Abstract
Background:
Symptom tracking for endometriosis has been validated with clinical benefits, however, there is limited evidence around the use of mobile apps for endometriosis symptom tracking.
Method:
We performed a survey of people with suspected or confirmed endometriosis in Aotearoa New Zealand to assess mobile app use for symptom tracking including which app is being used, how frequently symptoms are tracked, which features are most important and which features would be desired. We also explored willingness to share symptom data with clinicians and/or researchers.
Results:
A total of 188 survey responses were included. Mobile apps were used for symptom tracking by 83/188 (44.1%), with only 13 of 188 (6.9%) reporting they would not consider use of an app. Of current app users, 51.5% reported logging symptoms at least weekly. The most frequently desired features included tracking of specific symptoms (such as periods, pain, bowel symptoms, mental health symptoms), other tracking (such as medications and diet) and general app usability. Of those who use or would consider using an app 77.7% reported they would be comfortable sharing data with clinicians, and 76.1% reported they would be comfortable sharing anonymous data with researchers.
Discussion:
Almost half of participants reported using an app to track symptoms, and almost all reported they would consider use. Around three in four patients would be willing to share this data with clinicians and researchers, and therefore further focus on the utility of these apps may benefit patients directly, their relationship with healthcare providers and be utilised for further endometriosis research.
Background
Endometriosis is a common condition affecting women and gender diverse people. Worldwide, the prevalence of endometriosis is estimated at 10%. 1 Aotearoa New Zealand may be similar to Australia, where data suggests that 6%–11% of women are affected. 2 The gold standard for diagnosis of endometriosis is surgery,1,2 which makes estimating prevalence complex, as it is estimated around half of women with suspected endometriosis undergo surgery. 2 In many cases surgery may not be the recommended first line treatment; a provisional diagnosis of endometriosis is typically made on clinical history, examination and imaging modalities such as ultrasound or MRI.1,3,4 Clinical suspicion of endometriosis is an inconsistent predictor of disease; of women undergoing laparoscopy for suspected endometriosis, 18%–77% have surgically confirmed disease. 5 The nature and severity of symptoms varies widely between patients, with little correlation between surgical extent of disease and patient reported symptoms.1,3 The two most common reasons treatment is sought is pain and infertility. 1 Pain can include period pain (dysmenorrhoea), pain with intercourse (dyspareunia), pain related to bowel motions (dyschezia), chronic pelvic pain and others; patients also often report bowel symptoms such as bloating and diarrhoea, or bladder symptoms such as pain with urination, and a wide range of other symptoms.1,3
Keeping track of this complex array of endometriosis related symptoms has been the topic of previous studies, and symptom diaries have been validated. 6 There is limited research, however, of symptom diaries in mobile application form. 7 Numerous mobile applications have been developed for recording health symptoms in general 7 ; however as endometriosis symptoms are typically related to the menstrual cycle, the ability to track cycles and relate cycles to other symptoms is critical. Despite this, the crossover between pain and symptom tracking apps and menstrual tracking apps to date is limited. 8
The utility of mobile apps for self-management of chronic pain and other chronic conditions has been demonstrated.9,10 The mobile application platform enhances the use of symptom diaries by being easily accessible, allowing reminders to log entries and allowing real time visualisation and summaries of data, including interactive graphs and calendars. In addition, relevant information can be suggested to the user. Where patients choose to share data with clinicians, longer term data can be used to enhance the patient-physician relationship and guide treatment options, or review response to treatment. 11 Furthermore, the collection of pain and other symptom data over time in the research domain is notoriously complex to collect; where patients are already recording this information and consent to participation in research, there is potential for anonymised data to be used as a tool to assess treatments and therapies. Such apps frequently lack input from users, clinicians and researchers when being developed,8,12 and therefore may be of limited use in one or more of these settings.
Our study examines whether patients with endometriosis in Aotearoa New Zealand are currently using mobile apps to track endometriosis symptoms; what symptoms they are tracking; how effective they perceive the apps to be; and whether they would feel comfortable sharing information with clinicians or researchers.
