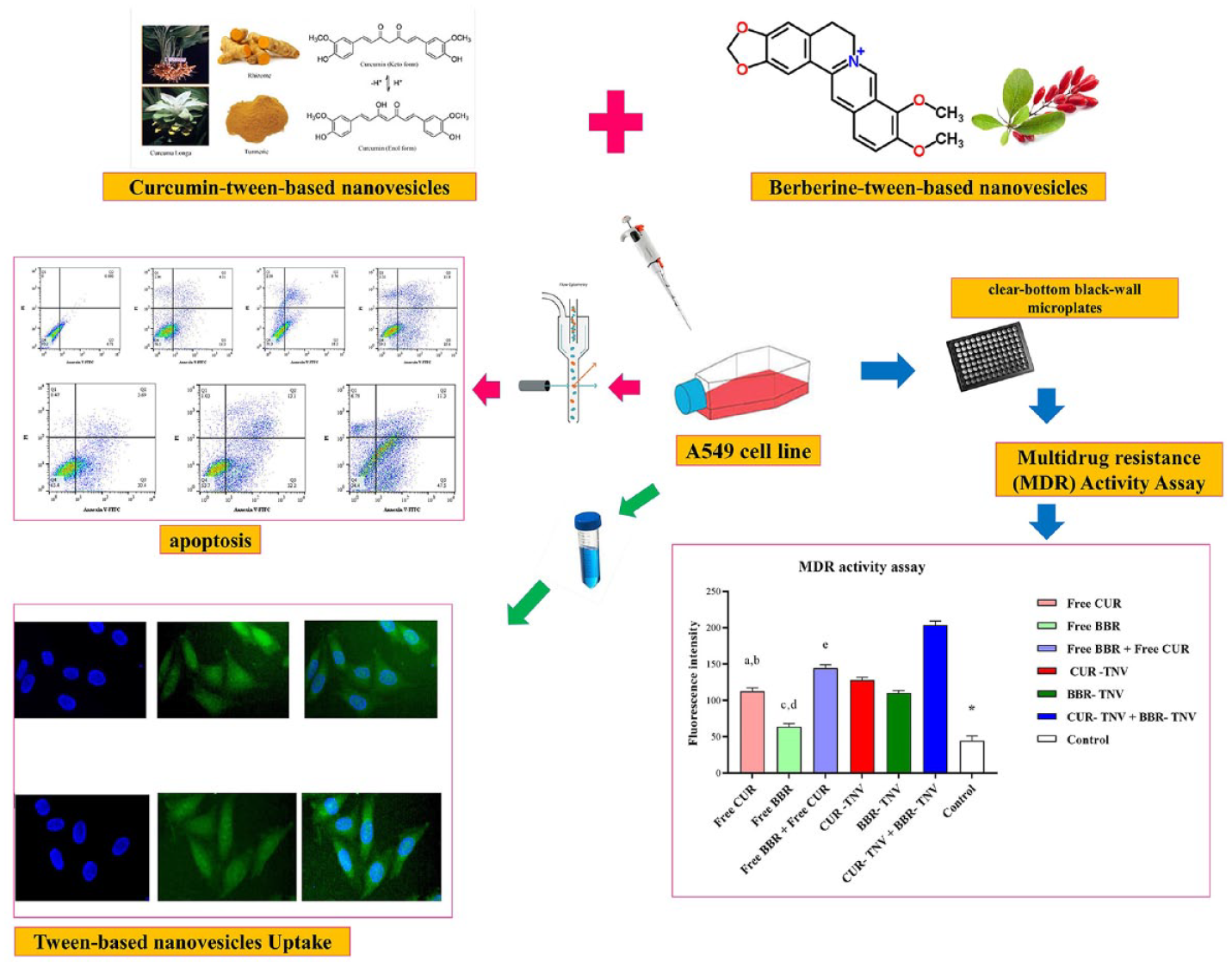
Abstract
Given the numerous adverse effects of lung cancer treatment, more research on non-toxic medications is urgently needed. Curcumin (CUR) and berberine (BBR) combat drug resistance by controlling the expression of multidrug resistant pump (MDR1). Fascinatingly, combining these medications increases the effectiveness of preventing lung cancer. Their low solubility and poor stability, however, restrict their therapeutic efficacy. Because of the improved bioavailability and increased encapsulation effectiveness of water-insoluble medicines, surfactant-based nanovesicles have recently received a great deal of attention. The current study sought to elucidate the Combination drug therapy by herbal nanomedicine prevent multidrug resistance protein 1: promote apoptosis in Lung Carcinoma. The impact of several tween (20, 60, and 80) types with varied hydrophobic tails on BBR/CUR-TNV was evaluated. Additionally, the MDR1 activity and apoptosis rate of the BBR/CUR-TNV combination therapy were assessed. The encapsulation effectiveness of TNV was affected by the type of tween. With the TNV made from tween 60, cholesterol, and PEG (47.5: 47.5:5), more encapsulation effectiveness was attained. By combining CUR with BBR, especially when given in TNV, apoptosis increased. Additionally, when CUR and BBR were administered in combination, they significantly reduced the risk of MDR1 development. The current work suggests that the delivery of berberine and curcumin as a combination medication therapy via tween-based nanovesicles may be a potential lung cancer treatment.
Introduction
The second most common cancer and the leading cause of cancer-related mortality globally is lung cancer. 1 Treatment choices for lung cancer are surgery and, later, adjuvant chemotherapy including primarily cisplatin along with mitomycin C, ifosfamide, and vinca alkaloids (vindesine, vinorelbine, and vinblastine), too etoposide, gemcitabine, and taxanes such as paclitaxel and docetaxel. Only 26% and 8% lung cancer’s patients are detected at stages I and II, respectively, whereas the patients with stage III and stage IV are diagnosed more as 28% and 38%, respectively. 2
There is hence an essential need for more research on lung cancer and non-toxic therapies.3,4 A unique strategy to stop or stop the spread of cancer using natural substances is known as cancer chemoprevention. A novel source of cancer chemoprevention has emerged from the phytochemicals found in herbs, such as berberine (BBR) and curcumin (CUR). By causing DNA damage, inducing apoptosis, and scavenging free radicals, these substances have the ability to stop the development and spread of cancer.5,6 The phytopolylphenols BBR and CUR, which were isolated from the roots of Rhizoma coptidis and Curcuma longa, respectively, are well known for their wide range of therapeutic effects, including those against atherosclerosis, cardiovascular disorders, inflammation, diabetes, and cancer. By reducing cell growth and inducing apoptosis, curcumin has anti-tumor effects on lung cancer.4,5,7 Multidrug resistant pump (MDR1), also known as P-gp, is an ATP-dependent transporter, and has an important role in the absorption and disposition of drugs such as anticancer drugs, antivirals, and antibiotics in cells. The different expression of MRP proteins has been linked to the poor prognosis and poor outcome of many cancer types; it is estimated that MDR accounts for 90% of cancer-related deaths in patients undergoing chemotherapy.8,9 Overexpression of MDR1 in lung cancer, reduces the efficacy of antitumor drugs by boosting the efflux of the drugs, resulting in shorter tumor-free survival and overall survival (OS) times in patients with lung cancer. 10
In order to reverse multidrug resistance (MDR) in human lung cancer, curcumin reduces the overexpression of the P-glycoprotein.11,12 Breast, ovarian, lung, prostate, liver, and leukemia are just a few of the tumors that berberine has anti-tumor effect against. By cell cycle arresting at the G0/G1 phase and inducing p53-mediated intrinsic apoptotic pathway, berberine may be able to slow the development and spread of lung cancer cells.13,14 The expression of proteins linked to drug resistance, such as multidrug resistance protein 1, is regulated by berberine, which has strong chemosensitizing properties that overcome drug resistance and victimize cancer cells to conventional chemotherapy drugs like cisplatin, doxorubicin, tamoxifen, and sunitinib.15,16 Berberine also modifies cell resistance pathways and increases the cytotoxic effects of cancer drugs. It’s interesting to note that combining berberine with curcumin is more effective at preventing the spread and growth of cancer cells in the breast, liver, lung, blood, and bone cancers. 17 However, the poor stability and relatively low solubility of berberine and curcumin under physiological conditions substantially restrict their therapeutic efficacy and bioavailability. 