Abstract
Joint replacements provide pain free movement for the injured or our aging population. Current prothesis mainly consist of hard metal on metal, or ceramic femoral head on ultra-high-molecular weight polyethylene (UHMWPE). In this study, a rodent fracture model was used to test the influence of wear debris from a high-performance polymer (polyimide MP-1™). Saline, MP-1™ Low Dose in Saline (1%), or MP-1 High Dose (2%) in Saline was injected directly into a standard closed unilateral femoral fracture in 12-week old Sprague Dawley rats (n = 25) for 1, 3 and 6 weeks. Endpoints included radiography, micro-computed tomography, mechanical testing and paraffin histology. No adverse effects from the wear particles were observed from the current study based on radiology, mechanical or histological data. Although the particles were present, histological analysis revealed a progression in healing between the Polyimide treated groups and the non-treated saline control groups over the duration of 1, 3, and 6 weeks, with no inhibition from the particles. The MP-1™ wear debris generated are larger than 1 µm thus are not able to be engulfed by macrophages and cause osteolysis. This family of polymers (polyimides) may be an ideal material to consider for articulating joints and other implants in the human body.
Introduction
In the USA alone, it is projected that total hip and total knee revisions will grow by 137% and 601% respectively, between 2005 and 2030. 1 Due to an increase in our aging population as well as a more active lifestyle, the need for new and durable implant bearing materials is warranted. Bearing materials like X-linked Polyethylene, ceramics and metal have a number of limitations and can wear over time. 2 The wear particles generated from these bearing systems have proven to be a major cause of early joint failure. 3 One alternate material that has recently been investigated is the polymer polyimide (MP-1™) as a potential advancement from the more common conventional materials used to date. 4
Polyimide MP-1™ is a high temperature thermoset polymer, that consists of an aromatic backbone molecular chain interconnected by ether functional groups. This chemical structure allows stability at very high temperatures, chemical resistance and radiation damage, and durability to creep and fatigue. 5 MP-1 is self-lubricating with no wear.
Like with many articulating joint materials, the production of wear debris throughout the lifetime of the implant is always a concern and a crucial issue to the longevity of total joint replacements, due to the subsequent tissue reaction to the debris. Submicron-size particulate debris generated by mechanisms of wear, abrasion, adhesion, and fatigue can induce the formation of an inflammatory response to initiate resorption of the polymer particles by macrophages. This process may promote a foreign-body granulation tissue response that can invade the bone-implant interface, thereby promoting osteolysis. Ultimately this can result in progressive local bone loss that compromises the fixation of the device.
MP-1™ when tested in a hip simulator 4 produced debris particles that were all larger than 2 microns. The need to investigate the biological reaction of these particles is warranted.
Such particles cannot be engulfed by macrophages, so they do not develop inflammation. Another area of interest is in trauma fractures where all devices (nails, screws and plates) are produced from metal (Titanium, Stainless Steel). These devices require predrilled holes for screw fixation. Use of trauma devices made out of MP-1™ do not need pre-drilled holes. These can be drilled whilst in the body which allows many intra-operative options. Drilling in the body however may leave debris of material which must be inert and not cause inflammation.
This study evaluated the effects of the first generation of polyimides MP-1™ wear debris on the healing behavior of bone when placed directly into a standard rodent fresh fracture model. 6
The study addressed the influence of wear debris of MP-1™ on the healing of fresh femoral fractured bone. Sterilized wear debris of MP-1™ was injected in the area of the fractured bone, with 2 different concentrations compared to a control of saline with no debris. The effect of the MP-1™ debris on healing was tested in two aspects: (1) Did the debris effect the healing rate of the bone (up to 6 weeks); (2) Did the debris effect the tissues around the bone and the newly formed bone or cause any inflammation or osteolysis of the bone.
These two effects were examined by radiographs, mechanical strength and histology.
In case both points showed negative results (inertness, no effect) the MP-1™ debris can be considered as biocompatible and inert.
Methods
Generation of particles
The samples required for this study were the smallest particles achieved by cryogenic milling and grinding of molded MP-1™ cured material.
Ten grams of cured MP-1™ material were supplied to “Super-fine Ltd” (Gallil Industrial Park, Israel). The samples were cut to small pieces and ground using a cryogenic mill under liquid Nitrogen. After reaching the smallest size available the particles were tested in ethanol to determine the particle size and size distribution (Malvern laser test). Particles were also examined under a Dino-Lite microscope (AnMo Electronics, Taiwan) and images were obtained using Dinocapture 2 software.
Particles were washed in Absolute ethanol, dried and steam sterilized.
Surgery
Seventy-five 12-week old female Sprague-Dawley rats (Animal Resources Centre, Perth, Australia) were used weighing an average of 260 g. Ethical approval was obtained through the Animal Care and Ethics Committee of the University of New South Wales [ACEC#: 17/88A]. Animals were acclimatized for at least 7 days prior to surgical procedures. Rats were randomly divided into 3 groups (n = 25): I – Saline control group, II – MP-1™ Low Dose in Saline (1%), and III – MP-1™ High Dose in Saline (2%) as per Table 1. Rats were sacrificed at 1 week (n = 3/group), 3 weeks (n = 11/group) and 6 weeks (n = 11/group) and both left and right femurs were radiographed, and micro-computed tomography performed to evaluate callus formation.
Study design.