Methods
An online survey was created using the Qualtrics platform. The full survey is included in Supplemental Appendix 1.
The survey and study were reviewed and received ethical approval by the Human Research Ethics Committee at the University of Otago (Reference H22/155). Consultation with Māori was also undertaken through the Kaitakawaenga Rangahau Māori at the University of Otago, prior to distribution of the survey. The survey was assessed by the University of Otago Department of Obstetrics, Gynaecology and Women’s Health Endometriosis Community Advisory Group. The group gave feedback on the suitability of the survey questions and additional questions to ask from their own experiences.
The survey was distributed by email via New Zealand patient support networks: Endometriosis NZ, Insight Endometriosis and Endo Warriors Aotearoa. The survey was opened on 15 January 2023 and closed on 29 January 2023.
Inclusion criteria included: either a surgical diagnosis of endometriosis or a suspicion of endometriosis based on symptoms (self-reported by the participant); age 16 or older; and pre-menopausal. After obtaining consent and determining eligibility, the survey collected demographic information as free text and non-mandatory fields including age, ethnicity and gender.
Participants were asked whether they currently use an app to track pain and other endometriosis symptoms. Those that currently use apps were asked which applications they use, a rating (1–5), how frequently they track symptoms, what the best features of the app are; what features they would like to see, and whether they currently share information from the app with healthcare providers. Those that don’t currently share information with healthcare providers were asked if they would feel comfortable doing this to help guide their own treatment. The final question asked whether they would feel comfortable sharing non-identifiable information with research teams to help endometriosis research.
Those that did not report current use of an app were asked whether they use paper or another type of journal or diary to track symptoms. They were also asked whether they would consider using a mobile app if it could track what they wanted. Those that reported they would consider using an app were asked what the most important features would be; and whether they would feel comfortable sharing information with healthcare providers to guide their own treatment, and non-identifiable information with research teams to help endometriosis research.
Free text results were read, and content analysis was used to group responses into themes. Descriptive statistics were reported as frequencies, and percentages with 95% confidence intervals and bivariate analyses were carried out using Microsoft Excel.
Results
Participant characteristics
A total of 193 responses were received; five of these were excluded as they did not fit the inclusion criteria outlined above (two respondents answered ‘no’ to a surgical or clinical diagnosis of endometriosis; three did not respond to one or more of the inclusion criteria questions). Therefore 188 responses were included in the analysis.
Age ranged from 16 to 47 years (mean 29.7, median 29, standard deviation 6.8). Gender (recorded as free text) was reported as female for 178 responses (94.7% [91.4%–97.9%]), male for 2 responses (1.1% [0%–2.5%]) and ’non-binary’, ‘gender diverse’ or another gender for 8 responses (4.3% [1.4–7.1%]).
Ethnicity was reported by respondents as Māori (n = 23, 12.2% [7.5%–16.9%]), Pacific (n = 4, 2.1% [0.1%–4.2%]), NZ European (n = 162, 86.2% [81.2%–91.1%]) or another ethnicity (n = 21, 11.2% [6.7%–15.7%]). More than one ethnicity was able to be selected.
A surgical diagnosis of endometriosis was reported by 125/188 (66.5% [59.7%–73.2%]) of respondents, with 63/188 (33.5% [26.8%–40.3%]) reporting suspected endometriosis.
Current users of mobile symptom tracking apps
Current use of mobile apps was reported for symptom tracking by 83/188 (44.1% [37.1%–51.2%]) respondents. By diagnosis this included 55/125 (44.0% [35.3%–52.7%]) respondents with a surgical diagnosis of endometriosis, and 28/63 (44.4% [32.2%–56.7%]) respondents with suspected endometriosis.
Frequency of logging symptoms was answered by 68/83 (81.9%) of respondents. Symptoms were logged multiple times daily by 4/68 (5.9% [0.3%–11.5%]), daily by 12/68 (17.6% [8.6%–26.7%]), multiple times weekly by 14/68 (20.6% [11.0%–30.2%]), weekly by 5/68 (7.4% [1.1%–13.6%]), multiple times per cycle by 18/68 (26.5% [16.0%–37.0%]) and monthly by 15/68 (22.1% [12.2%–31.9%]). This information is shown graphically in Figure 1.
