Abstract
For millennia, aloe vera (AV) and eucalyptus oil (EO) have been recognized as natural sources of healing and have been utilized for medicinal purposes in the realm of health. As an attempt to treat pressure sores, AV and eucalyptus oil were added as supplements to biocompatible and biodegradable poly (ethylene oxide) (PEO) polymer to synthesize nano and micro fibrous wound dressings by the electrospinning process. Additive solubility in polymeric matrix is the key parameter to achieve the synthesis of homogeneous fibers with controlled release of therapeutic oils, cure and humidity; therefore, lecithin as herbal (soybean) based emulsifier was used to control additive/polymer solubility. In this study, fibrous dressing in mat form with antioxidant activity was successfully obtained with the addition of natural AV and EO in PEO polymer solutions through electrospinning technique. Subsequently, the synthesized fibers were examined via scanning electron microscopy (SEM), thermogravimetric analysis (TGA), moisture absorption and UV-Vis spectroscopy. SEM imaging demonstrated the formation of randomly-oriented and beadless fibers with size of 0.48 ± 0.23 µm out of PEO/AV/EO/Lecithin blend and also with the addition of lecithin, fiber thicknesses were observed to be increasing. Moisture absorption analysis revealed that the weight of fibrous mat was affected by the humidity of the ambient environment. Relative humidity for 7 days ranged between 32% and 37% and it was observed that lecithin content increased the moisture retention rate by 50%. Uv-Vis results suggested that a more regular performance has been achieved with lecithin being involved in terms of timely manner changes; therefore, the contrast of samples between hours and days became more distinctive. PEO/AV/EO/Lecithin nanofiber also indicated antibacterial ability against Escherichia coli with approximately 18.5 mm diameter of inhibition. This research proves that the potential for developing biocompatible wound dressings with long-lasting moisture to the wound is possible through the use of these natural healing agents made homogeneously distributed through structure by the use of emulsifier.
Introduction
Pressure sores (also known as bedsores or decubitus ulcers) are defined by the National Pressure Ulcer Advisory Panel (NPUAP) as “localized damage to the soft tissue, usually over a bony prominence”. 1 They are wounds that appear on the skin and underlying tissues due to long-term or repeated pressure, particularly in areas of the body where bony prominences are present. Decubitus ulcers can have negative effects on a patient’s psychology, physical abilities, and social life.2,3 Decubitus ulcers can impede the healing of wounds and are frequently accompanied by excruciating pain and secondary infections, which will lengthen treatment and worsen the prognosis for the patient. Since there is now no primary treatment to prevent and hasten the healing of decubitus ulcers with adequate outcomes, management of these ulcers is still being researched. Without combining topical antibiotics, the primary treatment attempts to relieve pressure on the ulcer, administer systemic antibiotics in the event of a serious secondary infection, and treat wounds using topical medications, debridement, and surgery if necessary. 4 Penicillins, cephalosporins, aminoglycosides, macrolides, and quinolones are among the frequently used systemic antibiotics . Debridement is the process of removing necrotic tissue with a scalpel, gauze, tweezers, scissors, or another tool. Debridement is necessary to give the ulcer adequate vascularization, which will hasten the healing process. 5 Following the prolonged non-healing of the wound, surgical intervention becomes necessary in the subsequent process. And the aim of treating pressure sores is to relieve the patient’s pain and to heal the wounds as quickly as possible in a non-surgical way without leaving any scars.