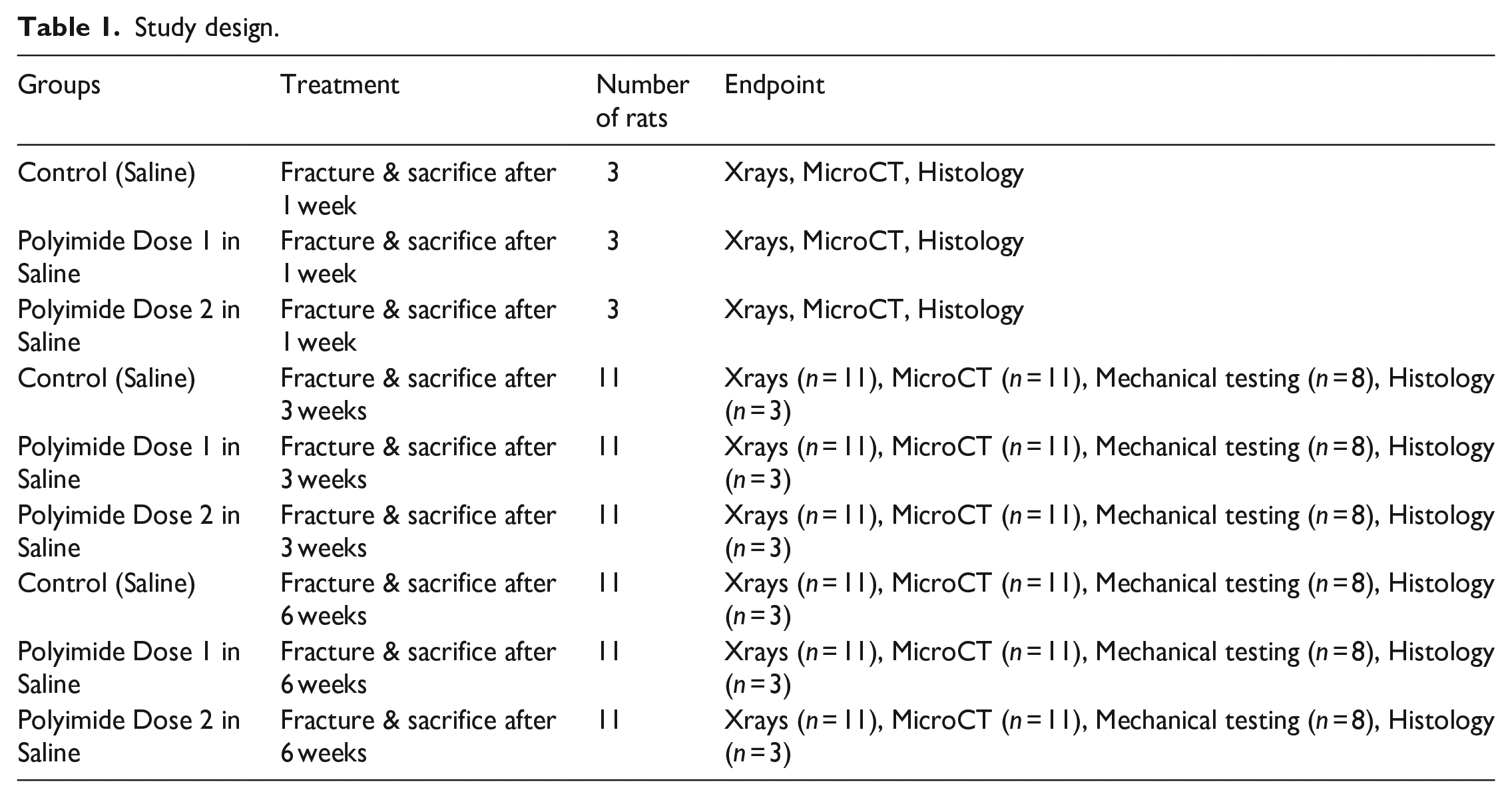
Three animals from each group per time point were randomly selected for histology and eight were used for mechanical testing from the 3- and 6-week groups. Sample size distribution for mechanical testing was alpha 0.05 and beta 0.8.
Following gaseous anesthesia, a standard closed unilateral fracture of the right femur was produced in each animal using a custom made three-point bending fracture device. The device is based on a parallel jaw nose pliers. 4.0 mm notches are cut on both jaw tips. 15 mm long, 3.6 mm diameter (n = 3) stainless steel bars spaced 6 mm apart are positioned on the edges of 1 jaw and on the center of the reciprocal jaw of the pliers in a manner such that the whole configuration delivers a parallel 3-point bending force to the bone when it is placed between the jaws.
Under sterile conditions, the right knee was flexed and a 1 cm incision was made medial to the patella. The longitudinal fibers of the quadricep muscles were divided medially, and with the knee in extension, the patella was dislocated laterally, exposing the femoral condyles. A 1.1 mm diameter stainless steel Kirschner wire (Zimmer, Australia) was inserted using a Surgical Z-Wire driver (Amscφ Hall Surgical z-wire driver) between the condyles and introduced into the canal, inserting it in a retrograde fashion up the shaft exiting through the greater trochanter. An incision was made over the greater trochanter to gain access to the proximal end of the wire. Distally, the k-wire was cut flush with the cortex of the patellofemoral grove and buried underneath the bone so as not to interfere with knee motion. The pre-pinned femur was placed in abduction and external rotation. The mid-shaft of the femur was placed between the 3-points in the jaw of the custom-made three-point bending device, and the jaws were rapidly closed to deliver a force that results in a standard closed fracture being created (Figure 1). All wounds were closed in layers with 3-0 Vicryl.
