Abstract
In the current study, Cnicus benedictus extract was loaded into electrospun gelatin scaffolds for diabetic wound healing applications. Scaffolds were characterized in vitro by mechanical testing, cell culture assays, electron microscopy, cell migration assay, and antibacterial assay. In vivo wound healing study was performed in a rat model of diabetic wound. In vitro studies revealed fibrous architecture of our developed dressings and their anti-inflammatory properties. In addition, Cnicus benedictus extract-loaded wound dressings prevented bacterial penetration. In vivo study showed that wound size reduction, collagen deposition, and epithelial thickness were significantly greater in Cnicus benedictus extract-loaded scaffolds than other groups. Gene expression studies showed that the produced wound dressings significantly upregulated VEGF and IGF genes expression in diabetic wounds.
Introduction
Diabetes is a chronic metabolic disorder that affects a large population worldwide. One of its common complications is the development of chronic wounds, which pose a challenge for effective healing and can result in severe consequences such as infections and amputations.1,2 Diabetic wounds pose a significant health challenge, as individuals with diabetes may experience delayed healing and increased infection risks. Elevated blood sugar levels impair circulation and immune function, making wounds more susceptible to complications. Proper wound care, glycemic control, and medical attention are crucial to prevent severe consequences and promote healing. 3
Wound dressings have been found to be an efficient approach for managing diabetic wounds. Natural products have been extensively researched for their potential to promote wound healing. Cnicus benedictus, commonly known as blessed thistle, has historical medical utility. Its potential anti-inflammatory and digestive properties have been explored in traditional medicine. Some studies suggest it may support liver health and exhibit antioxidant effects. However, more research is needed to fully understand its medical benefits and applications.4,5 Cnicus benedictus extract (CBE) contains bioactive compounds like terpenoids, flavonoids, and polyphenols, which have anti-inflammatory, antioxidant, and antimicrobial properties.6,7
Electrospinning is a widely used technique for producing nanofibrous materials for wound dressings. Gelatin is a natural polymer derived from collagen, which is popular for electrospinning due to its biodegradability, biocompatibility, and ease of processing. Electrospun gelatin nanofibers provide a scaffold for cell adhesion and proliferation, as well as enhance the delivery of bioactive molecules to the wound site, thereby promoting wound healing.8,9 Therefore, the combination of CBE with electrospun gelatin nanofibers has the potential to develop an effective wound dressing for diabetic wounds, where the extract’s antimicrobial and anti-inflammatory properties can help prevent infection and reduce inflammation.
This study aims to investigate the effectiveness of CBE-loaded electrospun gelatin wound dressings in promoting the healing of diabetic wounds.
Methods and materials
Preparation of CBE-loaded electrospun dressings
Gelatin (type A, Sigma Aldrich, USA) was dissolved in acetic acid (Glacial, Merck, Germany) at 35 wt.% final concentration for 24 h. Then, CBE (ethanolic extract from leaves, purchased from Iranpharma co ltd, Tehran, Iran) was added into the gelatin solution at 20 v/v% and mixed for 12 h. Finally, the CBE/gelatin solution was loaded into a 10-ml syringe. By applying a positive high voltage (19 KV), the polymer jet was formed and nanofibers were deposited onto an aluminum foil. The polymer feeding rate was 0.5-0.8 ml/h and the turning rate of the mandrel was 15-17 cm. Next, the scaffolds were cross-linked in glutaraldehyde vapor for 30 min and washed with distilled water three times. Finally, the scaffolds were incubated in vacuum chamber in order to get rid of residual glutaraldehyde.
Cell viability assay
The viability of L929 cells cultured onto the CBE-loaded gelatin scaffolds (CBEGEL) and CBE-free gelatin scaffolds (GEL) was investigated using Alamar blue assay (Sigma Aldrich, USA). 10 Briefly, L929 cells were seeded onto the scaffolds at the density of 7000 cells/scaffold in a 96-well plate and cultured for 7 days. On days 2, 5, and 7 cell viability was assessed using the protocol provided by the manufacturer.
Scanning electron microscopy (SEM)
The microstructure of CBEGEL and GEL scaffolds was assessed using a SEM instrument after coating them with gold with a sputter coater for 250 s. Imaging was performed under 25 kV accelerating high voltage via PHILIPS XL30 SEM. 11
In vitro wound healing (Scratch assay)
CBEGEL and GEL scaffolds were cut into 150 mg pieces and then immersed in 10 ml DMEM-F12 media containing 1% antibiotics (Gibco, USA) and kept for 5 days. Then, the culture media was taken, filtered, and kept at 4°C until use. Then, L929 cells were cultured in a 24-well plate until they reached 90% confluence. Then, a linear scratch was made roughly through the midline of each well and cells debris was washed with phosphate buffered saline (PBS). Then, CBEGEL and GEL scaffolds extract was added onto the cells and they were cultured for 72 h. At 48 and 72 h, the appearance of the wounds and their contraction was assessed. 12
Antiinflammatory assay
The potential anti-inflammatory activity of CBEGEL scaffolds was assessed with GEL scaffolds and control groups by seeding murine macrophage cells onto the scaffolds and stimulating them with 1 µg/ml lipopolysaccharide (LPS). 13 Finally, the concentration of pro-inflammatory cytokines including IL6 and TNF-a in each well was assessed using an ELISA kit.
Tensile strength analysis
The ultimate tensile strength of CBEGEL and GEL scaffolds was assessed by cutting the electrospun scaffolds into 5 × 2.5 cm2 pieces and measuring their thickness. Scaffolds were fixed into the jaws of the machine (Instron, USA) and a 500 N force was applied at an extension rate of 1 mm/min until the scaffolds failed. Ultimate tensile strength was calculated using stress-strain curves.
Water vapor permeability test
The water vapor permeability through the CBEGEL and GEL scaffolds was investigated using a method as described before. 14 Briefly, scaffolds were cut into squares with 2 cm diameter and were used to cap the opening of bottles containing 10 ml distilled water. Then, the bottles were incubated at 37°C for 12 h. Then, the evaporation of water through the scaffolds was measured and water vapor permeation was calculated using the following equation.
Permeation (mg.cm−2 .h−1) = W/AT where W is water weight loss, A is surface area, and T is the time of incubation.
