Abstract
Eucalyptus species are known for their decongestant and antimicrobial properties, mainly due to their essential oil. However, significant challenges persist in maintaining the stability and durability of the essential oils’ effectiveness. This study examines the use of polymeric hollow fibers for encapsulating Eucalyptus essential oil (EE) to improve its stability and effectiveness in treating nasal congestion. The EE was obtained by hydro-distillation method, and analyzed using gas chromatography-mass spectrometry (GC-MS). A dialysis cartridge containing polysulfone hollow fibers was used to load the EE, and their permeability, morphology, and stability were assessed. Fourier-transform infrared spectroscopy (FT-IR) and headspace sampling with gas chromatography-flame ionization detection (GC-FID) were employed to monitor EE release and the stability of EE-loaded hollow fibers. GC/MS analysis identified 20 major components with 1,8-cineole being the predominant compound at 59.32%. Physicochemical characterization of hollow fibers revealed complete permeability to EE. FT-IR spectra suggested potential interactions between EE and the fibers. Release studies indicated that over 80% of EE was released from the fibers within 180 min. Headspace analysis confirmed the presence and stability of 1,8-cineole in the loaded fibers. The stability test demonstrated no significant changes in the EE-loaded fibers over 6 days. This study indicated that the capillary properties of the EE-loaded hollow fibers facilitate oil loading, and headspace sampling provides a more efficient analysis. The successful and stable release of EE from the fibers, highlights the potential of hollow fibers for controlled drug delivery. However, conducting more accurate experiments can help deduce more logical results.
Introduction
Nasal congestion is a bothersome condition accompanied by the perception of a lack of airflow in the nasal passages and is a secondary symptom in many diseases of the upper respiratory system. The main mechanisms involved in the pathophysiology of nasal congestion include mucosal inflammation with increased nasal secretions, tissue edema, and venous congestion. Moreover, physical problems related to the structure of the nasal passages and changes related to sensory perception can lead to nasal congestion. 1 Sinusitis, otitis media, and sleep disorders are among the most important complications of nasal congestion. Treatments are divided into three groups comprising decongestants (topical and oral), anti-allergic measures (environmental control, antihistamines, antihistamine/decongestant combinations, corticosteroids, mast cell stabilizers, immunotherapy), and nasal dilation devices. 2
In recent years, special attention has been paid to the use of medicinal plants due to their wide range of bioactive compounds and minimal side effects.
Eucalyptus spp. are plants from the Myrtaceae family which have been brought from Australia to other parts of the world. The leaves of the plant have a wide range of phytochemicals, mainly from essential oils, like phenolic compounds, terpenes, and flavonoids. The plant has many medicinal properties such as decongestant, expectorant, antimicrobial, and antiviral effects. Most medicinal properties of the plant are attributed to its essential oil which is beneficial in pharmaceutical, food, cosmetics, perfumery, and detergent industries. Furthermore, this plant is found to be effective in respiratory diseases such as sinusitis, bronchitis, asthma, COPD, nasal congestion, etc.3,4 The species used in this research is Eucaliptus galbie which is mentioned in fewer sources. Studies have shown the antimicrobial effects of the plant. 5 1,8-cineole is a saturated monoterpene in Eucalyptus essential oil (EE). It has a pleasant taste and smell, making it popular in various industries, including perfumery and cosmetic industries. This component has many pharmacological effects, the most important of which are anti-inflammatory and antioxidant properties. Therefore it can be used in many diseases, such as cardiovascular diseases, Alzheimer’s disease, indigestion, and respiratory diseases. Although 1,8-cineole has shown good effects in preclinical studies, its application is practically limited due to its chemical instability. 6
Although medicinal plants are popular and safe to use, providing them in an effective and convenient manner poses a major challenge for researchers. Hollow fibers have recently attracted a lot of attention in the medical field due to their suitable features such as high loading capacity, porosity, and low weight. 7 Hollow fibers are cylindrical tubes with a porous or semi-porous wall, typically made from a polymeric material. Their hollow core allows for the encapsulation and controlled release of substances, while their porous wall facilitates diffusion. 8 These fibers are like small tubes that have the potential for loading different drugs. 9 Since the wall thickness and diameter of hollow fibers can be modified according to the diffusion coefficient; the uniformity of diffusion and the speed of drug release can be controlled in them. Furthermore, the encapsulated drug can be physically protected inside the hollow fibers. Medicines can reach the target organ directly through hollow fibers. 