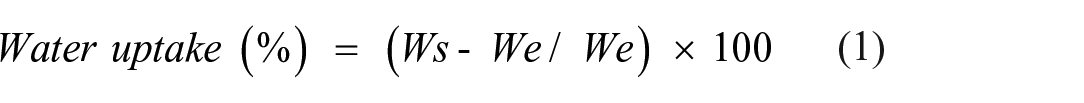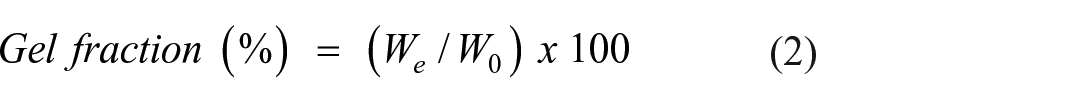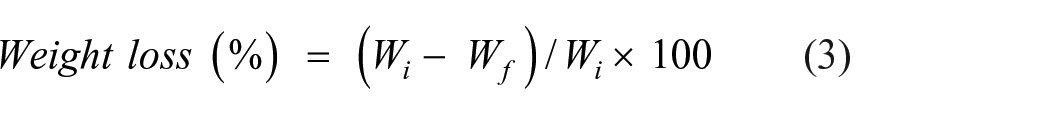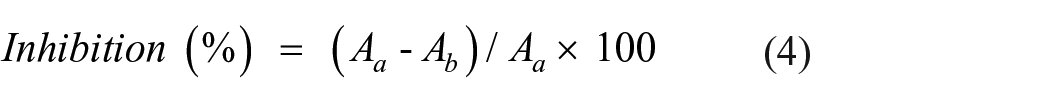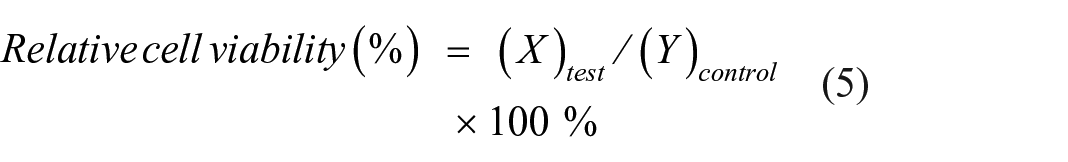Abstract
Vanillin loaded-physically crosslinked hydrogel membranes made of PVA/chitosan/itaconic acid (PVA-CS-IA) were prepared using freezing-thawing (F-T) cycle method. To ensure the entanglement of PVA-CS-IA chains, three F-T cycles were repeated. The polymeric chains entanglements were confirmed and characterized by different instrumental characterizations. Physicochemical properties for example, swelling ratio, mechanical characteristics, gel fraction percentage (GF%), hydrolytic degradation, and thermal stability of PVA-CS-IA membrane were discussed in detail. The findings showed that the swelling ratio, mechanical characteristics, and hydrolytic degradation of the crosslinked membranes enhanced with increasing CS-IA contents in membranes composition; however, GF% gradually declined with CS-IA content. Additionally, cell viability test using HFB-4 cell line and antimicrobial activity against Staphylococcus aureus and Escherichia coli were evaluated using MTT assay and the bacterium growth inhibition percentage method; respectively. Notably, with varying incubation durations and membrane concentrations, all examined constructed hydrogels showed significant cell survival percentages. The findings supported the notion that produced hydrogel membranes might be used in a professional setting as antibacterial dressings or biomaterials for quick wound healing rate.
Keywords
Introduction
Keeping the wound bed moist is one of the ideal conditions for quicker and better healing process. 1 In order to prevent microbial growth and maintain a moist environment, wound dressings must absorb excess exudates and liquids from the wound surface.Traditional wound dressing, such as cotton and gauze, cannot, however, achieve this criterion because cotton soaks up all wound exudates, leaving the wound surface extremely dried; additionally, the cotton adheres to newly formed epithelial cells, leaving the patient with excruciating pain and a slow healing process. 2 As a result, applying wound dressing via hydrogel membranes shall be more practical. In this regard, a hydrogel is a potential biomaterial for use in the manufacture of wound dressings. 3 Due to its solvent-free composition, biocompatibility, and nontoxicity, a physical method of crosslinking such as repeated freezing-thawing (F-T) cycles is more acceptable for medical demands than the conventional chemical crosslinking procedures.In addition, comparing to chemically cross-linked hydrogels, the physically generated gel has a great capacity to absorb water and is elastic in nature. 4
A common water-soluble polymer used in biomedical applications is polyvinyl alcohol (PVA), due to its benefits, for example, including being non carcinogenic, nontoxic, bio adhesive, and biodegradable with the ease of film-forming ability, it is also employed for many pharmaceutical purposes. 5 However, using PVA alone results in a rigid membrane with insufficient elasticity, which limits its utility as a polymeric membrane for wound dressing. 6 In order to enhance the overall biological characteristics of the resulting crosslinked composite membranes, PVA is therefore utilized preferentially in blends with other natural polymers. PVA-based hydrogel dressings provide a promising use as wound dressings and drug delivery systems, through delivering medications, DNA, growth factors, nanoparticles (NPs), and proteins to hasten the healing of wounds. 7 To treat infected wounds, for instance, mesoporous silica NPs and zinc oxide NPs were added to PVA hydrogels loaded with antibiotic cephalexin. 8
Poly-β-(1-4)-D-glucosamine, known as chitosan (CS), is a cationic marine polysaccharide made from a deacetylated derivative of chitin. It exhibits hemostatic activity, is non-toxic, biocompatible, and is biodegradable. Also, chitosan is produced cheaply, has fascinating ecological properties, and is widely available as a natural polymer.9,10 Numerous researchers have worked to improve the mechanical properties of chitosan hydrogels since they are insufficient for some biomedical applications. Because of their exceptional physical and biological characteristics related to their capacity for self-healing, biodegradability, biocompatibility, and antibacterial activity, self-healing materials made from chitosan have garnered a lot of attention. Preparation of a mixture of chitosan and PVA can lead to better mechanical qualities to overcome the drawback of both PVA as a rigid membrane and chitosan as a low mechanical properties membrane.11,12 It has been found that PVA-chitosan complex hydrogel dressings quicken wound healing. Electrospinning was used to create PVA-chitosan-hyaluronic acid (HA) dressings that were crosslinked with glutaraldehyde and included growth hormone. The resulting dressing is non-toxic, hydrophilic, and offers a sustained-release mechanism. Two days after the very release, 64% of the growth hormone was released through the nanofibers. 13