Bacterial penetration assay
The penetration of bacteria through CBEGEL and GEL scaffolds was assessed using a method as described before.14,15 Briefly, 10-ml empty bottles were sterilized and filled with Brain Heart Infusion (BHI) broth (Sigma Aldrich, USA) bacterial culture media and capped with CBEGEL and GEL scaffolds (cut into circles with 5 cm diameter) and kept for 3 and 5 days. The bottles capped with sterile cotton ball and open bottles served as the control groups. At the predetermined time points, the optical density of samples was read at 600 nm.
Release assay
Release of CBE from CBEGEL scaffolds was assessed using a method as described before. 16 In summary, 150 mg of electrospun scaffolds underwent immersion in 15 ml of PBS within sealed containers at 37°C for a duration of 10 days. Initially, a standard curve for CBE was established by gauging the optical density of the substance in PBS at its maximum wavelength of 345 nm. At various time intervals, a pre-determined volume of the release medium was extracted, and its optical density was measured at 345 nm. Subsequently, fresh PBS was introduced to replenish the release medium. The obtained data points were then applied to the standard curve of the compound, enabling the calculation of cumulative drug release.
In vivo study
Wound healing potential of CBEGEL scaffolds were compared with sterile gauze and GEL scaffolds in male Wistar rat model of diabetic wounds. All animal studies were approved by the ethics committee of the Jinzhou Medical University. Nine diabetic rats were randomly divided into three groups (three animals in each group). The animals in each group were anesthetized via intraperitoneal injection of ketamine and Xylazine. Then, the dorsal skin of the animals was shaved and washed with betadine and ethanol 70%. Then, an excisional wound (1.5 cm in diameter) was created using a surgical scissor. Then, CBEGEL and GEL scaffolds were applied on the wound site and fixed in place using an adhesive bandage. The control group was treated with sterile gauze. On week 1 and 2, the macroscopic appearance of the wounds was assessed using a digital camera. The animals were kept for 14 days and then sacrificed for histopathological studies. Briefly, the wound tissues were harvested, fixed in formalin, embedded in paraffin, and sectioned. Finally, the tissue slides were stained with H&E and Mason’s trichrome.
Gene expression studies
The tissue expression levels of VEGF and IGF genes were examined on day 14 after the animals sacrifice wound tissues were harvested for RNA extraction using the RNeasy Mini Kit (Sigma Aldrich, USA). Subsequently, the SuperScript III First-Strand Synthesis System (Thermofisher, USA) was employed to convert isolated RNA into complementary DNA (cDNA). Specific primers for VEGF and IGF genes were designed, avoiding self-complementarity, and a real-time PCR setup with Power SYBR Green was utilized for gene amplification. PCR cycling conditions included denaturation, annealing/extension, and melting curve analysis. Data analysis involved normalization to an internal control, employing the 2 −ΔΔCt method for relative quantification. Experiments were conducted in triplicate, and statistical significance was determined using appropriate tests. Finally, relative mRNA expression was calculated relative to the wound tissues in the negative control group. 17
Statistical analysis
The data was examined using Graphpad Prism software, and the one-way analysis of variance (ANOVA) approach was employed for analysis. Furthermore, Tukey’s Honestly Significant Difference (HSD) test was employed to pinpoint the particular group distinctions that were statistically significant following the one-way ANOVA.
Results and discussion
Cell viability assay