10 In addition to their protective features, hollow fibers provide several advantages for drug delivery systems. They can facilitate targeted delivery, allowing medicines to reach the target organ directly, thereby minimizing systemic side effects. The surface properties of hollow fibers can also be tailored to optimize drug adsorption and release profiles. Furthermore, the compatibility of these fibers with various pharmaceutical agents is crucial for their efficacy. It is also important to explore the interaction between the drug and the hollow fiber materials, as this can significantly influence drug release behavior and the overall efficiency of the delivery system. Understanding these aspects would provide a clearer rationale for the selection of hollow fibers in the context of this study. Finally, including comparative analyses with existing drug delivery methods could highlight the specific advantages and innovations offered by the use of hollow fibers in this research . Examples of utilizing hollow fibers in medicine include using nylon hollow fibers to deliver chlorhexidine to control infections caused by intrauterine contraceptive devices, 11 utilizing cellulose acetate hollow fibers for loading tetracycline to address periodontal diseases, and using polylactic acid hollow fibers to contain levonorgestrel.12,13 There are also investigations that used electrospun nano-fibers for herbal drug delivery with topical and systemic applications.14,15
In this research, polymeric hollow fibers (polysulfan) developed in dialysis cartridges were used for drug delivery of Eucalyptus essential oil in order to improve nasal congestion, and their physicochemical characteristics were studied.
Method and material
Eucalyptus essential oil (EE) preparation and TLC study
The plant leaves were obtained from a botanical market in Kerman province, Iran, and identified as Eucalyptus galbie by a botanist, Bahonar University. Herbarium samples of the plant were deposited in the Herbarium Center of Kerman Pharmacy, Department of Pharmacognosy (KF1703). The EE was extracted using the hydro-distillation method with a Clevenger apparatus. Initially, the plant was finely ground and sifted through a mesh sieve. Approximately 100 g of the powdered plant material was macerated in water and left in a dark, cool place for 24 h. The essential oil extraction took place over a 4-hour period. The obtained essential oil was then dehydrated using anhydrous sodium sulfate and stored at −20°C during the experiments. 16 Using the thin layer chromatography (TLC) method, Eucalyptus essential oil and 1,8-cineole in toluene were spotted on a GF254 silica gel plate and developed in toluene:ethyl acetate (7:93). To visualize the spots, a reagent of 5% vanillin and sulfuric acid in ethanol was applied, and the plate was placed in an oven at 100°C for 10 min.
Phytochemical studies
The plant underwent phytochemical screening to detect the presence of various secondary metabolites, such as alkaloids, flavonoids, saponins, steroids, tannins, and terpenoids.17,18
Gas chromatography-mass spectrometry (GC-MS) analysis
GC/MS analysis was conducted on EE to identify its components and measure their concentrations. The major representative compound, 1,8-cineole, was used for calculations in the study. The analysis was carried out on an Agilent 6890 GC and a series selective mass detector Agilent 5973N. Helium gas was used as a carrier gas at a flow rate of 1 mL/min and the temperature program of the column was set as follows: The initial oven temperature was 50°C, held for 5 min and increased to 240°C at a rate of 3°C per minute. Subsequently, the temperature was increased to 300°C with a thermal gradient of 15°C per minute and stopped for 3 min. The total program duration was 75 min. Mass spectra were obtained in electron impact mode (EI); with a unization voltage of 70 ev and a mass range was 35–450 m/z. The scanning speed was set at 1 scan per second. A mixture of n-alkanes (C8–C28) was used under these conditions to calculate linear retention indices (such as Kovats Index, I) for each compound. Identification was performed by comparing the obtained mass spectra and chromatographic peaks corresponding with those in NIST and Wiley Libraries. 19
EE calibration curve for release and stability experiments
The calibration curve for EE was established using the spectrophotometry method, specifically a spectrophotometer Shimdzu. The maximum wavelength of EE (λ max) was determined to be 266.17 nm by scanning absorbance within the range of 200–400 nm. Various dilutions of EE were prepared to create a calibration curve based on absorbance at λ max (266.17 nm). This process was repeated three times, twice on the same day and once 24 h later, to verify precision. The relative standard deviation (RSD) was calculated for all absorbance values obtained to ensure the repeatability of the experiment. The linearity of the curve was confirmed at each step.