13 Due to their higher encapsulation efficiency, controlled release of both hydrophobic and hydrophilic drugs, biodegradability, biocompatibility, extended blood circulation, and ability to target a particular area, vesicular carriers used in drug delivery systems (DDS) have attracted a great deal of interest in recent years. Non-ionic surfactants in an aqueous phase self-associate to form molecular clusters known as surfactant-based nanovesicles. Because they are more biocompatible, biodegradable, and less toxic than liposomes, non-ionic surfactants -based nanovesicles have been used extensively in drug administration in recent years. In addition, surfactant-based nanovesicles offer many fundamental advantages over liposomes, such as better stability, relatively inexpensive surfactants, and ease of synthesis in a variety of chemical, industrial, and biological applications.18 –20 Non-ionic surfactants -based nanovesicles not only slow down the release of the loaded agents but also regulate the release rate and pattern via formulation the bilayer composition and cholesterol amount. These carriers can easily be prepared by the self-assembly of non-ionic surfactants in bilayer structure to form vesicles with or without cholesterol as an additive. Non-ionic surfactants with a high hydrophilic–lipophilic balance (HLB) such as tween are possibly to be micelle forming surfactants and require additives to obtain suitable molecular geometry and hydrophobicity for bilayer vesicle formation. Membrane additives are added to the formulas, so as to stabilize the non-ionic surfactants -based nanovesicles. The most common additive utilized in non-ionic surfactants -based nanovesicles is cholesterol, which is known to change the fluidity of the chains in the bilayer and, if present in adequate concentrations, to remove the gel to liquid phase transition, resulting in less leakiness of the vesicles. Cholesterol increases membrane micro viscosity and on-ionic surfactants -based nanovesicles stability.18,21 –24 Adding of hydrophilic polymers to non-ionic surfactants -based nanovesicles such as derivatives of poly ethylene glycol (PEG), could provide steric-stabilization of the nanovesicles, enabling them to achieve a prolonged circulating time without opsonizing, recognizing and clearing by the mononuclear phagocytic system, and as a result improve therapeutic efficacy.25 –27
In recent years, Maiti et al. demonstrated that the combined chemopreventive effects of berberine and solid lipid curcumin particles increased cell death more effectively than the effects of either compound alone in human cultured glioblastoma cells. 17 In order to determine if berberine and curcumin loaded in tween-based nanovesicles (TNV) formulations are effective in treating A549 human lung cancer cells, the following research was conducted: Combination drug therapy by herbal nanomedicine prevent multidrug resistance protein 1: promote apoptosis in Lung Carcinoma
Materials
Tween (tween 20, tween 60, and tween 80) was obtained from DaeJung Chemicals & Metals, South Korea. Cholesterol, berberine, curcumin, fluorescent label (Dil, 2-[3-(1,3-dihydro-3,3-dimethyl-1-octadecyl-2H-indol-2-ylidene)-1-propen-1-yl]-3,3-dimethyl-1-octadecyl-3H-indolium, monoperchlorate), phosphate-buffered saline (PBS), dialysis bag ((MW = 8–14 kDa), paraformaldehyde solution, and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Polyethylene glycol (DSPE-mPEG 2000) was obtained from Lipoid GmbH, Germany. Chloroform, methanol, and dimethyl sulfoxide (DMSO) were acquired from Merck Chemical Co. (Darmstadt, Germany). The A549 cell line was purchased from Pasteur Institute Cell Bank (Tehran, Iran). Medium RPMI-1640 (Dulbecco’s Adjusted Eagle Medium), trypsin-EDTA, Trypan blue, fetal bovine serum (FBS), DAPI (4′,6-Diamidino-2-phenylindole dihydrochloride), and penicillin/streptomycin (PS) 100 X were acquired from Gibco, ThermoFisher Scientific (Waltham, MA, USA). A fluorometric MDR assay (AB112142) was obtained from Abcam, Cambridge, United Kingdom. Annexin V-FITC flow cytometry kit was obtained from Affymetrix Biosciences, ThermoFisher Scientific.
Methods
Preparing BBR / CUR loaded tween-based nanovesicles
The thin film hydration technique was used to prepare tween-based nanovesicles formulas. Three kinds of nonionic surfactants (including tween 20, tween 60, and tween 80) were utilized to prepare BBR-loaded tween-based nanovesicles (BBR- TNV) and CUR-loaded tween-based nanovesicles (CUR- TNV). Synthesizing and screening were performed for BBR- TNV and CUR- TNV for controlled release, particle size, and higher entrapment efficiency parameters. For optimizing the results and determining the optimum circumstances, the tests were conducted as follows: (1) Evaluating the effect of various kinds of a tween with different hydrophobic tails on drug-loaded tween-based nanovesicles. (2) Five percent polyethylene glycol was added to the formulas for achieving long blood circulation and improving drug stability. To increment membrane rigidity, cholesterol was utilized as a stabilizer for TNV formulation as a result of its capability at cementing the leaking space in the bilayer membranes.22,28,29
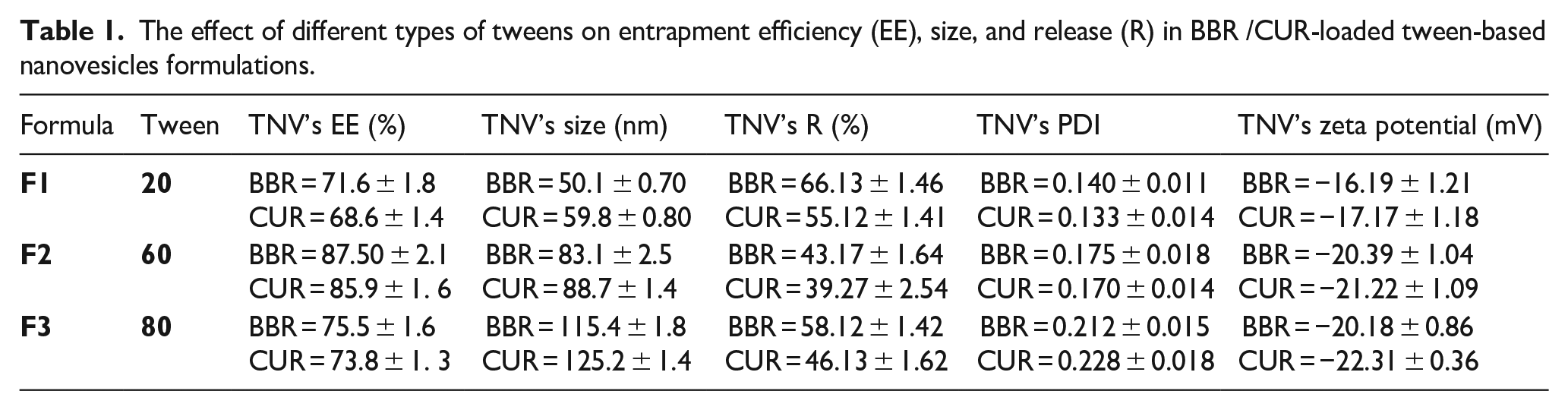
Accurately weighed quantities of lipid mixture including non-ionic surfactants and cholesterol (1:1 molar ratio, total weight of lipid mixture kept constant as 100 mg) and drugs (berberine and curcumin, 10 mg) were dissolved in chloroform/methanol mixture (2:1v/v). Then, the total mixture was transferred into a 500 mL round bottomed flask. The organic solvent was evaporated by rotary evaporator (Heidolph, Germany) at temperature of 50°C until the thin dry film was formed. The dried thin layer films were hydrated with 6 ml of PBS, pH 7.4, and the flask was kept rotating at 60°C with gentle shaking on a water bath. To sonicate the created formulas, a probe sonicator was used for half an hour for reducing the mean size of vesicles. Then, separation of free unloaded BBR and CUR from BBR-TNV / CUR- TNV was performed through a dialysis bag diffusion method against PBS at 4°C for 1 h (MW = 8–14 kDa). Table 1 represents the compositions of all formulations.18,22,29,30 Adding fluorescent label DIL to the lipid phase at 0.1% mole, the cellular uptake was evaluated.