Any part of the body where constant pressure and compressive forces are applied for an extended length of time is susceptible to developing decubitus ulcers. The development of decubitus ulcers may also be influenced by additional risk factors. Vulnerability to pressure ulcers arises from a confluence of exterior elements (pressure, friction, shear force, and moisture) and internal causes (such as fever, starvation, anemia, and endothelial dysfunction).6 –8
Few ailments have undergone as many tried therapies as decubitus ulcers. Historically, numerous chemical therapies have been employed, including poultices of vegetables, enzymes, vitamins, cod liver oil, dried blood plasma, different precious and non-precious metals, chlorophyll, sugar, and salt. Mechanical therapies encompassed electric lamps, ultraviolet light, hyperbaric oxygen, rubber rings, sawdust beds, and various pressure beds and padding. 9 Currently, the management of decubitus ulcers relies on four basic modalities: pressure alleviation and prevention of further ulcers, wound care, surgical intervention, and nutritional support. It’s crucial to care wounds once an ulcer forms. Cleaning, efficient drainage, and absorption are essential components of wound care that guarantee successful healing while safeguarding the surrounding skin. At this point, surgical pads and wound dressings become important materials for cleaning and protecting the wounded area.
Moisture is crucial for wound healing. Attempting to heal wounds in a dry manner can cause more tissue loss. 10 In 1962, George D. Winter reported that keeping wounds moist accelerates the healing process, contradicting those who argued that wounds should heal by drying out. 11 Over time, other researchers have emphasized the importance of moist healing and have developed nanomaterial-based wound dressings. They have used hydrogels, 12 alginates, 13 hydro fibers 14 and hydrocolloids 15 as moist wound dressings. An ideal wound dressing should provide excellent tissue compatibility, maintain moisture on the wound surface, allow for air flow, exhibit antibacterial properties, be biocompatible, exhibit high mechanical properties, but at the same time, not exhibit toxic properties, not stick to the wound, and not cause allergic reactions.12,16 Researchers have been developing wound dressings utilizing natural medicinal plants and essential oils to promote rapid and infection-free wound healing. 17
Thousands of years ago, oils, honey, and wine compresses were used to provide moisture for wound healing. 18 Aloe vera (AV), which has been known as a natural healing source since 3500 BC, is known to improve wound healing processes and reduce inflammation. 19 AV contains more than 70 compounds and these compounds have been reported to possess properties such as antifungal, antidiabetic, 20 antibacterial, 21 antiseptic, antiviral, 22 antioxidant, 23 anticancer, 24 anti-inflammatory, 25 and wound healing.26,27 Aloe polysaccharides, which are found in AV, are the most important compounds supporting wound healing. 27 Pereira et al. 28 have developed hydrogel films using alginate and AV gel for wound healing and drug delivery systems. AV has increased the hydrophilicity of the film surface and it has been shown to meet the basic properties required for wound healing. Yin et al. 29 have conducted research on the antibacterial properties of polycaprolactone/chitosan/a. vera (PCL/CS/AV) nanofiber membranes. It was observed that AV nanofibers increased the hydrophilicity and pore size. PCL/CS/AV nanofibers exhibited high antibacterial properties due to the AV effect, and it was shown that they could be suitable wound dressings for acute wounds. Jouybar et al. 30 have examined tissue healing by producing AV-coated poly-L-lactic acid (PLLA) nanofibers. They have investigated the effect of AV gel and non-gel on the healing process. Vaseline gauze was used as a control sample. It was observed that gel-coated nanofibers accelerated the healing time. Eucalyptus is an aromatic and medicinal plant from the Myrtaceae family. It is one of the most commonly cultivated species of tree resins with over 700 species worldwide. Eucalyptus leaves have many biological activities, including antioxidant, antibacterial, antifungal, and anti-inflammatory properties. 31 Kumar et al. 32 have produced natural fabrics with leaf extracts of Calotropis gigantean, Eucalyptus glolubus, and Syzygium aromaticum buds, fortified with rhEGF (REGEN-D™ 60) for use as wound dressing materials. They have demonstrated the role of fabrics in wound healing through experiments on guinea pigs. They exhibited high antibacterial activity against gram-negative and gram-positive bacteria. Natural extracts have increased wound healing rate. Darwish et al. 33 have encapsulated EO into a dextran sulfate/chitosan hydrogel coated with AV. The developed hydrogel exhibits biological effects and antibacterial properties against gram-positive bacteria, making it a promising candidate for wound dressings.