Preparation and physicochemical characteristics evaluation of hollow fibers
Determination of weight, length, and morphology
A dialysis cartridge containing polysulfone hollow fibers was acquired from Meditex Company in Tehran, Iran. The hollow fibers utilized in this study exhibited the following characteristics: an average weight of 2.15 ± 0.18 mg, a length of 23.43 ± 0.76 cm, a thickness of 0.007 mm, and an external and internal diameter of 0.25 and 0.15 mm, respectively. Eleven fibers were randomly selected and weighed accurately using a 4-digit analytical balance. Their length was measured with a precise ruler, and the thickness of some fibers was estimated using a caliper. Finally, the mean weight, length, and thickness of the hollow fibers were calculated. The study of the morphology of hollow fibers was conducted using a Scanning Electron Microscope (FESEM).
Permeability test
As an initial step to evaluate the permeability of the hollow fibers, 1 mL of eucalyptus solution (Barij Essence Company. Iran) containing 1,8-cineole (28 mg/mL) was introduced to one end of the fibers. The observation of a color change at the opposite end provided a rapid, albeit qualitative, indication of the Eucalyptus solution’s ability to penetrate the fiber structure. While acknowledging the inherent limitations of this visual assessment, this simplified approach was initially adopted due to constraints in available facilities for more sophisticated permeability testing. To further explore this characteristic, the fibers were then placed vertically in a colored ethanol solution, allowing for observation of capillary absorption of the dye after 1 and 48 h. 20 Moving forward, we recommend that future research incorporate quantitative methodologies to achieve a more accurate and comprehensive understanding of the fibers’ permeability properties. In the subsequent step, we measured the weight change of the hollow fibers after they were loaded with EE. To minimize potential errors caused by the difficulty of individually placing each fiber into a test tube, we twisted a bundle of 20 hollow fibers around a thin tube, creating a spring-like structure for better handling during measurement. The average initial weight of each bundle was recorded as 2.15 ± 0.18 mg (n = 3). The bundle was then carefully placed vertically into 2 mL of a 10% EE solution in ethanol and stored in the refrigerator for 24 h, during which the weight change was meticulously documented. The average weight of the bundles was approximately 15.03 ± 0.06 mg.
FT-IR spectrum
The FT-IR spectra of EE, empty hollow fibers were compared to EE-loaded hollow fibers in the range of 400–4000 cm−1.
Release study
To investigate the release of EE in a liquid environment, a package of 20 accurately weighed fibers was loaded with essential oil as described in the permeation test. To minimize potential bias, the 20 fibers were randomly selected from the prepared batch. All fibers were visually inspected prior to selection to ensure uniformity (e.g. diameter, absence of visible defects). Fibers meeting these criteria were assigned a number, and 20 numbers were chosen using a random number generator to select the fibers for the release study. These loaded fibers were then placed in a sealed container containing 30 mL of a methanol to distilled water solution (1:1 ratio). Samples were taken at 0, 5, 10, 15, 30, 60, 90, 120, 180, 240, and 300 min, and their absorbance was measured using a UV spectrophotometer. The percentage of EE release was determined by calculating the absorbance and utilizing the slope of the calibration curve. 21
Identification of 1,8-cineole peak using GC-FID (direct injection)
After establishing a suitable method to identify 1,8-cineole using GC-FID and obtaining the corresponding peak for this compound, the EE was also analyzed using the same GC-FID method to assess its composition.
Headspace sampling and analysis using GC-FID
A 20,000 ppm solution of 1,8-cineole was analyzed using the headspace method. Two bundles of 20 hollow fibers were then loaded with a 10% solution of 1,8-cineole and EE in HPLC methanol separately. Both bundles were kept in the refrigerator for 24 h, before being analyzed through headspace sampling using GC-FID. The analyses were conducted using a headspace auto sampler (PAL RSI) coupled with an Agilent 7890B gas chromatogram (GC). The headspace temperature was set at 190°C, slightly above the boiling point of 1,8-cineole (177°C). The GC device was equipped with a flame ionization detector and an HP-5 capillary column (30 m × 0.25 mm × 0.25 μm; Agilent, USA). The oven temperature was programmed the same as the GC-MASS analysis of EE. The detector and injector temperatures were 280°C and 250°C, respectively. The carrier gas was N2 (99.99%) at a flow rate of 0.5 mL min−1.