The effect of different types of tweens on entrapment efficiency (EE), size, and release (R) in BBR /CUR-loaded tween-based nanovesicles formulations.
Determining the encapsulation efficiency
Spectroscopic measurements were performed for evaluating the drug release profile and the entrapment efficiency over time.31,32 UV-spectrophotometry technique was used to estimate the quantities of CUR and BER loaded into TNV formulas at 430 and 345 nm (λmax), respectively. BBR /CUR-loaded TNV was combined with isopropyl alcohol for allowing quick shedding of entrapped BBR / CUR and lysing of the membranes, hence, encapsulation efficiency is approximated. To estimate the drug encapsulation efficiency (EE %) equation (1) was used:
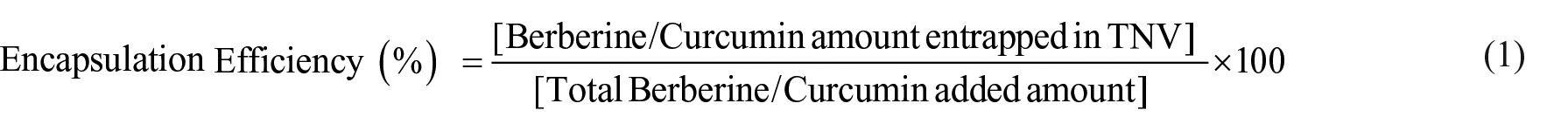
Particle diameter analysis
ZetaSizer Nano ZS device (Malvern Instruments Ltd, UK) was used to determine the mean diameter, polydispersity index (PDI) and zeta potential of tween-based nanovesicles formulas by the dynamic light scattering (DLS) technique. Light scattering was monitored at a 90° angles and a temperature of 25°C was maintained during the measurement. This device measures the particle size distributions in the range above 2 nm. A proper scattering intensity was generated by diluting all specimens (1:100) with deionized water before measurement to avoid multiple scattering events, due to it has a vital role to reduce antiparticle interactions. The polydispersity index (PDI) is a parameter to define the particle size distribution, which is expressed as dimensionless number. The value of Polydispersity Index may vary from 0.01 (mono dispersed particles) to 0.5 to 0.7, whereas, PDI Index value >0.7 indicated broad particle size distribution of the formulation. PDI can also show nanoparticle aggregation throughout the particle sample.33 –35
In vitro release assessment and drug release kinetics from tween-based nanovesicle formulations
Using the dialysis membrane technique, the berberine and curcumin release profiles from all TNV formulas were studied (F1–F4). The dialysis bag was used to wrap the TNV samples (MW = 8–14 kDa). Then, the dialysis bags were dipped in a phosphate buffer (comprising 2% Tween 80 for imitating a physiological setting). At various time intervals and up to 72 h, 3 ml were withdrawn and replaced by an equivalent volume of fresh medium. To determine the quantity of released BBR and CUR, the UV-Visible spectrometer was used at 345 and 430 nm, respectively. To achieve the kinetics of release, the release kinetic models were tailored (0, 1st, 2nd order, Higuchi model, and Hixson-Crowell model) based on our former studies by comparison of the values of correlation coefficients for selecting the best-fitted release model18,22,29
In vitro cytotoxicity study by the MTT assay
The in vitro cytotoxicity of the free “berberine/curcumin” and BBR-TNV/CCUR-TNV against an A549 non-small-cell lung cancer was determined by the MTT assay. Trypsinizing and seeding were performed for exponentially growing cells in 96-well plates at 10,000 cells per well. The cells were exposed to serial concentrations, including free-BBR solution, free-CUR solution, free-BBR + free-CUR physical mixture, BBR-TNV, CUR-TNV, and the co-administration of BBR-TNV + CUR-TNV. Then, the MTT technique was used to determine the treated cells’ viability, as follows: For removing the media, the cells were incubated with 200 mL of 5% MTT solution per well, and the dye was metabolized into colored-insoluble formazan crystals at 37°C for 4 h. The formazan crystals were then dissolved in 180 mL/well DMSO at room temperature for 30 min while shaking continuously. To measure absorbance at 570 nm, an Epoch Microplate Reader (Biotech, USA) was used.
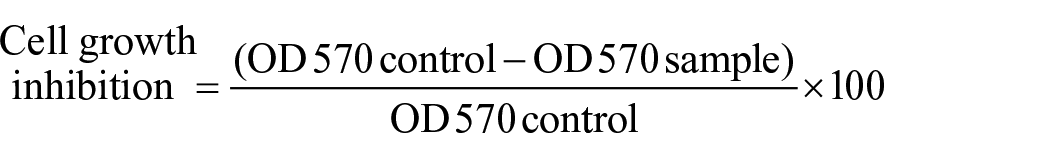
All the experiments were performed in triplicate. The cells treated with a culture medium were utilized as a control. The cytotoxicity of CUR and BBR alone and in combination was expressed as IC50. This is defined as the drug concentration required to inhibit cell growth by 50% relative to the control. These values were calculated by nonlinear regression analysis of the response curves via GraphPad Prism 6. The cell growth inhibition on OVCAR-3 cells was calculated as follows:
The Combination Index (CI) value was calculated for appraising combined CUR and BBR. This index was obtained by Chou and Talalay’s equation using CompuSyn software:

Where a is the IC50 of BBR combined with CUR at concentration b, A is the IC50 of BBR without CUR, and B is the IC50 of CUR without BBRL. According to Chou and Talalay’s equation, when CI < 1, the interaction of two drugs is synergistic; when CI = 1, the interaction of two drugs is additive; and when CI > 1, the two drugs are antagonistic18,22,29
Nanocarrier uptake studies
An investigation of in vitro cellular uptake of the BBR-TNV and CUR-TNV optimal formulations was performed on seeded A549 cells at 100,000 cells/well. The cells were treated with DIL-labeled CUR-tween-based nanovesicles (DIL- CUR-TNV) and DIL-labeled BBR-tween-based nanovesicles (DIL- BBR-TNV) while incubating for 3 h. A549 cells were then washed three times with PBS and fixed with paraformaldehyde solution. Ultimately, incubation of the cells was performed with a DAPI solution for 15 min, and then imaging was performed with a fluorescence microscope18,31
Evaluation of Annexin V-fluorescein isothiocyanate (FITC) and propidium iodide (PI)
