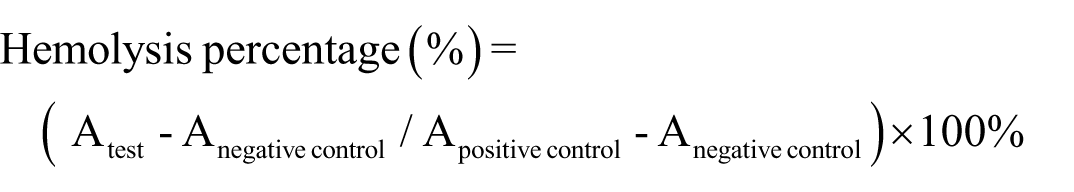Abstract
The management of critical-sized bile duct defects remains a significant clinical challenge, as current options like autologous grafts and synthetic stents provide mechanical support but lack the bioactivity needed for functional regeneration. To address this, we developed a novel tissue-engineered artificial bile duct using a photocrosslinkable gelatin methacryloyl-sodium alginate (GelMA-SA) hybrid hydrogel. This biomimetic artificial bile duct synergizes the superior cell-adhesion of GelMA with the mechanical robustness of SA. A systematically optimized GelMA-SA formulation demonstrated ideal physicochemical properties, including suitable mechanical strength, controlled degradation kinetics, and a conducive microarchitecture for cell growth. Comprehensive in vitro studies confirmed the excellent biocompatibility of artificial bile duct, promoting the proliferation of biliary epithelial cells. Our findings establish this GelMA-SA hybrid hydrogel-based artificial bile duct as a promising repair solution. It effectively bridges the gap between structural support and biological functionality, offering a versatile platform technology for biliary reconstruction and tubular tissue engineering.
Introduction
The biliary system, particularly the extrahepatic bile duct, serves the critical physiological function of conveying bile from the liver to the duodenum, which is indispensable for lipid digestion and the elimination of metabolic waste. 1 However, this vital conduit is susceptible to a range of pathologies, including cholangiocarcinoma, benign strictures, traumatic injury, and congenital disorders like biliary atresia, which can result in significant tissue loss or obstruction.2,3 The subsequent morbidity and potential mortality underscore the severity of biliary diseases and highlight a formidable challenge in modern hepatobiliary surgery: the reconstruction of long-segment bile duct defects. When primary end-to-end anastomosis is not feasible, the clinical options become severely limited, creating a pressing and unmet need for effective biliary restoration strategies.
The current therapeutic arsenal for managing significant bile duct defects remains suboptimal, with each strategy harboring inherent drawbacks that ultimately hinder functional tissue regeneration and long-term patency. The clinical gold standard often involves the use of autologous tissue grafts, such as vascularized jejunal or gallbladder patches.4–7 While providing a viable conduit, this approach is plagued by donor site morbidity, limited tissue availability, and fundamental pathophysiological mismatches.7,8 The implantation of intestinal mucosa into the biliary environment frequently incites chronic inflammation, mucus hypersecretion, and biliary sludge accumulation, predisposing patients to recurrent cholangitis, stone formation, and anastomotic strictures. Interposition of an artificial bile duct (ABD) in place of the resected extrahepaticbile duct could reconstruct the physiological biliary tract, which may solve the various problems associated with choledochojejunostomy. 9 Attempts at developing ABD have been made for a long time. Commonly used non-degradable biomedical materials for ABD include polytetrafluoroethylene, Dacron, silk, cobalt-chromium-molybdenum alloy, fluororubber, and non-degradable polyurethane. 10 Long-term implantation of non-degradable ABD in vivo inevitably leads to fibrous tissue hyperplasia around the implantation site, resulting in intimal hyperplasia and biliary stricture, however, an ABD has not been successfully put into clinical use yet.