To create a concentration-AUC curve, three bundles of 20 hollow fibers, were carefully weighed, and placed in EE dilutions (in 20, 50, and 100 EE µL/mL methanol HPLC) separately, and the weight changes of the fibers were measured, and then these fibers were analyzed using the headspace method with a GC-FID device. Finally, a plot of concentration versus AUC was generated. 22
EE-loaded hollow fibers stability test
Three accurately weighed bundles, each containing 20 fibers, were immersed in 1 mL of a solution comprising 50 μL of EE for a duration of 24 h to facilitate optimal absorption. The selection of the hollow fiber bundles was performed using a methodology akin to that outlined in section 2–7 of the release study. Following this exposure period, the fibers were carefully transferred to specialized vials designed for the GC-FID apparatus, which was maintained at a constant temperature of 40°C for subsequent analysis. The samples were analyzed on days 0, 3, and 6 through the headspace method using the GC-FID device. The area under the curve (AUC) corresponding to 1,8-cineole was extracted from each chromatogram to evaluate the stability of the formulation.
Results
Phytochemical studies
Phytochemical screening revealed the presence of tannins, flavonoids, and terpenoids in the plant. Alkaloids, steroids, and saponins were not detected at this stage of analysis. These preliminary results suggest a diverse phytochemical profile for the plant; however, further studies are required for a more precise and quantitative assessment of the chemical composition (Figure 1).

Phytochemical screening of Eucalyptus galbie to identify the presence of alkaloids, flavonoids, saponins, steroids, tannins, and terpenoids.
GC/MS analysis of EE analysis
The yield of essential oil extraction was 1.1 mL/100 g of dried Eucalyptus leaves. The GC/MS analysis led to the identification of 20 major components, representing 95.42% of the total oil. The main components of EE were 1,8-cineole (59.32%), α-pinene (5.62%), p-cymene (4.88%) limonene (4.45%), and globulol (4%) (Table 1).
GC/MS analysis of Eucalyptus essential oil (EE).

MH: hydrocarbon monoterpene; MO: oxygenated monoterpene; SH: hydrocarbon sesquiterpene; SO: oxygenated monoterpene.
Calibration curve of EE
The maximum wavelength (λmax) of 266.17 nm for the eucalyptus essential oil (EE) was determined based on a screening process conducted over a wavelength range of 200 to 400 nm. Following this, the absorbance of different concentrations of EE was measured three times at this λmax, which led to the creation of a concentration-absorbance curve. The results showed a linear relationship between EE concentration and absorbance (Y = 0.0017X + 0.0245; R2 = 0.9963) (Figure 2).

Concentration-absorbance curve of Eucalyptus essential oil (λmax = 266.17 nm).
GC-FID chromatogram of EE and 1,8-cineole
Figure 3 displays the GC-FID chromatograms of EE and 1,8-cineole, showing a slight difference in RT(11.57 and 12.19 min in pure form and EE respectively). This variance is probably caused by the presence of other compounds in EE that could interact with 1,8-cineole and influence its retention time.

GC-FID chromatogram of: (a) 1,8-cineole (RT = 11.57 min) and (b) Eucalyptus essential oil (1,8-cineole RT = 12.19 min).
Physicochemical characteristics of hollow fibers
The average weight of 11 fibers was 2.15 ± 0.18 mg, their average length was 23.43 ± 0.76 cm, and the thickness of the hollow fibers was determined to be 0.25 ± 0.05 mm. Hollow fibers showed complete permeability to EE. Weight changes occurred as a result of the absorption of essential oil into the hollow fibers, driven by capillary property. Figure 4 shows an image of hollow fibers by field emission scanning electron microscopy (FESEM) analysis.

Field emission scanning electron microscopy (FESEM) images taken of (a) cross-section and (b) outer surface morphologies of the hollow fiber membranes.