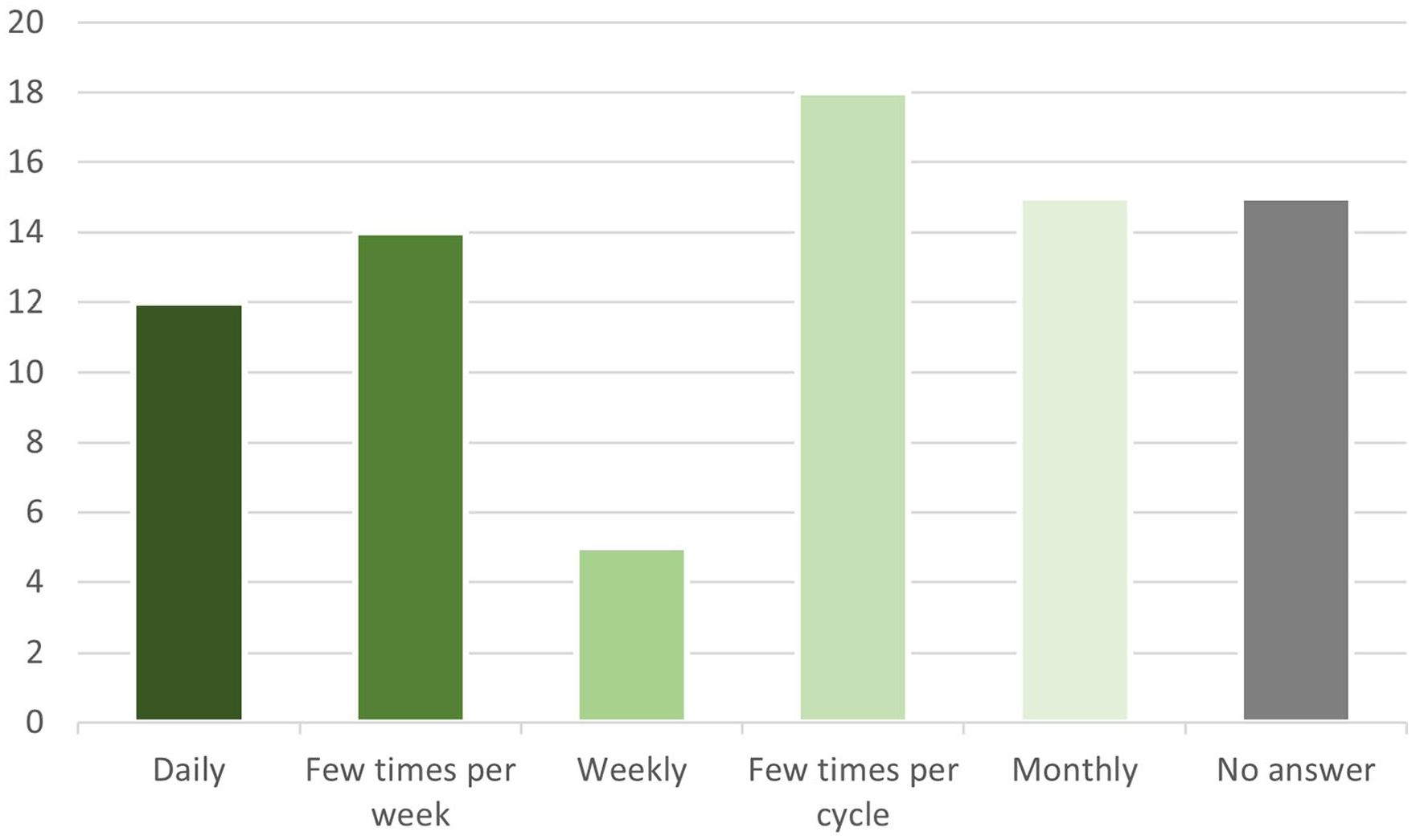
Frequency of symptom tracking amongst patients with confirmed or suspected endometriosis who use mobile apps for symptom tracking (vertical axis – number of patients; horizontal axis – frequency of app tracking).
