being used in consumer products. However, these analogues may exert similar adverse effects on the reproductive system and their epidemiological studies and toxicological data are still scarce.
Most of the world population is still widely exposed to BPA, due to its large use in the production of polycarbonate plastic and its release into foods and beverages. Recently, the prevalence of BPA analogs in the environment, foods, consumer products and human urine samples have been reported.
50
With high degrees of structural similarities to BPA, these analogues may potentially have a similar endocrine-disrupting capacity and the potential to exert adverse effects on the reproductive system. Numerous studies have investigated the reproductive toxicity of BPA and its analogues in rodents. Unlike rodents, an animal model that more reliably simulates endometriosis pathogenesis and pathophysiology is only possible in NHPs.
51
Studies indicate that amongst the NHPs, olive baboons (
Conclusion
Though endometriosis is a common disorder of unknown etiology, research indicates a relationship between bisphenol A exposure as well as its concentration in body fluids and the occurrence of endometriosis in women. The studies reviewed here provide insufficient toxicological and epidemiological data to characterise and determine the reproductive effects of BPA and its analogues.
2
The potential mechanisms of BPA and its analogues in contributing to human disease are unclear. Thus, considering that the incidence and/or prevalence of reproductive health problems associated with endocrine disruption has increased worldwide, further detailed studies using NHPs, in particular,
Footnotes
Acknowledgements
The completion of this review was made possible by the joint efforts and contributions of many people and organisations. We extend our heartfelt thanks to the academics and scientists whose work established the groundwork for the studies covered in this review. Their commitment to advance research in the subject has helped shape our understanding of the possible linkages between Bisphenol A with its analogues and endometriosis in non-human primates.
Data availability statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