Results showed that on day 2, control group had significantly higher cell viability than CBEGEL and GEL groups. However, on day 7, CBEGEL group had significantly higher cell viability than other groups. It could be that the Cnicus benedictus extract contains compounds that are biologically active and can promote the growth and multiplication of cells. This substance may have been slowly released from the scaffolds that were loaded with the extract, allowing them to gradually influence the cells. Moreover, the scaffolds made of gelatin may have also played a role in providing a nurturing environment for cell growth and division, thereby enhancing cell viability. Another possibility is that the Cnicus benedictus extract could have triggered the production of signaling molecules or growth factors known to support cell survival and replication. 18 It is important to note that the initial discrepancy in cell viability between the control group and the extract-loaded scaffolds groups on day 2 suggests that the extract-loaded scaffolds may have initially caused some degree of cell death or toxicity. Nonetheless, this effect was most likely temporary because the extract-loaded scaffolds eventually led to higher cell viability by day 7.
SEM imaging results
Results (Figure 2) showed that CNEGEL and GEL scaffolds had a web-like microstructure with a random distribution of nanofibers. Average fiber size for CBEGEL and GEL scaffolds was measured to be around 759.65 ± 243.10 nm and 876.21 ± 158.31 nm, respectively. The web-like microstructure of the scaffolds allows for increased surface area and porosity, which are desirable properties for tissue engineering applications. The random distribution of nanofibers also contributes to the scaffolds’ mechanical stability and flexibility, making them suitable for use in a wide range of tissue engineering applications. 19 The difference in fiber size between the two scaffolds is also noteworthy. The larger fiber size in the GEL scaffold may indicate that it has a more porous structure, which could potentially enhance its ability to support cell growth and proliferation. On the other hand, the smaller fiber size in the CBEGEL scaffold may indicate a more tightly packed structure, which could provide greater mechanical support. Overall, these results suggest that both CNEGEL and GEL scaffolds have desirable microstructural properties for tissue engineering applications. The differences in fiber size between the two scaffolds may offer distinct advantages in certain contexts, and further research is needed to fully understand how these differences may impact cell behavior and tissue growth. 20
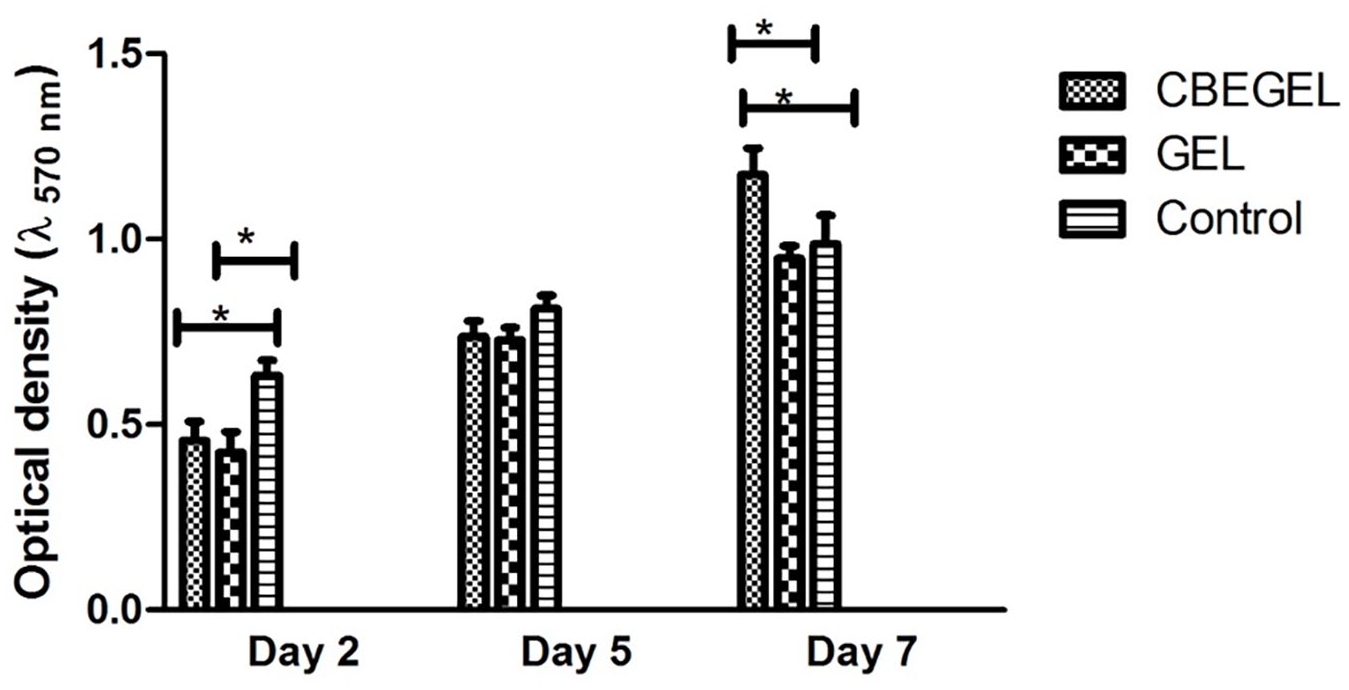
Alamar blue assay with L929 cells cultured on CBEGEL and GEL scaffolds.

SEM images of CBEGEL and GEL scaffolds.
In vitro wound closure
Results (Figure 3) showed that L929 cells cultured with the extract of CBE-loaded scaffolds could fill the scratch site to a significantly higher extent than other groups. Cnicus benedictus extract has various possible ways to enhance fibroblast cell migration activity. One of these ways is by activating specific signaling pathways involved in cell migration. This can be done by bioactive compounds in the herbal extracts that stimulate the PI3K/Akt signaling pathway, which is known to be related to cell migration and invasion.21,22 Another possible way is by modifying extracellular matrix (ECM) components. The polyphenol components of this extract has been found to increase the expression of certain ECM proteins such as fibronectin and collagen.23,24 These proteins provide a supportive environment for cell migration. Moreover, the extract might also cause the release of growth factors such as vascular endothelial growth factor (VEGF) and platelet-derived growth factor (PDGF).4,18,25 These growth factors are known to stimulate cell migration and angiogenesis. Apart from these mechanisms, the extract may have anti-inflammatory properties. These properties could potentially reduce inflammation and scarring at the wound site. This can lead to efficient fibroblast migration and wound healing. However, further research is necessary to fully understand the pathways and mechanisms underlying these effects.26–28
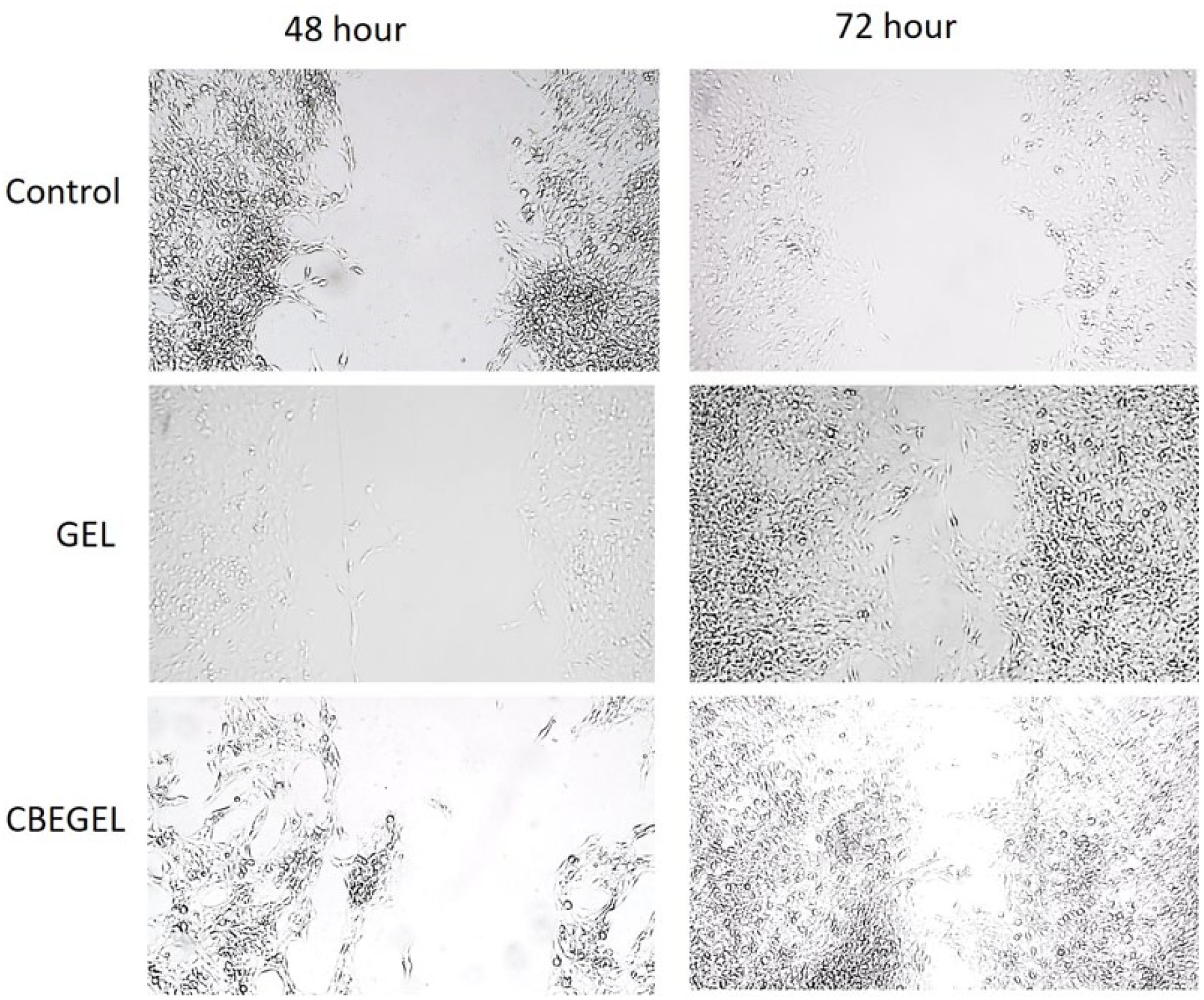
In vitro wound closure with L929 cells cultured with the extract of CBE-loaded and CBE-free gelatin scaffolds.