When asked which mobile app is used, 61 of the 83 respondents using apps provided a response. The most common mobile apps used to track symptoms were ‘Flo’ (n = 19; 31.1% [19.5%–42.8%]) and ‘Clue’ (n = 15; 24.6% [13.8%–35.4%]), which were given an average rating of 3.5/5 and 3.9/5 respectively. Less frequently reported apps included: ‘Period Tracker’ (n = 5; average rating 3.8/5); ‘Apple Health’ (n = 5; average rating 4.4/5); ‘Fitbit’ (n = 3; average rating 3.7); ‘Fertility Friend’ (n = 2; average rating 4.0); ‘Qendo’ (n = 2; average rating 4.5/5); and a further ten applications not reported by more than one respondent (‘b’, ‘Garmin connect’, ‘Life’, ‘Maya’, ‘Medisafe and Manage My Pain’, ‘My Calendar Period Tracker by Simple Design’, ‘Ovia’, ‘Qualtrics’, ‘Samsung Health’ and ‘Taking Charge of your Fertility’).
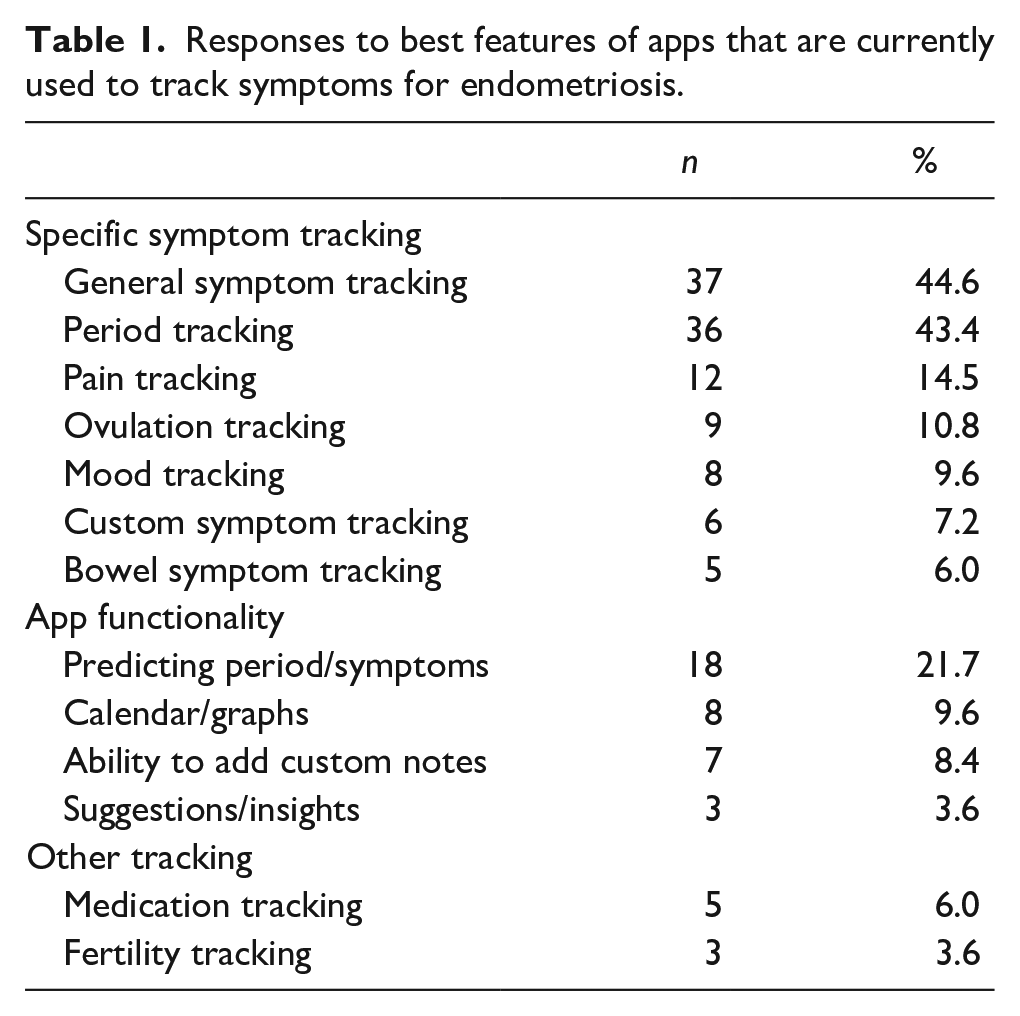
When asked the best features of currently used apps, responses generally fell into three categories: specific symptom tracking, app functionality and other tracking. The three most common responses were general symptom tracking (37/83; 44.6% [33.9%–55.3%]), period tracking (36/83; 43.4% [32.7%–54.0%]) and prediction of periods/symptoms (18/83; 21.7% [12.8%–30.6%]). All responses are outlined in Table 1.