FT-IR spectrum
Figure 5 shows the FTIR spectra of EE, empty hollow fibers, and EE-loaded hollow fibers. The FTIR spectrum of EE exhibits bands at 887.161 cm−1 (is assigned to C=C bending), 1167.57 cm−1 (assigned to C–O stretching), 1214.78 cm−1 (assigned to C–O stretching), 1305.84 cm−1 (assigned to C–O stretching), 1375.77 cm−1 (assigned to S=O stretching), 1633.02 cm−1 (assigned to C=C stretching), and 1737.19 (assigned to C=O stretching). These bands are absent in the spectra of empty and EE-loaded hollow fibers. Additionally, in the spectrum of EE-loaded hollow fibers, some bands were apparent at 738.34 cm−1 (assigned to C-H bending) and 1773.36 cm−1 (assigned to C=O stretching), which were absent in EE or empty hollow fibers; spectra. In general, the disappearance or rearrangement of these bands in the EE-loaded hollow fibers could indicate the involvement of the functional groups in other bonds implying a potential reaction between hollow fibers and EE.
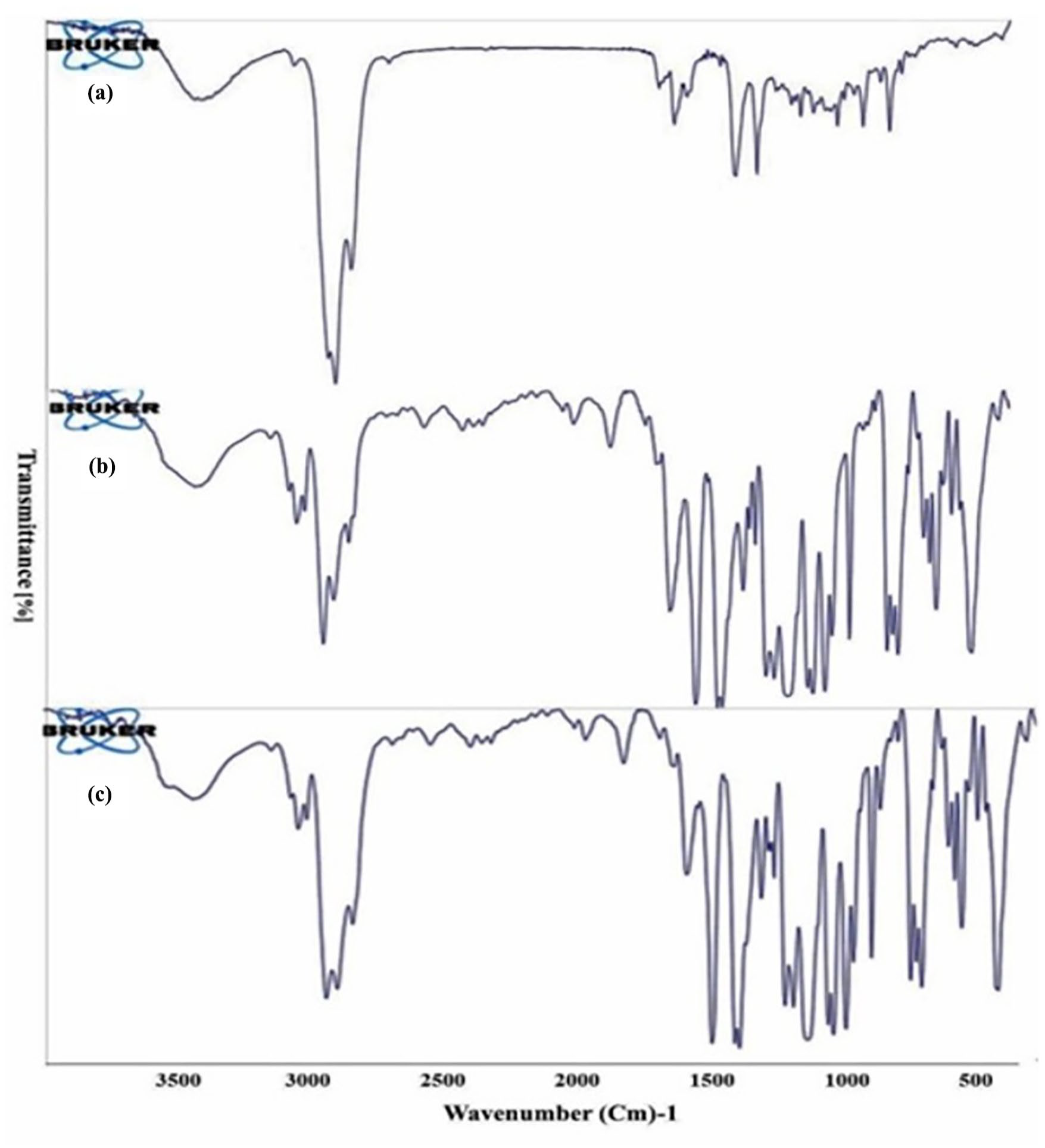
FT-IR spectrum of: (a) Eucalyptus essential oil, (b) empty hollow fibers, and (c) Eucalyptus essential oil-loaded hollow fibers.