Anti-inflammatory assay
Results (Figure 4) showed that macrophage cells cultured on CBE-loaded scaffolds released significantly lower amounts of TNF-a and IL6 compared with the cells in other groups. Cnicus benedictus extract has the potential to modulate inflammatory responses in various ways. One such mechanism is through its antioxidant properties, where bioactive compounds such as flavonoids and phenolic acids present in the extract scavenge free radicals and reduce oxidative stress, which is known to contribute to inflammation. 25 The extract can also inhibit the production of pro-inflammatory cytokines such as IL-6, TNF-α, and IL-1β, which are crucial mediators of inflammation, as demonstrated in vitro studies.25,29,30 Moreover, the extract can regulate inflammation by influencing the immune system. It has been shown to stimulate the activity of NK cells and antibody production, thereby regulating the immune response and inflammation. Additionally, the extract may inhibit the activity of enzymes like COX-2 and iNOS, which play a role in the production of inflammatory mediators.4,25,30
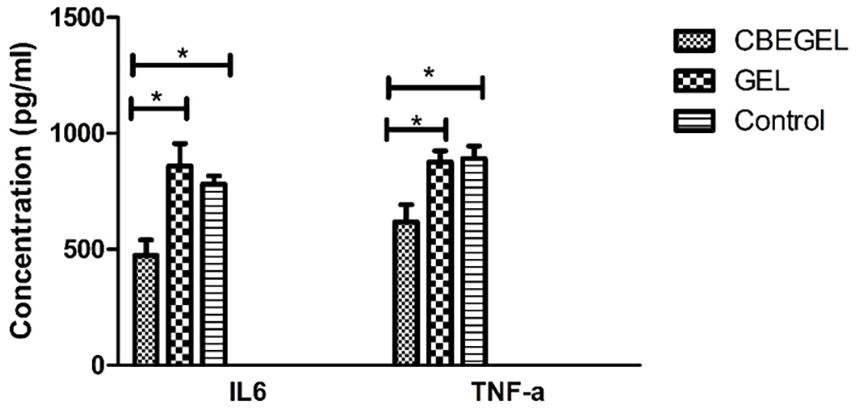
Anti-inflammatory activity of CBEGEL and GEL scaffolds.
Ultimate tensile strength measurement
Results showed that incorporation of CBE into the matrix of gelatin scaffolds did not significantly change their ultimate tensile strength. CBEGEL and GEL wound dressings had an ultimate tensile strength around 1.820 ± 0.530 MPa and 1.931 ± 0.266 MPa. In wet conditions, the ultimate tensile strength of CBEGEL and GEL wound dressings was 0.93 ± 0.15 MPa and 1.06 ± 0.23 MPa, respectively (Figure 5). The mechanical characteristics of wound dressings are vital since they determine the capability of the dressing to serve its intended purpose of wound protection and promotion of healing. Wound dressings should be able to resist mechanical forces, including movement or compression, without tearing or breaking. If a wound dressing is weak, it may not withstand daily activities’ forces, causing it to displace and, ultimately, hinder the healing process. On the other hand, a strong wound dressing may not conform well to the wound surface, thereby slowing down the healing process. Moreover, wound dressings’ mechanical properties are crucial for their ease of application and handling. A rigid wound dressing may be hard to apply in areas with uneven surfaces, while a soft or flimsy one may be challenging to manage or size. Hence, during wound dressing design and development, considering their mechanical properties is crucial to ensure they meet the necessary performance requirements and support effective wound healing.31–33