To fulfill the stringent requirements for an ideal ABD, the selection of an appropriate biomaterial is paramount. In this regard, gelatin methacryloyl (GelMA) has garnered significant attention due to its inherent bioactivity.11,12 Derived from collagen, the predominant component of the native extracellular matrix, GelMA retains cell-adhesive motifs (e.g., RGD sequences), which profoundly support cell attachment, proliferation, and viability.13,14 Furthermore, its photo-crosslinkable nature permits precise fabrication of complex 3D structures via UV-light. 15 However, pristine GelMA hydrogels often suffer from relatively low mechanical strength and rapid degradation kinetics, which are inadequate for providing sustained mechanical support in the dynamic biliary environment.16,17 To address these limitations, we introduced sodium alginate (SA), a natural polysaccharide renowned for its excellent biocompatibility and superior mechanical properties. 18 SA can form robust hydrogels and effectively slow down the degradation rate. Nevertheless, its inherent bio-inertness and lack of specific cell-binding sites often result in poor cell-material interactions.19,20 Therefore, we hypothesize that a strategic hybrid of GelMA and SA will yield a synergistic hydrogel system. The exceptional bioactivity of GelMA and the enhanced mechanical robustness and controlled degradation imparted by SA, thereby creating an optimal biomimetic microenvironment for biliary tissue regeneration.
Building upon the rational design of the GelMA-SA hybrid hydrogel, this study aims to develop, fabricate, and comprehensively evaluate a novel ABD for the regeneration of critical-sized biliary defects. We hypothesize that the GelMA-SA hybrid hydrogel will provide an optimal three-dimensional microenvironment that synergistically combines enhanced mechanical integrity with superior bioactivity, thereby effectively facilitating rapid and functional bile duct regeneration in vivo. To rigorously test this hypothesis, our research is structured around three specific aims. First, we will systematically fabricate a series of GelMA-SA hydrogels with varying compositional ratios. These materials will be thoroughly characterized to assess their key physico-chemical properties, including microstructure, swelling behavior, mechanical strength, and in vitro degradation profiles, to identify the most optimal formulation. Second, the in vitro biocompatibility and biofunctionality of the selected hybrid scaffolds will be evaluated using biliary epithelial cells.
In conclusion, this work presents the first comprehensive investigation into a GelMA-SA hybrid hydrogel-based ABD, representing a significant stride towards a regenerative solution for biliary defects. We anticipate that this biomimetic scaffold will not only address the critical limitations of current therapies but also establish a versatile platform technology with potential applications in other tubular tissue engineering realms, such as vascular and tracheal reconstruction.
Materials and methods
Materials
GelMA (EFL-GM-90) was provided by Suzhou Engineering For Life Co., Ltd. (China). Sodium alginate ((C6H7NaO6)n) and Calcium chloride (CaCl2) were purchased from Beijing Solarbio Science & Technology Co., Ltd. (China). The photoinitiator 2-hydroxy-1-[4-(hydroxyethoxy)phenyl]-2-methyl-l-1-propanone (Irgacure 2959, ≥98.0%, MW = 224.25 g/mol) and the bile juice (pH 7.4) were obtained from Shanghai Yuanye Bio-Technology Co., Ltd. (China). The CCK8 detection kit and Calcein/PI Live/Dead Viability/Cytotoxicity Assay Kit were provided by Shanghai Biyuntian Biotechnology Co., Ltd. (China).
Preparation of GelMA-SA hybrid hydrogel
The GelMA-SA hybrid hydrogel had two components: 10% (w/v) GelMA and 1% (w/v) SA, 10% (w/v) GelMA and 2% (w/v) SA. First, 1 mL 10% (w/v) GelMA was dissolved in distilled water at 37°C and then added 0.5 mL of 1% (w/v) or 2% (w/v) SA. Next, 0.5% (w/v) photo-initiator (Irgacure 2959) was added into the mixed solution for inducing photocrosslinking. 10% (w/v) CaCl2 solution was prepared as an ionic crosslinking agent. All the solutions were sterilized and stored at 4°C before usage.
Preparation of GelMA-SA hybrid hydrogel ABD
The ABD molds customized by Shenzhen Xinglong Hardware and Plastic Products Co., Ltd. (China) with an inner diameter of 3 mm and an outer diameter of 5 mm and an inner diameter of 5 mm and an outer diameter of 8 mm. The customization of ABD molds in a range of sizes is designed to meet the diverse repair requirements of bile duct injuries.