This study focuses on the effect of polarities of additives and PEO polymer on the structure of electrospun fibers and their properties. PEO is known to be a polar polymer 33 which is soluble in water and adsorbs water molecules. However, AV has partial polar and nonpolar parts that makes it partially soluble in water while EO is nonpolar and insoluble in water.34,35 Using AV and EO with PEO polymer to form homogeneous fibers requires the use of emulsifying agent and for this concept it has to be biocompatible.
Biocompatible nanofibers were synthesized by electrospinning a blend of AV and EO with polyethylene oxide (PEO), a biocompatible polymer along with and without emulsifier. The physicochemical properties of the synthesized nanofibers were characterized with SEM, thermal analysis, UV-Vis and moisture absorption. Additionally, antibacterial properties were systematically evaluated. The synthesized nanofibers were subjected to characterization processes. It is believed that this study will contribute to the treatment of various wounds, including pressure ulcers, by reducing the risk of infection through preventing the adhesion of dressing material and accelerating the wound healing process.
Laboratory scale samples were successfully prepared through existing device settlement; however, manufacturing fibrous dressings in large scale is complicated and expensive process. There are available production infrastructure for other industries; however, involving natural oils and emulsifiers along with polymer makes the process more complicated for this specific area and custom made production line might need to be focused on. And this can be future project to consider.
Materials and methods
Materials
PEO – polyethylene oxide with Mw~200,000 was obtained from Sigma Aldrich. Lecithin consisting of 94% phosphatidylcholine was also supplied from Sigma Aldrich. AV extract was obtained from mature AV plants. 100% pure EO was supplied from Onka Farma. Deionized water was supplied through the device produced by Human Corporation through New Human Power and used throughout the study. Lecithin was sourced from a company that produces it for the food and pharmaceutical industries. Phosphate-buffered saline (10× PBS solution) was obtained from Ecotech Biotechnology firm.
Methods
Preparation of aloe vera
The fully matured leaves of naturally grown AV were cut and cleaned by washing with deionized water, and the epidermal layer of the leaves was meticulously removed to extract the inner gel. The inner gel was divided into small pieces and placed into vials where they were homogenized by an electromagnetic stirrer at 1000 rpm for 3 h. The homogenized inner gel was then filtered through a 125 mm filter paper from Macherey–Nagel to prepare the AV extract.
Preparation of nanofibers
A 20% (w/w) PEO solution was dissolved in deionized water. To obtain a homogeneous solution, it was stirred for 4 h at room temperature. In literature, high AV and/or EO concentrations (15%–25%) have been considered for different matrix materials in variety of structures.28,36 –39 And some reports indicate inactivity of the additives under 5% (w/w) loading. 28 Therefore, 20% (w/w) total additive amount was fixed in this study. AV loaded nanofibers were prepared by mixing PEO solution with 20% (w/w) AV for 2 h in a magnetic stirrer to homogenize. EO loaded nanofibers were prepared by stirring PEO solution with 20% (w/w) EO for 3 h. And AV/EO mixed samples were prepared by mixing PEO solution with 10% (w/w) AV and 10% (w/w) EO by stirring for 4 h. Stirring times were defined according to the homogeneity of the solution. 1% (w/w) of soy lecithin added drop by drop in 30 min by magnetic stirring.40 –42 The electrospinning process was carried out at room temperature using a 19–21 kV voltage range, 0.3 mL/s flow rate, and a 15 cm distance between the tip and collector copper plate in a setup consisting of a syringe pump (New Era Pump Systems Inc.), high voltage power supply (PHYWE Systeme GmbH & Co. KG), copper collector, and 10.0 mL syringe. After electrospinning, the obtained nanofibers were carefully removed from the copper plate and stored under suitable conditions for analysis.
Characterization
Structural characterization of the fibers was performed by scanning electron microscope (SEM, Gemini 500, Zeiss). The fibers were collected directly on the copper plate. Fiber size analysis was performed by measuring fiber diameter using Image J microscope software.