Responses to best features of apps that are currently used to track symptoms for endometriosis.
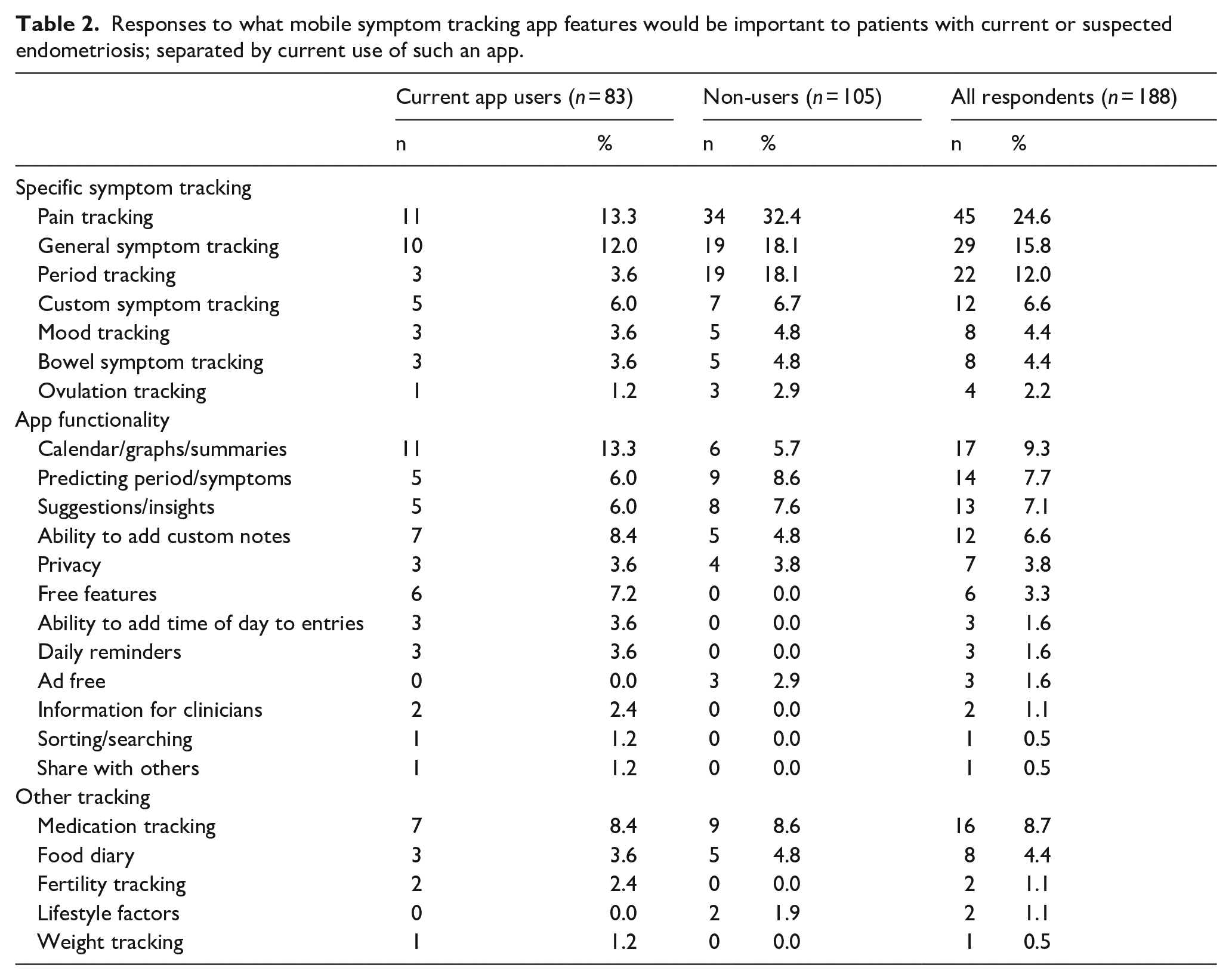
When asked what features respondents would like to see, the most common three responses were ‘Pain tracking’ (n = 11; 13.3% [6.0%–20.5%]), ‘Calendars/Graphs/Summaries’ (n = 11, 13.3% [6.0%–20.5%]) and ‘General symptom tracking’ (n = 10; 12.0% [5.0%–19.1%]). All responses are shown in Table 2, again grouped as specific symptom tracking, app functionality and other tracking.