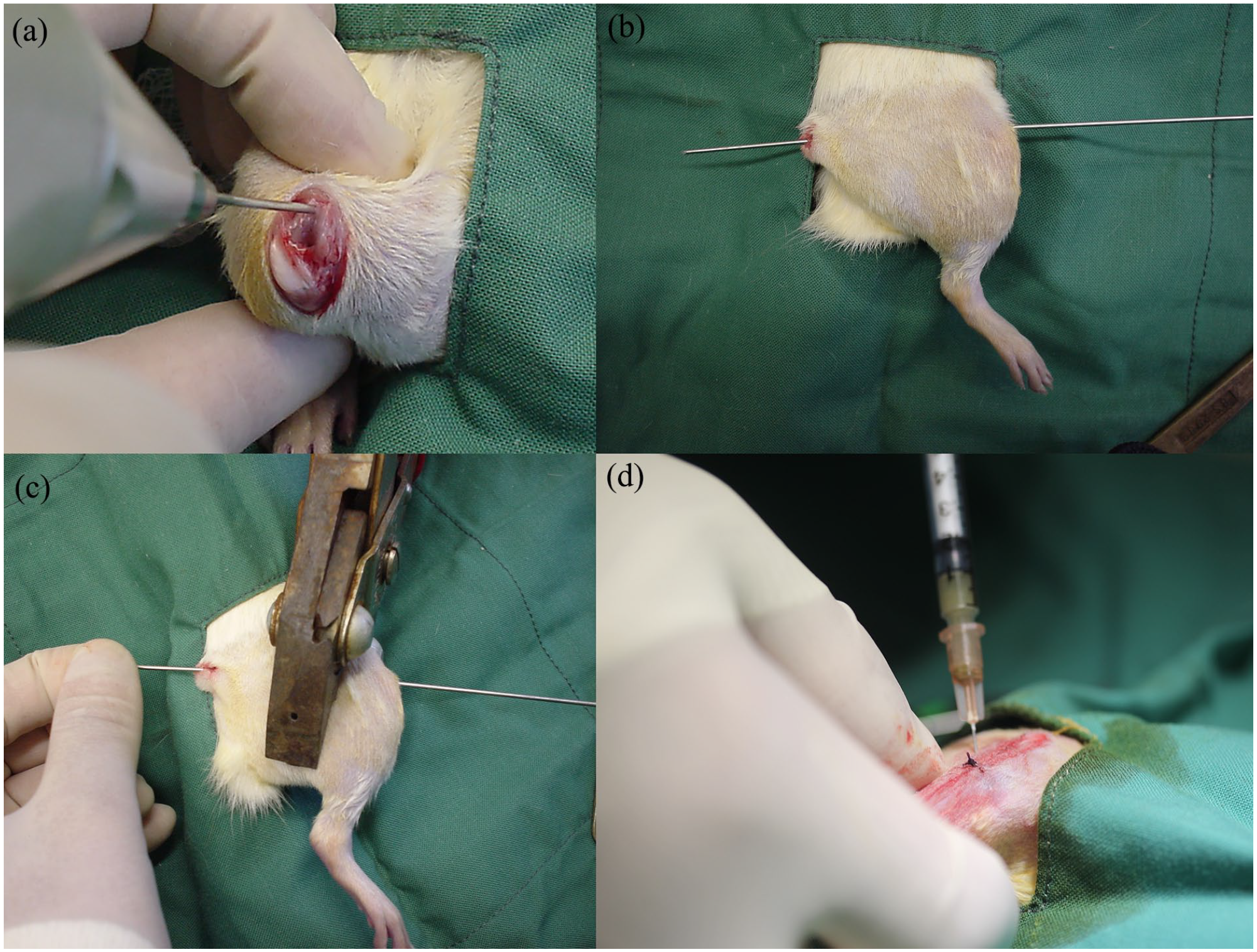
Creation of fracture. (a) Kirschner wire is driven between the condyles using a driver. (b) Retrograde extension of the Kirschner wire up the shaft and exiting through the greater trochanter. (c) Custom-made three-point bending device is positioned over the midshaft and a closed fracture created. (d) Injection of suspension into fracture site.
The particles in a saline carrier or control solution (0.2 mL) was percutaneously introduced into the fracture site using a 23G needle, and by palpation, maneuvered until the tip of the needle was at the fracture site, before injecting directly into the site of the nominated solution.
All rats received buprenorphine (0.1 mg/kg) and Hartmann’s solution (10 mL) following the procedure for post-operative pain relief and hydration respectively. Recovery was on a heat mat and all animals were monitored whilst allowing them to recover spontaneously.
The left femur served as the contralateral control. Rats were grouped housed with no restrictions on movement and given food and water ad libitum and monitored daily.
In addition, the regional lymph nodes, heart, kidney, liver, lungs and spleen sections were also harvested and placed into formalin for standard histopathology. These were reviewed for the presence of any foreign particles and adverse reactions.
Radiography
Harvested left and right femurs were X-rayed in the anteroposterior and lateral views using a Faxitron (Faxitron, Wheeling, IL) and high resolution digital plates (AGFA, Sydney Australia).
Mechanical testing
Femurs allocated for mechanical testing were biomechanically tested in three-point bending using Material Testing System 858 Mini Bionix (MTS Systems Corp, Minneapolis MN).6,7
Prior to testing, any remaining tissue was carefully removed. Specimens were placed on a custom-made 3-point bending jig with a span of 15 mm and loaded at the widest part of the callus in a saline bath at room temperature. The custom jig ensures consistent alignment of the axis of the bone with the axis of the testing machine. The displacement rate was 2 mm/min. The load versus displacement was recorded. The mechanical data was analyzed with a two-way ANOVA (treatment and time). Post hoc comparisons were performed using a Games Howell post hoc comparison (IBM SPSS Statistics) for Windows, version 25 (IBM Corp., Armonk, N.Y., USA).
Decalcified histology
Samples for histology were fixed in neutral buffered formalin (10%) for 48 h and decalcified in 10% formic acid solution at room temperature. The decalcified, embedded samples were sectioned to 5 microns using a microtome (Lecia, Germany). Slides were stained with hematoxylin and eosin (H&E) and Tetrachrome. The stained sections were examined under light microscopy (Olympus, Japan). Images of the histology were digitally captured (Olympus DP72 Camera) and used to assess callus formation and general tissue response to the presence of the test implant materials at the fracture site in a blinded manner to treatment and time.
Results
Particle size analysis
The average size of the particles DV/50 was 30–40 microns and size distribution were 3 microns (Figure 2). Figure 3 demonstrates particle size distribution at x200 magnifi-cation.