Thermal analysis of nanofibers was carried out by thermogravimetric analysis (TG,Perkin Elmer). Nanofibers were heated in nitrogen atmosphere from 50°C to 600°C. The measurement results were analyzed and plotted using Origin Pro 2023.
UV-Vis spectroscopy (UV-1800, Shimadzu) was used to measure the absorbance of additives in the nanofibers. Nanofibers were incubated in phosphate-buffered saline (PBS) at 37°C and measurements were taken at half-hour intervals for 2.5 h, followed by measurements at 1-h intervals for 3 h after 24 h. The data were analyzed and plotted using OriginPro 2023.
The moisture uptake capacities of the fibers were measured using a precision balance from Weightlab Instruments for 7 days. Changes in the weights of the fibers were recorded according to the ambient humidity on measurement days. The data were analyzed and plotted using OriginPro 2023.
Antibacterial test
Antibacterial test was conducted using the gram-negative bacterium Escherichia coli (E. coli, ATCC 25922). One of the better markers from a cleanliness standpoint was E. Coli. 43 The disc diffusion method was used to evaluate the generated materials’ antibacterial properties. This procedure involved cultivating the bacteria in Mueller Hinton agar broth liquid medium and incubating them for 24 h at 37°C. After that, the bacteria from the solid agar were moved to the nutrient agar plates, and a seed culture was created by cultivating the bacteria for 24 h at 37°C. The densities were then adjusted in accordance with Mc Farland 0.5. A slice of our sample, measuring 2 cm × 2 cm, was extracted and placed on nutrient agar plates together with 100 μL of a fresh culture for the growth inhibition assays. The samples were then incubated for 24 h at 37°C. The diameter of the inhibitory zones was used to gauge the sample’s antibacterial properties.
The standard ISO 22196, which is titled “measurement of antibacterial activity on plastics and other non-porous surfaces,” was also used to determine antibacterial activity. Samples are subjected to a predetermined quantity of germs in this standard. Targeted sample and control sample serve as comparisons. Following a 24-h period, the number of bacteria is counted, and the antibacterial activity of the targeted sample is determined by the logarithmic difference between the control and the sample. A sample is considered to be exhibiting antibacterial activity if the logarithmic difference is more than 2.
Results and discussion
The morphology and size of PEO, PEO/AV/EO and PEO/AV/EO/Lecithin nanofibers were investigated using SEM. SEM images of the samples are given in Figure 1 along with fiber diameters. Smooth and randomly oriented nanofibers were successfully obtained for all samples, indicating that the additives were homogeneously mixed in the PEO solution. Mean averages of the obtained nanofibers’ diameters are 0.25 ± 0.12 µm, 0.27 ± 0.1 µm, and 0.48 ± 0.23 µm, respectively. Lecithin is also known as a thickening agent in addition to being an emulsifier; therefore, addition of lecithin is assumed to affect not only solubility of different phases but also their viscosity. For the samples with the same composition, viscosity is directly proportional to the achieved diameters. 44 Results show that lecithin based thickening is consistence, even though the AV additive is known to reduce the diameter of the nanofibers.45,46 The diameters of PEO/AV/EO/Lecithin nanofibers are larger than other nanofibers due to the effect of lecithin.