The induction of apoptosis in A549 cancer cells was determined via flow cytometry assay. The cells were cultured (105 cells per well for 24 h) and treated to the IC50 concentration of free-BBR, free-CUR, free-BBR + free-CUR physical, BBR-TNV, CUR-TNV, and the co-administration of BBR-TNV + CUR-TNV for 48 h. Then, the cells were trypsinized and washed twice with cold PBS. The cells were then exposed with an apoptosis quantitation kit to stain the cells with annexin V-FITC and propidium iodide. Annexin V-FITC binds to the membrane phospholipid phosphatidylserine on the surface of apoptotic cells and emits green fluorescence. Propidium iodide (PI) is a fluorescent intercalating agent that penetrates the dead cells, emitting red fluorescence. PI is useful to differentiate necrotic, apoptotic, healthy, and dead cells. Finally, the apoptosis rate of A549 cancer cells was obtained using the BD FACSCalibur flow cytometer (D Biosciences, USA) and analyzed by FlowJo software (FlowJo LLC, Ashland, OR, USA) to determine the percentage of apoptotic and necrotic cells in each sample. In the plot of flow cytometry, Q4, Q3, Q2, and Q1correspond to live cells (Annexin V-FITC-/PI-), early apoptosis (Annexin V-FITC+/PI-), late apoptosis (Annexin V-FITC+/PI+), and necrotic cells (Annexin V-FITC-/PI+), respectively. The cells not receiving any drug treatment were considered the control33,36,37
Multidrug resistance (MDR) activity assay
A fluorometric MDR assay was performed to assess the synergistic impacts of curcumin and berberine on Multidrug resistant pump (MDR1) activity. Thus, the activity of MDR1 was determined utilizing a kit (AB112142, Abcam, Cambridge, United Kingdom). A fluorescent MDR indicator assayed these pumps’ activity in this kit. This hydrophobic fluorescent dye molecule rapidly penetrates cell membranes and becomes trapped in cells. After a short incubation, the intracellular free dye concentration significantly increased. This dye is extruded in the MDR1-expressing cells via the MDR transporters; therefore, the cellular fluorescence intensity is reduced. Nevertheless, its cellular fluorescence intensity significantly increases by blocking its extrusion via an agent such as curcumin or berberine that interferes with the MDR1 pump activity. Seeding A549 cells into clear-bottom black-wall microplates, they were then incubated for 24 h and treated with drug- or tween-based nanovesicles formulations, including free CUR solution, free BBR solution, free BBR + free CUR physical mixture, BBR-TNV, CUR-TNV, and co-administration of BBR-TNV + CUR-TNV. The cells not receiving any drug treatment were considered controls. Then, adding 100 l of MDR dye-loading solution to each well, incubation was performed at room temperature for 1 h in darkness. To detect the cells’ fluorescence intensity, an EPOCH Microplate spectrophotometer (Synergy HTX, Biotech, USA) was used at an emission wavelength of 525 nm and an excitation wavelength of 490 nm. The intensity of fluorescent dye inside cells was detected through the assay. The MDR (pump) activity was indicated by a reduction in the dye intensity38 –40
Statistical analysis
GraphPad Prism 6 software was used to perform statistical data analyses, which were expressed as mean ± SD. The two independent groups and multiple samples were compared using a student t-test and an ANOVA test, respectively. The p-value of less than 0.05 was significant.
Results
Effects of various kinds of surfactant on BBR /CUR loaded tween-based nanovesicles
To find the best BBR/CUR-TNV formulation for achieving better entrapment efficiency, controlled release (pH 7.4 and temperature of 37°C), and small vesicle size, various formulations were evaluated (Table 1). The stability of tween-based nanovesicles was enhanced by adding cholesterol (1:1 molar ratios) to the formulation, which functions as a rigidifying agent.41,42 The effectiveness of encapsulating curcumin and berberine was found to be enhanced by increasing the hydrophobic chain of the used tween (Table 1). Formula utilizing tween 60 (equation (2)) achieved the most encapsulation efficiency among all formulations. Despite possessing the same alkyl chain with tween 60, tween 80 had a lower encapsulation efficiency than tween 60. Therefore, we selected the formula 2 for further analysis. Table 2 shows the effect of PEG on entrapment efficiency, size, and release in equation (2). As shown in this Table, the F4 formula with 5% PEG had a smaller diameter, higher drug entrapment, lower drug release, and a smaller polydispersity index (PDI) than the F2 formula. In terms of greater encapsulation effectiveness and sustained drug release, F4, which consists of tween 60 and cholesterol: PEG (47.5:47.5:5 molar ratios), has chosen as the preferred formulation for further studies.
The effect of PEG on entrapment efficiency, size, and release in BBR/CUR-loaded tween-based nanovesicles formulations.
In vitro release assessment and drug release kinetic from tween-based nanovesicles formulations
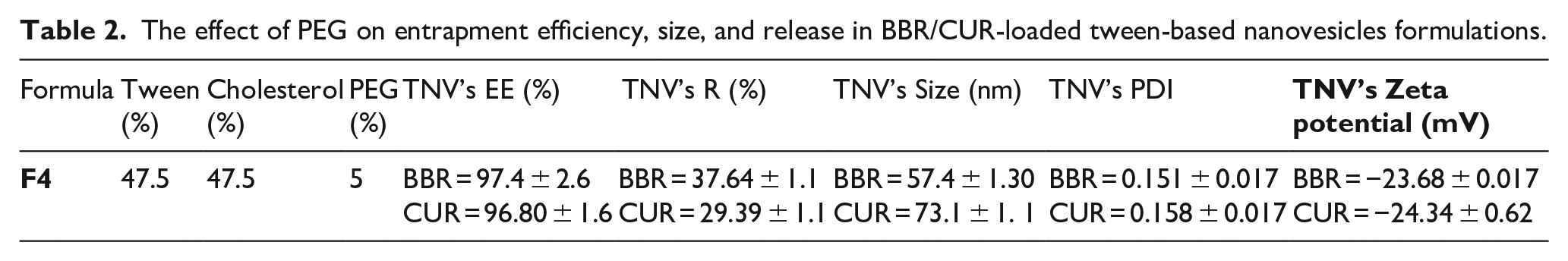
Figure 1 displays the outcomes of a 72-hour release profile of CUR and BBR from TNV in PBS (pH 7.4) at 37°C. In the ideal formulation (F4) for CUR and BBR, respectively, about 29.39% and 37.64% of the loaded medicines were released. Evidently, CUR and BBR’s release profiles were biphasic, with the faster release phase coming first, followed by a slower release phase. For the BBR/CUR-loaded TNV, the correlation coefficient (R2) is shown in Table 3. The higher Regression coefficient (R2) showed that the CUR and BBR releases from the TNV equations are best matched to Higuchi’s correlation, according to the data. The mathematical analysis of the BBR/CUR release data included zero-order, Hixson-Crowell, first-order, and Higuchi’s equations, as shown in Figure 2.
The in vitro release profile of berberine and curcumin from tween-based nanovesicles optimum formula.
Release kinetics data of berberine and curcumin from tween-based nanovesicles optimum formula.