Responses to what mobile symptom tracking app features would be important to patients with current or suspected endometriosis; separated by current use of such an app.
When asked if a previous mobile had been used, there were 15 responses. Previously used apps included ‘Flo’ (n = 8), ‘Clue’ (n = 3), ‘Fitbit’ (n = 2), ‘Apple Health’ (n = 1) and ‘Kindara’ (n = 1). Reasons for stopping use were listed as lack of features (n = 6, 40.0% [15.2%–64.8%]), cost (n = 5, 33.3% [9.5%–57.2%]), Ease of use (n = 5, 33.3% [9.5%–57.2%]) and one respondent reported each of privacy concerns, not enough focus on endometriosis and no ability to predict cycles/symptoms.
Non-users of mobile symptom tracking apps
Of those who don’t currently use a mobile tracking app, 92/105 (87.6% [81.3%–93.9%]) reported they would consider using an app if it tracked symptoms well.
When asked what the most important features of a mobile symptom tracking app would be, the most common responses were ‘Pain tracking’ (n = 34; 32.4% [23.4%–41.3%]), ‘General symptom tracking’ (n = 19; 18.1% [10.7%–25.5%]) and ‘Period tracking’ (n = 19; 18.1% [10.7%–25.5%]). All responses are shown in Table 2.
Use of a paper or other journal
Use of a paper or other journal was reported by 53/188 (28.2% [21.8%–34.6%]) respondents; those who did not use a mobile app were significantly more likely to use a paper or other journal, compared with those who did use a mobile app (37/105, 35.2% [26.1%–44.4%] vs 16/83, 19.3% [10.8%–27.8%], p = 0.016).
Sharing of information
When asked about sharing information from a symptom tracking app with a healthcare professional to guide their treatment, of those using an app 39/83 (47.0% [36.3%–57.7%]) reported currently sharing data from the app they use and 64/83 (77.1% [68.1%–86.1%]) reported they would feel comfortable sharing such data. Of respondents who do not currently use an app, 82/105 (78.1% [70.2%–86.0%]) said they would feel comfortable sharing this data if they were to use an app (there was no significant difference in comfort with data for healthcare professionals between app users and non-app users [p = 0.872]). In total, 146/188 (77.7% [71.7%–83.6%]) would feel comfortable sharing information with healthcare professionals.
When asked about sharing information from a symptom tracking app in a non-identifiable way with research teams for endometriosis research, 62/83 (74.7% [65.3%–84.1%]) of current app users and 81/105 (77.1% [69.1%–85.2%]) of non app users (no significant difference in comfort with data for research between app users and non-app users, p = 0.697, total 143/188; 76.1% [70.0%–82.2%]) reported they would feel comfortable sharing this information.
