Abstract
Despite advancements in therapeutic techniques, restoring bone tissue after damage remains a challenging task. Tissue engineering or targeted drug delivery solutions aim to meet the pressing clinical demand for treatment alternatives by creating substitute materials that imitate the structural and biological characteristics of healthy tissue. Polymers derived from natural sources typically exhibit enhanced biological compatibility and bioactivity when compared to manufactured polymers. Chitosan is a unique polysaccharide derived from chitin through deacetylation, offering biodegradability, biocompatibility, and antibacterial activity. Its cationic charge sets it apart from other polymers, making it a valuable resource for various applications. Modifications such as thiolation, alkylation, acetylation, or hydrophilic group incorporation can enhance chitosan’s swelling behavior, cross-linking, adhesion, permeation, controllable drug release, enzyme inhibition, and antioxidative properties. Chitosan scaffolds possess considerable potential for utilization in several biological applications. An intriguing application is its use in the areas of drug distribution and bone tissue engineering. Due to their excellent biocompatibility and lack of toxicity, they are an optimal material for this particular usage. This article provides a comprehensive analysis of osteoporosis, including its pathophysiology, current treatment options, the utilization of natural polymers in disease management, and the potential use of chitosan scaffolds for drug delivery systems aimed at treating the condition.
Keywords
Introduction
The skeletal system is a vital component of the human body, comprising several tissues that contribute to its structure and function. 1 Approximately 60% of human bone is formed of inorganic mineral content, with hydroxyapatite being the most often recognized. The remaining 30% is comprised of an organic matrix, which is made up consisting of collagen, proteoglycan, and lipid. Additionally, bone contains approximately 10% cells and blood vessels, all of which play crucial roles in its maintenance and overall health. 2 The skeleton serves several crucial roles inside the human body. Firstly, it provides structural support, ensuring the integrity, and stability of the body. Additionally, it offers support to vital organs housed within its framework. Furthermore, the skeletal system serves as a reservoir for essential minerals and contributes to the creation of blood.3,4 The complex and specialized dynamic tissues undergo continuous metabolism and remodeling throughout an individual’s lifespan in order to sustain a robust skeletal framework for various physiological processes. Bone metabolism encompasses a complex interplay of various fundamental bone cells, including osteocytes, osteoblasts, and osteoclasts, which individually or collectively play pivotal roles in maintaining the equilibrium between bone breakdown and formation.5,6 The remodeling of bone commences with the activation of osteocytes through mechanosensory stimuli, subsequently leading to the recruitment of osteoclasts to the diseased areas of bone. 7 In addition, the recruitment of MSCs and bone progenitors is observed, resulting in the specialization of MSCs into osteoblasts. The aforementioned process finally culminates in the mineralization of the osteoid matrix, hence facilitating the development of new bones. Altering any of these stages has the potential to result in bone metabolism disorders. 8
Some prevalent bone related ailments are osteoporosis, osteomyelitis, Paget’s syndrome, osteosarcoma, and arthritis. 9 Osteoporosis is a degenerative condition affecting the body’s skeleton, which leads to a reduction in bone density and degeneration of the microarchitecture of the bone tissues. This leads to heightened vulnerability to fractures and enhanced fragility of the bones. 10 It is a lifelong progressive disease characterized by the gradual loss of bone mass. Osteoporotic patients commonly experience limited or moderate symptoms prior to the onset of osteoporotic fragile fractures. 11 The fractures give rise to a range of unfavorable outcomes, encompassing persistent discomfort, impairment, depressive symptoms, diminished standard of life, and higher mortality. 12 The worldwide incidence of osteoporosis has appeared as a significant public health concern due to the increasingly rapid aging of population. On a global scale, the occurrence of osteoporosis is estimated to affect over 200 million individuals, with approximately 9 million fractures due to fragility occurring each year. 13 The numbers presented above depict the simultaneous increase in the older population facing a decline in their general standard of life and the rising socioeconomic burden. 14 However, the existing pharmacological and surgical interventions employed for the treatment of osteoporosis are deemed inadequate. Hence, it is evident that there exists an urgent requirement and growing desire for novel pharmaceuticals targeting osteoporosis as well as nanomaterials aimed at enhancing the healing process of fragility fractures.
In modern times, there has been a discernible surge in the exploration of the application of polymers derived from naturally occurring substances for various medicinal and biological purposes. 15 These applications encompass pharmaceutical delivery systems, engineering tissues, wound treatment, and packaging for food, among others.16,17 Naturally occurring polymers encompass two main categories: protein-based and polysaccharides-based. In contrast to polysaccharides, proteins exhibit amino acids sequences that are commonly associated with cellular attachment through integrin-binding domains. In contrast, it is necessary to augment cell adhesion and osteoconductivity in polymer scaffolds through chemical surface changes, the blending of osteoconductive substances, or the inclusion of integrin-binding sequence or cell adhesive proteins. The polymers of natural origin that are frequently investigated for biomedical applications encompass chitosan, collagen, alginate, peptides, and other more compounds.18,19 Chitosan is a widely utilized polymer in the field of medical research, ranked as the second most prevalent after cellulose. It is classified as a naturally existing amino polysaccharide. 20 The solubility in water of chitosan is attributed to its polycationic properties, enabling it to effectively connect to negative-charged surfaces, including mucous membranes, due to its bioadhesive nature. Consequently, it augments the adherence to the mucosal surface, thus prolonging the duration of drug molecule interaction for enhanced penetration. 21 The compound exhibits remarkable biocompatibility and minimal toxicity owing to its molecular and structural similarities to endogenous glycosaminoglycans. 22 Chitosan undergoes rapid biodegradation, resulting in the formation of nontoxic byproducts known as amino sugars, which are readily absorbed by the human body. 23 Moreover, the properties and effects of chitosan in biological systems can be altered through appropriate physical and chemical modifications of its chemical makeup. 24 The favorable utilization of chitosan in many practical domains can be facilitated by modifications in its molecular weight (MW), degree of deacetylation (DD), cross-linking, covalently bonded functional groups, as well as the incorporation of coordinated anion or polyanion. 25
A substantial amount of research has been dedicated to the investigation of chitosan-based nanostructures as potential therapeutic interventions for bone-related ailments. The objective of this review is to present a comprehensive analysis of the latest advancements in the field of pharmaceutical delivery systems based on chitosan, focusing on their application in the treatment and control of osteoporosis. The forthcoming prospects of chitosan based nanosystems are also emphasized, aiming to provide new opportunities for the development of optimal materials to aid in the cure of bone diseases.
Osteoporosis
Osteoporosis is a pathological condition affecting bone metabolism, which manifests as a prolonged decline in trabecular bone mass and an elevated vulnerability to bone fractures. The hormonal changes that occur after menopause, together with the natural process of aging, disrupt the equilibrium of bone homeostasis.26,27 The aforementioned diseases lead to atypical bone remodeling characterized by an elevated rate of bone turnover and reduced osteogenesis activity, which eventually results in loss of bone and degeneration of the microarchitecture. The global prevalence of osteoporosis has become a significant medical and social worry owing to the increasing age of the general population. The current approach to managing osteoporosis mostly involves the administration of pharmaceutical interventions, specifically anti-resorptive medicines and anabolic agents.28,29 Nevertheless, the utilization and effectiveness of these treatments are hindered by several factors, including the occurrence of various adverse effects and the lack of patient adherence resulting from prolonged usage. 30 The development of more efficacious rehabilitation techniques for osteoporosis is essential. Therapeutic approaches utilizing nanomaterials with pronounced osteogenic and osteo-conductive properties have emerged as highly intriguing possibilities in the realm of osteoporosis management.31,32 In the past few years, there has been growing acceptance of the potential of injectable hydrogels and nanoparticles as potential materials for the treatment of osteoporosis. Extensive research has been conducted on hydrogels and drug-loaded nanoparticles as local delivery systems in order to get accurate and long-lasting drug release. 33