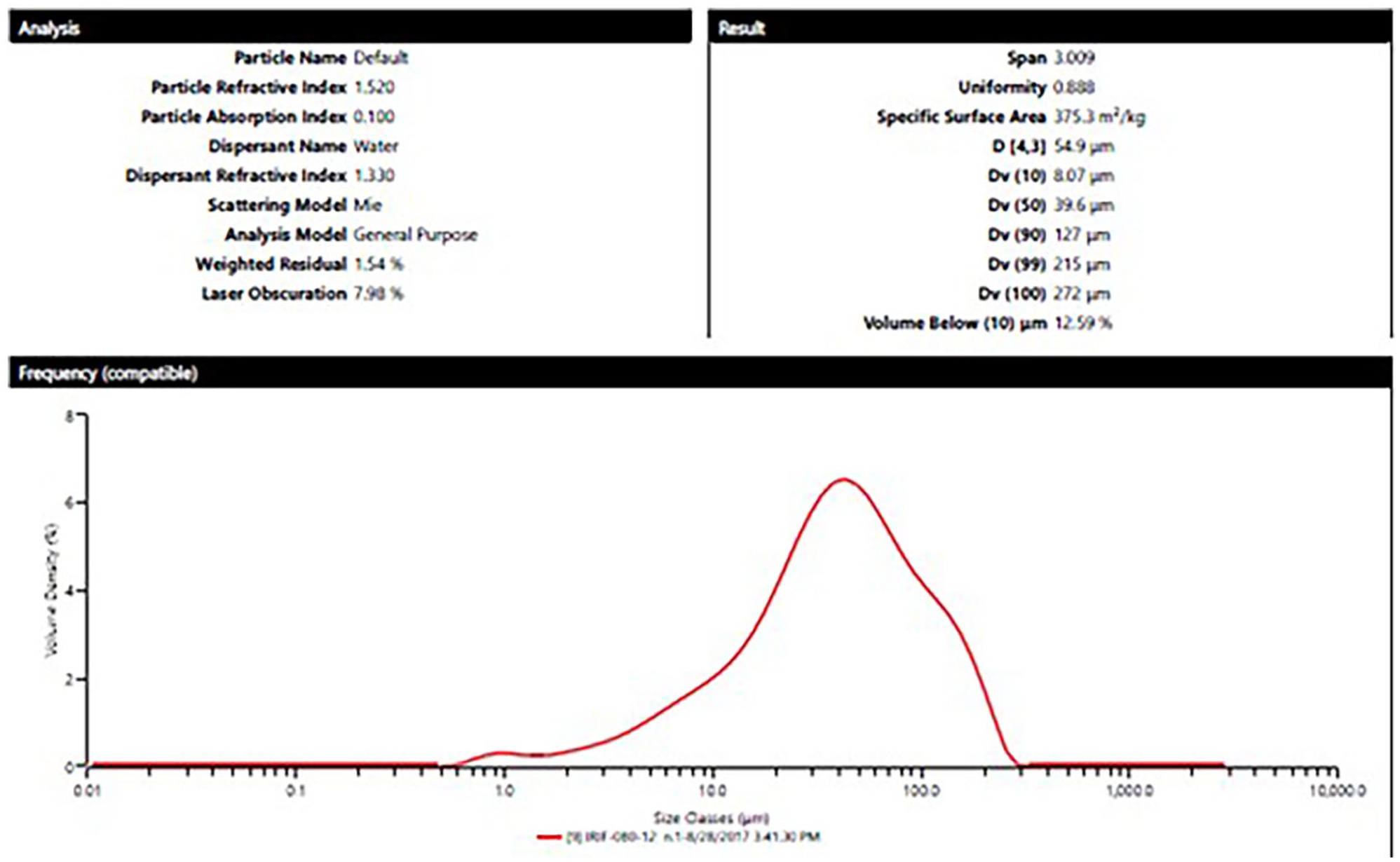
Average size and size distribution of MP-1™ milled particles. The average size was 30–40 microns.

MP-1™ particles after milling under x200 magnification (The black bar is 0.2 mm).
The material is MP-1™ a medical grade of polyimide proved for medical use. The polyimide is based on 4 monomers dissolved and cured to fine powder which is dried and cured at 400°C and at high pressure under compression molding.
This material was proved to have no wear and no friction, high modulus, high durability and full biocompatibility to human body, thus suitable to be used as medical devices specially in articulating joints.
MP-1™ is used as liner and femoral ball in hip implants (THR), Maxillofacial implant, dental implant and as trauma nail, plates and screws. MP-1™ is a high thermal resistant thermoset polymer from the polyimide family, consisting of an aromatic backbone molecular chain which is interconnected by ether functional groups. This chemical structure confers stability at very high temperatures (exceeding 400°C), resistance to chemicals and radiation damage, and durability to creep and fatigue. MP-1™ can be processed by conventional techniques such as compression molding to blocks and rods followed by machining or near net shape compression molding in specific designed mold allowing a broad design and manufacturing flexibility. MP-1 has been reported to have a tensile strength based on ASTM D638 of 108 MPa and a tensile modulus of 3748 MPa with a tensile elongation at break of 8.9%. The compressive modulus is 3900 MPa and the flexural modulus is 4272 MPa. 4
Surgery
Surgery was completed without any adverse events. One rat had a comminuted fracture and was excluded from the study (I – Saline control group, 6 weeks). All rats tolerated the surgery and were ambulating within the first day of post-operative monitoring. Normal gait and joint movement was observed in all animals. All rats gained weight over the course of the study.
Limb harvesting
The animals were euthanized, and the left and right femurs were harvested and photographed using a digital camera at their allocated time points. The general integrity of the skin incisions as well as the macroscopic reaction of the underlying subcutaneous tissue was normal. No adverse reactions or infections were observed. No migration of MP-1™ particles was observed in the subcutaneous tissues.
Radiography and MicroCT
Anteroposterior (AP) and Lateral (LAT) radiographs revealed no adverse reactions. Fracture calluses were visible at 3 and 6 weeks. The Polyimide particles were not visible on either the anteroposterior or lateral views. Radiographs and micro-CT did not show evidence of osteolysis. The fracture healing pattern appeared to progress in a normal fashion with or without MP-1™ particles when viewed with both imaging modalities.