Different concentrations of hybrid hydrogel including GelMA (10% w/v) and GelMA-SA (10% w/v and 1% w/v or 10% w/v and 2% w/v) were tested to generate GelMA-SA hybrid hydrogel ABD. Immediately after synthesised, GelMA and GelMA-SA hybrid hydrogel ABD were further injected into the ABD molds then polymerized by UV exposure. The ABD were exposed to UV light (365 nm wavelength, 6.5 mW cm−2 power) for 30 s to induce crosslinking of GelMA and GelMA-SA. These ABD were stored at CaCl2 solution were sterilized after UV light treatment for 5 min before use.
Physicochemical characterization of GelMA-SA hybrid hydrogel ABD
Morphology of the GelMA-SA hybrid hydrogel ABD
Take the sample and directly attach it to the conductive adhesive. Then, use the Quorum SC7620 sputtering coating instrument to apply gold plating for 45 s, with a gold plating current of 10 mA. Subsequently, use a scanning electron microscope (SEM, ZEISS Sigma 360, Germany) to capture the morphology morphologies of GelMA-SA hybrid hydrogel ABD. During the morphology capture, the acceleration voltage is 3 kV.
Chemical structure characterization of GelMA-SA hybrid hydrogel ABD
Fourier transform infrared spectroscopy (FTIR, Thermo Fisher Scientific Nicolet iS20, USA) was utilized to identify the presence of various functional samples in the GelMA-SA hybrid hydrogel ABD. The resolution is 4 cm−1, the scanning times are 32 and the test wavenumber range is 400–4000 cm−1.
Compression test of GelMA-SA hybrid hydrogel ABD
The GelMA-SA hybrid hydrogel ABD were removed from the ABD molds and stored in PBS at 4ºC for 24 h to achieve equilibrium swelling. The samples were then compressed at a rate of 1 mm/min using a biomechanical testing machine (Meters Industrial System CMT6103, China).
In vitro swelling and degradability test of GelMA-SA hybrid hydrogel ABD
The freeze-dried GelMA-SA hybrid hydrogel ABD were weighted to obtain the dry weight (W0). Then, the lyophilized stents were immersed in 1 × PBS, and the weight (WS) of samples were measured after swelling for 30 min, 1, 2, 4, 8, 12, 24 and 48 h. The swelling ratio was calculated by the following equation 21 :
The freeze-dried GelMA-SA hybrid hydrogel ABD were immersed in 1 × PBS for 24 h to achieve swelling equilibrium which were weighed as initial weight (Wi). Then, the swelling equilibrium GelMA-SA hybrid hydrogel ABD were immersed in bile juice (PH 7.4) at 37°C, with the bile juice being replaced every 2 days. The weight (Wt) of ABD was measured after degradation for 3, 7, 14 days. The weight retention ratio was calculated by the following equation:
In vitro biocompatibility of GelMA-SA hybrid hydrogel ABD
In vitro cell cytocompatibility and proliferation assay
To evaluate the cytocompatibility of GelMA-SA hybrid hydrogel ABD, mouse extrahepatic bile duct epithelial cells (HyCyte, China) at a concentration of 5 × 105 cells/mL were seeded onto the 24-well plate and incubated for 24 h. These cells were cultured in HyCyte® mouse extrahepatic bile duct epithelial cells complete medium. Next, the cell culture medium was replaced with sample extracts, and the cells were incubated for 1 and 2 days with media changes. Live/Dead staining was conducted at days 1 and 2 days. After staining, the cells were gently rinsed with PBS three times and observed using an image scanning system (Keyance BZ-X800LE, Japan). For cell proliferation assessment, mouse extrahepatic bile duct epithelial cells cells (5 × 103 cells/well) were added to each well of a 96-well plate and incubated for 24 h. Next, the cell culture medium was replaced with sample extracts, and the cells were incubated for 1, 3, and 5 days. At each time point, 10 μL of CCK8 solution was added to each well and incubated for 2 h in a CO2 incubator. The optical density was measured at 450 nm using a microplate reader (Thermo scientific, Multiskan GO, USA), and the cell proliferation assessment was determined based on absorbance values at days 1, 3, and 5.