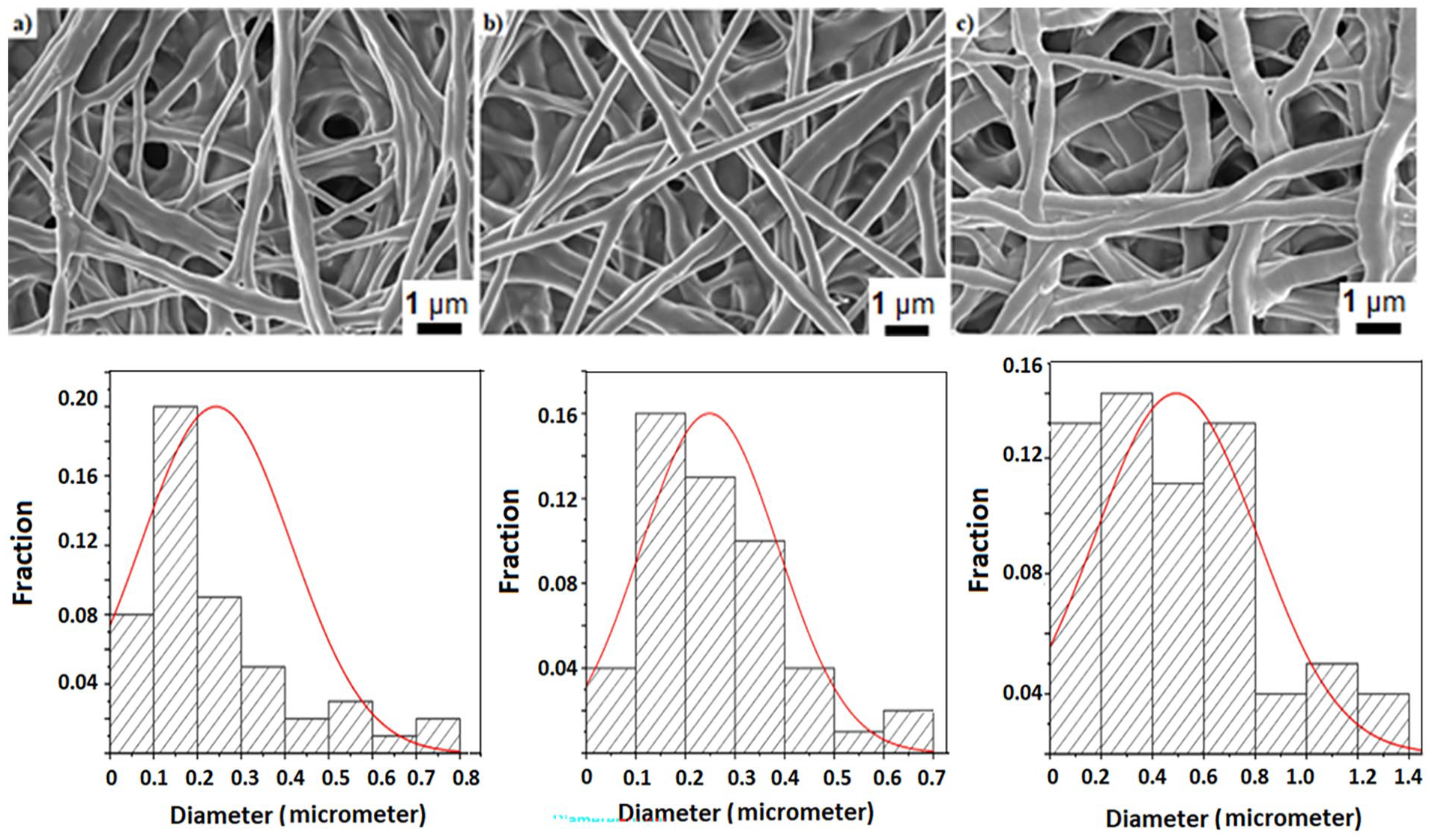
SEM images of (a) PEO, (b) PEO/AV/EO, (c) PEO/AV/EO/Lecithin nanofibers along with associated fiber size distribution graphics underneath.
The porosity between nanofibers, as observed in the SEM images, provides air and moisture permeability, which is effective in wound healing. 30 As the diameter of the nanofibers decreases, the surface area increases. This leads to a more porous structure, which is crucial for cell growth and proliferation.36,47 However, as the nanofiber thickness increases, packing density decreases48,49 and allows better air and moisture permeability.