Mechanical testing
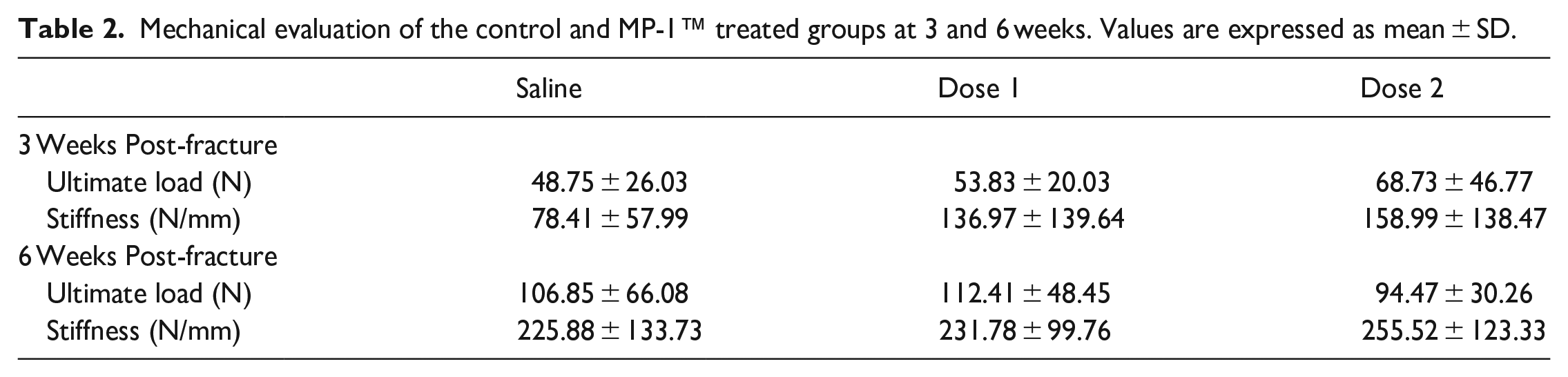
Samples allocated for mechanical testing failed in the midshaft through the original fracture site under 3-point bending. The mechanical properties that is, peak load and stiffness of the fractured femurs increased with time. No statistical differences in the ultimate load or stiffness was observed between the control group and MP-1™ treated fractures at any time point. The mechanical properties assessed using 3-point bending are summarized in Table 2.
Mechanical evaluation of the control and MP-1™ treated groups at 3 and 6 weeks. Values are expressed as mean ± SD.
Histology
Histology of saline treated fractures (Group I) at 1 week, revealed a fracture callus filled predominantly with fibrous tissue and cartilage, and some woven bone (Figure 4). By 3 weeks cartilage and new bone formation demonstrated closing of the fracture gap. The cortex had remodeled by 6 weeks in all cases (Figures 5 and 6). For Groups II and III at 1 week, the polymer debris was observed within the fracture callus surrounded by some multi-nucleated cells, however these were isolated and localized (Figure 7). The presence of the particles at either dose did not appear to inhibit the fracture healing process or elicit an inflammatory response. The particles were also birefringent under polarized light (Figure 8). Although at 3 and 6 weeks, MP-1™ debris was present in some of the fracture sites callus, healing of the fractures progressed normally. At 3 weeks the fracture callus was predominantly filled with cartilage and remodeling bone closing the fracture gap via endochondral ossification. By 6 weeks, some MP-1™ particles were present in the fracture site surrounded by multi-nucleated cells, however this did not appear to inhibit the healing process. Remodeling of the fracture callus continued at 6 weeks (Figure 9).
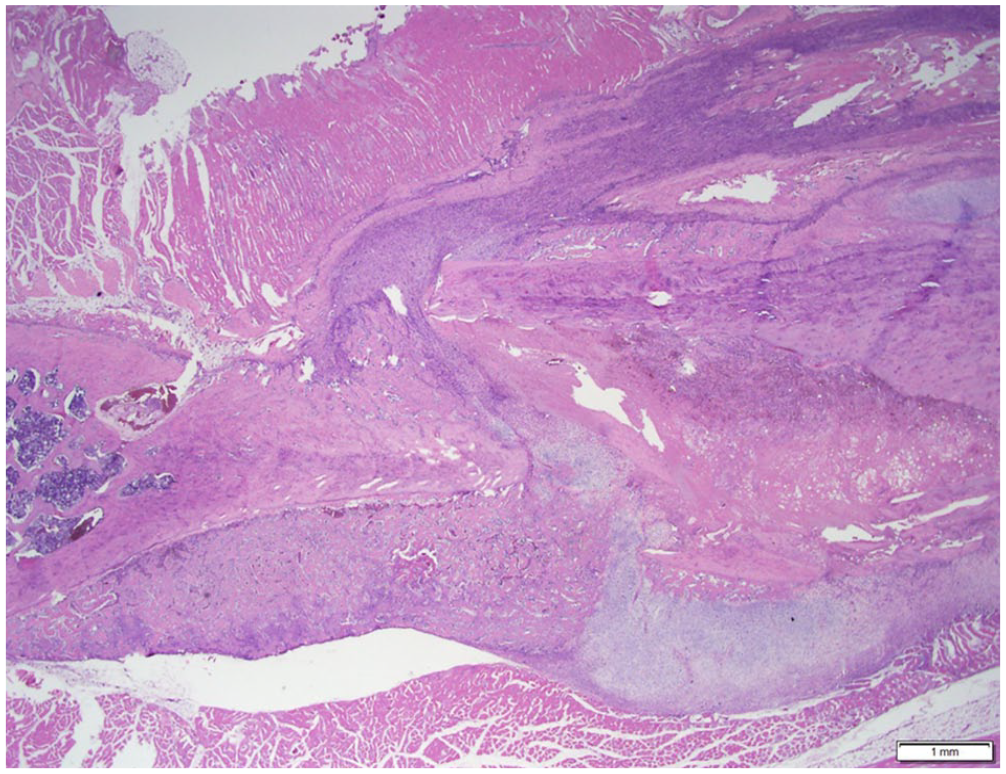
One week Saline: The fracture callus was predominantly filled with fibrous tissue and cartilage, and some woven bone (H&E).