In vitro hemolysis test
Hemolysis assay was performed to evaluate the in vitro hemocompatibility of the GelMA-SA hybrid hydrogel ABD.22,23 Blood samples were collected from the ear marginal vein of rabbit, then red blood cells (RBCs) were separated from the rabbit blood samples by centrifugation. The RBCs were washed with sterile isotonic saline for three times, diluted to a concentration of 2% (v/v) and mixed with an equal volume of normal saline containing different concentrations of powdered GelMA-SA hybrid hydrogel ABD ranging from 400 to 1600 μg/mL. Pure normal saline and distilled water were also mixed with 2% RBCs solution and served as negative control and positive control, respectively. The mixtures were left at ambient condition statically for 4 h and then centrifuged to remove the liquid supernatants. Finally, the liquid supernatants were observed, photographed and measured by microplate reader (Thermo scientific, Multiskan GO, USA) at a wavelength of 540 nm. The hemolysis percentage (%) was calculated by following equation:
Statistical analysis
The data are expressed as mean ± SD. Statistical significance was analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. Significance was determined at *p < 0.05 and **p < 0.01. All experiments were conducted in triplicate.
Results
Fabrication and structural of GelMA-SA hybrid hydrogel ABD
Size-tunable ABD were successfully fabricated via dual-crosslinked hydrogels of GelMA and GelMA-SA precursor solutions within customized molds (Figure 1(a) and (d)). Macroscopic inspection confirmed concentric lumen geometry and uniform wall thickness for both Type I (3 mm/5 mm) and Type II (5 mm/8 mm) constructs, with no evidence of delamination or bubble entrapment (Figure 1(b) and (e)). SEM analysis of 10% GelMA ABD, Type I (3 mm/5 mm) and Type II (5 mm/8 mm) constructs respectively (Figure 1(c) and (f)), revealed an obvious tubular structure.

Fabrication and structural of GelMA-SA hybrid hydrogel ABD: (a, d) computer-aided design schematics illustrating the two standardized ABD geometries: Type I (ID 3 mm, OD 5 mm) and Type II (ID 5 mm, OD 8 mm), (b, e) macroscopic digital photographs of ABDs immediately after demolding, and (c, f) representative scanning electron micrographs of the 10% GelMA luminal surface after critical-point drying, showing a denser fibrillar microstructure; scale bar = 1 mm.
Representative SEM images (Figure 2) revealed a clear transition in surface micro-architecture with increasing alginate content. The 10% GelMA control presented a dense, sheet-like fibrillar morphology with scarce visible pores, consistent with single-network methacrylated gelatin hydrogels. In addition, unlike the smooth surface observed in the 10% GelMA group, the addition of 1% SA and 2% SA resulted in a rougher microstructure. In the 10% GelMA/1% SA group, discrete alginate‑rich nodules can be observed within the matrix (indicated by white arrows in Figure 2), and the pores appear slightly larger and more irregular, indicating a modest increase in porosity. Discrete alginate-rich nodules were formed within the matrix. Otherwise high-magnification inspection reveals that the incorporation of 2% SA led to a slight but observable increase in porosity compared to the control and 1% SA groups. Further elevating SA to 2% produced a highly interconnected porous network that is expected to facilitate nutrient diffusion and cellular infiltration during biliary tissue regeneration. These data confirm that dual-cross-linking of GelMA and alginate enables independent tuning of micro-topography while preserving the macroscopic ABD geometry (Figure 1(c) and (f)).

SEM image of GelMA-SA hybrid hydrogel ABD.