Bone remodeling process
In order to attain desirable therapeutic effectiveness, it is imperative to comprehend the underlying molecular processes and genetic factors that contribute to osteoporosis, together with alterations in the milieu of the bone. The pathogenesis of osteoporosis can be primarily attributed to an imbalance in the processes of osteogenesis and osteoclastogenesis. 28 Bone is a complex tissue that experiences continuous modelling and remodeling processes facilitated by the activation of many cell types. 34 Osteoclasts are derived from hematopoietic cell precursors and play a substantial part in the process of bones resorption. Conversely, osteoblasts are derived from MSCs and contribute to bone production. 35 Osteocytes, which are osteoblasts that have completed the process of terminal differentiation, are situated within mineralized bone and provide a crucial function in governing the temporal and spatial aspects of bone remodeling. 36 Bone remodeling is a complex physiological process that consists of five distinct phases: activation, resorption, reversal, creation, and termination. The activation phase primarily encompasses the initiation of the remodeling signal through the upregulation of RANKL expression in osteocytes located at locations of bone injury. 26 This upregulation facilitates the attraction, differentiation, and fusing osteoclast precursors that ultimately results in the development of osteoclasts. Osteocytes are accountable for the conversion of mechanical strain messages into biological impulses, which subsequently trigger the process of bone remodeling. The role of parathyroid hormone is to support the maintenance of calcium homeostasis and initiate bone remodeling through its interaction with receptors located on osteoblasts. 37
During the resorption phase, the recruitment of osteoclasts occurs at the site of remodeling. The osteoblast is involved in the secretion of cytokines and structural metalloproteases, which play a pivotal role in promoting osteoclastogenesis. As a consequence, there is a progression of unmineralized osteoid breakdown and the subsequent development of spots for osteoclast attachments. 38 During the phase of reversal, the process of bone resorption assisted by osteoclasts is concluded, and the bone undergoes preparations for osteoblast-mediated bone synthesis. This preparation involves the elimination of collagen residue by the mononuclear cells. 39 During the formation stage, osteoblasts release bone matrix, also known as osteoid, which is subsequently deposited at the sites of bone resorption to counterbalance the loss of bone. 40 The primary highlight of the formation phase is the maturation and specialization of osteoblast progenitor cells and the production of chemicals that facilitate bone growth, such as type I collagen, alkaline phosphatase, lipids, and proteoglycans. 41 In the final stages, the process of remodeling ends when the resorbed bone is entirely replaced by freshly produced bone in an equivalent quantitative proportion (Figure 1). The process of mineralization of osteoid is the concluding stage in the bone production phase, wherein hydroxyapatite crystals undergo growth within matrix vesicles and then adhere to type I collagen. Osteoclasts undergo programmed cell death within a span of several weeks, whereas the apoptosis of osteoblasts occurs over the course of several months. The progressive acceleration of bone remodeling processes will result in a decline in bone density.38,42
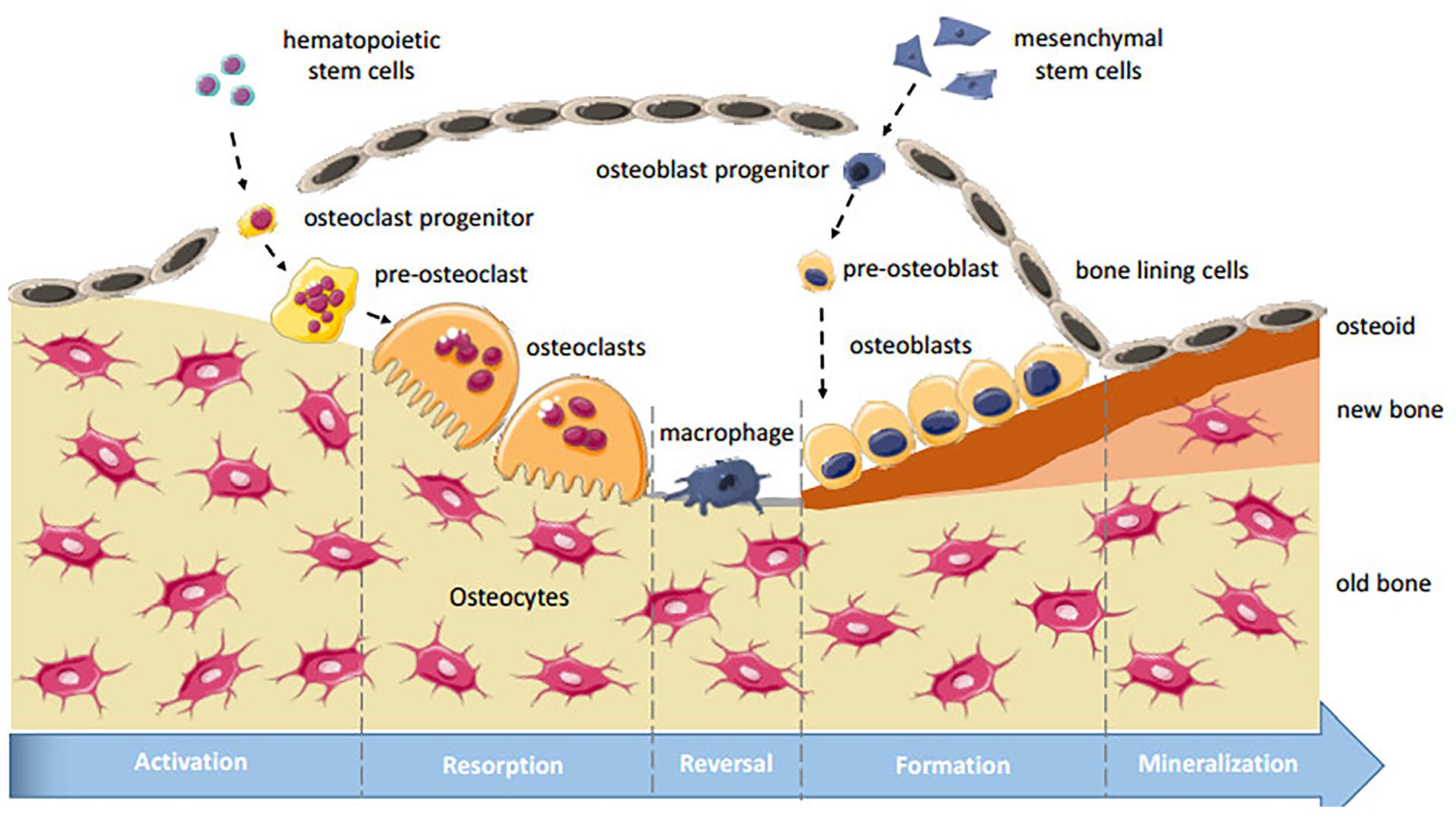
The bone reconstruction process and its most common types of cells that contribute to bone remodeling. The figure shows the five phases of bone remodeling: 1. Activation step; 2. Bone resorption period; 3. Reversal period; 4. Bone formative stage; and 5. Termination phase.
Various routes facilitate bone regeneration by regaining the equilibrium between osteoblasts and osteoclasts, so promoting the production of new bones at the sites of injury. The Wnt/β-catenin signaling system is involved in various cellular determinations throughout the entire process of bone healing. Wnt signaling, facilitated by β-catenin, contributes to the restoration of bone fractures by enhancing the formation and functioning of osteoblasts. 44 The Notch signaling pathway is a ligand-receptor signaling system that facilitates cell proliferation, differentiation, their survival, and determination of cell destiny. The activation of this pathway occurs when notch ligands engage with their receptors. This is a potential route that directly stimulates bone cells known as osteoblasts, leading to bone formation.45,46 The bone morphogenetic protein (BMP)/transforming growth factors-β (TGF-β) pathway is a significant route involved in the process of bone regeneration. Bone morphogenetic proteins (BMPs) are a member of the transforming growth factor-beta (TGF-β) class, playing a vital part in the process of osteogenesis. 47 BMPs have been shown to induce endochondral bone growth in mice. However, assessing the efficacy of BMPs in foetal and adult subjects poses challenges. BMP2 is a subject of interest within the realm of clinical research due to its demonstrated efficacy in the process of bone regeneration. The BMP2 plays a crucial role in promoting bone regeneration through its ability to stimulate MSCs and osteoblast progenitor cells, ultimately resulting in the creation of callus.48,49 Figure 2 illustrates several more pathways that play significant roles in the mechanism of bone regeneration.
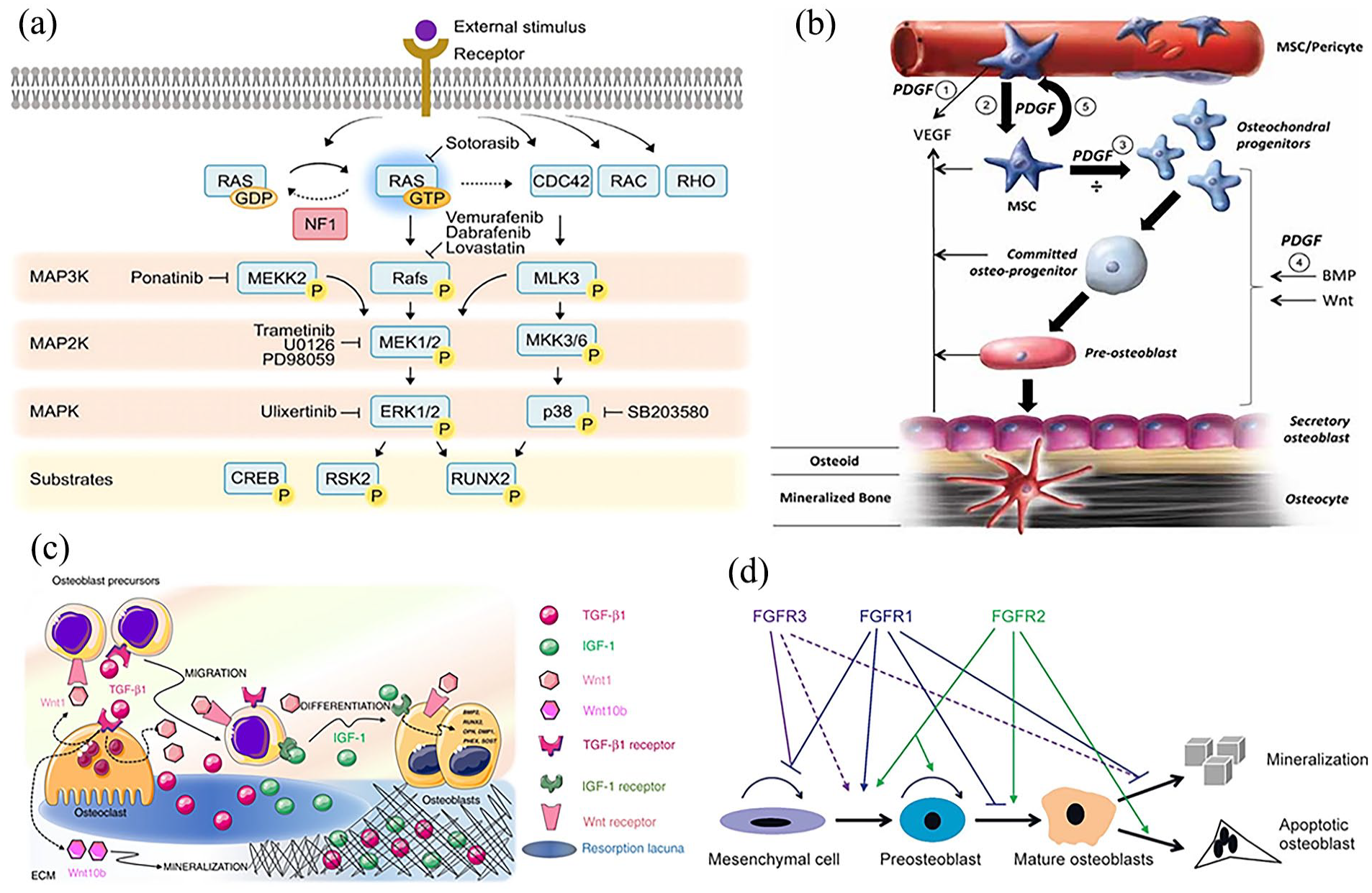
Different pathways that contribute to bone regeneration by reinstating the equilibrium between the roles of osteoblasts and osteoclasts, hence promoting the production of new bone at the site of injury: (a) mitogen-activated protein kinase (MAPK) pathway regulating cellular processes like proliferation, differentiation, migration, and cell death, which are vital for bone formation, 50 (b) platelet-derived growth factor (PDGF) an external factor regulates cellular processes in the skeletal system, stimulating cell division, development, and blood vessel formation, ultimately increasing the population of MSCs, 51 (c) insulin-like growth factor (IGF) pathway is crucial for osteoblast proliferation and differentiation, with IGF1 and IGF 2 being the only members with similar biological traits and expression within osteoblasts, and 52 (d) fibroblast growth factor (FGF) pathway is crucial for bone cell formation, maintaining proliferation and differentiation of fibroblast osteoblasts, and regulating signaling pathways in osteoprogenitor cells. 53
Role of bone cells in bone remodeling
Bone remodeling is a dynamic process that involves the coordinated activity of three primary cell types, namely the osteoclast, osteoblast, and osteocyte. These cells are essential for the maintenance of the integrity of structure and functional properties of bone tissue. Osteoclasts are the cellular entities accountable for the process of bone resorption. MSCs are distinguished from hematopoietic stem cells and exhibit a tight relationship with macrophages and monocytes. 54 The process of osteoclastogenesis encompasses the transformation of bone marrow precursors into fully-activated osteoclasts with multiple nuclei. 55 The efficacy of this procedure is contingent upon the presence of M-CSF and RANKL. The process of osteoclastogenesis is initiated by the binding of M-CSF to its receptor, known as colony-stimulating factor-1 receptor (C-FMS). 56 This connection induces an increase in the activity of the transcription factor c-FOS, which subsequently triggers the activation of RANK expression. The activation of the receptor activator of nuclear factor kappa-B (RANK) by its ligand, RANKL, initiates a cascade of events that leads to the synthesis and release of several proteins and enzymes that facilitate the process of osteoclastogenesis. 57 The activation of the IκB kinase (IKK) complex is initiated by the process of phosphorylation of κB-inducing kinase (NIK). The IKK complex facilitates the activation of NF-κB by the process of ubiquitination and subsequent destruction of its suppressor molecule, IκB. The transcription factor NF-κB has the ability to initiate the activation of NFATc1, which subsequently undergoes a process of self-amplification to activate genes associated with osteoclastogenesis.58,59