For the majority of applications, crosslinking chitosan is necessary to increase its resistance to acids, alkalis, and other chemicals; nevertheless, the networks thus created have a lower adsorption capacity than the linear polymer. 14 The chemical modification of chitosan with acids has been investigated as an innovative alternative way to create unique hybrid materials made of synthetic polymers and natural polysaccharides in order to increase the adsorption capacity for application in metal and dyes uptake and other pharmaceuticals preparation.12,15 Chitosan has been transformed into a variety of multipurpose adsorbents according to the amount of capacity needed for use. 16 Research on the preparation of chitosan composites for adsorption applications has increased in response to the evaluation of chitosan to capture metallic ions from natural and sea waters. 17 A search of the Scopus database for “Chitosan modifications for adsorption application” produced almost 122 research papers among the years 2015 and 2019. 18 Due to its being a biocompatible and simple to copolymerize, itaconic acid (IA) is frequently used for the synthesis of pH-sensitive hydrogels marketed as drug delivery systems.19,20 When this acid is added to hydrogels, even in very little quantities, the ionization of COOH groups causes a significant alteration in the hydrogels’ swelling behavior at the appropriate pH levels. Chitosan possesses innate biological characteristics when it is ionized.21,22
The focus of this research is to create HPNs from chitosan and PVA that would maintain a desirable level of swelling while also improving the mechanical qualities of pure chitosan hydrogels. The new method, in contrast to other literature findings, involved dissolving chitosan in itaconic acid. Ionic crosslinks between the chitosan macromolecules are created in acidic environments when the two COO− groups of itaconic acid bind with the NH+3 ions.The mechanical characteristics of chitosan are improved by this ionic crosslinking with dicarboxylic acid. 23
One of the most often used flavoring ingredients is vanillin (4-hydroxy-3-methoxybenzaldehyde), which is derived from the seeds of Vanilla planifolia plant. It is frequently used in foods, beverages, cosmetics, medicines, and other products.24,25 It is significant that vanillin has been identified as a vital bioactive chemical with anti-inflammatory, antiviral, antibacterial, antifungal, nephroprotective, and anticancer activities. It is used to treat a variety of illnesses, from skin cuts to sickle cell anaemia. 26 Additionally, it has been discovered that vanillin and its synthetic equivalents regulate gene expression, restrain the overproduction of inflammatory chemicals and free radicals, and promote tissue regeneration.27,28 Xiong et al. 29 offered hydrogels made by the freeze-thaw cycle approach and exhibited strong antioxidant qualities, outstanding mechanical and biological qualities, and the capacity to stop the growth of S. aureus and E. coli when made with PVA, CS, and V as raw materials. Amir et al., 30 evaluated the possibility of wound healing with chitosan-PVA hydrogel membranes. The membranes, which are enhanced with nanoparticles and reinforced with nanocellulose and vanillin, demonstrate strong mechanical strength, hydrophilic properties, good moisture retention, swelling behavior, biodegradability, and significant antimicrobial activity.
This study aims to prepare physically crosslinked vanillin loaded-PVA-CS-IA hydrogel membranes to construct and analyze possible topical wound dressings as self-sterilizing biomaterials. Moreover, CS’s solubility and antibacterial capabilities were improved by modification with IA, while PVA’s physicochemical qualities were enhanced by mixing with CS-IA. Finally, the resultant physically crosslinked PVA/CS/IA/vanillin composite membranes were bio-assessed in vitro in terms of its antimicrobial activity and cell viability (%).
Materials and methods
Materials
Polyvinyl alcohol (PVA) (98.9% hydrolyzed, average Mwt. 72,000 g/mol) was purchased from ADVENT CHEMBIO PVT LTD, Mumbai, India. Chitosan (CS) with (Mwt. of 1.30 × 105 g/mol and deacetylation degree of 85%) was purchased from Carl Roth, Germany. DMSO, vanillin, and itaconic acid were purchased from Sigma-Aldrich Chemie GmbH, Steinheim, Germany. This work was conducted entirely with distilled water.
Experimental part
Modification of chitosan onto itaconic acid derivative
The modification of chitosan onto itaconic acid was adapted according to the reported procedure of Abdelaal et al. 31 Typically, 0.5 g of CS was suspended in 30 mL of DMSO and 1.0 g of itaconic acid was also added to the stored mixture under stirring for 24 h at 60°C. The suspension pH was kept between 10 and 12 using 15% NaOH aqueous solution. Itaconic acid-modified CS was collected and dried after overnight at 40°C under reduced pressure. 32
Preparation of Vanillin loaded-PVA-CS-IA hydrogel membranes
PVA-CS-IA composite hydrogels were prepared using a freezing-thawing (F-T) cycle. Briefly, solutions containing 10% (w/v) PVA, (CS-IA = 0%, 10%, 20%, and 30%, (w/v)) CS-IA, and (0% or 5%) of vanillin solution were prepared in DW and 1% (v/v) acetic acid. The mixture was vortexed for 1 h before being poured into a Petri dish. Then, for three successive cycles, it was frozen at 18°C for 18 h and thawed at room temperature at 25°C for 6 h. The formed composite membranes were dried at 50°C in vacuum oven for 6 h (Scheme 1).
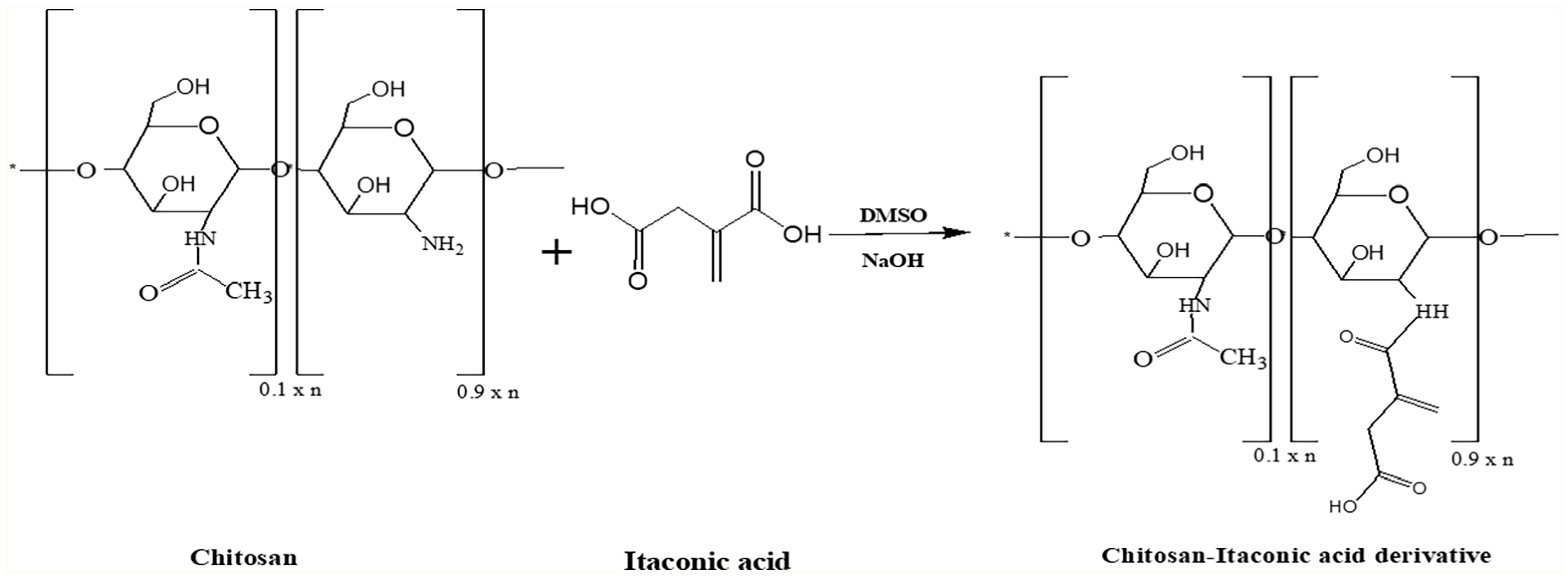
Modification reaction of chitosan with itaconic acid.