FTIR of GelMA-SA hybrid hydrogel ABD
FTIR analysis (Figure 3) confirmed successful incorporation of SA into the GelMA network. The amide II band at 1535 cm−1 remained constant across all formulations, verifying retention of the gelatin backbone. A gradual increase in absorbance at 1239 cm−1 (C–O stretching of alginate guluronate units) was observed with rising SA content. The peak shifted to the right and became narrower, while the peak at 545 cm−1 corresponding to unreacted methacrylate C=C bonds decreased only modestly, indicating efficient UV-induced cross-linking without complete exhaustion of double bonds—an observation consistent with preserved hydrogel ductility.

FTIR of GelMA-SA hybrid hydrogel ABD.
Swelling ratio and weight retention ratio of GelMA-SA hybrid hydrogel ABD
The time-dependent swelling behavior of 10% GelMA, 10% GelMA/1% SA, and 10% GelMA/2% SA hydrogels is shown in Figure 4(a). All formulations demonstrated rapid water uptake (>90% of final swelling) within the first 4 h, indicating that the ABD scaffolds can achieve full hydration shortly after implantation, a prerequisite for conformal sealing of bile duct defects. As summarized in Figure 4(b), the 10% GelMA control reached an equilibrium swelling ratio (ESR) of 587 ± 5% within 24 h. In comparison, the 10% GelMA/1% SA and 10% GelMA/2% SA groups showed slightly lower equilibrium values of 564 ± 60% and 554 ± 56%, respectively. Although the absolute differences among the three groups are moderate, statistical analysis revealed that the SA-containing hydrogels exhibited significantly reduced equilibrium swelling compared with the 10% GelMA control (p < 0.05). These results indicate that incorporation of SA into the GelMA network, particularly at higher concentration, restricts water uptake and decreases the maximum swelling capacity of the hydrogels.

(a) Swelling ratio, (b) ESR, and (c) weight retention ratio of GelMA-SA hybrid hydrogel ABD.
The weight retention ratios of the three hydrogel formulations—10% GelMA, 10% GelMA/1% SA, and 10% GelMA/2% SA—were monitored over 14 days (Figure 4(c)). All samples exhibited minor variations within the first 3 days, followed by a gradual divergence. At day 14, 10% GelMA showed the lowest retention (89.06 ± 0.89%), whereas 10% GelMA/1% SA maintained 91.33 ± 1.10%. The 10% GelMA/2% SA group demonstrated the highest stability, retaining 93.54 ± 1.02% of the initial weight, indicating minimal degradation. These findings suggest that dual crosslinking with SA substantially enhances structural stability during the degradation period.
Mechanical property of GelMA-SA hybrid hydrogel ABD
Incorporation of ionically cross-linked alginate significantly enhanced both stiffness and strength of the ABD constructs (Figure 5). The 10% GelMA control exhibited a compressive modulus of 8.4 ± 0.6 kPa. Addition of 1% SA increased compressive modulus to 38.1 ± 8.1 kP (p < 0.05), while 2% SA further raised to 59.7 ± 10.8 kPa (p < 0.01). The increased in modulus is attributed to the synergistic interpenetration of a rigid ionic alginate network within the covalent GelMA matrix, which effectively dissipates load and delays crack propagation. Then, the compressive strength of 10% GelMA ABD was 116.6 ± 5.8 kPa, 10% GelMA/1% SA and 10% GelMA/2% SA were 130 ± 0.0 kPa and 253.3 ± 30.6 kPa respectively. These values fall within the lower range of native porcine common bile duct (120–150 kPa), indicating that the 10% GelMA/2% SA formulation provides sufficient mechanical integrity to resist physiological bile pressure (~2–3 kPa) while maintaining compliance for surgical handling.