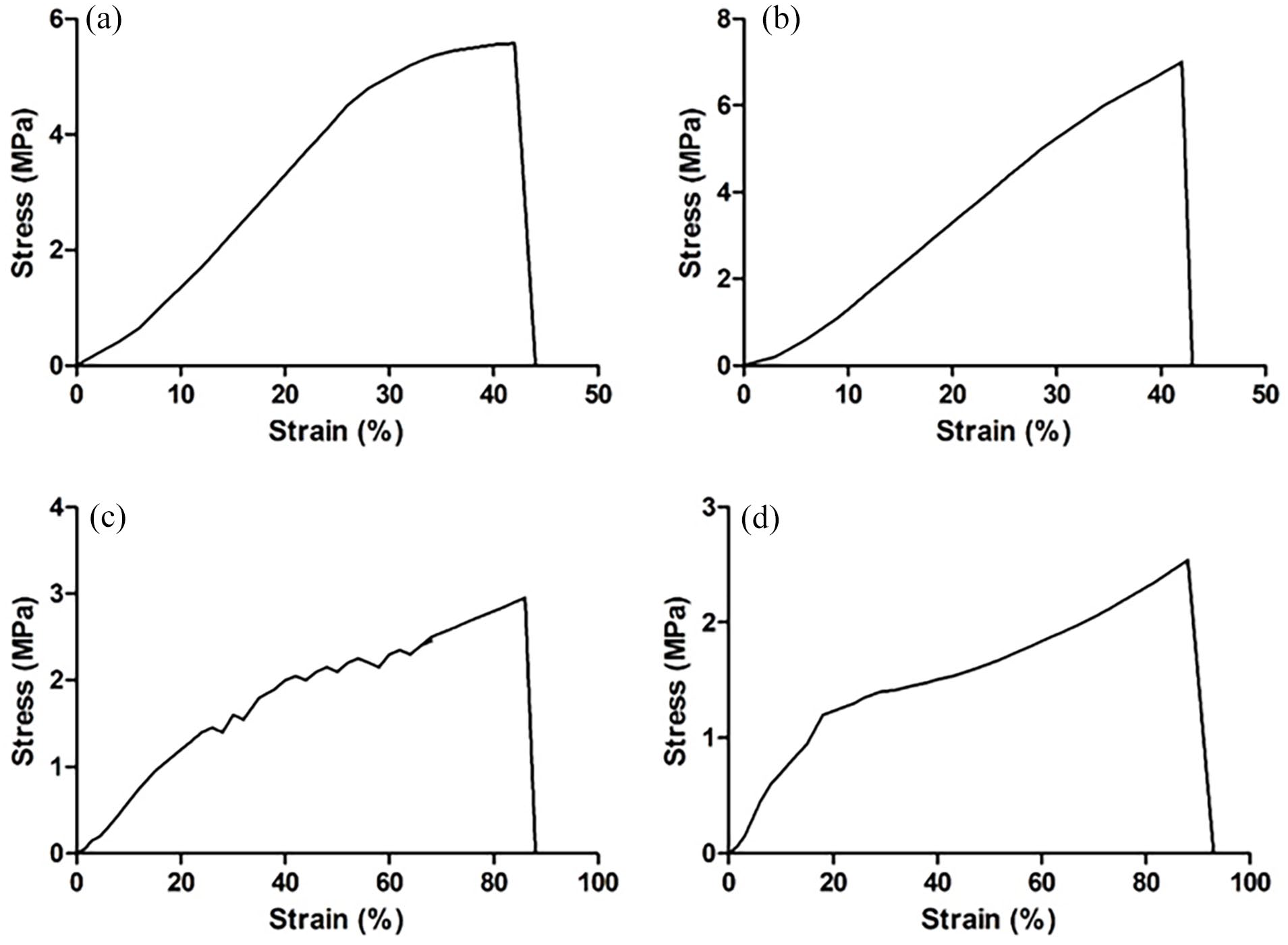
Stress-strain curve of (a) CBEGEL (dry scaffolds, (b) GEL (dry scaffolds), (c) CBEGEL (wet scaffolds), and (d) GEL (wet scaffolds).
Water vapor permeation test
Results showed that CBEGEL and GEL wound dressings had almost 28.10 ± 5.75 mg/cm2/h and 33.50 ± 4.78 mg/cm2/h of water vapor permeation, respectively. The differences were not significant. The electrospun fibers have a highly porous structure that allows for the diffusion of gases and fluids, which is necessary to maintain a healthy wound environment. The pores in the electrospun dressing allow oxygen to enter the wound and carbon dioxide to exit, promoting gas exchange and creating an optimal environment for wound healing. Moreover, electrospun wound dressings can also be designed to have controlled porosity and pore size, which can help regulate the flow of gases and fluids. This can be beneficial for wounds that require a specific level of moisture, such as those with high exudate levels.9,34,35
Bacterial penetration assay
Results (Figure 6) showed that CBEGEL group had significantly lower optical density (which indirectly implies less turbidity) than cotton ball-capped bottles, open bottles, and GEL group, indicating that CBE has somehow blocked the invasion of bacteria through the dressings. While the exact mechanism behind the antibacterial effects of CBE is still not completely understood, there are several possible explanations. One possible mechanism is the prevention of the synthesis of bacterial cell walls, which could cause the death of the bacteria. Studies have demonstrated that Cnicus benedictus extract has a variety of bioactive compounds, such as cnicin, which are capable of interfering with bacterial cell wall production. Another possible mechanism is the inhibition of bacterial protein synthesis. Flavonoids, including apigenin and luteolin, are present in Cnicus benedictus extract, which may be able to bind to bacterial ribosomes and stop protein synthesis.36,37 Indeed, the blockade of bacterial penetration via electrospun dressings have been shown before. In this regard, Salehi et al. showed that kaolin-loaded dressings could successfully prevent bacterial invasion. 38 Samadian et al. 14 showed that cellulose acetate/gelatin-based electrospun dressings could block the migration of electrospun matrices.
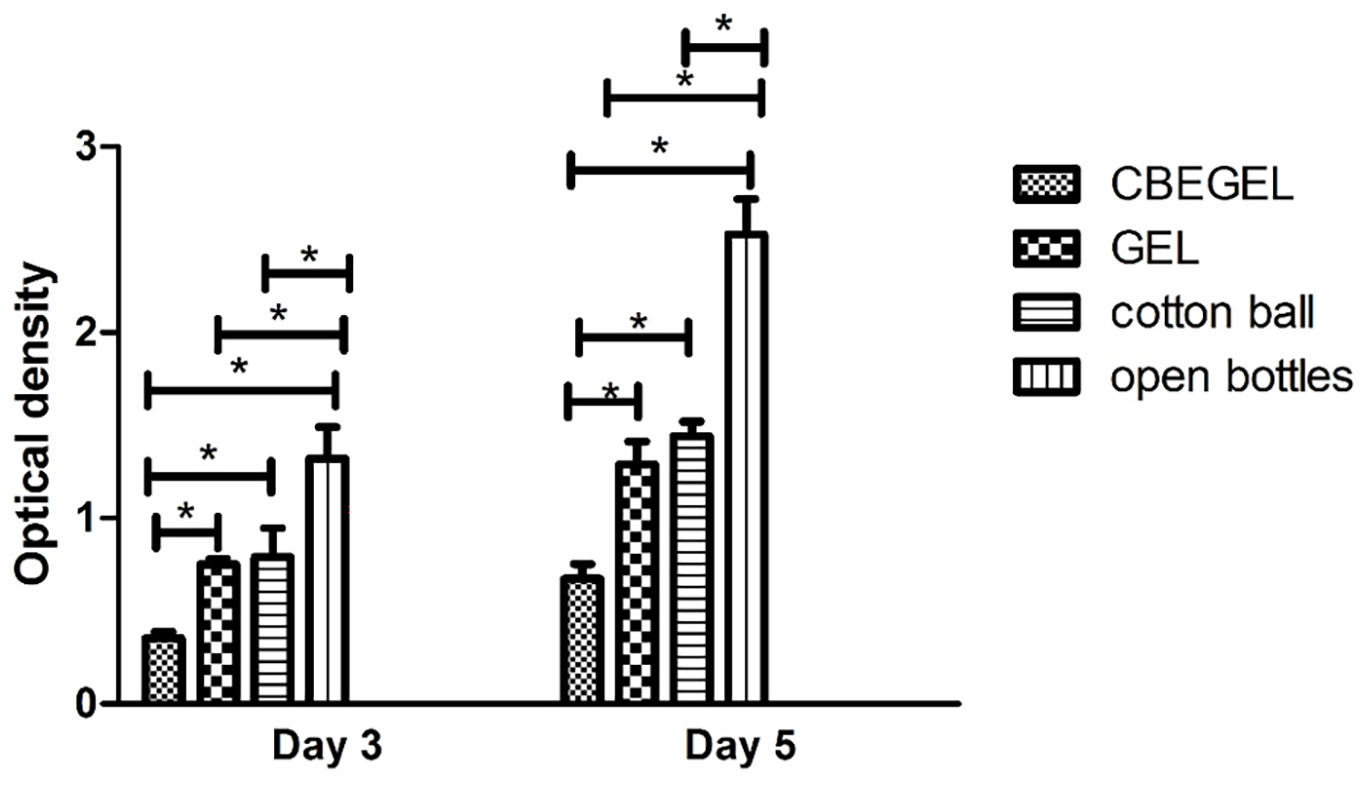
Optical density measurement of BHI growth medium capped with CBEGEL and GEL electrospun scaffolds.