Methods
Water uptake
The weight of the dried samples was calculated after drying the membrane samples at 50°C in an oven for 6 h to test the water uptake of PVA-CS-IA-V hydrogel membranes (We). The dried samples were weighted (Ws) at predetermined intervals after being soaked in distilled water, stored at 37°C, and incubated. Using the following equation 33 the water uptake of PVA-CS-IA-V hydrogel membranes was calculated using equation (1).1,22
Where, (Ws) represents the weight of the swollen sample and (We) represents the weight of dry sample.
Gel fraction (%) of obtained hydrogels
PVA-CS-IA-V hydrogel membranes were first dried for 24 h at room temperature with sterile conditions, then dried for another 24 h at 50°C in an oven and weighted (W0). After that, dried samples were maintained in DW for 24 h to reach an equilibrium swelling weight (Ws). The hydrogel membranes were then weighted again after immediately drying at 50°C in the oven (We). The given equation was used to calculate the gel fraction GF(%) in Lu et al. 33
Where, We is the weight of the sample after it has swollen and dried, W0 is the weight of the membrane after it has dried.
Hydrolytic degradation
The degradation rate or weight loss (%) was calculated via a technique described by Xiao et al. 34 Briefly, 10 mL of PBS, or phosphate-buffered saline (0.1 M, pH 7.4) was used to soak 2 × 2 (cm) dried membranes. The membranes were removed at regular intervals, blotted with soft papers to remove the extra water, and dried in ambient conditions. The bulk was also weighed at regular intervals. With Wi being the membrane′’s initial mass and Wf being each piece that was removed from the medium then dried in an oven, and then weighed, the weight loss (%) was computed using the following equation:
Bio-evaluation tests
Antimicrobial activity test
Depending on the bacterial strain, antibacterial agents can either kill germs or prevent them from reproducing. The antibacterial activity can be carried out through different methods, including broth or agar dilution, well-diffusion, and disc-diffusion.35,36 According to the reported method of Hassan et al., 2 the antibacterial activity of the produced PVA-CS-IA-V were assessed in this work where E. coli (NCTC10418) and Staphylococcus aureus (ATCC 25923; ATCC) isolates used in an in vitro test of antibacterial activity were standardized. In brief, 1% LB broth medium was used to dilute the previously renewed bacteria suspensions up to 100 times. After sterilizing at 121°C for at least 20 min, 100 μL of diluted suspension containing 0.1 g of the test sample was cultured in 10 mL of Tryptone medium. After that, the mixture was shaken for 18 h at 37°C. and the percentage of bacterial growth inhibition was obtained by measuring the culture medium’s absorbance at 600 nm with visible spectroscopy.
The following equation was used to compute the inhibition (%):
Where, Ab and Aa represent, respectively, the absorbance of the bacterial culture in the absence and presence of the tested substance.
In vitro cytotoxicity test
Colorimetric MTT assay was used to assess the cell viability % of tested membranes of (PVA, PVA-V, PVA-CS, PVA-CS-IA-V (90:10), PVA-CS-IA-V (80:20), and PVA-CS-IA-V (70:30)) affected the viability of HFB-4 (normal skin melanocyte of human) cells.37,38 The attached cells were placed in sterile 24-well flat-bottom plates at a density of 1.0 × 105 cells/well and treated with all tested substances at disc weights of (1.0, 2.0, 3.0, 4.0, and 5.0 mg/mL) in triplicates. The cells were then incubated for 2, 4, and 6 days at 37°C in a 5% CO2 incubator. The following formula was used to determine the cell viability %, as compared to control (i.e., untreated cells):
Characterization
Ft-Ir
On an EQUINOX 55 instrument, vacuum dried samples of freeze-thawed PVA-CS-IA-V membranes were investigated by FT-IR (BRUKER, Germany). The dried test materials were blended with infrared grade KBr, ground, and then pressed to create translucent KBr-discs. The FTIR spectrums were acquired by capturing 64 scans at a resolution of 2 cm−1 between 4000 and 400 cm−1.
Mechanical properties
Using a tensile test machine (model: AG-I/ 50N-10KN, Japan), the maximum tensile strength and the elongation-to-break (%) of PVA-CS-IA-V composite hydrogel membranes were measured. PVA-CS-IA-V membranes were shaped into a dog-bone-specific design (6 cm long, 2 cm wide at end, and 1 cm at middle). For determining the load for each sample, the study was carried out at a stretching rate of 20 mm/min with a pre-load of 0.5 N. Before analysis, the thickness of the membrane samples was measured using a digital caliper.
SEM investigation
Analytical-SEM (type: JEOL, JSM-6360LA, Japan) with 10 kV voltage for secondary electron imaging was used to examine the surface and inner structure of lyophilized hydrogel membrane samples. Using a freeze-dryer to dehydrate the xerogel membranes, they were then coated with Au using an ion sputter coater (model: 11430, USA, combined with vacuum base unit or SPi module control, model: 11425, USA).