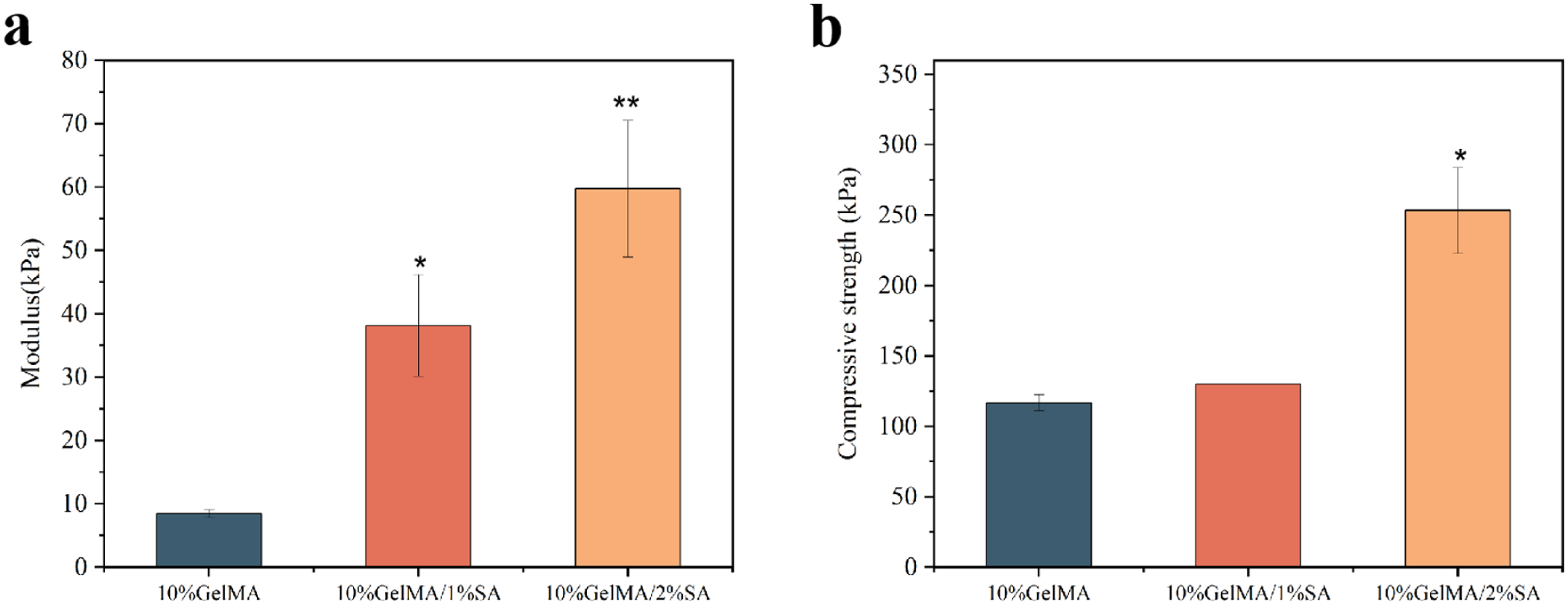
(a) Compressive modulus and (b) strength of GelMA-SA hybrid hydrogel ABD.
Cell viability assessment of GelMA-SA hybrid hydrogel ABD
Cell proliferation was evaluated by CCK-8 assay after incubation of mouse extrahepatic bile duct epithelial cells with extracts from 10% GelMA, 10% GelMA/1% SA, and 10% GelMA/2% SA ABD for 1, 3, and 5 days (Figure 6). For all three formulations, OD values increased progressively over time, indicating continuous cell growth. On day 1, OD values were low and comparable among the groups, suggesting similar initial cell viability. By day 3, OD values had increased in all groups with only modest differences between formulations. On day 5, both SA-containing groups showed slightly higher OD values than 10% GelMA, with the 10% GelMA/1% SA group exhibiting the highest mean OD and the 10% GelMA/2% SA group displaying a similar but marginally lower level. Overall, these results indicate that incorporation of SA does not compromise the viability of mouse extrahepatic bile duct epithelial cells and may modestly enhance their proliferative activity.

CCK8 analysis of GelMA-SA hybrid hydrogel ABD.
Live/Dead staining was performed to visualize cell viability and morphology in mouse extrahepatic bile duct epithelial cells (Figure 7). At both 24 h and 48 h, the majority of cells in all groups (Control, 10% GelMA, 10% GelMA/1% SA, and 10% GelMA/2% SA) exhibited strong green fluorescence, indicating high viability, while red fluorescence (dead cells) was negligible. As shown in the images, cell density increased noticeably from 24 h to 48 h across all groups, with cells displaying a healthy, spreading morphology. The fluorescence images confirm that the incorporation of SA did not induce cytotoxicity or inhibit cell attachment, as the cell density and viability in the SA-loaded groups were visually comparable to those in the Control and pure GelMA groups.