The FTIR spectrum of nanofibers recorded in the 4000–500 cm−1 range was indicated in Figure 2. PEO nanofibers’ FTIR spectra revealed two peaks at 2880 and 1340 cm−1, which were attributed to CH2 wagging vibration and C-H stretching, respectively. The asymmetric and symmetric stretching of C–O–C were linked to the distinctive triplet peak at 1145, 1095, and 1058 cm−1. Furthermore, the stretching vibration of the molecular bonds C–O–C and C–C was identified as the cause of the band at 840 cm−1. These findings closely matched those found in earlier research.50,51

FTIR spectra of (a) AV, (b) PEO, (c) PEO/EO, and (d) PEO/EO/Lecithin nanofibers.
The distinctive broad band of phenolic compounds at 3330 cm−1, which corresponds to the stretching modes of the several -OH groups, was visible in the FTIR spectra of AV and EO dominated the spectra of all samples. Moreover, C=O and C=C ring stretching may be responsible for the maxima at 1745 and 1603 cm−1, respectively. 52
The peak at 3391 cm−1 is typically attributed to O–H bonds of phenols and alcohols. 53 The alkane group stretching (CH2) and the carbonyl group (C=O ester bond) vibration in EO and lecithin were caused by the bands that were already present at 2923–2855 and 1736 cm−1, respectively. The peak at 2923 cm−1 indicated the presence of –C–H (CH2; Vibration mode: Asymmetric and symmetric stretching). The peak at 1607 cm−1 corresponds to N–H (NH2), C=C (Alkene; Vibration mode: Bending, stretching). The peak at 1516 cm−1 is indicated to N=O (NO2) and C=C (Aromatic) stretching. The bond at 1405 cm−1 is attributed to –C–H (CH3) bending. The peak at 1269 cm–1, 1057.98 cm−1 can be assigned to the C–X, SO, C–N (NH2), C–O (OH, COOH) stretching. 53 These outcomes verified that AV was successfully incorporated into the PEO fiber mats following the electrospinning procedure.
Thermogravimetric analysis was conducted to evaluate the thermal stability of nanofibers. TGA curves of PEO, PEO/AV/EO and PEO/AV/EO/Lecithin nanofibers are presented in Figure 3. It shows the mass loss in the temperature range of 50°C–600°C. The weight losses were evaluated in three stages: the first stage was due to the loss of water or solvent from the samples, the second stage was due to thermal decomposition, and the third and final stage was due to carbonization of the material. 31 Pure PEO nanofibers degraded by 92% in the temperature range of 160°C–250°C. When TGA curves were analyzed, the first decomposition temperatures of PEO/AV/EO and PEO/AV/EO/Lecithin nanofibers were 164°C–285°C and 165°C–345°C, respectively, with degradation rates of 85% and 95%. The significant weight loss observed in the temperature range of 160°C–350°C was attributed to the breaking or fragmentation of the molecular structure. 54 Adding lecithin to the solution increased the decomposition temperature of the nanofibers, which enhanced their stability. 38

TGA curves of PEO, PEO/AV/EO, and PEO/EO/Lecithin nanofibers.
UV-Vis spectroscopy was performed to measure the concentration of the materials. The purpose of the UV-Vis measurement is to determine the absorption of the volatile content into the nanofiber structure and their stability in time. 55 The UV-Vis spectra of PEO, PEO/AV/EO and PEO/AV/EO/Lecithin nanofibers scanned between 200 and 700 nm wavelength are shown in Figure 4. The absorbance values of the nanofibers are 229, 234, and 237 nm, respectively. The addition of essential oils and emulsifier to PEO nanofiber has increased the absorbance values. In Figure 4(b), no peak is observed in the measurements taken at 26 h, which indicates that volatile content has completely left the system. On the other hand, with the addition of emulsifier structure is preserved even after 26 h which indicates that the release of volatile content like moisture and oils slows down with the addition of emulsifier. This can be an advantage to control the release of wound healing content from the patch that provides longer use time of the patch on the patient.

UV-Vis spectra of PEO, PEO/AV/EO, and PEO/AV/EO/Lecithin nanofibers.