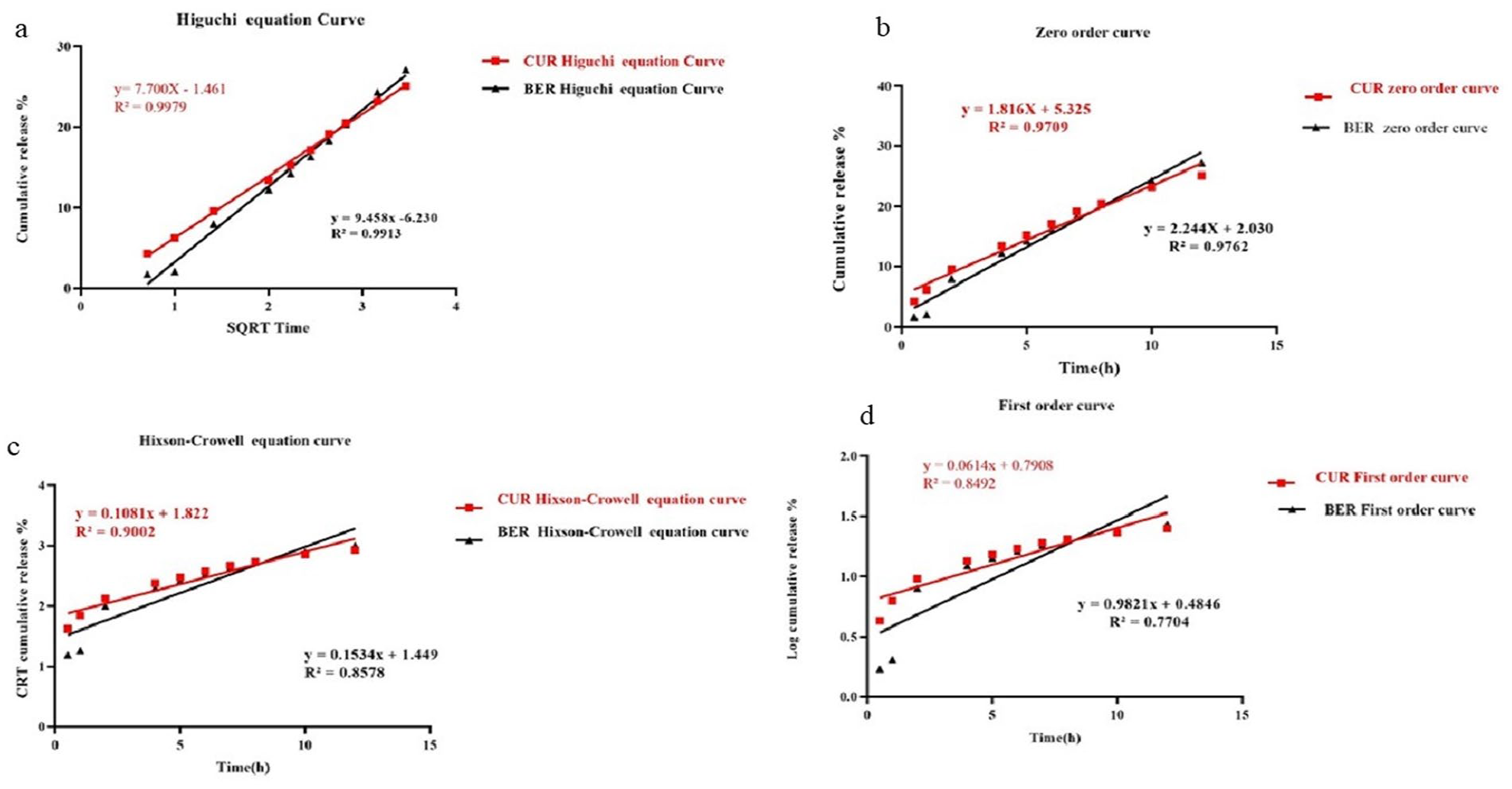
Berberine and Curcumin comparative plots. (a) Higuchi (SQRT) release kinetics, (b) Zero order release kinetics, (c) Hixson–Crowell model, and (d) first order release kinetics.
Cell viability was further decreased by curcumin and berberine combination drug therapy, especially when administered in tween-based nanovesicles formulations
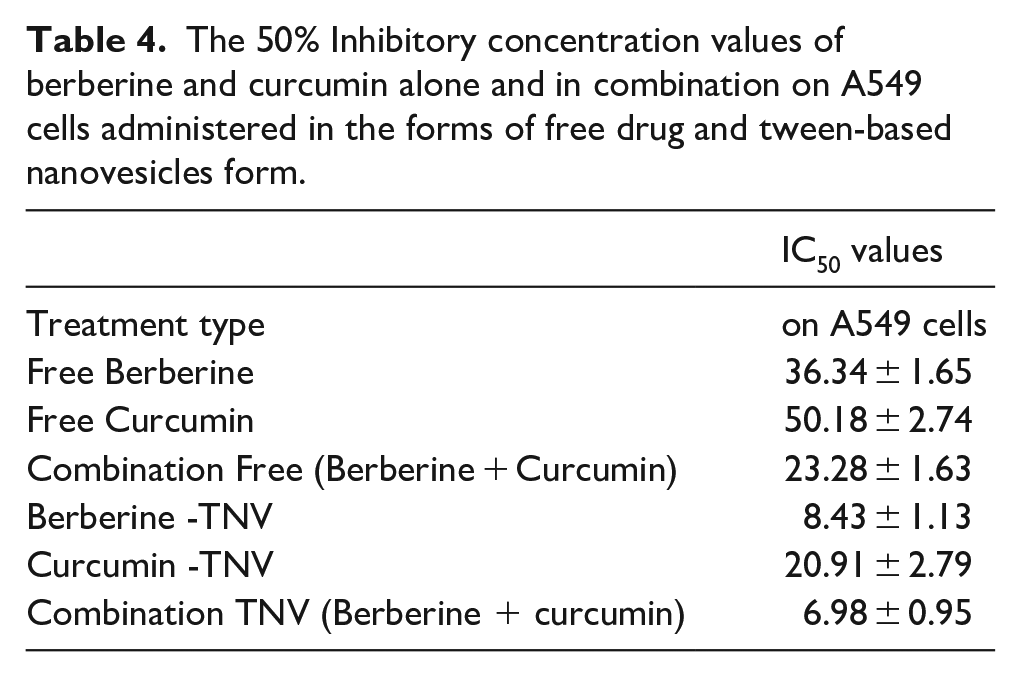
On A549 cells, the following were found to be the inhibitory effects of each curcumin and berberine as free compounds and tween-based nanovesicle formulations: Curcumin and berberine dose-response experiments using the MTT test were initially performed. The free-form and TNV formulations individually inhibited the proliferation of A549 cells in a dose-dependent manner, as seen in Figure 3. The IC50 values for various compounds are evaluated in Table 4. In delivering the medicines CUR and BBR to A549 cells, tween-based nanovesicles were extremely effective (Figure 3(b)). When CUR and BBR were administered to the A549 cell in TNV form as opposed to free CUR and free BBR solutions, the concentrations of the drugs were found to be reduced by 2.4 and 4.3-fold, respectively (Table 4). Then, the IC50 concentrations were utilized to create the fixed ratios for succeeding combination experiments and to determine the combination index (CI). The results of the combination research used to determine the synergistic anticancer effects of curcumin and berberine are shown in Table 5. Figure 4(a) and (b) show the dose-response curves for the combined therapy with curcumin and berberine on the A549 cell line. In A549 cells, a combination of curcumin and berberine therapy resulted in a greater reduction in cell viability than either compound alone. When the CUR and BBR were administered in TNV formulations instead of a free solution, the combined treatment regimen was considerably more effective (p-value of less than 0.05) (Table 4). Consequently, the therapeutic potential was increased by combining CUR and BBR. The combination index analysis of the CUR and BBR interactions in the A549 cell is shown in Figure 4(c) and (d). Curcumin and berberine were combined in both free and TNV forms, and the CI values were both less than one, demonstrating the two medicines’ synergistic effect in reducing cell growth (Table 5; Figure 4(c) and (d)).