Discussion
The findings of this survey suggest that almost half of people with confirmed or suspected endometriosis are currently using mobile apps to track their symptoms; with many more reporting they would considering using an app if it tracked symptoms well; only 6.9% (13 of 188 respondents) reported they would not use an app. Of those who currently use an app, 23.5% reported logging symptoms at least daily, and 51.5% reported logging symptoms at least weekly. Of those who use or would consider using an app 77.7% reported they would be comfortable sharing data with clinicians for their own management, and 76.1% reported they would be comfortable sharing data with researchers.
The vast majority of applications listed are not specific endometriosis symptom tracking apps; the most commonly listed apps (Flo, Clue and Period Tracker) are period tracking apps, with many of the remainder being general health tracking apps that have period tracking features (Apple Health, Fitbit, Samsung Health). The only endometriosis specific app reported was Qendo (n = 2), which was also the highest rated app used by more than one respondent. The most desired features were relatively consistent amongst those currently using apps and those who would consider using apps, with pain tracking, general symptom tracking and period tracking being the three most common answers. Other desired features were broadly broken down into specific symptom tracking (such as bowel symptoms and mood symptoms), other tracking (such as medication, food and weight) and improved application function (including calendars and graphs, suggestions and insights, daily reminders, ability to customise symptoms and add custom notes, free features and being ad free and improved privacy). The most common reasons to stop the use of an app were lack of features, cost of the app and the app not being easy to use. Studies have repeatedly shown that consumer input into mobile app development is often under-utilised7,8; increased availability and awareness of high quality and free endometriosis specific symptom tracking apps should place high value on consumer input to ensure that these features and others important to consumers are addressed.
The results of the survey raised the issue of barriers to access to apps, with cost being listed as a barrier and improved free features and ad-free apps being listed as desired features by both those who currently use an app and those who would consider use. As the utility of these apps increases and becomes an important health tool, the issue of equity becomes a concern if cost is a barrier. Our study is not adequately powered to comment on differences in ethnicity with app access, however, we do note that Māori and Pacific patients reported less use than non-Māori/Pacific.
Obtaining clinically and/or scientifically useful pain data over time is notoriously difficult; if patients are already recording this information in mobile apps for their own use and are willing to share the data, there is a great potential for clinicians and researchers to utilise this data. Multidisciplinary clinics for the management of endometriosis are increasingly being utilised,3,13,14 the use of long term symptom data across multiple domains (menstrual, pain, bowel, diet and psychological) presents significant potential to enhance patient management in this setting, as well as assess response to specific treatments. In a research domain, such data could be utilised to explore symptom patterns and interactions, correlate to surgical phenotyping or other investigations, assess response to treatment and a range of other possibilities. Further research into data extraction from such apps and utility in clinical and research domains is needed.
The present study is a small patient survey and therefore has several limitations. Subjects were recruited by patient advocacy and support groups in Aotearoa New Zealand, and therefore the results may not be reflective of other populations. Respondents to an online survey may represent a population that are more likely to utilise phone apps and therefore the true proportion of people with endometriosis who utilise an app may be overestimated. The survey used free text data with content analysis, and therefore the full extent of some responses may not be fully reflected. This study was not powered to demonstrate use of mobile apps across different ethnic groups, gender diverse groups, age brackets or disabilities; all of which should be considered if use increases in clinical or research domains.
This survey suggests that nearly half of people with suspected or confirmed endometriosis currently utilise a symptom tracking mobile app; and that nearly all would consider use of an app. Just over three in four would be comfortable sharing information with clinicians to help guide their own treatment, or with researchers to contribute to endometriosis research. Current use is largely limited to general period tracking apps rather than endometriosis specific apps, with the most frequently desired features being tracking of specific symptoms (such as periods, pain, bowel symptoms and mental health symptoms), other tracking (such as medications and diet) and general app usability.
Supplemental Material
sj-pdf-1-pev-10.1177_22840265231193486 – Supplemental material for A survey exploring the use of symptom diary mobile apps for endometriosis
Supplemental material, sj-pdf-1-pev-10.1177_22840265231193486 for A survey exploring the use of symptom diary mobile apps for endometriosis by Simon M Scheck, Bryndl Hohmann-Marriott, Gary Faughnan and Claire Henry in Journal of Endometriosis and Pelvic Pain Disorders
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