Weight measurements were carried out for 7 days to analyze the holding capacity of PEO, PEO/EO, PEO/EO/Lecithin, PEO/AV/EO and PEO/AV/EO/Lecithin fibrous mats. Fibrous mats are affected by ambient humidity, and an increase in ambient humidity results in an increase in their weight. 56 While an increase in ambient humidity should result in an increase in weight for the PEO/EO mixture, no increase was observed. This is thought to be due to the volatility of EO at room temperature. Lecithin seems to increase stability of the EO in the fibrous mats. Furthermore, an increase in AV provides better results than lecithin in the sample. Figure 5 shows the weight change graphs of PEO, PEO/EO, PEO/EO/Lecithin, and PEO/AV/EO nanofibers according to ambient humidity. Table 1 was demonstrated the relative humidity on the days of measurement of PEO/AV/EO/Lecithin. According to the results shown in Figure 5, with natural oil additives fibrous mats perform more controlled weight loss which can be associated with higher moisturization performance of the mats. 35 Table 1 shows the relative humidity for the 7 days of measurement. Each humidity measurement was taken at noon time to avoid fluctuations. Relative humidity change measured looks within 15% difference between the highest and the lowest times. It is considerable amount; hence, some samples weight changes are beyond those limits which can be explained with the components of the fibrous mats.

Weight changes of PEO, PEO/AVPEO/EO, (d) PEO/AV/EO and PEO/AV/EO/Lecithin nanofibers according to ambient humidity on different days.
Relative humidity on the days of measurement of PEO/AV/EO/Lecithin.

Table 1 shows the relative humidity for the 7 days of measurement.
The antibacterial test results of PEO/AV/EO/Lecithin nanofiber were analyzed in Figure 6. As shown, an inhibition zone was observed for the targeted sample (Figure 6(b)) compared to the plain PP control sample (Figure 6(a)) and the diameter of inhibition is approximately 18.5 mm. Because of its antifungal, anti-inflammatory, antiviral, and antibacterial properties against a variety of illnesses, AV is utilized in skin-related treatments. AV’s antibacterial qualities have also shown strong antimicrobial efficiency against gram-negative E. coli. 39 These results are in coherence with bacteria growth measurements where antibacterial activity was measured for control and the targeted samples were 1.45 and 2.3, respectively.
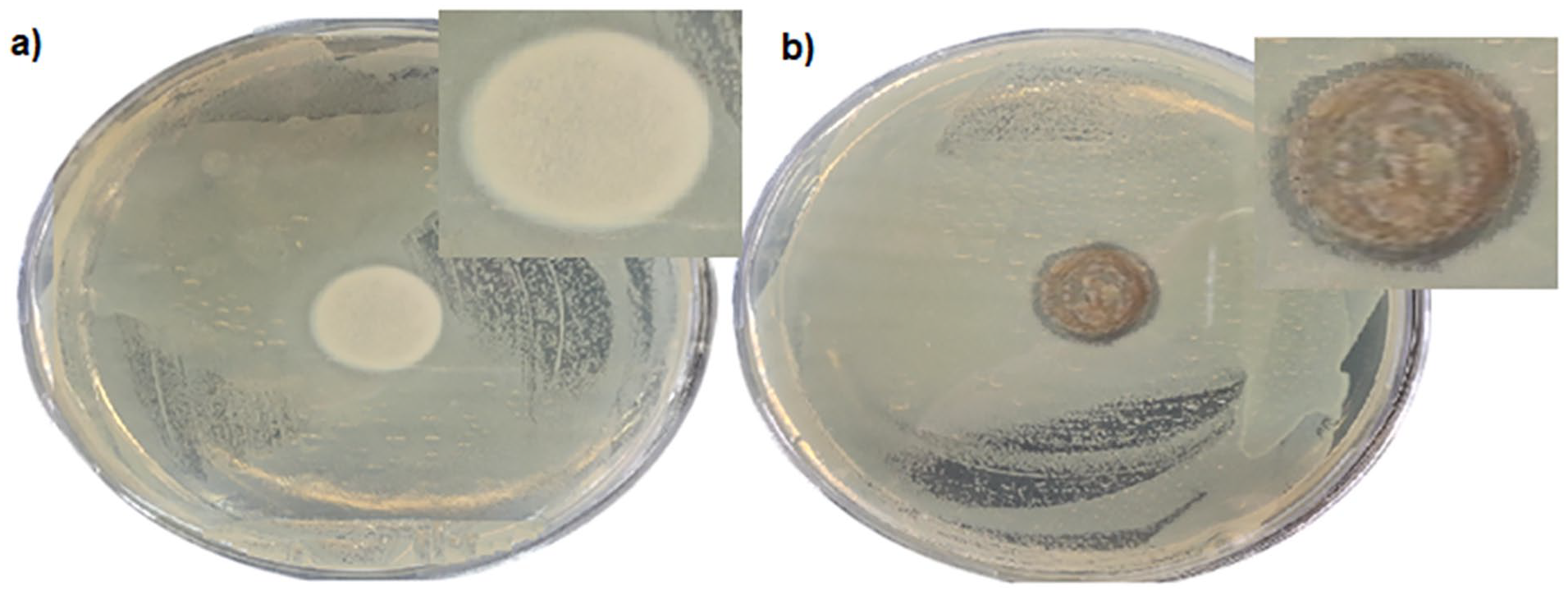
Antibacterial test results of (a) PEO fibrous mat and (b) PEO/AV/EO/Lecithin fibrous mat.
In Table 2, the results of this study were compared with the results of other literature studies. In this study, using the AV and EO along with lecithin, nanofibers provide long-lasting humidity in relation to ambient humidity.
Literature review about wound dressings.

Conclusion