Osteoclasts, which exhibit a morphology resembling that of jellyfish, adhere to bone surfaces and establish a sealing zone. The resorptive activity is subject to the impact of cathepsin K. Osteoblasts, which are accountable for the process of bone production, are derived from MSCs. 60 The Wnt/β-catenin pathway is of significant importance in the regulation of osteoblastic development. Wnt inhibitors like as Dkk-1 and sclerostin exert a negative regulatory effect on this pathway. 61 Three critical transcription factors implicated in the process of osteoblast development include Runx2, osterix, and β-catenin. 62 Mature osteoblasts are responsible for the secretion of an organic matrix, which subsequently undergoes mineralization to facilitate the formation of bone tissue. The differentiation process of osteoblasts plays a crucial role in determining the rate and efficiency of bone production. 63 Osteocytes, comprising more than 90% of cellular components in bone, are specialized osteoblasts that play a crucial role in the process of bone remodeling. These cells exhibit resemblances to neural cells and facilitate the transmission of mechanical loading signals within the bone. 64 Osteocytes demonstrate the expression of factors implicated in phosphate metabolism, hence indicating the possibility of matrix mineralization. Cytokines such as Dkk1, sclerostin, RANKL, and OPG are released by these cells, and they play a critical part in retaining bone equilibrium by impeding the differentiation of osteoblasts and the creation of bone.65,66
Nanomaterials in treatment of osteoporosis
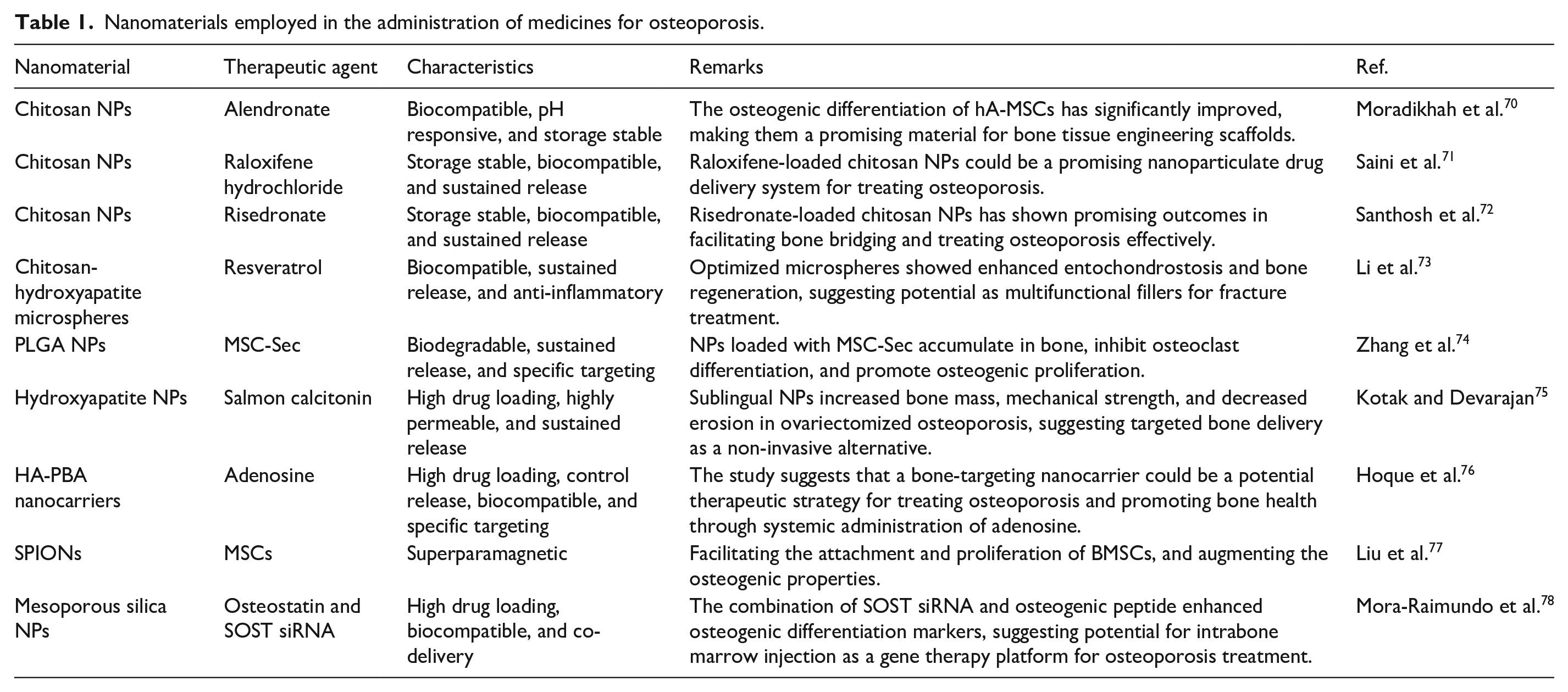
The utilization of nanomaterials in the field of bone regeneration has shown a substantial increase in recent times. The term “nanomaterial” refers to a substance that possesses a dimension inside the nanometer scale, often ranging from approximately 1 to 100 nm. The characteristics of nanomaterials undergo significant alterations as a consequence of the higher surface-to-volume ratio. 67 Nanoparticles have demonstrated significant potential as precise and effective vehicles for medication delivery. Nanomaterials have the potential to be utilized in the development of drug delivery systems that specifically target bones, enabling the loading of therapeutic compounds such as medicines and vesicles. 12 Furthermore, it is possible to alter them in order to exhibit intrinsic therapeutic properties. Pharmacological intervention is a prevalent therapeutic approach for the management of osteoporosis. For an extended period, this therapeutic approach has encountered challenges related to the distribution of medication to unintended targets and the occurrence of unfavorable consequences. In order to tackle these concerns, scientists have been attempting to fabricate nanoparticles possessing the capacity for precise targeting, thereby serving as efficacious medication delivery systems. However, the issue of off-target delivery continues to be a significant constraint that hinders the broader implementation. 68 There exist two approaches for achieving bone-targeting functionality: a strong affinity for hydroxyapatite, exemplified by bisphosphonates and oligopeptides, or a selectivity towards cells associated with bone. The distinctive P–C–P bond found in bisphosphonates, combined with the carboxyl group present in oligopeptides, imparts these molecules with a high selectivity for the apatite found in the tissues of bone. 69 Table 1 presents an overview of the application of nanoparticles in the therapeutic management of osteoporosis.
Nanomaterials employed in the administration of medicines for osteoporosis.
In contrast to traditional pharmacological interventions, gene therapy via RNA interference (RNAi) is an innovative strategy in the management of systemic osteoporosis. RNAi is an intrinsic biological mechanism that selectively destroys certain messenger RNA (mRNA) molecules, resulting in the subsequent reduction of the related proteins. The application of RNAi to suppress genes associated with osteoporosis is a potentially effective approach for the systemic treatment of this condition. RNAi treatments, albeit promising, face limitations such as shorter half-lives, rapid degradation rates, and inadequate penetration capabilities. The use of nanoparticles has been employed as a strategy to tackle these challenges, primarily focusing on safeguarding and transporting nucleic acid molecules.79,80 The field of nanomedicine has seen significant interest in the utilization of nanoparticles of mesoporous silica (MSNs). The great loading capacity of MSNs can be attributed to their mesoporous structure, which provides a huge surface area, as well as their good biocompatible and biodegradable properties. A novel delivery strategy based on MSN has been recently created for the efficient delivery of small interfering RNAs (siRNAs) specifically intended for targeting the SOST gene. The gene in question is responsible for the production of sclerostin, a protein that functions as a suppressor of osteogenesis. The use of a polyethylenimine coating to the MSN-based system of administration enhances its potential to simultaneously distribute SOST siRNA and osteostatin, an osteogenic peptide. The introduction of the MSN nanomaterials into the tissue of bones resulted in an increase in the expression of genes associated with osteogenesis, leading to enhancements in bone structure and alleviation of osteoporosis. 78
Several types of nanoparticles have inherent therapeutic properties, including the ability to suppress the formation of osteoclasts or enhance the development of fresh bone cells, known as osteogenesis. The therapeutic efficacy of inorganic nanoparticles in promoting bone regeneration in individuals with osteoporosis has been proven. The core-shell structure of HA-Iron oxide nanoparticles has been shown to possess dual capabilities in facilitating osteogenesis and suppressing osteoclastogenesis. 81 Nitric oxide (NO) plays a significant role in the process of bone regeneration. It has a substantial role in facilitating osteogenesis and mitigating the effects of osteoporosis. Nevertheless, the utilization of NO poses certain difficulties due to its limited duration of activity and extensive range of diffusion, spanning from 40 to 200 micrometers. Extensive research has been conducted on the subject of light-controllable release of NO. 82 In addition to their role as carriers for delivering pharmaceuticals and regulators with intrinsic anti-osteoporosis characteristics, tiny particles also serve as boosters in scaffolds for osteoporotic bone tissue engineering or injectable form of hydrogels for drug loading. This is achieved through a synergistic mechanism that promotes osteogenesis by enhancing physiochemical characteristics of these substances. 70
The utilization of nanoparticles in the context of osteoporosis presents several potential benefits, including convenient application, adaptability, rapidity, efficacy, adjustable multifunctionality, and limited invasiveness. To begin with, nanoparticles possess the inherent capability to undergo facile modifications or seamless integration with biological components, including growth factors. The aforementioned phenomenon can be ascribed to the inherent chemical features of the substances, which facilitate the adherence of cells and the development of stem cells. Moreover, the incorporation of inorganic nanomaterials as fillers has been shown to effectively manipulate and augment the mechanical characteristics of tissue engineering materials, including hydrogels and scaffolds. Nanostructures are highly suitable for the purpose of precisely administering anti-osteoporotic medicines and nucleic acids, hence promoting the repair of bone tissue affected by osteoporosis. 83 Notwithstanding these benefits, it is imperative to acknowledge the challenges pertaining to the implementation of nanomaterials in real contexts. To begin with, it is important to note that nanoparticles exhibit cytotoxic properties. 84 Therefore, it is necessary to conduct further research for the purpose to gain a better comprehension of the biological safety of nanoparticles in the context of osteoporotic bone regeneration, with a specific focus on their suitability for long-term therapeutic applications. Recently, polymers have garnered significant acceptance among scholars owing to their exceptional attributes such as biocompatibility, bifunctionality, and biodegradability. The aforementioned characteristics render it well-suited for various medical applications, such as biomedicine, pharmaceuticals, the engineering of tissues, and delivery systems for drugs.