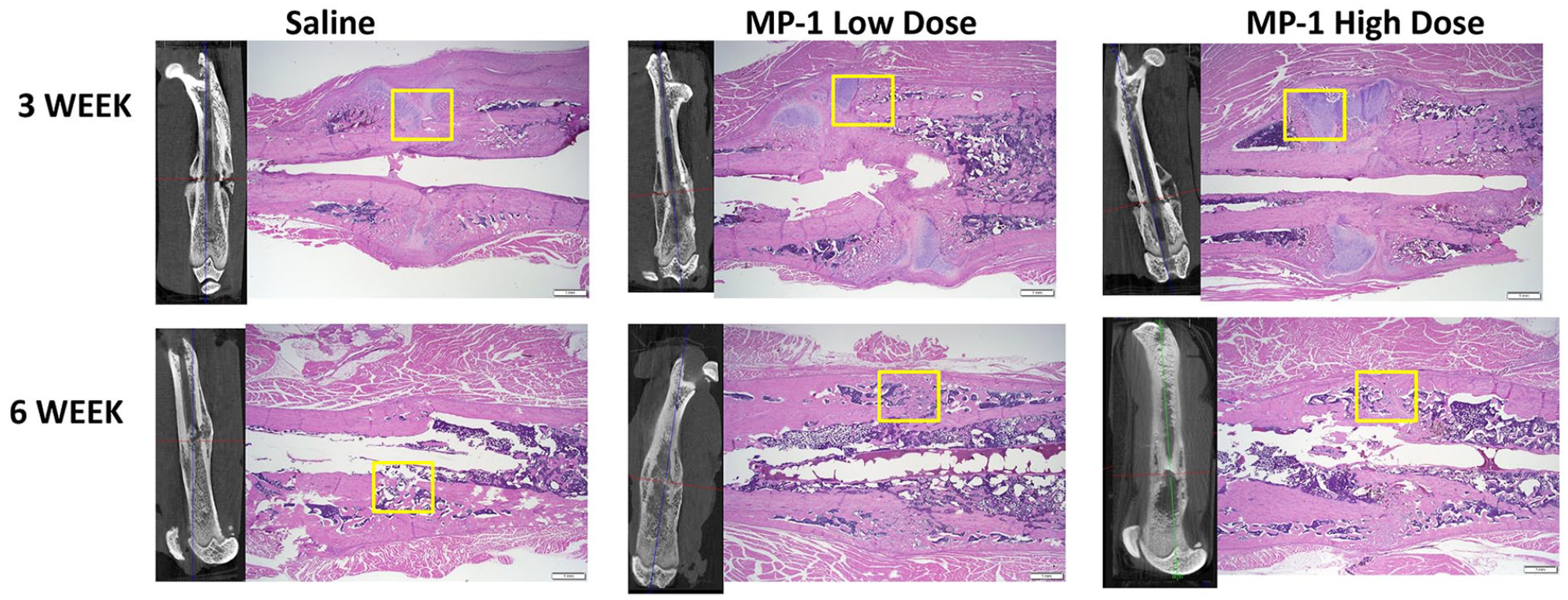
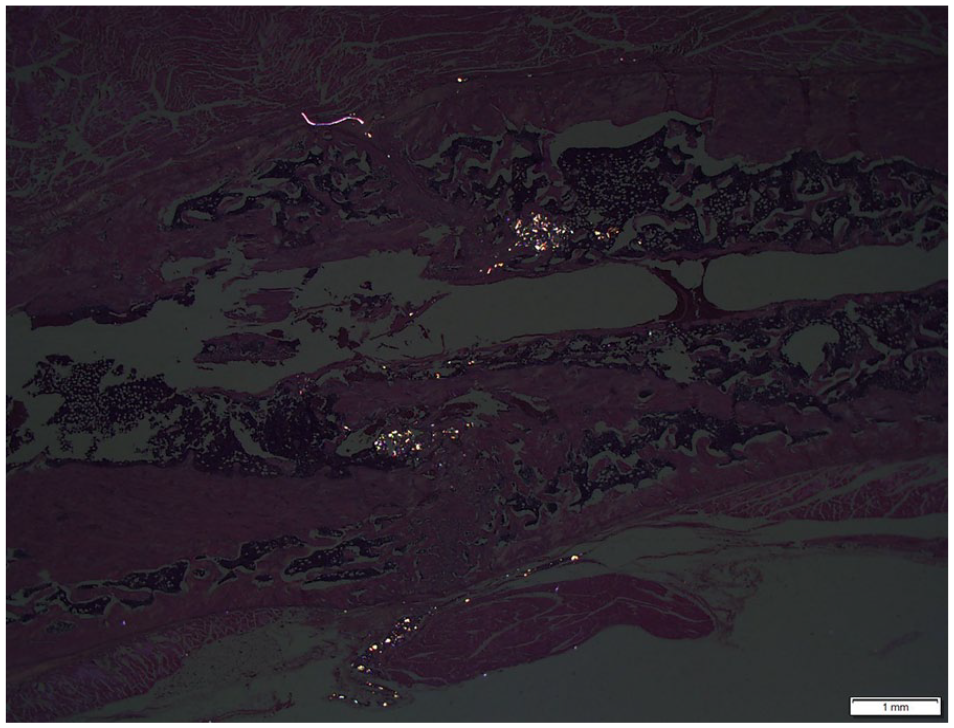
MicroCT and corresponding histology of the fracture site at 3 and 6 weeks for all 3 groups (1.25x, scale bar 1 mm). The yellow boxes are high magnification images represented in Figure 6.