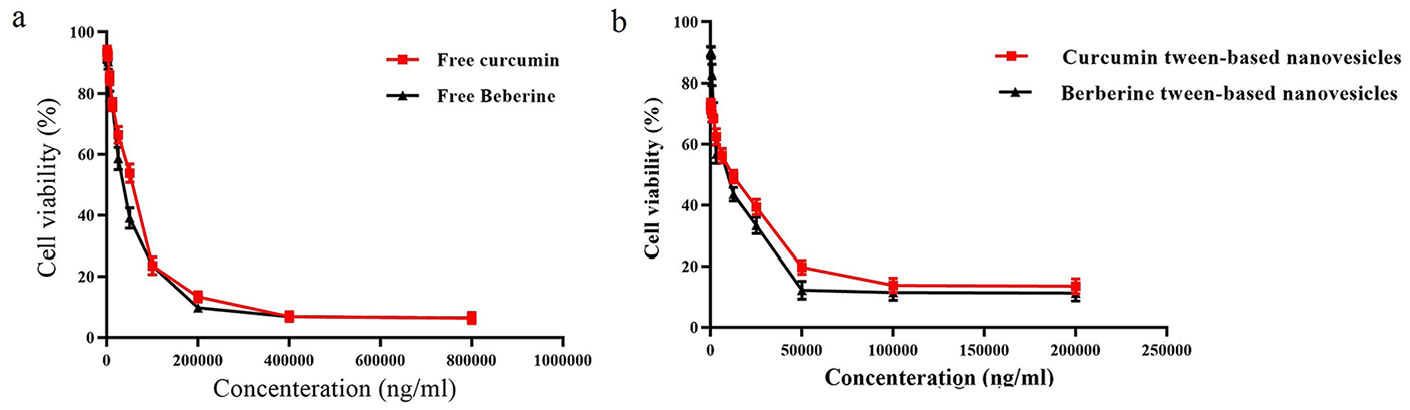
Inhibition of cell growth by berberine and curcumin individual as a drug-free form and drug tween-based nanovesicles form in A549 cell. (a) Free BBR versus Free CUR, (b) BBR - tween-based nanovesicles versus CUR - tween-based nanovesicles.
The 50% Inhibitory concentration values of berberine and curcumin alone and in combination on A549 cells administered in the forms of free drug and tween-based nanovesicles form.
Berberine and curcumin combination index against A549 Cell. CI < 1, synergistic; CI = 1, additive; CI > 1, antagonistic.

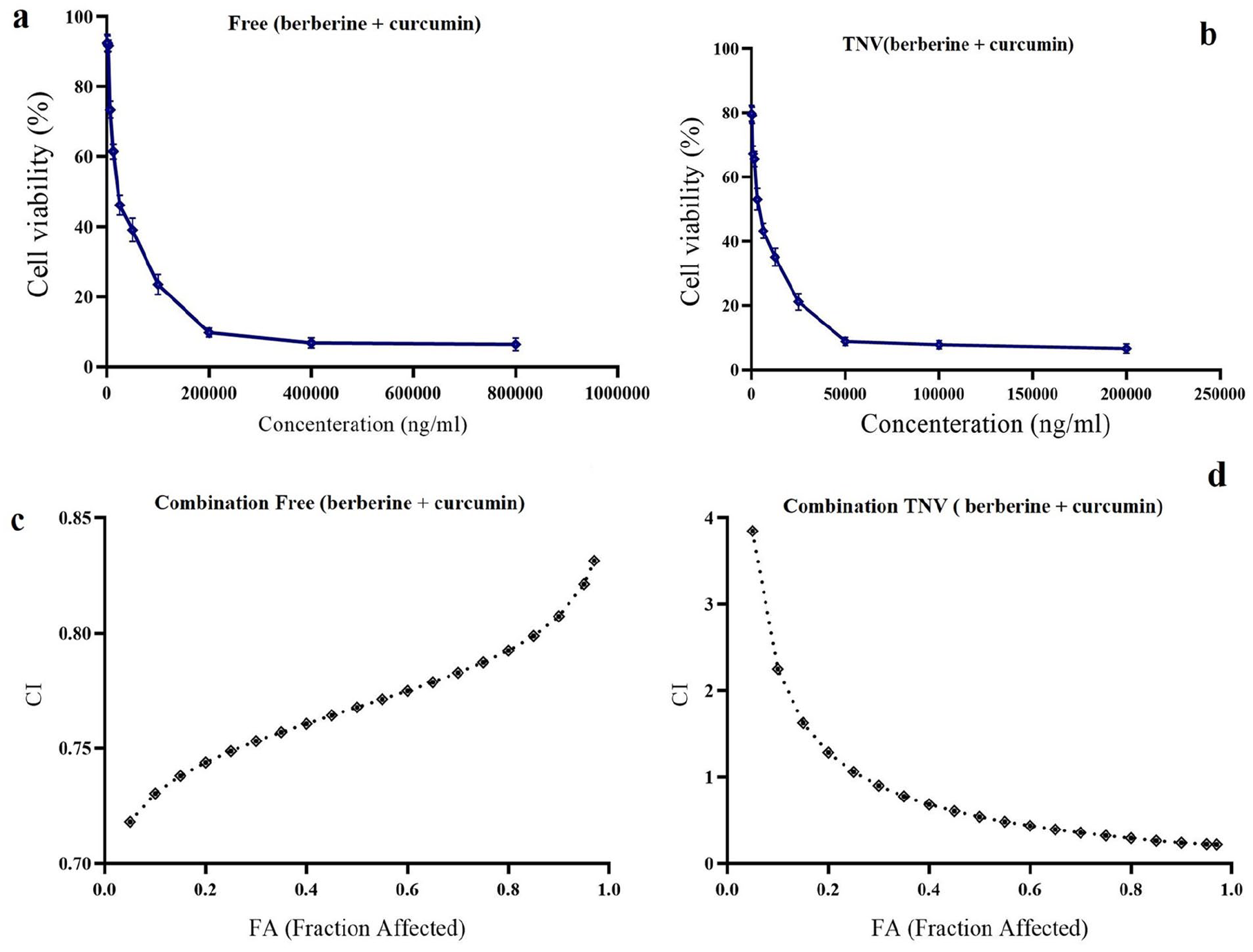
Analysis of synergy between berberine and curcumin for A549 cell. (a) Dose–response curve of free BBR + Free CUR, (b) dose–response curve of TNV (berberine + curcumin). CI values at different levels of growth inhibition effect (fraction affected; FA) (c) “Combination Free (berberine + curcumin),” and (d) Combination TNV (berberine + curcumin) in A549 cell).
Nanocarrier uptake studies
A549 cells that had been treated with DIL- CUR-TNV and DIL- BBR-TNV were examined using fluorescence microscopy to see how they were absorbed by the cells. The tween-based nanovesicle formulations effectively infiltrated the cancer cells, mostly into the nucleus, as shown in Figure 5(a) and (b). The entry of the drug-loaded tween-based nanovesicles into the cells appears to be greatly aided by endocytosis.