Live/dead analysis of GelMA-SA hybrid hydrogel ABD.
Hemolytic test of GelMA-SA hybrid hydrogel ABD
Extracts of 10% GelMA, 10% GelMA/1% SA and 10% GelMA/2% SA induced negligible hemoglobin release across the tested concentration range (Figure 8), with hemolysis ratios consistently below 1.2%. The absence of a dose-dependent increase and the clear separation from the positive control indicate that the UV-cross-linked hybrid networks do not adversely affect erythrocyte membrane integrity, supporting the hemocompatibility of the ABD constructs for in vivo applications.

(a) Hemolysis images and (b) hemolysis ratio of GelMA-SA hybrid hydrogel ABD.
Discussion
The management of bile duct defects remains a formidable challenge in hepatobiliary surgery, primarily due to the limitations of existing autologous grafts and synthetic prostheses, which often fail to provide a bioactive microenvironment conducive to functional tissue regeneration. In this study, we have engineered a novel ABD from a dual-crosslinkable GelMA-SA hybrid hydrogel, designed to emulate the native biliary extracellular matrix. Our findings robustly demonstrate that this biomimetic construct successfully integrates the superior cell-instructive properties of GelMA with the enhanced mechanical robustness by SA, thereby addressing a critical unmet need in biliary reconstruction.
The rationale for selecting a hybrid system stems from the inherent limitations of the individual components. While GelMA, a denatured collagen derivative, excels in supporting cell adhesion and proliferation through its intrinsic RGD motifs,13,24 its relatively weak mechanical properties and rapid degradation kinetics render it suboptimal for alone sustaining the dynamic biliary environment. 16 Conversely, SA forms strong, durable ionic networks but is notoriously bio-inert, hindering effective cell-material interactions. 25 The incorporation of ionically cross-linked sodium alginate increased the compressive modulus of 10% GelMA from 8.4 ± 0.6 kPa to 38.1 ± 8.1 kPa (1% SA) and 59.7 ± 10.8 kPa (2% SA), with compressive strength reaching 253.3 ± 30.6 kPa in the latter (Figure 5). Although physiological biliary pressure is low (~2–3 kPa), ABDs must also resist suture handling, transient compression, and peristaltic motion. 26 Native porcine bile ducts exhibit strengths of 120–150 kPa; the 10% GelMA/2% SA formulation thus approximates this range, providing a safety margin while maintaining compliance. This dual-crosslinked design balances robustness and flexibility, though in vivo validation is required to refine the optimal mechanical window. Regarding surgical handling, the improved mechanical toughness of the GelMA/SA double-network hydrogel is crucial for clinical translation. The material demonstrates adequate suture retention strength, suggesting that these ABDs can be implanted via standard end-to-end anastomosis using microsurgical sutures without identifying risk of tearing or leakage. Furthermore, the FTIR analysis (Figure 3) corroborated the successful integration of both polymers, with the retention of some unreacted methacrylate groups potentially contributing to the observed ductility and resilience of the hydrogel.
Beyond bulk mechanics, the micro-architectural evolution observed via SEM (Figure 2) is of paramount importance. The transition from a dense, sheet-like morphology in pure GelMA to a highly interconnected, porous network in the 2% SA group creates a topographical landscape that is expected to facilitate not only nutrient and waste diffusion but also enhanced cell infiltration and tissue integration. This structural advantage likely underpins the superior in vitro bioperformance of the hybrid scaffolds. The CCK-8 and Live/Dead assays (Figures 6 and 7) unambiguously revealed that the alginate-containing scaffolds, particularly the 2% SA variant, actively promoted the proliferation of biliary epithelial cells compared to GelMA alone. We posit that the enlarged pore size and the presence of negatively charged alginate chains, which may enhance the adsorption of serum proteins vital for cell spreading, create a more favorable niche for cellular activities. 19 The excellent hemocompatibility, with hemolysis ratios well below the 5% safety threshold (Figure 8), further underscores the clinical potential of this material by minimizing the risk of acute inflammatory responses upon implantation.