Polymers in osteoporosis management
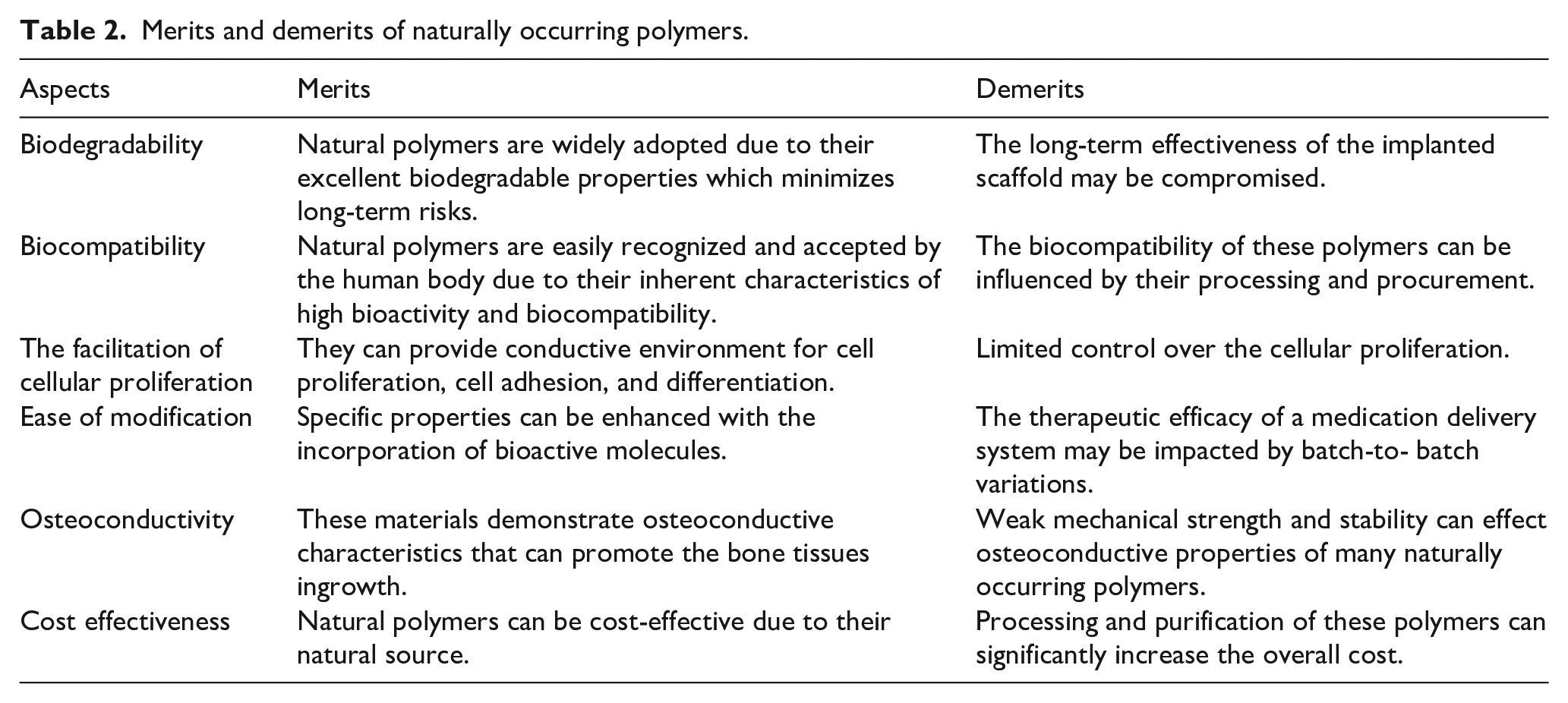
There has been a noticeable surge in interest surrounding the application of naturally occurring polymers in many medical and biological contexts in contemporary times. These applications encompass systems for delivering drugs, engineering tissues, wound treatment, among others. 85 Naturally occurring polymers can exist in two forms: protein-based or polysaccharide-based. In contrast to polysaccharides, proteins exhibit amino acid sequences which are commonly linked to cellular attachment through integrin-binding domains. In contrast, the augmentation of cell adhesion and osteoconductivity inside polysaccharide scaffolds necessitates the implementation of chemical surface changes, the amalgamation with osteoconductive substances, or the integration of integrin-binding sequences or cell adhesion proteins. Polymeric materials of natural origin that are frequently investigated for biomedical applications encompass chitosan, collagen, alginate, and peptides. 86 Among the various types of naturally occurring polymers, polysaccharides are particularly prevalent due to their intricate architectures and inherent propensity to form supramolecular network. 33 Some of the merits and de-merits of natural polymers are presented in Table 2. The presence of chemically active groups, such as hydroxyl, carboxylic acids, and amines, within the molecular composition of polysaccharides can confer several advantageous properties in vivo. These include an extended half-life within the organism, the ability to facilitate cellular proliferation, and the capacity to form hydrogels with minimal elicitation of inflammatory or immunological reactions. 87 This review aims to explore the potential of chitosan as a material for the management of osteoporosis.
Merits and demerits of naturally occurring polymers.
Chitosan
Chitosan, a type of carbohydrate bio polymer, has garnered significant attention from researchers worldwide, recognized as a well-researched natural biopolymer with significant potential. Chitosan is a polysaccharide derived from chitin, which is a biopolymer occurring naturally in shells and fungus, and is obtained through the process of deacetylation. 88 The significant level of interest in chitosan stems from its exceptional attributes of high biocompatibility, biodegradability, biological activity, and low toxicity. Figure 3 encompasses a variety of distinct physical, biological, and chemical properties of chitosan. The potential applications of this substance in many biomedical contexts includes its utilization as a vehicle for drug delivery, incorporation into wound care products, and integration with implant materials. Chitosan is recognized as a substance that facilitates the regenerative processes of both hard and soft connective tissues. 89 It has demonstrated independent and synergistic antibacterial properties, establishing its status as an antibacterial biopolymer. Due to its inherent characteristics and facile nanoparticle formation, the material in question can be utilized for the fabrication of diverse structures such as microfibers, beads, membranes, gel-like substances, sponge-like architectures, and scaffolds. The utilization of this material as the primary scaffold component appears to be very intriguing. 90 Extensive study has been conducted on the application of chitosan based scaffolds in the field of tissue engineering for bones. The primary objective of a scaffold is to facilitate the establishment and advancement of tissue ingrowth by offering a supportive framework and conducive surroundings. Ultimately, the scaffold is intended to degrade while simultaneously facilitating the substitution of new healthy tissues. 91
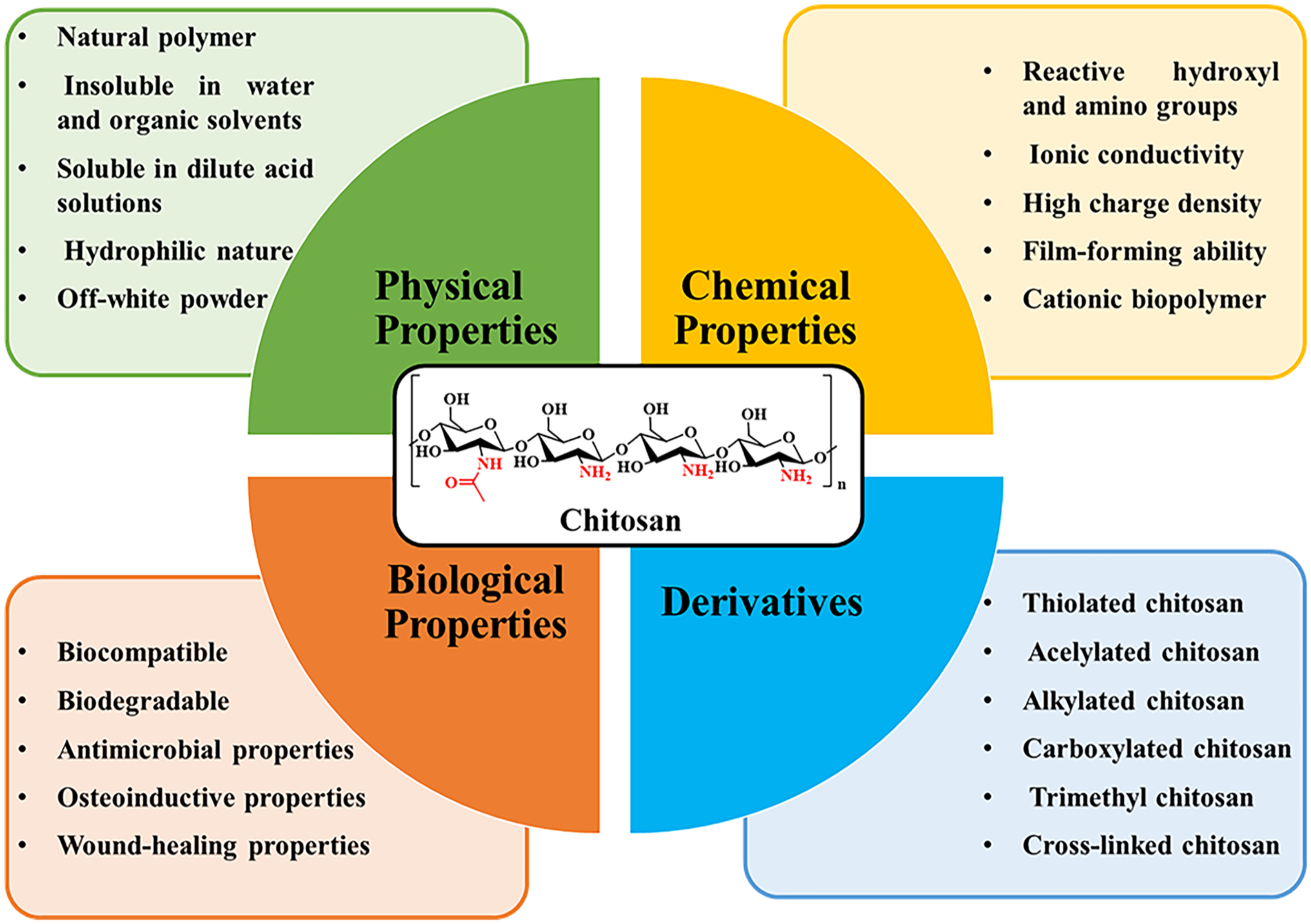
Different properties and derivatives of chitosan.
This biopolymer exhibits notable appeal as a bone scaffold due to its ability to facilitate the generation of hydroxyapatite, and it also promotes the adhesion and development of osteoblast cells. Numerous scaffolds have undergone extensive testing, employing a diverse range of fillers in their fabrication. Notable examples include carbon nanotubes, titanium oxide, polypyrrole, and zirconium nanomaterials.92,93 Without a doubt, calcium phosphates, particularly hydroxyapatite, are the most often employed fillers in chitosan composites for the purpose of bone regeneration. The extensive utilization of this substance can be attributed to the reason that hydroxyapatite contains the primary constituent of human skeletal structure. 94 As a result of the growing demand for biomaterials, scaffolds must possess the ability to release pharmaceutical agents along with their fundamental characteristics. Bisphosphonates are the pharmacological class of medications most frequently employed in the treatment of osteoporosis, a prevalent skeletal disorder. 95 Drug release of various categories of pharmaceuticals from chitosan scaffolds has the potential to facilitate accelerated recovery of patients following surgical procedures. Nevertheless, it is imperative that the administration of the medicine be limited to modest dosages owing to its pronounced toxicity at higher dosages. 96 Furthermore, the physical and chemical characteristics of chitosan are influenced by several parameters, such as its degree of deacetylation and its molecular weight. The structural flexibility of chitosan is a significant characteristic attributed to the existence of active hydroxyl and amino groups within its molecular chain. The flexibility for chemical alteration of each repeating unit is attributed to the existence of two hydroxyl groups at C-3 and C-6 positions and an amine group at the C-2 position. 24
The properties and effects of chitosan in living systems can be altered through appropriate physical and chemical modifications in its chemical composition. The favorable utilization of chitosan can be achieved by making modifications to its molecular weight, degree of deacetylation, cross-linking, covalently bonded active groups and moieties, as well as including coordinated anions or polyanions. These modifications can be applied in many practical domains where chitosan is implemented. By incorporating minor chemical groups into the structure of chitosan, such as alkyl groups like hydroxyl-propyl or carboxy-methyl groups, the solubility of chitosan can be significantly enhanced. 97 The cationic feature can be reverted by sulfonation, resulting in the introduction of an anionic characteristic. This modification leads to improved water solubility, enhanced paste flow, a higher water reducing ratio, and anticoagulation properties. The mechanical durability of the particles is enhanced by increasing the crosslinking of CS, for example, through the use of anions, dextran, formaldehyde, and glutaraldehyde. Highly cross-linked particles exhibit reduced swelling, decreased infiltration of water into their interior, and limited diffusion of drugs from their exterior. Crosslinking has the ability to decelerate the release of drugs and hinder sudden release. 98 The mucoadhesive characteristics can be enhanced by trimethylating the main amino groups of chitosan and by PEGylation or attachment of SH groups that interact with the cysteine rich area of the mucous glycoprotein. Enhancement of mucoadhesive properties can also be achieved through the creation of complexes including polyvalent anionic medicines, excipients, and polyvalent inorganic anions (Table 3). 99
Typical modification of chitosan through different chemical procedures.