Release study of EE from hollow fiber
The release curve of EE from polymeric hollow fibers in a liquid medium is depicted in Figure 6. As shown, the absorbance measurements of the samples indicate the amount of released EE within the time interval of 0–300 min (Figure 6). Around 50% of the oil is released after 60 min, and over 80% is released after 180 min.

Release curve of Eucalyptus essential oil from polymeric hollow fibers within 0 to 300 min (Mean ± SD, n = 3).
Headspace analysis
The GC-FID spectrum of a 20,000 ppm solution of 1,8-cineole was obtained using the headspace method. The peak of 1,8-cineole appeared at 11.86 min. In the chromatogram of 1,8-cineole-loaded hollow fibers, it was seen at 11.630 min, and in the chromatogram of EE-loaded hallow fibers, the peak appeared at 11.593 min.
AUC-concentration curve of EE
GC-FID analysis of hollow fibers impregnated with EE at 20,000, 50,000, and 100,000 ppm indicated that the retention time of 1,8-cineole was 11.583, 11.634, and 11.696 min, respectively(Figure 7). The weight changes of the fibers remained constant at around 0.1 g for all three EE concentrations.

Concentration – area under the curve (AUC) of hollow fibers loaded with Eucalyptus essential oil.
Stability test
During stability tests, there were no significant changes in the appearance of the EE-loaded hollow fibers on days 0, 3, and 6 after preparation. Based on AUC – concentration curve of EE, the weight changes of hollow fiber were approximately about 0.1 g. The results for 1,8-cineole retention time, the peak area, and the amount of EE retained in the hollow fiber (based on the AUC – concentration curve) analyzed by GC-FID and headspace sampling did not show a significant difference across the sampling time intervals (data not shown).
Discussion
Eucalyptus plants are commonly used in the treatment of inflammatory and infectious diseases, particularly those impacting the respiratory system, nasal congestion, asthma, and COPD. These benefits are mainly attributed to the essential oil of the plant. Despite the availability of various preparations of Eucalyptus essential oil, instability, and volatility present a significant challenge in their formulation.3,23 In this study, we aimed to study the use of polymeric hollow fibers, typically found in dialysis cartridges, to load the essential oil of a specific species of Eucalyptus, E. galbie, for inhaled drug delivery. These hollow fibers have small channels and are an ideal system for encapsulating drugs due to their high surface-to-volume ratio, exceptional flexibility in drug loading, and the ability to control the rate of drug release.9,10 The use of polymeric hollow fibers to encapsulate Eucalyptus essential oil (EE) offers a promising approach for improving the stability and efficacy of this natural medicines, especially for nasal congestion. The inherent instability and volatility of EE, responsible for its decongestant and antimicrobial effects, often hinder its practical application. By exploring a delivery system that can enhance the therapeutic potential of EE, we aimed to address this therapeutic gap and challenge. The hollow fibers used in this study had an average weight of 2.15 ± 0.18 mg, a length of 23.43 ± 0.76 cm, and a thickness of 0.007 mm, and external and internal diameter of 0.25 and 0.15 mm respectively. Importantly, the fibers demonstrated good permeability to EE, leading to weight changes as a result of EE absorption through capillary action. FESEM images (Figure 4) revealed the physical structure of the fibers. These physical measurements provide necessary information about the dimensions and capacity of the hollow fibers. The permeability to EE is a significant finding indicating that these fibers are suitable for loading essential oils. The observation that EE is absorbed due to capillary action ensures an efficient and passive loading mechanism, reducing the need for external pressure. The results of this study demonstrated the successful encapsulation of EE in hollow fibers, primarily due to the permeability of the essential oil facilitated by the polymeric structure of the hollow fibers. This structure also facilitated the release of the encapsulated material while protecting the essential oil from environmental factors that could compromise its quality. FT-IR analysis suggests potential interactions between EE and fibers (Figure 5), indicating that these interactions may contribute to the stability of the entrapped essential oil by preventing oxidation or evaporation of 1,8-cineole, the dominant essential oil compound identified in GC-MS analysis (Figure 3).