Our work builds upon and advances previous efforts in the field. Early attempts using non-degradable materials like PTFE or Dacron were plagued by chronic foreign body reactions, fibrous encapsulation, and stricture formation. 10 More recent tissue-engineered approaches, such as those using decellularized matrices or gelatin nonwoven fabrics, have shown promise but face challenges related to source variability, immunogenicity, and mechanical consistency.5,17 The GelMA-SA ABD presented here offers a synthetic, yet biomimetic and tunable alternative. Its rapid swelling kinetics (Figure 4) ensure prompt hydration, which is critical for achieving a conformal seal at the anastomotic site in vivo, while its controllable degradation profile (tailored via SA content) can be engineered to match the rate of neo-tissue formation, ensuring a seamless transition from scaffold support to native tissue function.
Notwithstanding its promising results, this study has limitations. A key limitation is that cellular evaluation used non-human models rather than primary human biliary epithelial cells or patient-derived bile duct organoids. Although our data indicate good biocompatibility, testing with human cells is needed to confirm clinical translational potential. Future studies will incorporate such models to assess cell–material interactions and long-term functional integration. Additionally the in vitro findings necessitate in vivo validation to determine the construct’s integration, regeneration of a multi-layered biliary wall, and long-term patency. 27 Further investigation into the host immune and fibrosis response and the development of more advanced, patient-specific fabrication methods are also essential. 27
Looking forward, several exciting research directions emerge from this work. The immediate next step is to conduct a rigorous in vivo evaluation in a large animal model (e.g., porcine) to validate the safety and efficacy of the optimal 10% GelMA/2% SA formulation. To further enhance regenerative outcomes, the hybrid hydrogel serves as a versatile platform for functionalization. Incorporating bioactive factors such as vascular endothelial growth factor (VEGF) to promote angiogenesis or hepatocyte growth factor (HGF) to mitigate fibrosis could significantly improve functional regeneration. Additionally, seeding the ABD with patient-specific stem cells or cholangiocytes prior to implantation could pave the way for a more advanced, cell-based therapeutic strategy. Finally, the fundamental design principle of this GelMA-SA hybrid system—combining a bioactive polymer with a mechanically reinforcing one—is highly translatable, suggesting its potential application in regenerating other tubular tissues, such as blood vessels, the trachea, or the intestine.
In conclusion, we have developed and comprehensively characterized a GelMA-SA hybrid hydrogel-based ABD that uniquely fulfills the dual requirements of mechanical support and biological functionality. This biomimetic scaffold represents a significant stride beyond current palliative options towards a truly regenerative solution for biliary defects. With continued development and validation, this technology holds substantial promise for transforming the clinical management of devastating biliary diseases.
Conclusion
In conclusion, we have successfully developed a biomimetic ABD based on a GelMA-SA hybrid hydrogel. This scaffold synergizes the bioactivity of GelMA with the mechanical strength of SA, demonstrating optimal physicochemical properties, excellent biocompatibility, and the ability to promote biliary epithelial cell growth in vitro. The 10% GelMA/2% SA formulation is identified as a promising regenerative strategy for repairing bile duct defects, effectively bridging the gap between structural support and biological functionality. This study lays a solid foundation for future in vivo applications and advanced tissue engineering solutions.
Footnotes
Acknowledgements
We would like to thank all members of our research team for their valuable contributions.
Ethical considerations
Ethics approval was not required.
Consent to participate
The study does not involve human participants or their data.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by grants from Tianjin Health Research Project, China (Grant No. TJWJ2023QN100) and Independent Innovation Science Fund Project of Characteristic Medical Center of the Chinese People’s Armed Police Force (Grant No. KYZZCX2419).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during this study are fully available within the article.