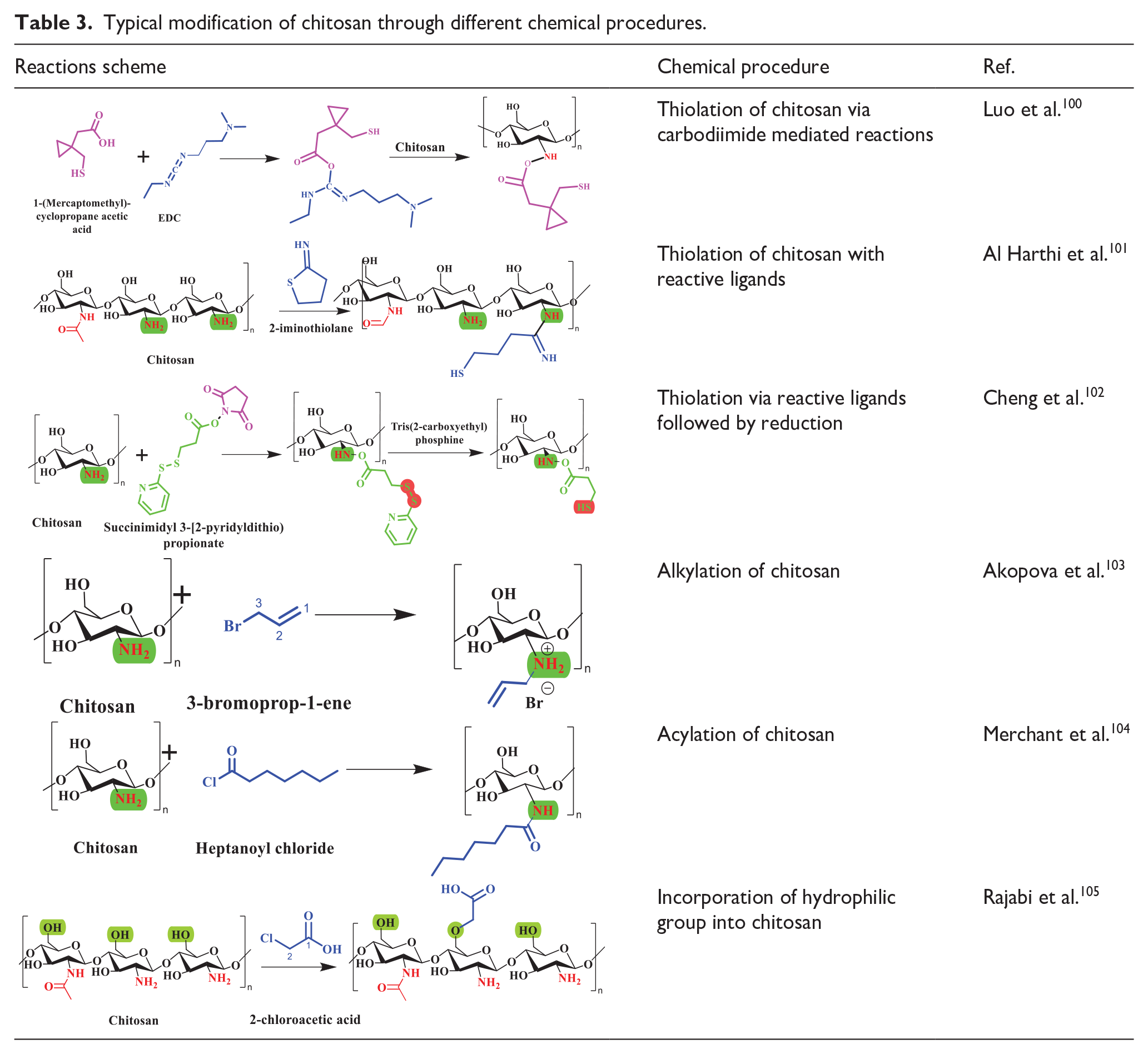
Chitosan may be effectively cross-linked using glutaraldehyde, which significantly improves its ability to rapidly create a hydrogel without the need for any potentially dangerous chemicals. The gelation characteristics of chitosan can be enhanced through thiolation. Thiolation significantly amplifies the permeability effect, sometimes more than 30 times. Trimethyl chitosan (TMC) is soluble across all pH levels and has been observed to decrease trans-epithelial electric resistivity by 25%–85%. TMC is an effective permeation enhancer due to its ability to disrupt tight junctions. Additionally, it enhances the paracellular transportation of the medications. 106 Despite having numerous functional qualities, chitosan is limited by its significant hydrophilicity, limited ductility, considerable swelling, and thermal instability. An important constraint to its usage is its low solubility. Chitosan is not soluble at the normal pH level found in the body (pH 7.4) and does not effectively improve absorption, which hinders its use in biomedical applications. Enhancing the solubility of chitosan is a vital aspect for safe utilization across many applications. 98 In the past few years, there has been a growing interest in the chemical derivatives of chitosan due to their enhanced capabilities in comparison to the unmodified form. These derivatives offer benefits in terms of the ability to dissolve, gel-forming properties, ability to design water-resistant derivatives with amphiphilic character. Over the years, many types of chitosan derivatives products have been created to meet the specific needs of diverse biological applications. 107
Synthesis of chitosan scaffolds
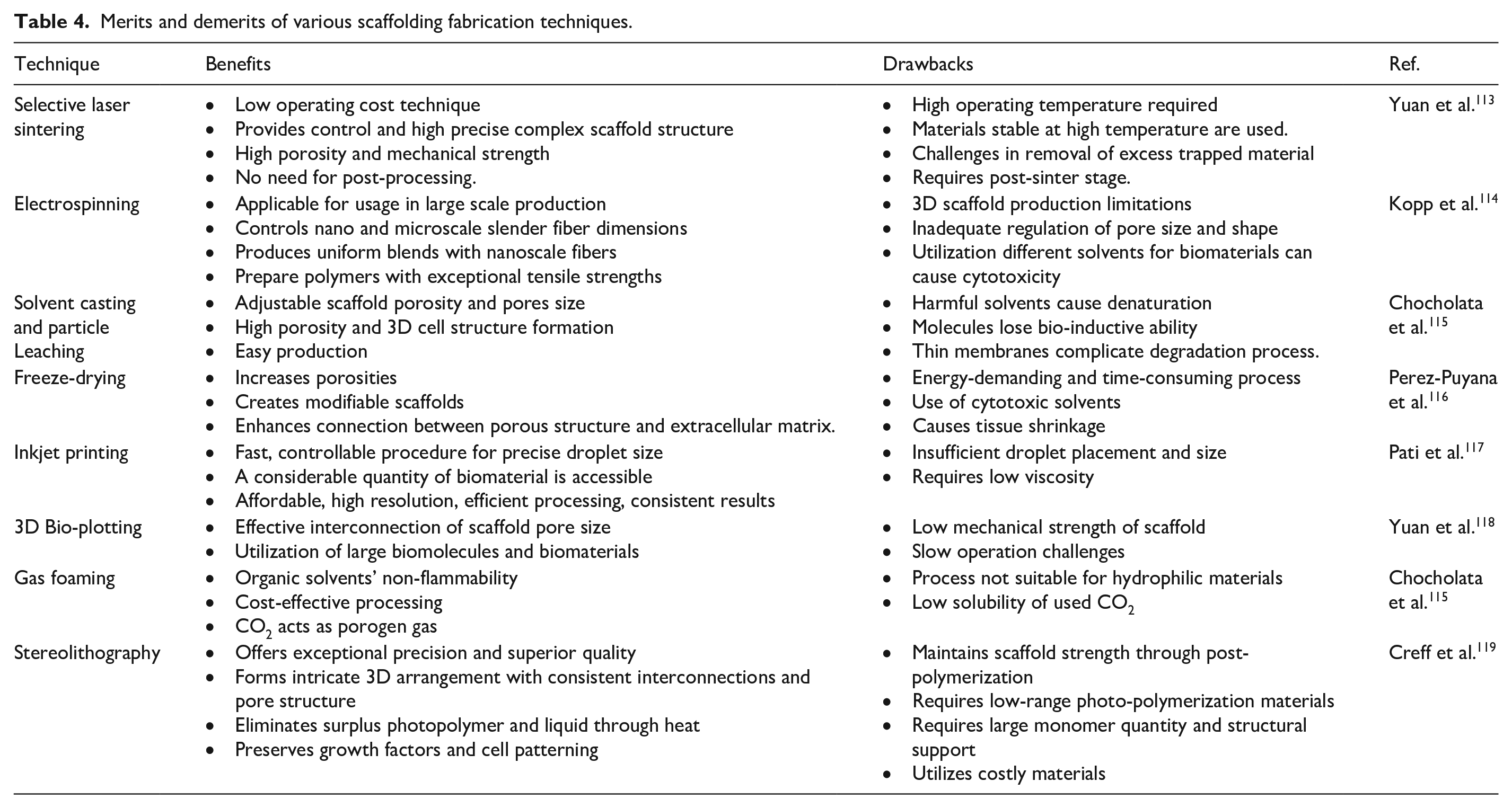
Chitosan scaffolds used in the management of bone diseases are designed to replicate the complex and ever-changing bone matrix found in living organisms. Hence, the selection of the construction procedure significantly influences the physiochemical and biological aspects of the scaffolds. 108 Chitosan scaffolds are fabricated employing a diverse range of processes. Several methods can be employed for the purpose of achieving certain outcomes. These methods include selective laser sintering, electrospinning, solvent casting, and particle leaching, freeze-drying, inkjet printing, 3D Bio-plotting, gas foaming, stereolithography, etc. (Figure 4).109,110 Table 4 details the specific benefits and drawbacks of each technique. Lyophilization is the prevailing methodology employed for the production of chitosan scaffolds among various other methods. When the chitosan solution is subjected to freezing, ice crystals are formed through the process of phase separation. Upon sublimation, the crystals vacate the original volume they occupied, resulting in the creation of pores. Achieving precise control over the temperature is imperative for the successful implementation of this technique. 111 One of the drawbacks associated with this phenomenon is the potential collapse of the pore’s structures in the absence of temperature regulation. The growth of bigger pores is a potential outcome, and the selection of solvents is constrained. Solvent-exchange/phase-separation, a technique reliant on the gelation of a solution through the introduction of an alkaline solution below its gelation threshold, can be employed. By employing these methodologies, it is possible to fabricate 3D scaffolds with diverse geometries. However, a significant drawback of these approaches is the inadequate mechanical robustness. The salt leaching technique entails the integration of porogens, such as NaCl, of specific dimensions into a chitosan matrix. Subsequently, the salt particles are removed through a series of water leaching processes, leading to the creation of porous CS scaffolds. 112
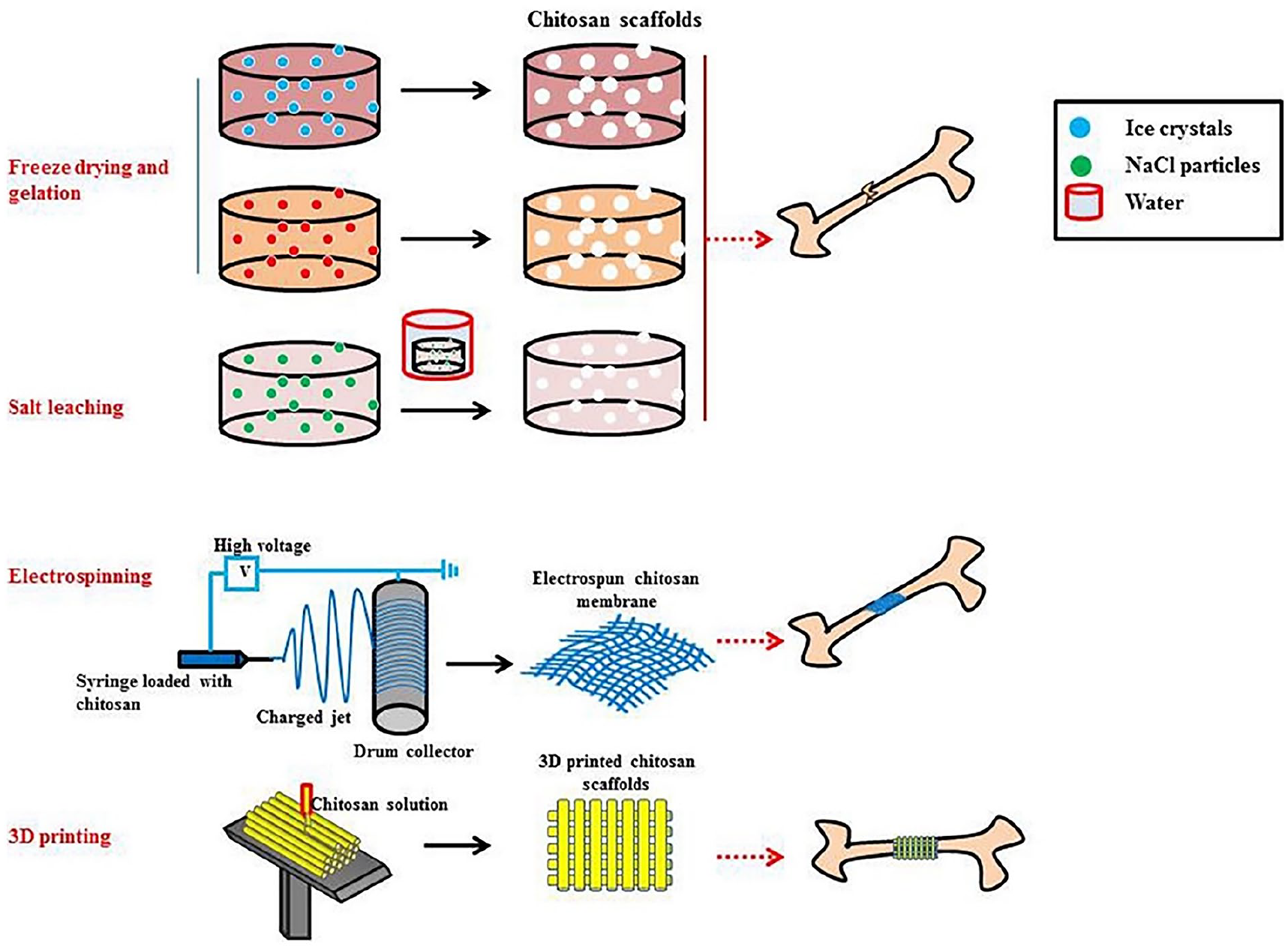
This diagram illustrates the prevalent fabrication techniques employed in the production of chitosan-based scaffolds, including the use of freeze-drying, freeze gelation, salt leaching, electrospinning, and 3-D printing.