In this study, smooth electrospun nanofibers were successfully obtained by using PEO as matrix material through electrospinning method. The obtained nanofibers were characterized using SEM, TGA, and UV-Vis. SEM analysis revealed that AV led to a decrease in the diameter of the nanofibers. Smaller nanofiber diameters provide greater surface area and porosity, allowing for air and nutrient permeability. It is highly important AV causes a decrease in nanofiber diameters while acting similar to lecithin. TGA results showed that AV increases the decomposition temperature, making the nanofibers more stable. EO enhances the absorption of the nanofibers by more than 100% compared to the PEO and AV + PEO fibrous samples. The moisture absorption property of the nanofibers is directly related to the ambient humidity. Increasing the ambient humidity can help maintain the moisture content of the nanofibers for a longer time; hence, emulsifier content has been proven to control water retention rate by 50% in this study. The antibacterial properties of PEO/AV/EO/lecithin nanofiber were also demonstrated against Escherichia coli. In conclusion, use of soy bean based lecithin in AV/EO/PEO system provides more homogeneous, stable and higher performance fibers to be used as wound dressings since it can absorb a lot of blood and exudates, prevent infections, and have good thermal stability with their porous and high surface area characteristics. The techniques used here may be appropriate for creating several biocompatible composite nanofibers from diverse materials to achieve comparable physicochemical characteristics for wound dressing or a variety of other uses. This kind of wound dressing will accelerate the healing process of the wound by preventing the adhesion of the material to the skin, providing moisturization and reducing the possibility of infection in the treatment of wounds by conducting clinical research in the future. To better understand how these nanofiber work in vivo, more research should be done. AV added nanofibers can also be beneficial for use in areas such as packaging when it needs to be used moist with its moisture retention feature.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Erciyes University Research Fund (Project No. FKA-2020-9978). We also would like to express our gratitude to TUBITAK for their support and assistance under the 2210-C Domestic Priority Areas Graduate Scholarship. We would like to thank the Erciyes University Office of the Dean for Research for providing the necessary infrastructure and laboratory facilities at the ArGePark research building and the Proofreading and Editing Office of the Dean for Research at Erciyes University for the copyediting and proofreading service for this manuscript.
Statement of research and publication ethics
The study is complied with research and publication ethics.