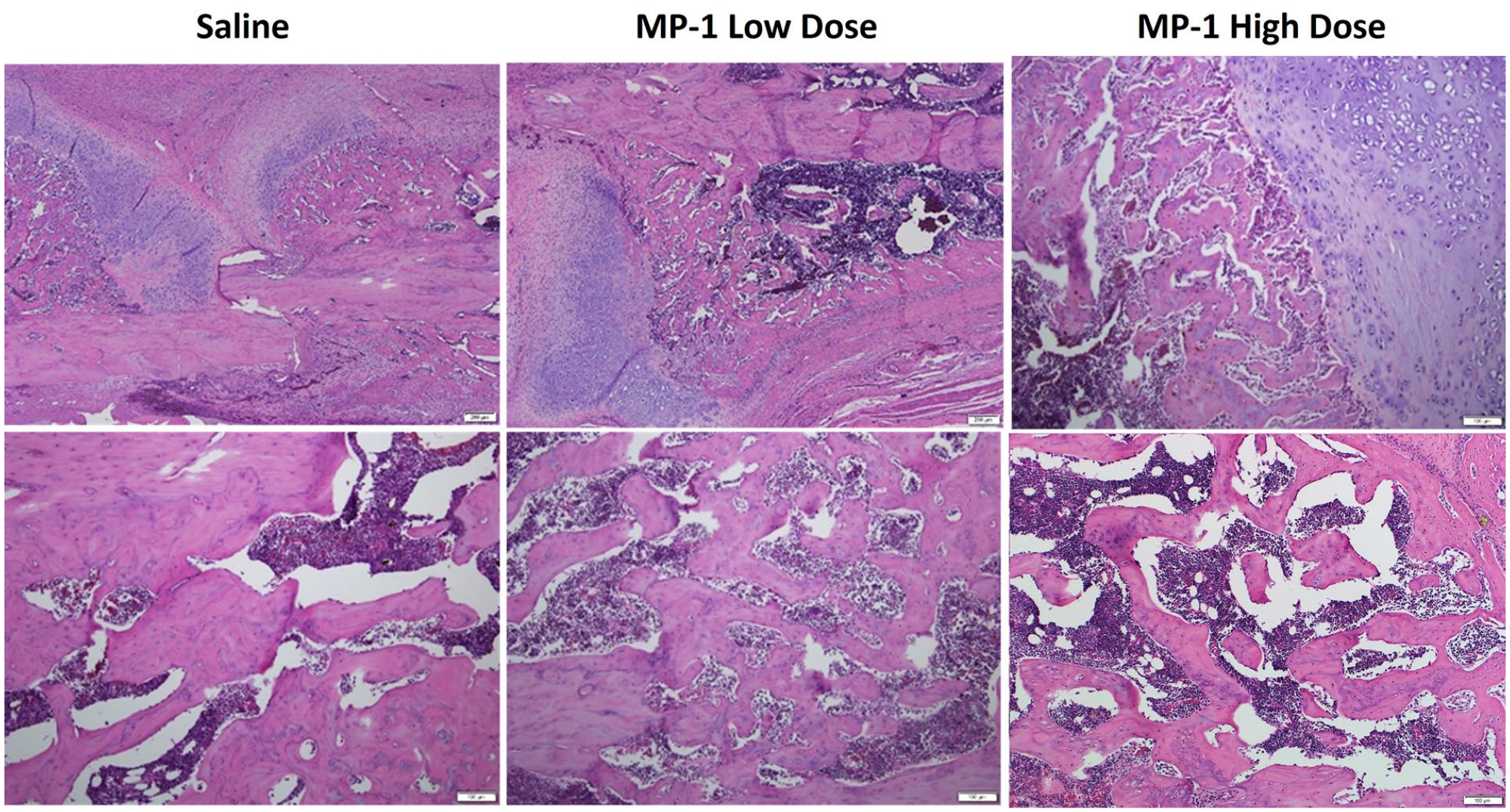
Higher magnification images of the healing progression within the fracture callus for all 3 groups at 3 (top row, 10x, scale bar 200 µm) and 6 weeks (bottom row, 20x, scale bar 100 µm).
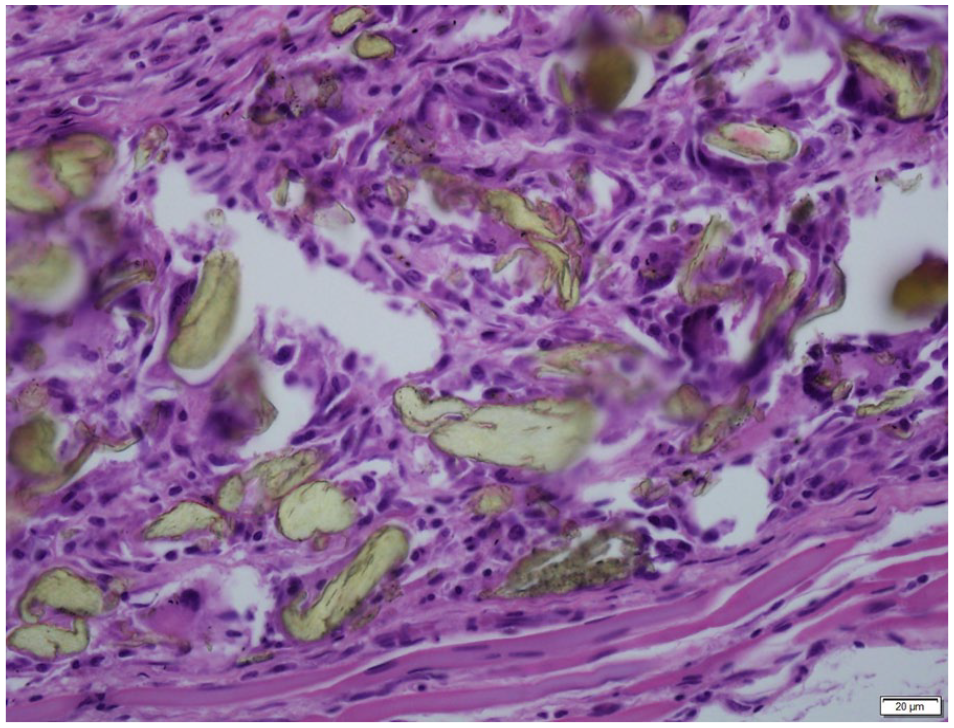
One week Polyimide Dose 1: The polymer debris can be seen within the fracture callus with the presence of some multi-nucleated cells. This however, did not appear to inhibit the fracture healing process (H&E).