Merits and demerits of various scaffolding fabrication techniques.
The electrospinning technique involves the conversion of a polymer solution into nanofibrous mats through the application of a strong electric force. The process of electrospinning operates based on a fundamental principle whereby the application of an electric charge surpasses the surface tension of the polymer solution, leading to the accumulation of a charged polymer jet on a metallic collector. The system consists of three primary components: a high direct current (DC) voltage source, a spinneret, and grounded collectors. The utilization of this adaptable methodology facilitates the generation of fibers at both the nano and micro scales.120,121 The MSN/CS nanofibrous membranes scaffolds were manufactured by Li et al. 122 using the electrospinning approach. The findings of their study demonstrated an increase in tensile strength, water retention capabilities, and promotion of osteoblast growth. Additionally, it was revealed that the utilization of electrospun MSN/CS tiny fibers as scaffolds is more advantageous compared to using solely electrospun CS nanofibers. This preference is attributed to the superior microenvironment offered by the former, which promotes cellular growth and emulates the extracellular matrix (ECM). Biomaterials present significant benefits in comparison to typical metallic implants with regards to cellular processes such as adhesion of cells, propagating, differentiation, proliferation, and others. The efficacy of scaffolds is contingent upon the physicochemical and biological characteristics of the constituent components. The scaffolds must possess a range of qualities that are appropriate for use in bone tissue engineering applications. The key factors to consider in this context are as enlisted in Figure 5. The properties and applications of scaffolds in bone tissue engineering (BTE) can be enhanced by adjusting these parameters through the incorporation of other polymers and ceramics into chitosan.
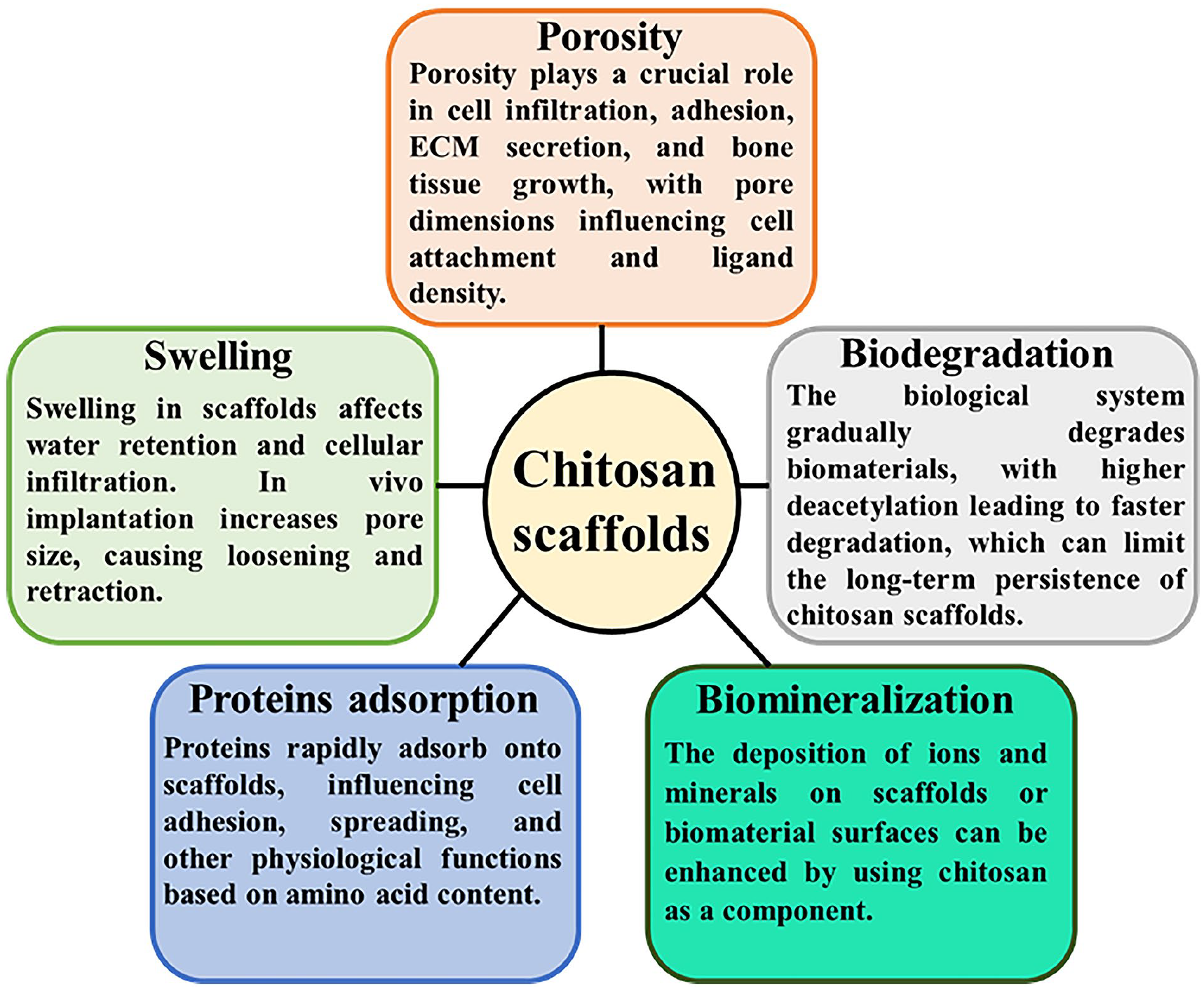
Chitosan scaffolds properties including porosity, water retention ability, protein adsorption, biomineralization, and biodegradation.
Mechanism underlying the release of drugs from chitosan-based scaffolds
The release of pharmaceuticals from CS-based scaffolds can be achieved through four separate mechanisms, namely diffusion, erosion, swelling, and stimulus-embedded systems (Figure 6). Diffusion-controlled release systems regulate drug release through solvent penetration or diffusion. They can be matrix or reservoir, with the drug enclosed within a polymeric membrane. 123 The drug core can be liquid or solid, and the membrane can be microporous or nonporous. A saturated drug core ensures consistent drug molecule movement through the membrane, as the force driving movement remains constant. High water solubility can affect the release of a drug due to the challenge of maintaining a saturated state. Matrix systems dissolve or disseminate drug within a polymer device, with the rate of release decreasing over time due to concentration gradients. 124 The release profile of diffusion-controlled systems is significantly influenced by the properties of the drug, such as such as physical size, loading level, diffusivity in the matrix phase, and solubility in the matrix. The drug release profile is also influenced by various parameters, including the geometry of the scaffold, which impacts the path length and accessible surface area for diffusion, as well as the water content and composition of the scaffold. 125 Samaneh and Saeed reported a chitosan-based scaffold for bone repair. 126 The scaffolds were successful in loading celecoxib, a model drug, due to their large surface area. The drug showed a biphasic release pattern, with a small initial burst followed by a persistent release lasting up to 14 days. However, an increase of hydroxyapatite content in the scaffold composition reduced the drug release percentage.