Thermal analysis by TGA
Thermogravimetric analysis (TGA), carried out on a 204 Phoenix TGA apparatus (NETZSCH, Germany) from 50°C–600°C at a heating rate of 10°C/min, was used to determine the thermal stability of dried PVA-CS-IA-V membranes.TGA thermograms were used to determine the onset temperature (Tonset). The temperature at the point of inflection or maximum rate of mass loss is known as the onset and is measured at the intersection of the baseline mass and the tangent drawn to the mass loss curve. 39
Results and discussion
FT-IR of Cs-IA modified derivative
FT-IR spectra of CS and CS treated with IA are displayed in Figure 1. The distinctive peaks approximately at ν 898 and 1158 cm−1 (antisymmetric stretching of the (C-O-C bridge) corresponding to saccharide structure) might be seen in FTIR spectra of pure chitosan (Figure 1). Chitin and chitosan both exhibit absorption peaks at ν 1654 and 1321 cm−1, which have been identified as amide I and III peaks, respectively. CH3 symmetric group deformation mode is responsible for the sharp peaks at ν 1382 and 1424 cm−1, while C-O stretching vibration in chitosan is responsible for the broad peaks at ν 1075 and 1033 cm−1. The stretching vibration of O-H and/or N-H, as well as intermolecular hydrogen bonding within the polysaccharide, should be attributed to the intense band at approximately ν 3434 cm−1. The typical C-H stretch vibrations have peaks at ν 2875 and 2922 cm−1. Further, CH2 groups in CS-IA derivative (b) contributed significant absorption at ν 1441 cm−1. While there are no bands at ν 1700 cm−1 connected to the carboxylic C=O group, there was an absorption at ν 1647 cm−1 related to the ketonic C=O group, showing that the carboxyl group engaged in the interaction was totally reacted. The C=O-NH group was associated with a high absorption at ν 1569 cm−1, whereas the CH2-bending and NH groups were associated with absorptions at ν 2924 and 3440 cm−1; respectively. These absorptions attested to how easily CS interacts with IA.
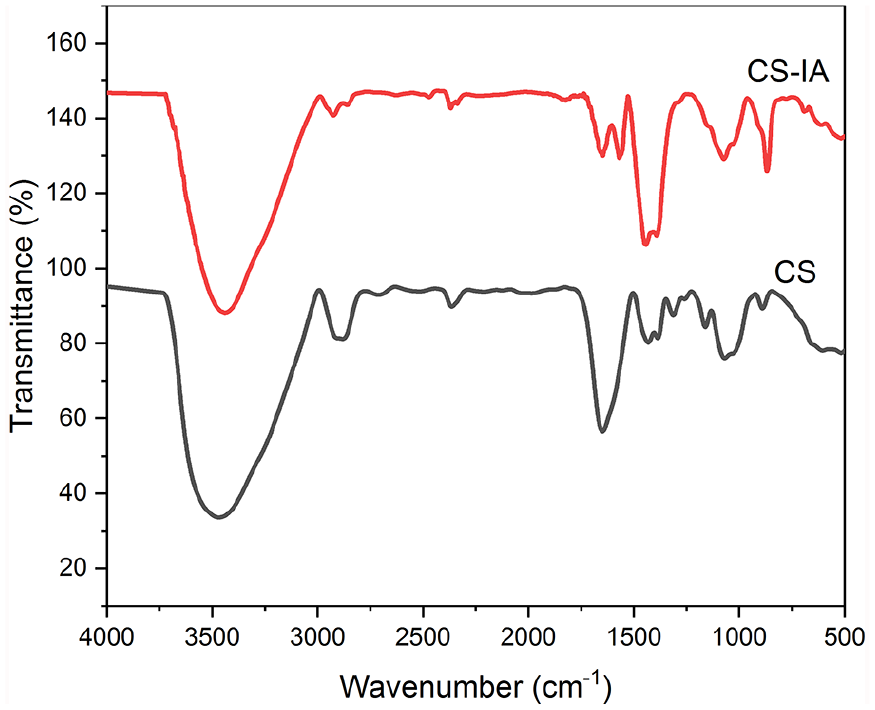
FT-IR spectroscopic analysis of itaconic acid–modified CS (CS-IA) membrane.
Swelling study of PVA-CS-IA-V hydrogel composite membranes
As shown in (Figure 2), shows the proportion of water held by PVA-CS-IA-V hydrogel membranes with various compositional ratios. As observed, when the content of CS-IA against PVA is increased in membranes composition, the swelling ratio (%) rise in a similar manner. This is a result of the strong hydrophilic capacity of chitosan derivatives to establish hydrogen bonds with water in the swelling medium. A high crosslinked and compressed structure of PVA membranes is achieved in absence of CS-IA (i.e., 100% PVA), however these membranes are unable to hold onto water, leading to limited swelling ability and a water uptake (%) of roughly 265%. Notably, the water uptake % dramatically increases to 370%, when CS-IA level reaches 30%. As a result, the wettability and hydrophilicity characteristics of hydrogel membranes are significantly improved by increasing the concentration of CS-IA in PVA membranes. These results are agree with those obtained findings by Martinez-Ruvalcaba et al. 40 ; they demonstrated that swelling characterization and drug delivery kinetics of poly acrylamide-co-itaconic acid/chitosan hydrogels, where the swelling ability increases with increasing of CS content. In addition, Wei et al., 41 prepared chitosan hydrogel dressings based on PAAc/CFCS/vanillin for diabetic wound healing, where the composite hydrogel has exhibited good adhesive qualities and enough swelling properties to stop quickly the bleeding from wounds.
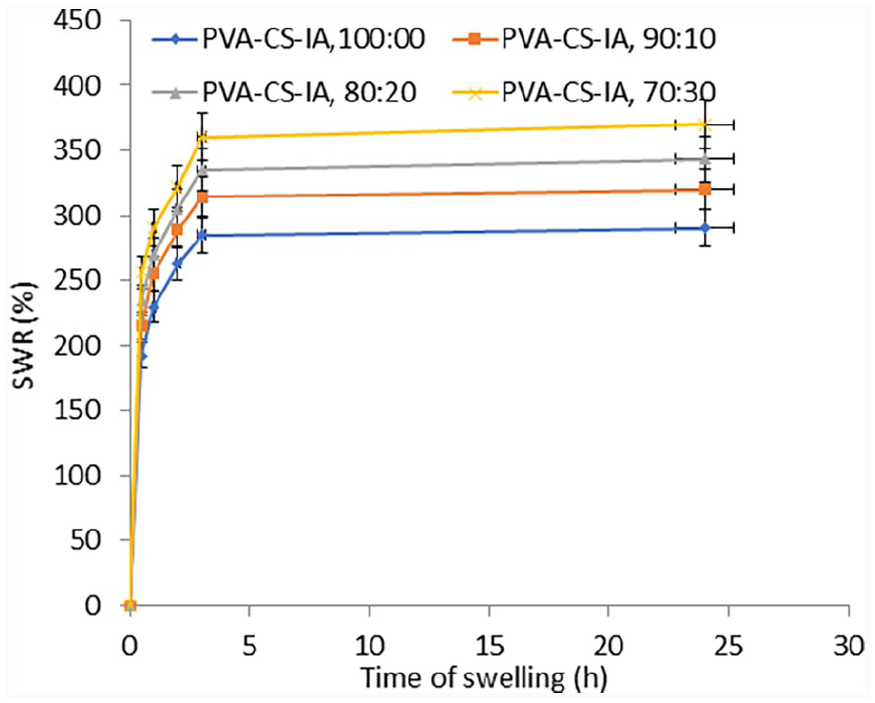
Effect of membrane composition on the swelling ratio% of (PVA-CS-IA) hydrogel membranes.