Release assay
Results (Figure 7) showed that CBE was released from the matrix of CBEGEL scaffolds in a sustained manner. Cumulative drug release was sustained for over 10 days of incubation in PBS. Drug release from tissue-engineered scaffolds involves a multifaceted interplay of mechanisms that collectively govern the controlled delivery of therapeutic agents. Diffusion, a fundamental process, guides the movement of drug molecules from the scaffold to surrounding tissues based on concentration gradients. 39 Biodegradable scaffolds contribute to drug release through controlled degradation, releasing encapsulated drugs as the scaffold breaks down. Surface erosion and chemical interactions between drugs and scaffold materials further influence release kinetics. 40 Swelling and contraction of certain materials, along with electrostatic interactions, play pivotal roles in modulating drug release. Additionally, smart scaffolds responsive to temperature and pH changes enable triggered drug delivery. This comprehensive understanding of the mechanisms underlying drug release allows for the tailored design of tissue-engineered scaffolds, providing a versatile platform for applications in regenerative medicine and controlled drug delivery with the potential to optimize therapeutic outcomes.41,42
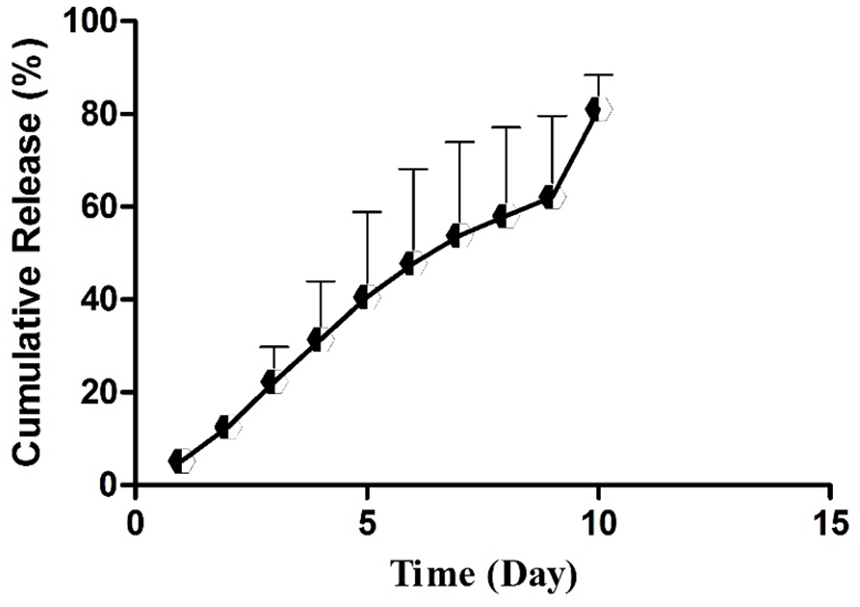
Release of CBE from the matrix of CBEGEL scaffolds during 10 days of incubation in PBS.
In vivo study