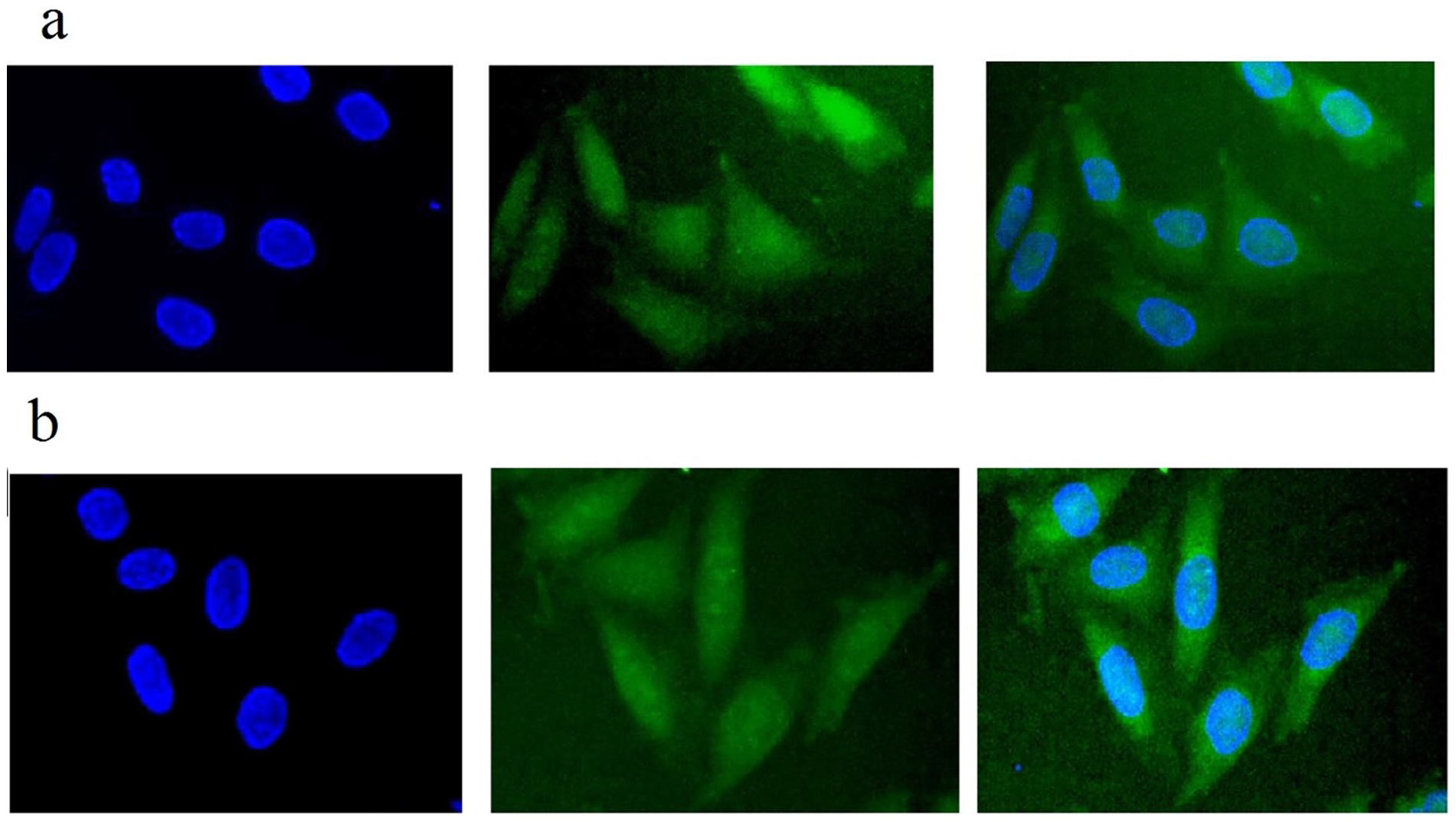
Cellular uptake images of A549 cells, were incubated with DIL-labeled CUR- tween-based nanovesicles and DIL-labeled BBR- tween-based nanovesicles for 3 h. (a) DIL- CUR- TNV and DIL- BBR- TNV, (b) DIL- BBR- TNV. DAPI stain (blue) was used for nucleus staining and DIL dye (green) was used for surfactant staining.
Combination drug therapy of curcumin and berberine demonstrated an augmentation in apoptosis rate than single-agent chemotherapy, particularly when administered in tween-based nanovesicles formulation
It was determined whether A549 cells had undergone apoptosis after being exposed to CUR and BBR alone or in combination (Figure 6). In cells exposed to free CUR or free BBR, early apoptosis occurred at a rate of 10.7% and 15.5%, respectively. When the cells were treated with CUR-TNV or BBR-TNV, however, the early apoptotic rate increased to 20% and 28%, respectively. Additionally, apoptosis was increased by combining curcumin and berberine compared to single agent treatment, especially when the drugs were given in drug TNV form (20% early apoptosis in free curcumin + free berberine solution vs 27.5% early apoptosis in CUR-TNV + BBR-TNV, p 0.05). The outcomes are consistent with the cytotoxicity test conducted on berberine and curcumin.
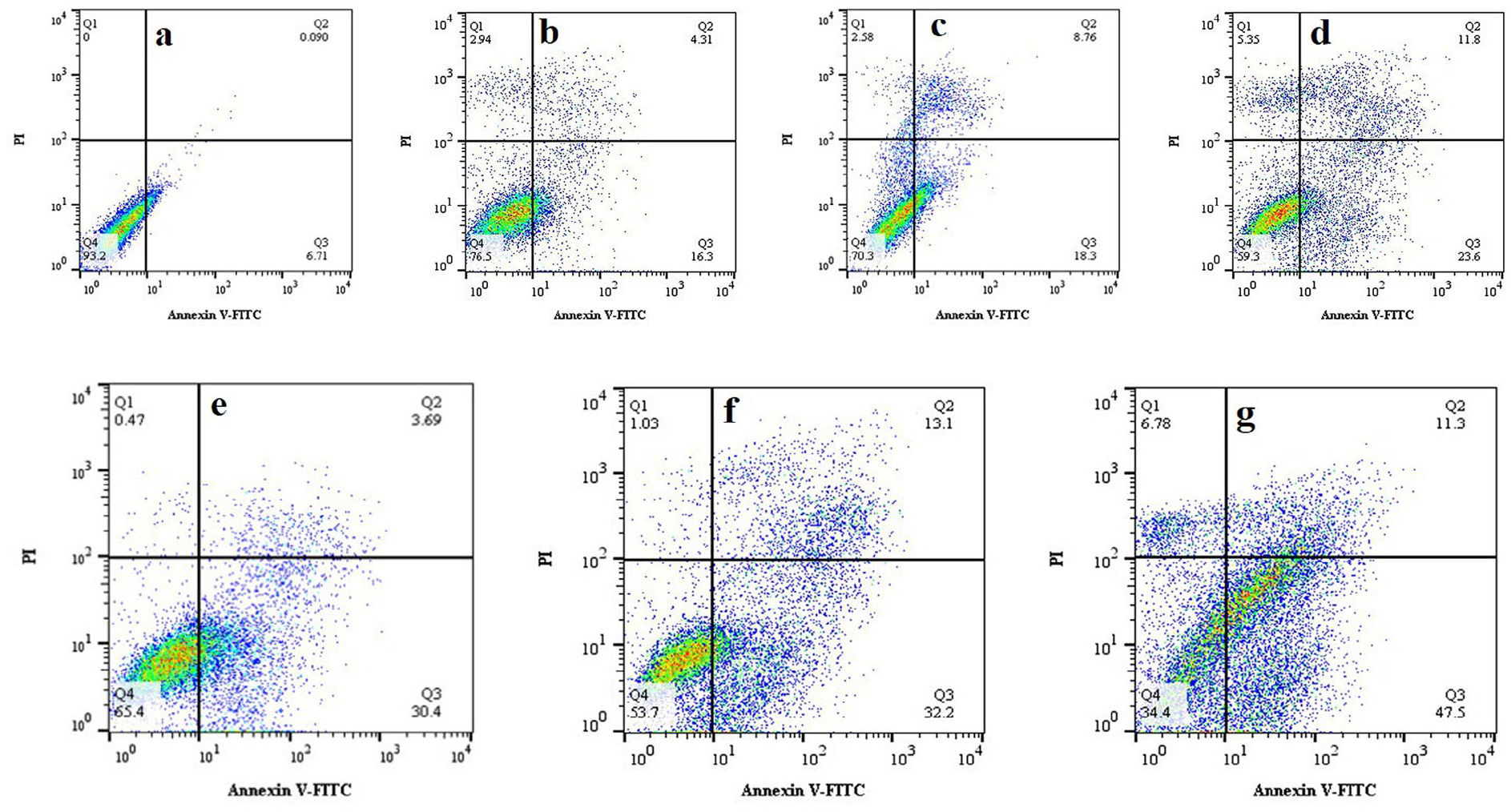
Cell apoptosis was analyzed with Annexin V-FITC kit following the treatment of cells with BBR and CUR, either as single agents or as a BBR + CUR combination, administered in aqueous solution or in the tween-based nanovesicle formulations. (a) Control, (b) Free Curcumin, (c) Free berberine, (d) Free Curcumin + Free berberine, (e) CUR-TNV, (f) BBR-TNV, and (g) “BBR-TNV+ CUR-TNV.”