Gel fraction (%) of PVA-CS-IA-V hydrogel composite membranes
The gel fraction of PVA hydrogel membranes against various CS-IA contents is shown in Figure 3. The findings demonstrate that the gel fraction of produced membranes decreases with increasing the CS-IA content. Since, PVA-CS-IA-V polymer hydrogel membranes were prepared by successive F-T cycles that become entangled based on the content of PVA. Additionally, by boosting the CS-IA content in PVA hydrogel, the crosslinking reaction and subsequent gelation process might be significantly slowed down. The gel fraction grows to its highest level, which is around 92%, in the absence of CS-IA content. This result is consistent with the outcomes of our previous study conducted by Kenawy et al. 36 ; who found PVA-quaternized chitosan blended hydrogels fraction decreased with increasing Q-chitosan. However, the GF% increased with increasing PVA content in the membrane’s blend.
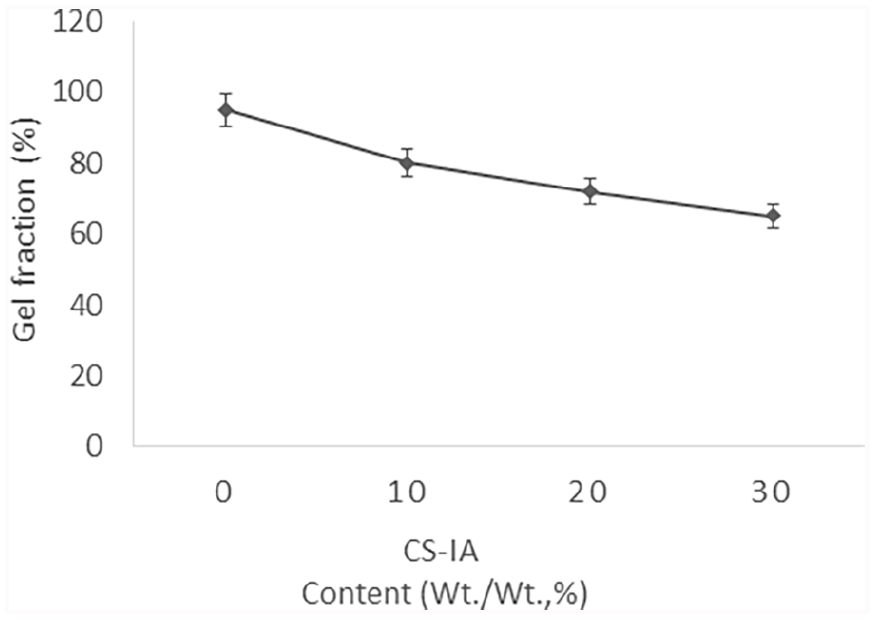
Effect of CS-IA content in PVA hydrogel membranes on GF %.
Hydrolytic degradation of PVA-CS-IA-V hydrogel composite membranes
Hydrolytic degradation of PVA with various CS-IA contents, membranes in PBS is depicted in Figure 4. The provided findings show that a high CS-IA component in the membrane composition causes a greater hydrolytic breakdown of PVA membrane. This could be because the membranes made of PVA-CS-IA-V are significantly degraded because of the cleavage of available PVA’s entanglement segments are broken down. These findings are explained by the observation that whereas PVA degrades very slowly, PVA-CS-IA degrades somewhat more quickly than CS-IA contents. Notable PVA hydrogels have lost almost 5% of their weight, while (PVA-CS-IA, 70:30) have lost almost 30% of their weight after 24 h. Additionally, PVA and CS-IA are nontoxic, and it has been suggested that the hydrogel formed from PVA and CS-IA, as well as the byproducts of its degradation, are nontoxic biomaterials. These latter results are in agreement with those from Kenawy et al., 42 who found that hydrolytic degradation rates of PVA-HES blended membrane increased gradually as HES contents in PVA hydrogel membrane increased.
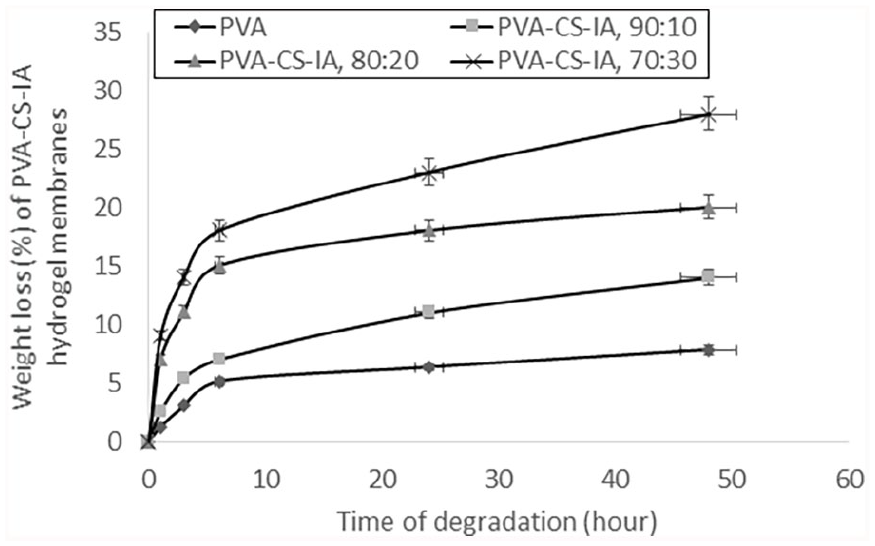
Effect of CS-IA contents of PVA-CS-IA-V hydrogel membranes on weight loss (%) through different degrading times in PBS (0.1 M, pH 7.4, at 37°C).