Wound closure assay (Figure 8) showed that on day 14, CBEGEL wound dressings had significantly higher wound size reduction than GEL and control groups. Statistically no significant difference was found between gel and the control group. Histopathological studies (Figure 9) showed that the wounds treated with CBEGEL wound dressings had decreased infiltration of pro-inflammatory cells compared at the wound bed compared with GEL and control groups. In the CBEGEL group, neovascularization has been occurred and the defect site was filled an organized network of collagen fibers. However, in the GEL and control groups the defect site was still empty and filled with a crusty scab. Histomorphometric studies (Figure 10) showed that percentage of collagen deposition and epithelium thickness in the CBEGEL group were significantly higher than GEL and control groups. However, statistically no significant difference was found between GEL and control groups. Gene expression studies (Figure 11) showed that the tissue expression levels of IGF and VEGF genes in the CBEGEL group was significantly higher than GEL group. Therefore, we can assume that at least part of the CBEGEL wound dressings’ healing activity could be due to the upregulation of these genes. Upregulation of IGF and VEGF plays a pivotal role in wound healing. IGF stimulates cell proliferation and tissue repair, while VEGF promotes angiogenesis, enhancing blood supply to the wound. Together, their coordinated upregulation accelerates healing processes, facilitating efficient tissue regeneration and restoration of damaged areas.43–45
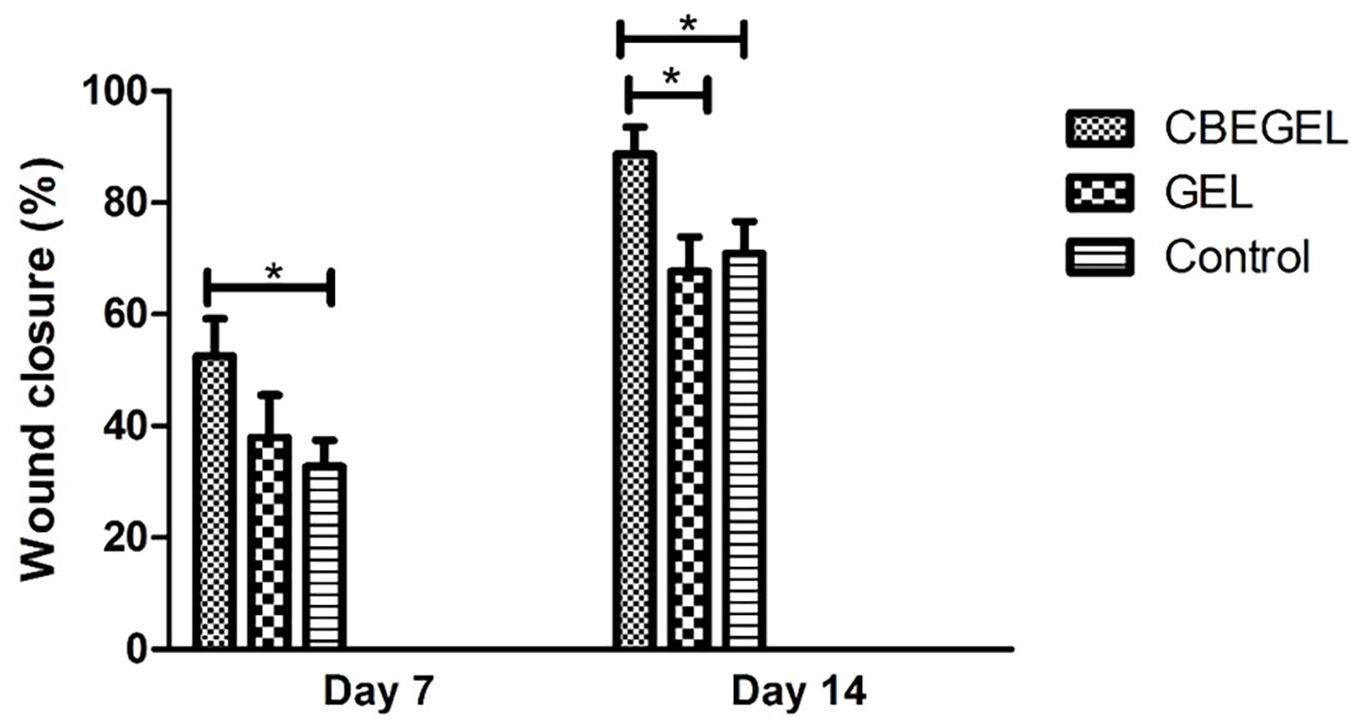
Wound size reduction in the rats wounds treated with CBEGEL and GEL wound dressings.
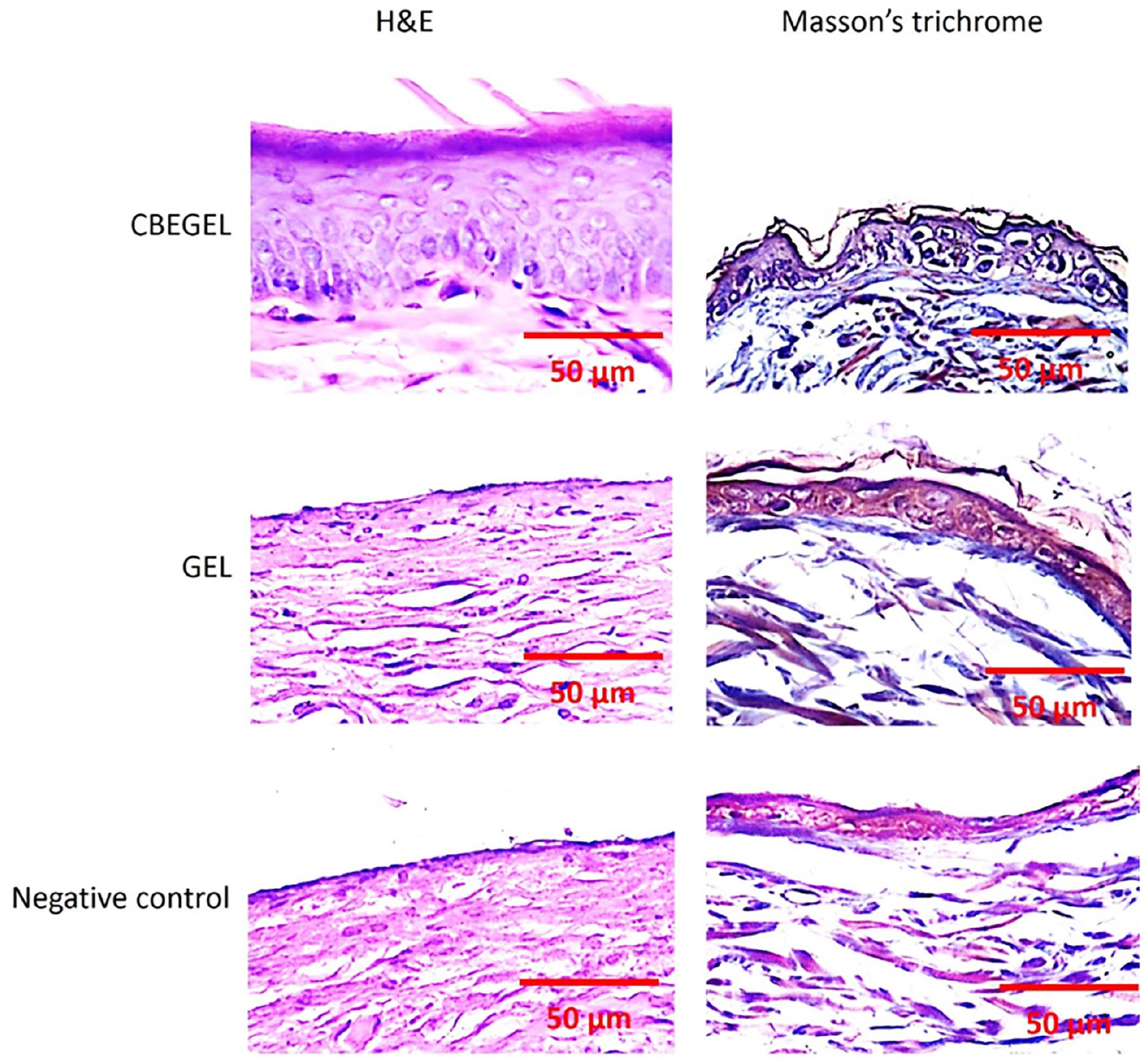
H&E and Masson’s trichrome (MT) staining images of diabetic wounds treated with CBEGEL and GEL wound dressings.