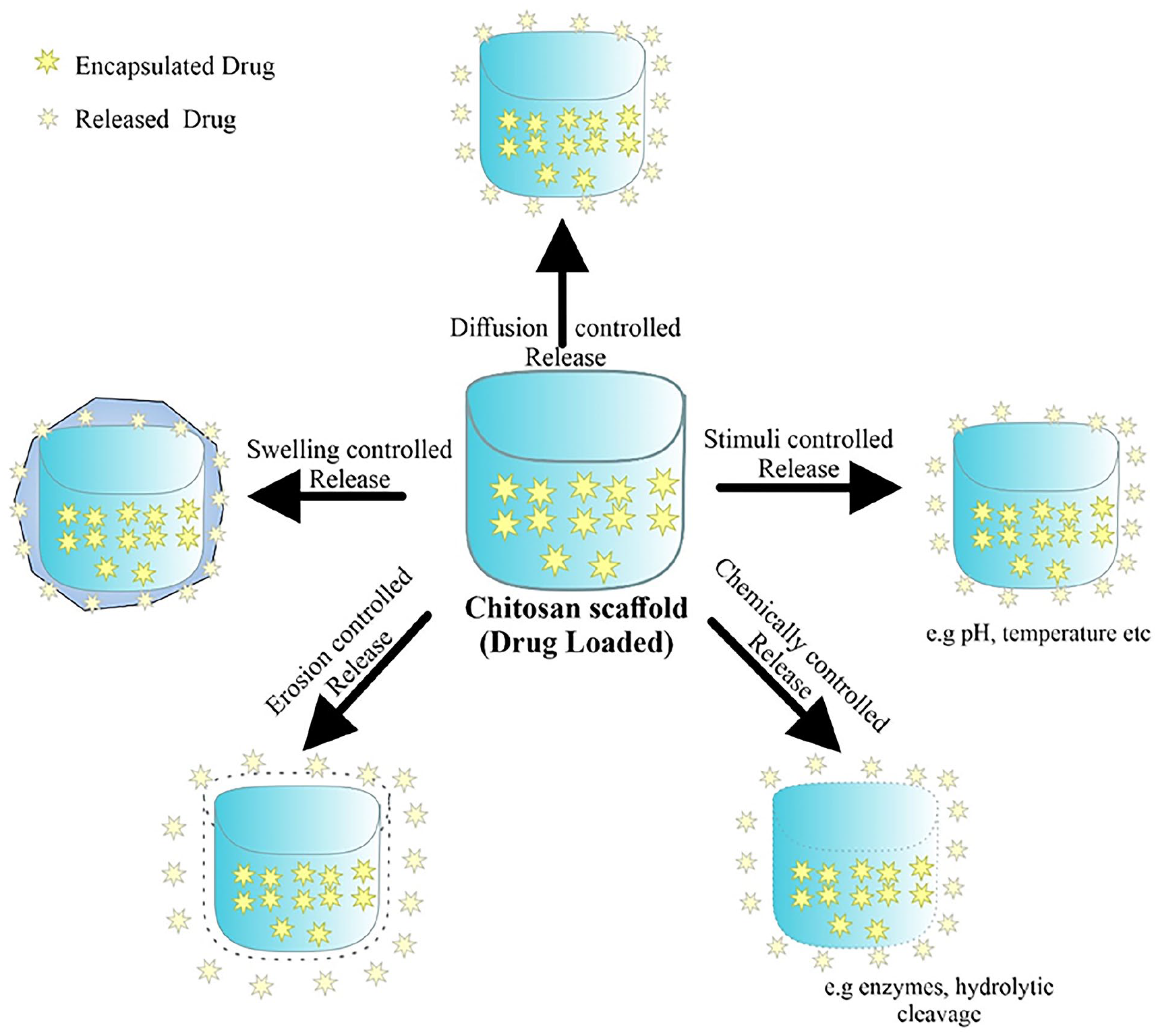
The mechanism underlying the controlled release of medicines from chitosan scaffolds. Chitosan exhibits promising potential as a drug/gene delivery system owing to its highly regarded biological, physical, and chemical characteristics. The release of pharmaceuticals from scaffold based on controlled substances primarily occurs through many processes.
Swelling-controlled systems utilize physical principles such as osmosis and swelling to control the release of bioactive components. The process begins with water-induced swelling of the polymeric structure, where the penetrating solvent interacts with the drug and polymer matrix, compensating for interactions between the drug and polymer matrix.126,127 Most swelling-controlled systems are hydrogels, where the drug is contained in its dry state. When water is present, the hydrogel undergoes swelling, reducing resistance to drug solute diffusion. The release rate is primarily determined by the polymer’s swelling rate, which is influenced by crosslink density and hydrophilicity. Protein delivery can be slowed down by the ionic contact and chain entanglement that occur between the protein and the matrix. 128 Gull et al., 129 successfully synthesized a hydrogel system with multiple response properties by employing chitosan and polyvinyl pyrrolidone. The manipulation of the concentration of tetraethyl orthosilicate, a cleavable cross-linker, allowed for the regulation of lidocaine release. This control was attributed to the delivery mechanism that is governed by swelling. 129
The erosion process of drug-loaded scaffolds is characterized by the depletion of material resulting from the detachment of monomer and oligomers from the polymer matrix. Erosion-based delivery is governed by chemical reactions that take place within the polymer matrix. Possible reactions encompass (1) the enzymatic or hydrolytic destruction of polymer chains attached to the main structure, (2) either irreversible or reversible reactions between a drug and the polymer that can be released, (3) erosion of the surface, and (4) decomposition of the entire bulk material. 130 Surface erosion can occur when the rate of water entering the polymer is significantly slower than the rate of bond hydrolysis. However, surface erosion exclusively takes place in enzymatic degrading systems when the rate of enzyme transport into the gel is slower than the rate of enzymatic degradation, due to the naturally high water content of hydrogels. In erosion, the rate-determining step is the chemical processes, which govern the overall rate of drug release. Gentile et al. 131 developed a chitosan-based scaffold with localized controlled-release properties to promote bone repair. The release of simvastatin showed an initial lag phase, followed by a rapid release phase with approximately 40% release, and then a linear increase over 10–28 days. The release of drug from scaffold follows a sigmoidal pattern, which corresponds to the degradation time-profile of the polymer. This indicates that the release of drug is controlled by the degradation of the polymer.
Stimulus-responsive long-term drug release techniques are sophisticated environment sensitive processes capable of responding to various biological stimuli, including pH, ATP, redox, enzymes and glucose as well as physical stimuli including light, temperature, magnetic forces, ultrasounds, and electricity. 132 The researchers Zhao et al. 133 successfully fabricated a scaffold that exhibits pH-responsive behavior, undergoing self-assembly at a pH of 7.5. The synthesis of the hybrid hydrogel system involved the combination of carboxymethyl with amorphous calcium phosphate, resulting in the formation of CMC-ACP hydrogels. The CMC-ACP hydrogels exhibited a notable enhancement in the effectiveness and advancement of BMP9-induced osteogenesis in vivo, while simultaneously inhibiting bone resorption during an extended period of ectopic presence.
Drug delivery systems are utilized to transport a diverse array of pharmaceutical substances to specific areas of absorption, hence enhancing the pharmacological efficacy of the medications. In addition, they effectively address challenges such as hydrophobicity, limited solubility, inadequate bioavailability, drug accumulation, and absence of selectivity. 134 The positively charged characteristics of chitosan enable various applications such as regulated drug release, muco-adhesion, on-site gelation, transfection, permeation improvement, and inhibition of efflux pumps. The aforementioned qualities collectively establish chitosan based scaffolds as a promising candidate for the targeted and regulated delivery of therapeutic chemicals to the site of a defect. The drug release profile can be modified by various factors, including the loading strategy, the matrix’s degradation capacity, and the precise amount of the pharmaceutical. 135 The local delivery of drugs through scaffolds has several advantages over systemic delivery, including a reduction in adverse effects and the possibility of overdose. Moreover, this methodology facilitates the enhanced and prolonged delivery of the pharmaceutical agent to the intended site, hence ensuring optimal concentration levels at the desired spot during the entirety of the healing process. Several methodologies have been devised to provide certain pharmaceuticals and bioactive compounds via chitosan-based scaffolds, with the aim of augmenting the process of bone repair. 136
Osteoporosis treatment using chitosan-based scaffolds
Osteoporosis is a skeletal condition characterized by an elevated susceptibility to severe fractures of the spine, hips, and wrists, resulting in substantial morbidity and mortality. The osteoporosis illness encompasses various factors, such as the process of aging, inadequate calcium intake, diminished testosterone levels, and genetic susceptibility. 72 The prevalence of osteoporosis condition is on the rise within the senior population. The careful consideration and utilization of suitable methodologies at distinct phases of the osteoporosis condition are imperative for the efficient therapy and administration of the ailment. In order to address this illness, various conventional therapies have been utilized, including the use of anabolic drugs such as parathormone and its counterparts, as well as antiresorptive therapy. 137 While the objectives of these therapies align in terms of reducing bone fractures and maintaining bone integrity, it is important to note that they all face significant challenges related to bioavailability. Drug delivery systems present a promising avenue for achieving enhanced and optimal therapeutic outcomes through the utilization of convenient formulations that offer improved oral bioavailability or enhanced patient compliance.
Oral methods of drug delivery have limited bioavailability and increased instability, hence significantly reducing the effectiveness of formulations that aim to adhere to cells or undergo oral absorption. However, it is imperative for an oral medication formulation to successfully traverse digestive barriers and attach to the brush boundary of the intestinal mucosa. Subsequently, translocated particles might be absorbed by intestinal cells through intracellular absorption, paracellular uptake mechanism, or phagocytic uptake facilitated by intestinal macrophages. Chitosan-based nanoparticles exhibit considerable potential as oral drug delivery systems within the realm of polymeric nanomaterials. This is primarily attributed to their capacity to induce the opening of closed junctions among cells in the intestinal epithelium, hence facilitating the paracellular transfer of medicines. 138 Risedronate sodium, a bisphosphonate molecule containing nitrogen, is widely regarded as an effective pharmaceutical agent for enhancing bone strength and impeding the progression of osteoporosis. Regrettably, the bioavailability of the substance is documented to be below 1% upon oral ingestion. Numerous formulations have been proposed as a means of addressing this constraint.139,140 Chitosan possesses an inherent positive charge, while risedronate exhibits a negative charge. Therefore, the formation of chitosan nanoparticles can be achieved by the ionic gelation technique, employing tripolyphosphate (TPP) as a cross-linker for the purpose of encapsulating the pharmacological agent within the chitosan nanoparticles. According to molecular modeling investigations and in vivo research, it has been observed that nanoparticles composed of risedronate-chitosan has the ability to selectively target and inhibit human farnesyl diphosphate synthase, a key enzyme in the mevalonate pathway. The formulation’s potential impact involves the inhibition of osteoclastogenesis and the subsequent prevention of bone resorption, hence promoting bone regeneration and augmenting bone density (Figure 7). 72
Various modified forms of chitosan are used for bone repair.