Mechanical properties of PVA-CS-IA-V hydrogel composite membranes
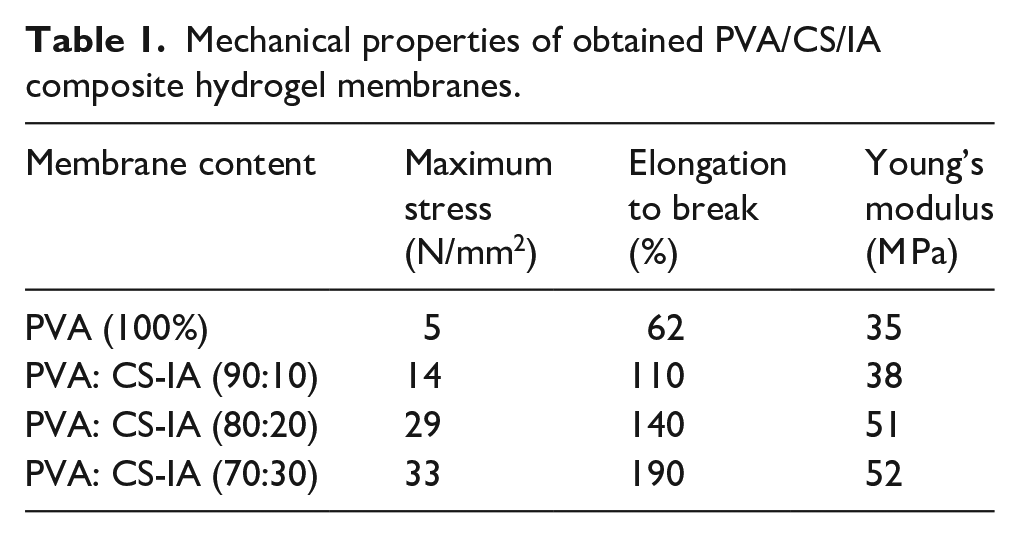
To ensure patient comfort and integrity during storage and handling, it is advantageous for targeted dressing materials to have strong mechanical strength with balanced degradation rate as discussed above. The maximum tensile strength and elongation at break have been measured to explore the blending influence of CS-IA on the mechanical characteristics of PVA hydrogel membranes (Table 1). The data is compiled and listed in Table 1 as a function of differed membrane composition ratios (PVA: CS-IA). As shown, the addition of CS-IA copolymer from 0% to 10%, 20%, and 30%, respectively, increases the elongation at break of physically entangled membranes from 62% to 110%, 140%, and 190%; respectively with increasing of CS-IA as unexpected. This is because CS-IA is more hydrophilic and easier to homogenize than PVA membranes. Also, it might be the cause of the increased elongation percentage and improvement in elasticity of hydrogel membranes. Due to the elastic nature of CS-IA, Young’s modulus and maximum stress onto hydrogel membranes also increase clearly with increasing inclusion of CS-IA into hydrogel membranes (Table 1). Thus, the already developed membranes are advised for use as dressings biomaterial for burns and wounds. These findings are in line with findings of Zhao et al., 43 which show that the amount of CMCs in hybrid membranes enhanced elongation at break and tensile strength. Moreover, Zhang et al., 44 prepared crosslinked CS films with 5% vanillin, where the results proved the slight beneficial effect of vanillin on the mechanical properties, particularly the elasticity and elongation-to-break of prepared CS films.
Mechanical properties of obtained PVA/CS/IA composite hydrogel membranes.
Surface morphology of PVA-CS-IA-V hydrogel composite membranes
Figure 5 shows the morphological surface structure of PVA membranes in comparison to various CS-IA regions. SEM images show a very homogeneous, smooth, compressed, pressed and nonporous shape-surface structure in absence of CS-IA (Figure 5(a)). However, the inclusion of CS-IA into PVA hydrogel in various concentrations (10%, 20%, and 30%, w/v) led to the creation of extremely minute pores at the surface, where formed surface-pores noticeably become larger with increasing CS-IA contents in the membranes (Figure 5(b)–(d)). The good homogeneity, hydrophilicity, or high degree of miscibility between two components of composite membranes (PVA and CS-IA) might be responsible for the morphological modifications. The strongly tangled PVA and the disorderly crystalline phase of PVA membranes were blended with various portions of CS-IA to produce an ordered crystalline phase and unified shape construction in case of 0% of CS-g-GTMAC. The results of current SEM investigation are in line with those that reported by Kamoun et al., 45 where who reported similar SEM findings for PVA-SA hydrogel blend membranes. Also, those obtained by Amir et al., 30 where the antibacterial effect of prepared wound dressing made from chitosan-PVA hydrogel enhanced with vanillin and mechanically was reinforced with nanocellulose incorporation and CuO-Ag nanoparticles. They also found that, incorporating of vanillin into CS-PVA hydrogels made the surface was more dense and smooth.

SEM micrographs of physically crosslinked PVA-CS-IA hydrogel membranes composed of (a) PVA (100%), (b) PVA-CS-IA (90:10), (c) PVA-CS-IA (80:20), and (d) PVA-CS-IA (70:30), (original magnification of all images ×5000 at 10 Kv).
Thermal stability of PVA-CS-IA-V hydrogel composite membranes
Figure 6 exhibits the influence of CS-IA incorporation in different ratios on the thermal stability of PVA membranes. As seen, all tested membranes regardless of their composition have lost the initial weight from 25°C to 250°C, due to evaporation of humidity, residual solvents, and water. Meanwhile, the second decomposition stage was significantly enhanced and prolonged, owing to the CS-IA incorporation; respectively. Where, the second decomposition stage of PVA membrane is recorded at 253°C–450°C with losing more than 58% of its weight. This stage was enhanced to 263°C–522°C, due to incorporation of CS-IA portion to membranes, with weight loss reaching 48%. These findings are consistent with reported TGA results of PVA-HES hydrogel membranes, due to HES incorporation in different portions by Kenawy et al., 34 and Kamoun et al. 36 Also, Suneetha et al., 46 have reported that the thermal stability of hydrogels made from fungus-derived carboxy methyl chitosan (FC)/vanillin (V)/PVA physically crosslinked by freeze-thaw method, has improved significantly by incorporation of vanillin.