1,8-cineole was used as a reference in the analyses to evaluate the EE loading capacity of the hollow fibers. The release curve (Figure 6) showed that about 50% of EE was released from the hollow fibers after 60 min and more than 80% after 180 min in a liquid medium. The release rate of EE is rapid in the first few minutes and gradually increases to reach a steady state. This rapid release profile is useful for applications that require a rapid therapeutic action. The fact that most of the oil is released in a relatively short time suggests that the hollow fiber matrix is effective in releasing EE and can serve as an efficient delivery system, ensuring the desired therapeutic effect. This rapid release profile is useful for conditions such as nasal congestion, where rapid therapeutic action is often required. In the second part, the content of the hollow fibers loaded with EE was sampled using the headspace technique. The use of headspace sampling in conjunction with GC-FID has been shown to be effective for monitoring EE release. This technique provides a sensitive method to assess the fluctuations and stability of EE, thus providing a better understanding of how the encapsulated oil behaves over time in the delivery system. Compared to the direct injection method, headspace sampling offers advantages such as time efficiency and prevention of column contamination by multiple components, thereby reducing the need for frequent column and injector replacement.24,25
The findings of the present study also showed that EE was successfully loaded into the polymeric hollow fibers (Figure 5) and released into ambient air while maintaining satisfactory stability (Figure 7). This is attributed to the capillary property of the hollow fibers leading to the loading of the essential oil and distinguishes this study from other similar investigations that used injection or vacuum pumps. While slight differences in retention time (RT) between pure 1,8-cineole (11.57 min) and 1,8-cineole in the EE sample (12.19 min) were observed in the GC-FID chromatograms (Figure 3), these differences were not unexpected. The presence of other compounds in the EE may contribute to slight variations in the interaction of 1,8-cineole with the chromatographic column, causing changes in its retention. Further investigation is needed to fully elucidate these matrix effects. The 1,8-cineole peak is clearly identifiable in the EE mixture, confirming that its signal is intact and detectable, an important finding because 1,8-cineole is considered the key active component in EE.
Headspace sampling confirms that 1,8-cineole (the volatile component) is present and can be detected after encapsulation in hollow fibers. Headspace analysis is a valuable method for evaluating the volatile components of the essential oil, which are crucial for its therapeutic effect. The presence of 1,8-cineole in the headspace indicates that it retains its volatile character even after incorporation into hollow fibers. This suggests that the encapsulation process of EE can contribute to effective therapeutic activity and prevent its degradation.
Furthermore, the present method uses GC-FID to analyze the vapor released from the hollow fibers, which distinguishes it from other research in inhalation formulation. GC-FID analysis of hollow fibers impregnated with EE at 20,000, 50,000, and 100,000 ppm showed that the retention times of 1,8-cineole were 11.583, 11.634, and 11.696 min, respectively (Figure 7). The fiber weight remained relatively constant at about 0.1 g for all three EE concentrations. This indicates that these fibers are able to absorb more essential oil at higher concentrations without physical degradation or major changes in their weight. Therefore, more EE can be loaded into the hollow fibers by increasing its concentration, which can be useful for dose control and improving treatment efficacy. However, these fibers have a loading capacity ceiling and may need to be optimized for loading. In this particular case, it has been shown that higher concentrations of the essential oil do not have a significant effect on the retention time, so the EE saturation limit may be around 20,000 ppm.
Stability testing showed no significant changes in the appearance of the EE-loaded hollow fibers over 6 days. Furthermore, the results for the retention time of 1,8-cineole, peak area, and amount of EE remaining in the hollow fibers (based on the AUC concentration curve) did not show any significant differences across the sampling time intervals (data not shown). The absence of visible changes and the constant 1,8-cineole peak area indicate good physical and chemical stability of the EE-loaded fibers under the tested conditions.