In order to enhance the pharmaceutical formulation of alendronate sodium, a bisphosphonate prescription, it was encapsulated in chitosan. This approach demonstrated the required therapeutic effect and achieved prolonged release of the drug. These features offer a means to mitigate the negative consequences of alendronate sodium following its oral intake. 142 Wu et al. 143 constructed a microsphere delivery system based on chitosan for controlled alendronate release and bone tissue development. The results indicated that chitosan-based scaffolds had better degradation properties, mechanical strength, controlled release, and improved osteogenic differentiation of adipose-derived stem cells by increasing calcium deposition and alkaline phosphate activity. The in vivo trial demonstrated greater efficacy of the scaffolds than autografts in fully restoring major bone defects within a period of 8 weeks, indicating their potential for localized delivery and bone regeneration (Figure 8). Raloxifene hydrochloride exhibits a limited oral bioavailability of 2%. This substance functions as an agonist for estrogen in bone tissue, effectively preventing the degeneration of vertebral bone by suppressing the function of cytokines that promote bone resorption. The bioavailability of raloxifene could be increased by encapsulating it in chitosan nanostructures. The in-vitro release investigation demonstrated an immediate and rapid release of the medication, which was subsequently followed by a gradual and prolonged release. An animal investigation shown that the plasma concentration of the medication was greater following intranasal delivery of nanoparticles compared to oral administration. The observed phenomenon can be attributed to the absorption of raloxifene hydrochloride through the nasal mucosa, facilitated by the mucoadhesive nature of chitosan nanoparticles. Hence, the administration of raloxifene chitosan NPs through the nasal route has great potential as a method for treating osteoporosis.71,144
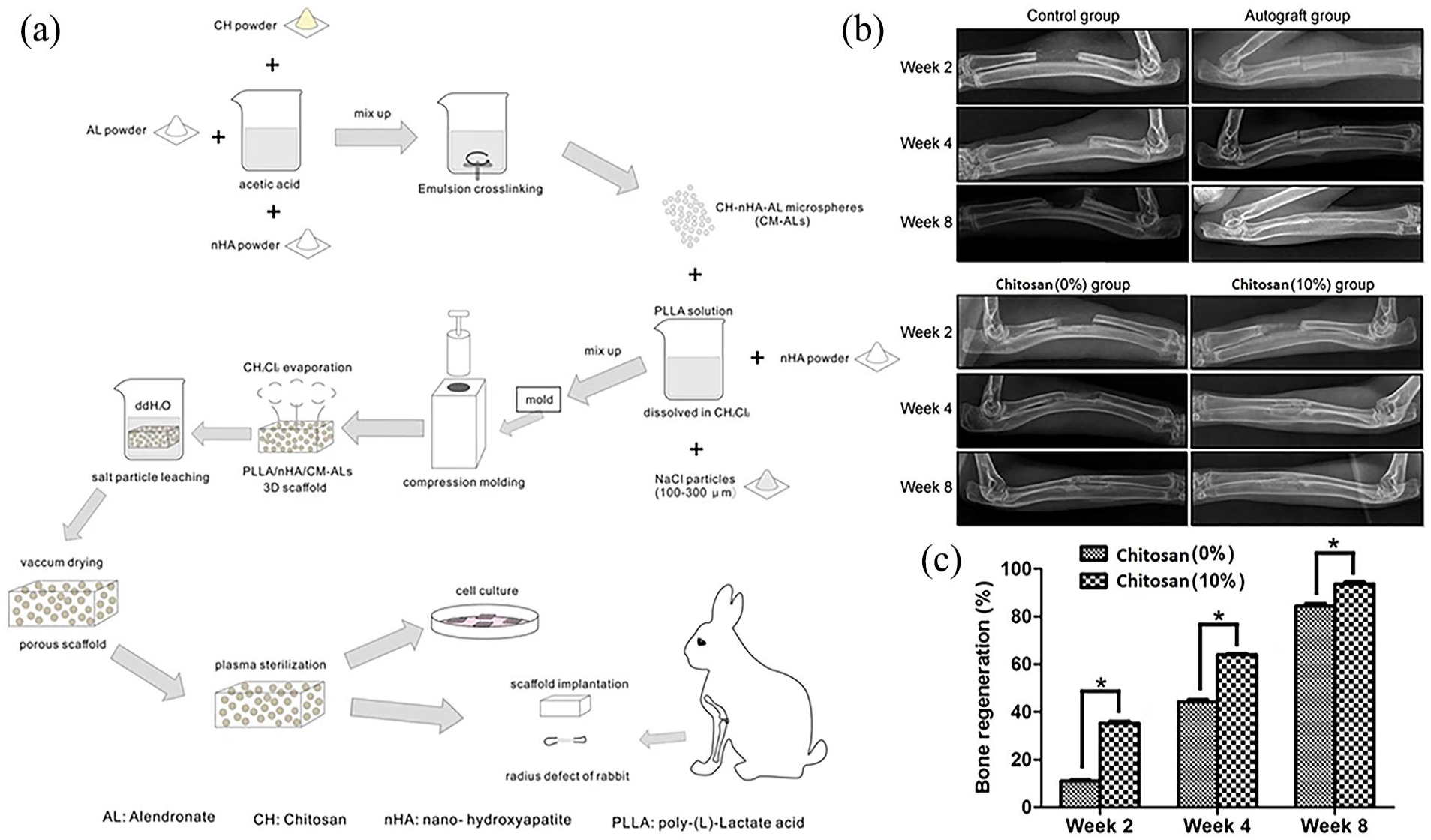
(a) Illustration demonstrates the preparation of chitosan-based scaffolds for large bone defects in a rabbit model. (b) X-ray analysis showed how bones regenerate at different time points after the implantation surgery. While some new bone formed in the control group, the bone did not fully heal. In the autograft group, the use of autologous orthotopic transplantation resulted in complete healing of the radius breaks within 8 weeks. In the chitosan (0%)-implanted group, there was clear new bone formation at week 8. However, there were few bone fracture lines observed, and the bone marrow cavities did not become interconnected. The chitosan (10%)-implanted group experienced the healing of bone defects and the formation of new bone between 4 and 8 weeks. (c) The semi-quantitative analysis showed that new bone formation was significantly higher in the chitosan (10%) group compared to the chitosan 0% group.
A chitosan-zeolite scaffold that is compatible with living organisms was created, followed by the absorption of the medicine used to treat osteoporosis. 96 Evidence demonstrated that the zeolite is uniformly dispersed across the entirety of the scaffold, leading to a notable influence on the consistent absorption and release of the medication from materials that could potentially be used in bone tissue creation. The zeolite scaffolds exhibited a drug retention capacity that was nearly double that of the hydroxyapatite scaffolds. The reason for this is that hydroxyapatite exhibits greater agglomeration, while zeolites include holes that allow for drug permeation. 96 Recently, Zhang et al., 145 combined melatonin (MT) with Chitosan and tri-block co-polymer made of poly (l-lactide) (PLA) and aniline pentamer (AP) in sequence as PLA-b-AP-b-PLA (PAP) to enhance the anti osteoporetic activity of melatonin. They prepared nanofibers and hydrogels formulations containing MT and chitosan to evaluate the effect of MT on MC3T3-E1 osteogenesis and mineralization. The findings demonstrated that chitosan@PAP nanospheres loaded with MT had beneficial osteogenic properties, both in laboratory settings and in living organisms, offering a viable approach for bone regeneration in individuals with osteoporosis. A biocompatible transdermal gel containing Risedronate (RS)-chitosan nanoparticles have been recently investigated for osteoporosis. The ex vivo Franz diffusion and in vitro cell uptake and distribution assays demonstrated significant cellular absorption and distribution, with a noticeable penetration of over 70% of the released drug in the mouse epidermal tissue. The research findings indicate that the prepared biodegradable chitosan-RS nanoparticles containing gel has the potential for treating osteoporosis and pain in clinical settings through passive targeting. 146 Extensive research has been conducted on the application of chitosan-β-glycerophosphate gels within the realm of biology, particularly for their potential in delivering medications that can be broken down by bacteria in the human colon. Chitosan-β-glycerophosphate hydrogels can function as a drug reservoir and release the drugs in a regulated manner. A study demonstrated the effectiveness of a chitosan-β-glycerophosphate hydrogel as a controlled release mechanism for fluoride in the gastrointestinal tract when taken orally. Low dosages of fluoride are known to promote the development of osteoblasts and decrease the likelihood of vertebral injuries in individuals with osteoporosis. 147
Evaluating the cytotoxicity of chitosan-based scaffolds
It is essential to evaluate the toxicity of chitosan-based scaffolds utilized for bone regeneration. This evaluation involves assessing many criteria, such as the integrity of cell membranes and the activity of mitochondria. The scaffold’s cellular milieu significantly influences its pro-regenerative capacity as a biomaterial. Several researchers have assessed the toxicity and biological compatibility of chitosan-based scaffolds, and the findings indicate that these scaffolds are non-toxic and biocompatible in both in vitro and in vivo settings. Gupta et al. 148 introduced dental pulp MSCs onto scaffolds made of both chitosan and chitosan integrated with vitamin D. The researchers then evaluated the cells metabolic activities after 72 h utilizing the colorimetric MTT test. The findings demonstrated the biocompatibility of both chitosan and chitosan scaffolds integrated with vitamin D, in comparison to the control group consisting of dental pulp mesenchymal stem cells (MSCs) in the growth medium. In a similar manner, Hu et al. 149 assessed the biocompatibility and ability to promote bone formation of scaffolds made from lanthanum phosphate and chitosan. When incubated with rBMSCs, these scaffolds did not exhibit any apparent toxicity or impact on cell shape. However, they did enhance the process of bone formation in a rat cranial injury model.
Conclusion and future prospects
Our understanding of osteoporosis has been steadily growing. However, the complex dynamics of bone activity and repair render simple methods rarely effective for their treatment. Consequently, certain targets have been established for the biomaterials employed in the treatment of various diseases. This article demonstrates the promise of chitosan scaffolds as therapeutic agents for bone treatment by providing a concise overview of the recent advancements in this field. Researchers employed chitosan, known for its sustainable characteristics and attributes, in combination with other polymers that are compatible with living organisms and can be naturally degradable. This combination led to the creation of films, nano/microparticles, and gels that can serve as drug-delivery systems for bone regeneration. Macromolecular engineering techniques can achieve precise modification of polymer properties, either by modifying polymers through (bio)conjugation or generating specific morphologies. As a result, these modified biomaterials will offer varying degrees of non-toxicity, biocompatibility, stability, target specificity, and biodegradability, hence serving as effective delivery methods for the management of osteoporosis. Whereas a wide range of different chitosan-based biomaterials have been investigated, it is apparent that this field demands significant attention to meet various requirements, such as biomaterial degradation, cytotoxicity, and their effects on both the environment and physiological functions. However, a few of these biomaterials are currently undergoing clinical trials. Hence, it is believed that the continued advancement of chitosan-based biomaterials would offer novel opportunities for effectively treating bone diseases.
Footnotes
Author contributions
YMW: Data gathering and organization; writing initial manuscript; JGT: Conception and design of the study; Review and finalization of the manuscript; Supervision and guidance of the work and finalization of the manuscript. All the authors read the final manuscript and agree to publish this work.
Data availability statement
Not Applicable. This is a review article and all relevant information is provided in the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
Not applicable. This is a review paper and do not involve direct research on humans or animals.
Consent for publication
“Not applicable” as this manuscript does not contain data from any individual person.