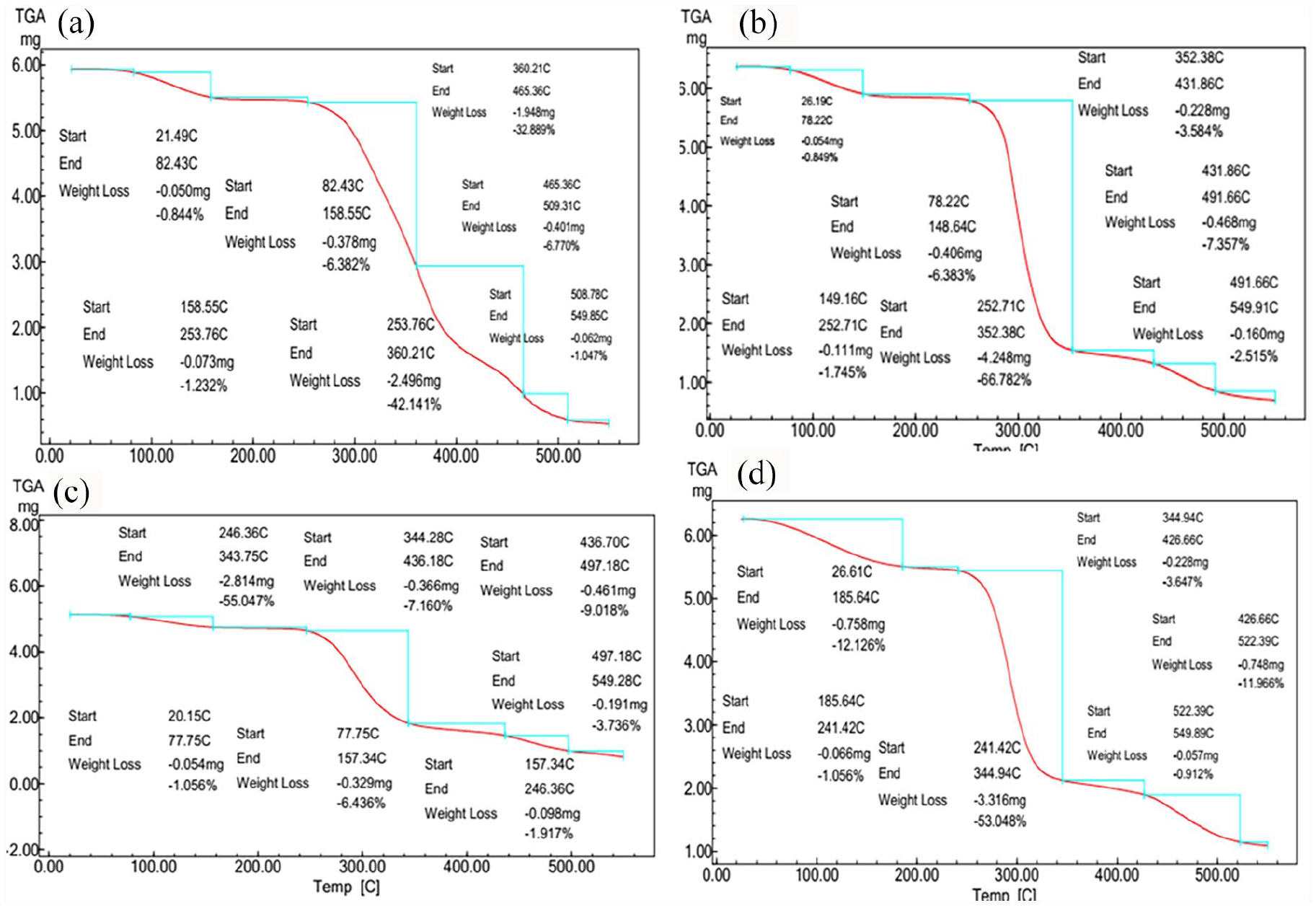
TGA thermographs of physically-cross-linked hydrogel membranes composed of (a) PVA (100%), (b) PVA-CS-IA (90:10), (c) PVA-CS-IA (80:20), and (d) PVA-CS-IA (70:30).
In vitro assessments
Antibacterial activity of PVA-CS-IA-V hydrogel composite membranes
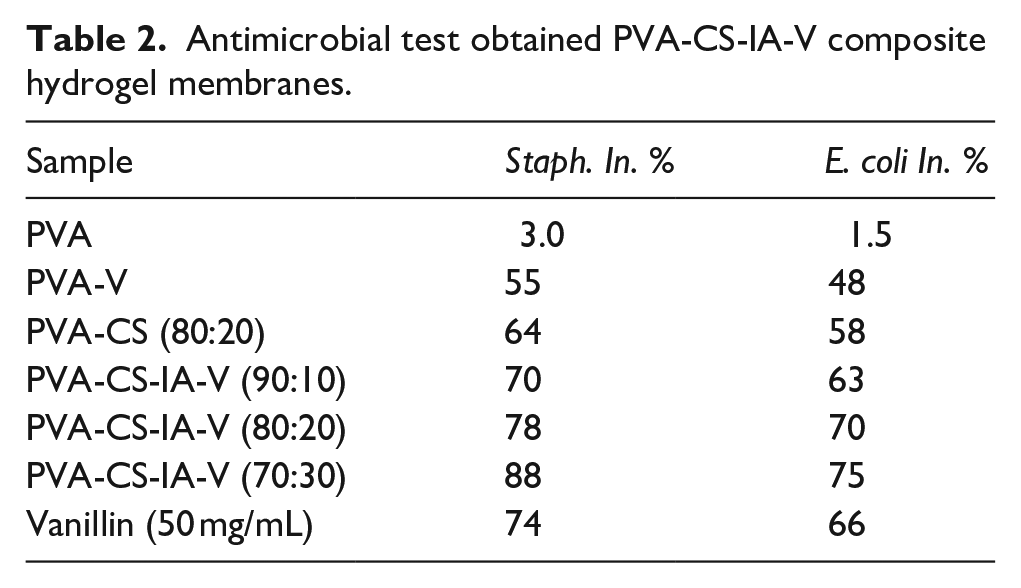
The potent activity might prevent wound infection with bacteria and speed up the healing process, when these materials used in vivo.35,47 Table 2 provides an overview of PVA-CS-IA-V membranes’ antibacterial efficacy against S. aureus (Gram positive) and E. coli (Gram negative). Pure PVA failed to exhibit an inhibition action, demonstrating that it lacks to antibacterial action. 48 Although CS possesses inherent antibacterial capabilities, the precise mechanism underlying its antimicrobial activity is unknown. 49 The method of action might be based on CS interaction with microbial DNA, which causes CS to enter the nucleus of bacteria and suppress mRNA and protein production.50,51 Also, Moreira et al., 52 indicated that the polycationic property of the molecule, which allows interaction and creates polyelectrolyte complexes with polymers produced at the bacteria cell surface, may be the cause of the CS’s antibacterial activity. The difference between S. aureus and E. coli in the inhibitory impact of the PVA-CS-IA-V membranes may be due to the structure of their cell walls being different.36,53 It was shown that increasing the amount of CS-IA in the tested membrane discs significantly increased the inhibition percentage which agreed with the results obtained by Kenawy et al. 53 on their work on the antibacterial activities of hydrophilic-modified nonwoven PET where they found that the quantity of biocidal activity increases with an increase in the CS grafted with IA concentration derivative. The antibacterial activity of PVA-CS-IA films was significantly enhanced by the incorporation of vanillin. This could be explained by the fact that vanillin naturally has antimicrobial properties. These results are in agreement with those obtained by Narasagoudr et al., 54 on their work on ethyl vanillin/chitosan/poly(vinyl alcohol) active membranes for food packaging applications where they found that on the basis of the inherent antibacterial activity of CS and EV, the improved antibacterial activity of CPEV mix films may be explained also noticed higher antibacterial activity in CS/PVA films with increasing CS contents.
Antimicrobial test obtained PVA-CS-IA-V composite hydrogel membranes.