The stability of the retention time suggests that there is no change in the way 1,8-cineole interacts with the chromatographic column over these days. The constant peak area indicates that 1,8-cineole remains stable and does not degrade significantly over time. Stability testing shows that the hollow fiber material effectively protects the EE from rapid degradation and the encapsulated EE can maintain its integrity and therapeutic potential for a reasonable period, at least for 6 days. This is important for ensuring effective product use and also for effective shelf life design.
The literature review indicates that no similar work has been done on Eucalyptus or any other plant essential oil. Cheung et al. 7 conducted a comprehensive study to investigate the loading capacity, release kinetics, and antibacterial effect of nylon hollow fibers. Their findings showed that these fibers can effectively accommodate various pharmaceutical formulations including solutions, oils, and suspensions. Drug release studies were conducted using deionized water, ethanol, and PEG 4000 as release media. The results showed that the drugs were released from the hollow fibers, followed by stabilization of the fiber weight, indicating a steady-state release rate. Interestingly, the study also showed that increasing the fiber length resulted in a decrease in the rate of drug release. This suggests that fiber length can be adjusted to control drug release and potentially reduce drug toxicity. In addition, the study evaluated the antibacterial properties of penicillin and streptomycin-loaded hollow fibers, showing significant bacteriostatic effects in vitro, highlighting the potential of these drug-loaded hollow fibers as effective antibacterial agents.
Goodson et al. 26 investigated the feasibility of using cellulose acetate hollow fibers for the topical delivery of tetracycline in a dental setting. In their study, they immobilized hollow fibers in a needle syringe, loaded the drug, and administered it around the tooth. By determining the release rate of tetracycline using spectrophotometry, they reported that this drug was released within 2 h and reduced bacteria in the treated area compared to the control area.
Kudo et al. 27 conducted an extensive study to evaluate the kinetics of drug release from hollow fibers and hollow fiber patches. In their study, they investigated the release characteristics of heparin and sodium chloride loaded from hollow fibers and reported preliminary laboratory results of drug-impregnated hollow fibers and hollow fiber fabric. The results showed that over the 4-week study period, chemicals in the hollow space impregnated with a vacuum chamber were continuously released from the hollow fiber models and the hollow fiber fabric for more than 2 weeks.
Eenink and colleagues loaded biodegradable hollow fibers with a 25% solution of radiolabeled levonorgestrel in castor oil using a dry-wet phase inversion spin technique. Drug release was evaluated in 40% ethanol solution and the findings showed that the wall structure affects the drug release rate from hollow fibers. Subcutaneous implantation of these hollow fibers in rabbits showed a sustained release pattern that maintained drug levels in rabbit blood for 210 days. 13
This study explored the loading of volatile phytochemicals into polymeric hollow fibers, emphasizing the capillary properties of the fibers without relying on specialized equipment like injection pumps or vacuum systems. This innovative approach sets our work apart from prior research in the field. We employed the headspace sampling method, which offers a more time-efficient alternative to traditional direct injection techniques.24,25 Additionally, spectrophotometry was utilized to evaluate the release of volatile compounds, while GC-FID analysis provided critical insights into the dynamics of vapor release from the hollow fibers.
Conclusion
Our findings indicate that alterations in the porosity of the hollow fibers could significantly impact the loading efficiency of essential oils. This study specifically targets the formulation of an inhalation product for drug delivery, setting it apart from earlier investigations. The evaluation of Eucalyptus essential oil (EE) loading into the hollow fibers at two stages confirmed the stability of the product, revealing a consistent release profile. Despite these promising results, further extensive studies are required to optimize loading conditions and enhance the capacity for essential oil incorporation in the hollow fibers. Additionally, it would be beneficial to design a sustained-release drug delivery system and to investigate polymers with varying wall densities and porosity characteristics. The cumulative evidence from this study suggests that polymeric hollow fibers hold great potential as a drug delivery system. The positive indicators regarding loading, stability, and drug release support the therapeutic application of this technology, particularly in enhancing the stability and effectiveness of natural remedies for conditions like nasal congestion. By focusing on the encapsulation of Eucalyptus essential oil, this research addresses a critical gap in the field, aiming to overcome the challenges posed by the instability and volatility of essential oils. In summary, this investigation underlines the potential for polymeric hollow fibers to advance the delivery of Eucalyptus essential oil, capitalizing on its therapeutic properties and addressing the need for improved systems in natural remedy applications. Future studies will aim to substantiate these findings further and explore innovative solutions in drug delivery.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