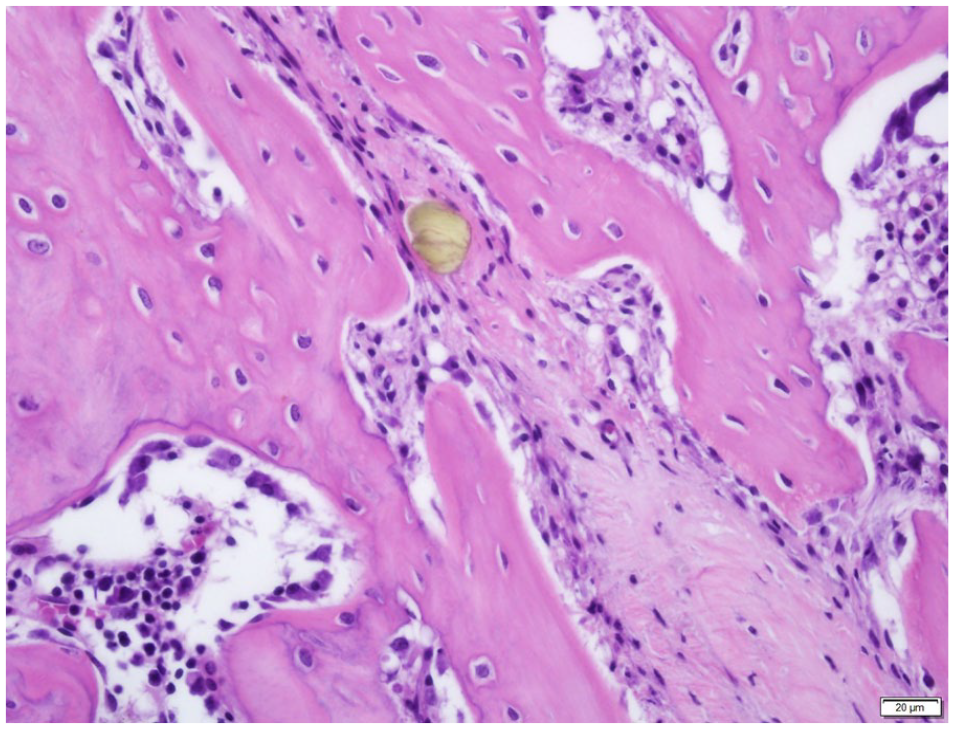
The particles are birefringent under polarized light and can be seen within the fracture site.
At 6 weeks MP-1 high dose, fracture healing progressed with no inhibition (H&E).
Discussion
Research into the biocompatibility of orthopedic materials is becoming ever more important as the use of implants steadily increases, the expectations of implant longevity and performance grow, and as new implants are developed and marketed. The effects of wear debris on bone cells, bone resorption and surrounding tissues is a crucial issue for orthopedic implant durability.
MP-1™ is the first generation of polyimides used in medical devices. Previous pre-clinical testing has shown that MP-1™ has very low wear, 4 lower than all other couples used in articulating joints in orthopedics such as metal, ceramic and X-UHMWPE. Fracture toughness was superior to that seen in ceramics and better wear resistance than polyethylene. In addition, minimal friction coefficient and a high chemical inertness and durability in fatigue and creep were also demonstrated.
Previous tests aimed at determining the safety of the material for articulating joints included Hip simulator, catastrophic impact test, stability tests and aging stability. After 5 million cycles in saline solution, hip simulator results demonstrated that MP-1™ exhibited an order of magnitude less wear than the UHMWPE control. Fluid analysis showed that the volume of particles from the MP-1™ material were significantly low (0.044 mm3 after 5 M cycles = 5 years). The size of the MP-1™ particles generated from the hip simulator was 1.9 to 23 µm whilst the size of UHMWPE particles was 0.694 µm and less (sub-micron).
Biocompatibility of MP-1™ has been tested according to ISO 10993-1 and included: Cytotoxicity, Extractable identification, Trace metals, hemolysis test, reverse mutation assay, mammalian chromosome aberration, mammalian micronucleus test, Intracut Reactivity, Sensitization, Acute Systemic Toxicity Mouse, Genotoxics I&II, Muscle implantation (4 and 12 weeks), Bone implantation (12 weeks) and chronic toxicity. All tests passed and were performed in full compliance with the principles of Good Laboratory Practice (GLP). A biocompatibility certificate was issued.
MP-1™ has also been implanted in THR in clinical trials for as long as 17 years with no infection or rejection.
The MP-1™ wear debris generated are larger than 2 µm thus are not able to be engulfed by macrophages, avoiding osteolysis or inflammation. All other materials produce sub-micron debris causing inflammation. Therefore, this family of polymers (polyimides) may be a material to consider for articulating joints and other implants in the human body.
Although preclinical testing demonstrated positive results and showcased MP-1™ in a positive light, an in vivo study into the biological reaction to this material was warranted.
The current study evaluated the effects of fracture healing in the presence of polyimide wear debris particles (2 doses) in a rodent fracture model compared to a saline control group. No statistical adverse effects from the wear particles were observed from the current study based on radiology, mechanical or histological data. Although the particles were present, histological analysis revealed a progression in healing between the Polyimide treated groups and the non-treated saline control groups over the duration of 1, 3, and 6 weeks, with no inhibition from the particles.
The rationale behind the high and low dosages was to see if a higher dose induces a more pertinent host response. Previous studies have shown that low doses of particles (Co-Cr, Ti, and UHMWPE) promote the proliferation of bone marrow stromal cells whilst high doses, mainly of Co-Cr, can lead to cell death8,9 hence the difference in doses was to see if a change in response was observed on healing.
The fact that no inflammation, no osteolysis, nor growth of fibrous cells were observed around the MP-1™ debris and the fact that no inhibition of the healing process occurred proves the biocompatibility of the MP-1™ wear debris in the body.
Due to its favorable properties, MP-1™ biomaterials is an attractive platform to develop many different novel bioactive devices. MP-1™ will be an alternative to metallic biomaterials in the orthopedic community for implantation and fracture fixation implants in the future.
One of the limitations of this study was that a clinical control material was not included in this study for comparison against the performance of MP-1™.
Conclusion
For the past two decades low-wear UHMWPE has been considered for total hip implants. However frequent failure of these protheses has been due to late aseptic loosening, migration and inflammation resulting in the need for revision surgeries. Due to an increase in an aging population, the demand for new and more durable implant bearing materials is warranted. This study showed that polyimides (MP-1™) when introduced into a fracture site at various doses did not inhibit fracture healing and could be a potential future material to consider.
Footnotes
Author contributions
WW, RO, AB, SS researched the literature, conceived the study and were involved in protocol development. WW and RO gained ethical approval. WW, RO, TW and CC performed the endpoints. RO wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AB and SS are employees of MMATECH.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has received funding from the European Union’s Horizon 2020 research and innovation program under grant agreement No.767901.
Guarantor
RO