Cell viability (%) by MTT assay
MTT assays on human HFB-4 normal skin melanocyte cells were carried out to verify the efficacy and compatibility of tested membrane composition for example, PVA, V, CS, and IA for using them as biomaterials (Figure 7). Our findings show that all prepared membranes reduce the toxic effects of HFB-4 cells, as compared to untreated cells. With a viability of roughly 93%, the maximum achievable toxicity is detected with all tested membranes at 5.0 mg/mL after 6 days of incubation. However, after being incubated for all the investigated durations, the vitality of the HFB-4 cells-maintained levels around 100% at doses of 1.0, 2.0, and 3.0 mg/mL. These results demonstrate the effectiveness of the approach for encapsulating V, CS, and IA within the PVA polymer, which may result in a decrease in their cytotoxicity without lowering the doses utilized. Additionally, it is observed that the produced membranes′ cytotoxicity towards the HFB-4 cells decreased over time when IA content and V increased, from PVA-CS-IA-V (90:10) to PVA-CS-IA-V (80:20) and PVA-CS-IA-V (70:30), compared to PVA alone. The higher concentration of IA and V in polymer complexes may be responsible for this improved outcome. These results corroborated those published by Tomić et al., 55 indicating that the addition of itaconic acid increases the cytocompatibility of the hydrogels, depending on the amount of IA in the gel, the copolymer samples demonstrated improved cell viability than PHEMA.
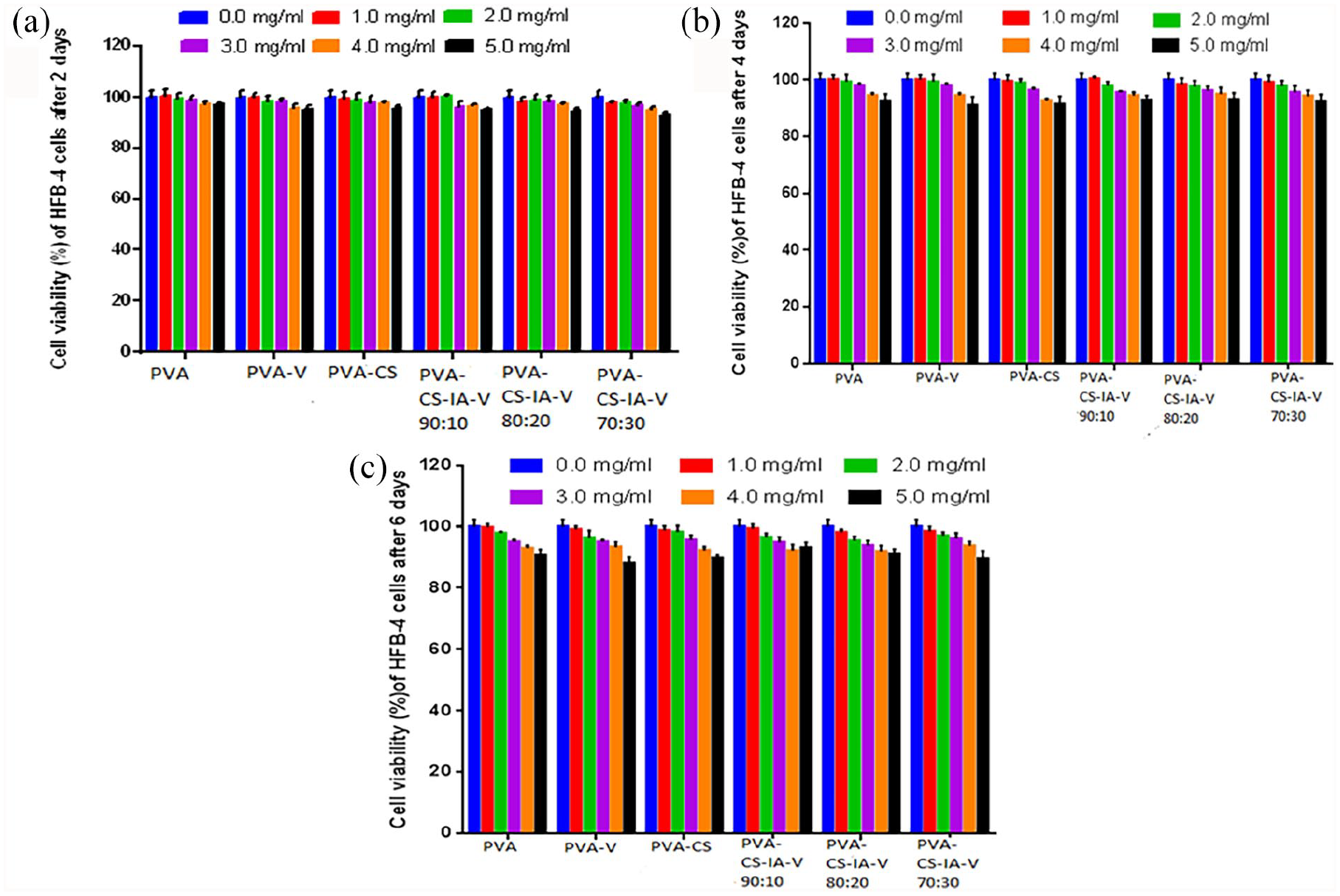
Cell viability by MTT assay of tested different concentrations of membranes (0, 1, 2, 3, 4, 5 mg/mL) onto HFB-4 cell line after (2, 4, 6 day) of incubation.
Conclusions
Hydrogel membranes composed of PVA-CS-IA-V were physically crosslinked and used as biomaterials for antibacterial wound dressings. PVA-CS-IA-V composite hydrogel membranes were characterized by FTIR, TGA, and SEM analyses, in addition the addition impact of different CS-IA and vanillin contents into PVA were assessed in terms of antimicrobial activity and toxicity. By adding more CS-IA and vanillin, the produced composite hydrogel membranes demonstrated a good antibacterial activity against both G+ and G− microorganisms, particularly noticed with vanillin inclusion. Furthermore, the inclusion of CS-IA and vanillin into PVA membrane significantly reduced the toxicity effect and enhanced the cell viability of tested membranes particularly with vanillin incorporation. The unique prepared PVA-CS-IA-V composite hydrogel membranes are also considered potential biomaterials and are good antibacterial, biodegradable, and cellular toxicity outcomes examined those materials for topical wound healing applications.
Footnotes
Acknowledgements
Not applicable.
Credit authors statement
SH: Experiments, data acquisition, interpretation of data, data analysis and wrote the original draft; EAK: Design of the work, draft revision, revised the final draft. YA-GM: Experiments and drafting microbiology part. EK: Supervision and revised the final draft. SME and AAE: Supervision. All authors have approved the manuscript before submission. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
Consent for publication
Not applicable.
Data availability
All raw data of measurements is available and could be shared when requested, corresponding author (EAK) is fully responsible for providing all data requested.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
Not applicable, no animal was used in this study.