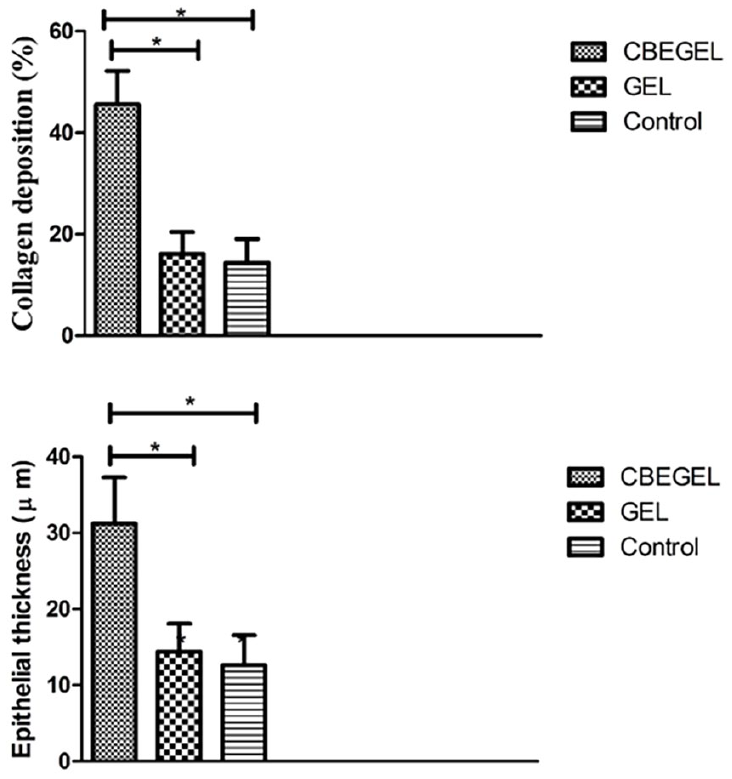
Histomorphometric studies in rats treated with CBEGEL and GEL wound dressings on day 14 after wounding.
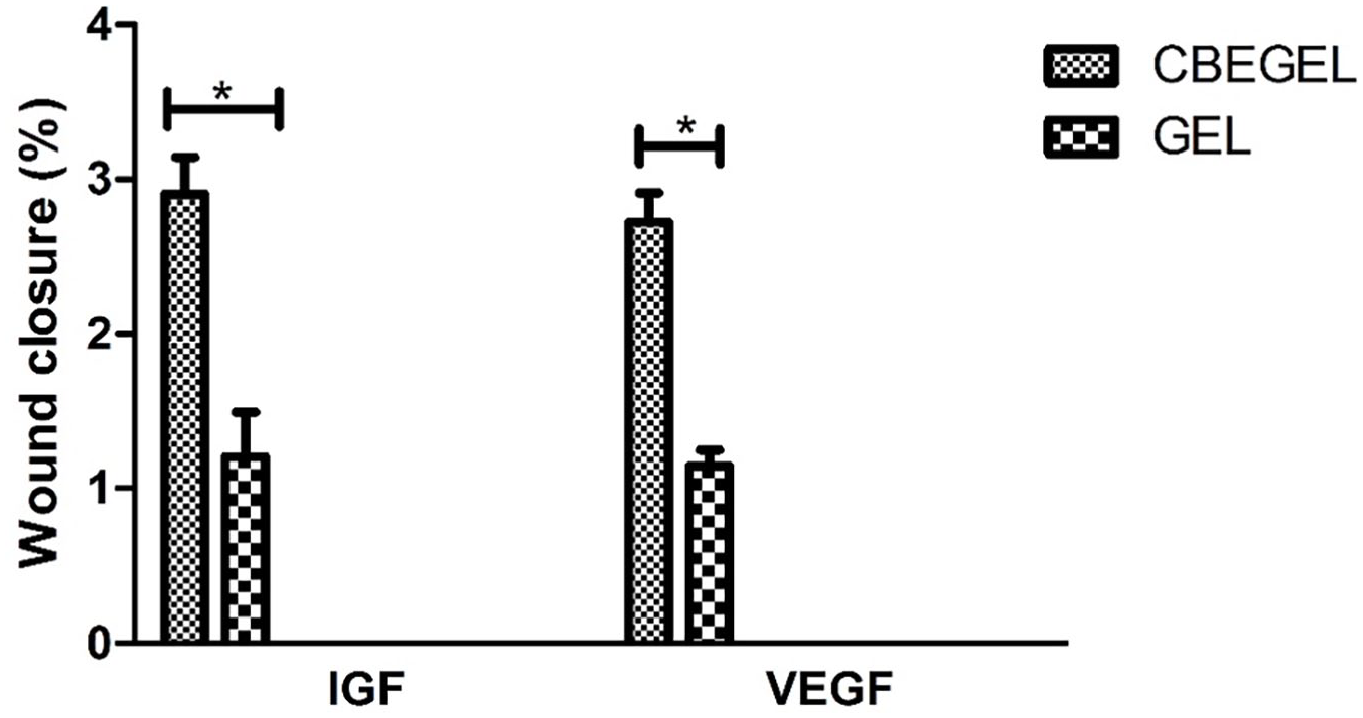
Relative mRNA expression of VEGF and IGF genes on day 14 after wounding.
CBE is a medicinal plant that has long been used for various therapeutic purposes, including the healing of wounds. This extract is believed to possess properties that may contribute to wound healing. One of the ways in which CBE can enhance wound healing is by mitigating inflammation. Although inflammation is a natural response of the body to injury or infection, excessive inflammation can hinder the healing process. CBE is rich in cnicin and polyacetylenes, which have anti-inflammatory effects, that could help reduce swelling and improve healing.25,46 Moreover, CBE is said to have antibacterial and antioxidant properties, which can further facilitate the healing process of wounds. 36 By preventing bacterial growth and eliminating free radicals that may cause cell damage, the extract may reduce the risk of infection and protect the tissues from further harm, resulting in more efficient healing. Although more research is necessary to comprehend the precise mechanisms of how CBE can aid in wound healing, its anti-inflammatory, antibacterial, and antioxidant properties make it a hopeful natural remedy for promoting wound healing.
Conclusion
In the current study, CBE was loaded into the matrix of gelatin scaffolds in order to improve diabetic wounds closure. In vitro studies showed that CBE-loaded wound dressings were not toxic against L929 fibroblast cells and improved their ability to close an in vitro wound model. Animal studies showed that CBE-loaded gelatin wound dressings promoted diabetic wounds healing to a significantly higher extent than other groups. Gene expression studies implied that at least part of the CBE-loaded dressings’ healing activity was mediated by upregulation of VEGF and IGF genes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