Combination drug therapy of curcumin and berberine, compared to the individual treatments, caused further reduced MDR1 activity particularly when administered in tween-based nanovesicles formulations
Using a fluorometric MDR assay, it was determined whether curcumin and berberine had any additive effects on MDR1 activity. The intracellular intensity of the fluorescent P-gp substrate in A549 cells increased in comparison to the control group due to free curcumin and free berberine’s ability to inhibit the function of MRP and MDR1 pump (Figure 7). In comparison to the untreated control, curcumin and berberine respectively boosted intracellular fluorescence intensity by 2.54 and 1.44-fold. However, after giving the cells treatments with CUR- TNV and BBR- TNV, respectively, the intracellular fluorescence intensity was increased to 2.9 and 2.5-fold. Additionally, CUR and BBR in combination with chemotherapy have been shown to be more effective than single-agent chemotherapy at reducing MDR1 activity. Additionally, this combination drug therapy trend was significantly more effective (****p-value less than 0.0001) when curcumin and berberine were delivered in TNV formulations as opposed to free drug solution (3.26-fold increase in intracellular fluorescence intensity in free curcumin + free berberine solution as opposed to 4.6-fold increase in intracellular fluorescence intensity in CUR- TNV + BBR- TNV). Apoptosis assay and cytotoxicity tests have supported these findings.
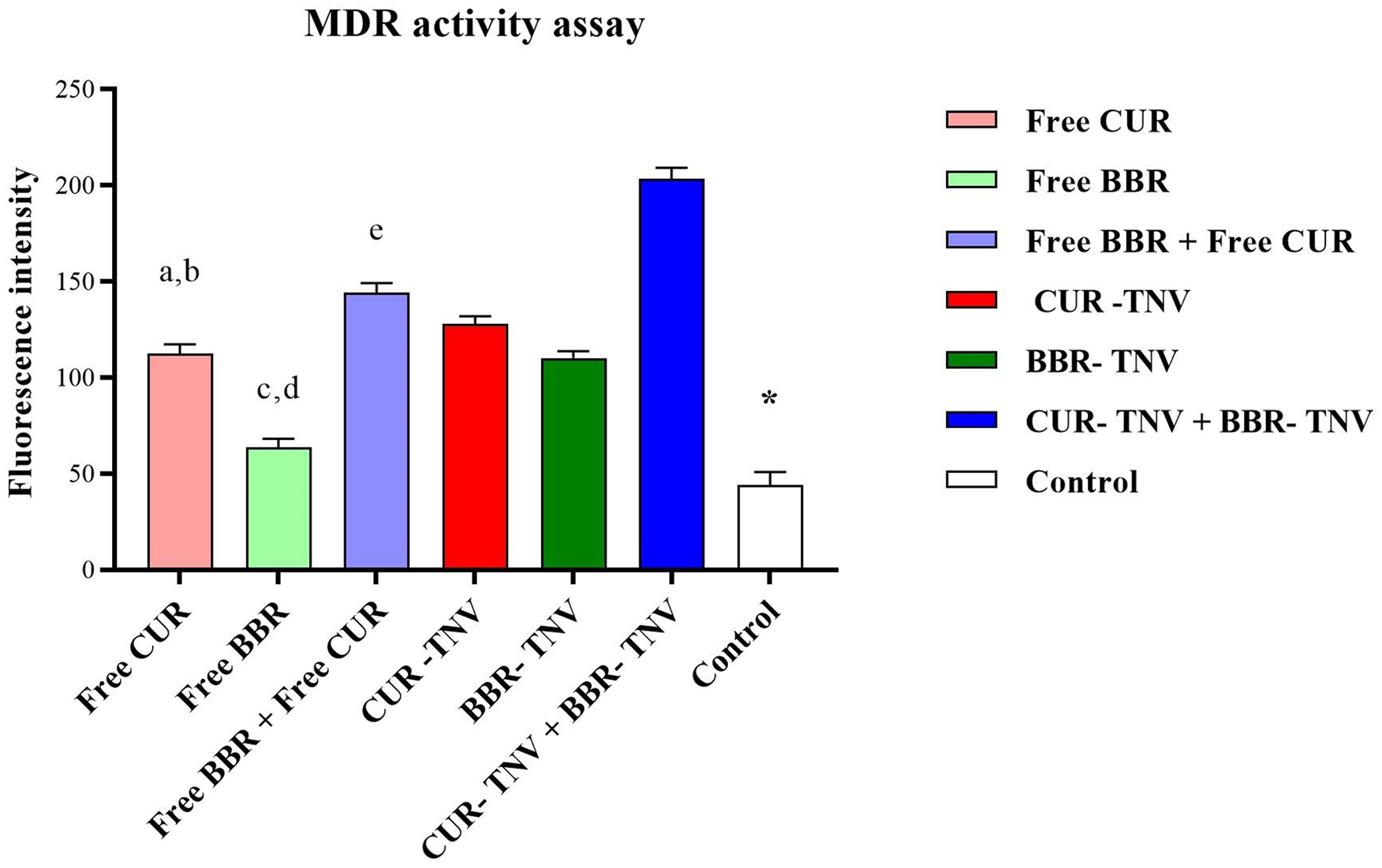
A549 cell was treated with berberine and curcumin either as a single agent or in combination when administered in aqueous solution and TNV formulations, and then P-gp activity was determined using a fluorometric MDR assay kit. *P < 0.001 versus control. (a) Free CUR versus “Free BBR+ Free CUR,” (b) Free CUR versus CUR-TNV, (c) Free BBR versus “Free BBR + Free CUR,” (d) Free BBR versus BBR-TNV, and (e) “Free BBR + Free CUR” versus “BBR-TNV+ CUR-TNV.”
Discussion
For both men and women, lung cancer continues to be the leading cause of mortality. More than 50% of patients with advanced non-small cell lung cancer get chemotherapy, which frequently fails. 43 According to the definition of cancer chemoprevention, it is a novel method for suppressing or reversing the development of cancer using natural substances like berberine and curcumin. The idea of chemoprevention has now been expanded to include all stages of cancer development, including cancer initiation and progression. Since phytochemicals from herbs have emerged as a novel source for cancer chemoprevention and as a chemotherapy medication adjuvant for reducing cancer initiation and progression via free-radical scavenging, apoptosis, and DNA damage,5,43,44 this has gained more attention. In this regard, BBR and CUR have demonstrated strong anti-cancer activity in a variety of malignancies. Each of the chemicals may also increase the sensitivity of the cancer cells to standard chemotherapy treatments. It is intriguing to examine the exertion of synergistic chemopreventive effects by combining renowned natural compounds, in particular BBR and CUR, on lung cancer cells because of their promising anticancer activities, distinctive chemical properties, multiple targets, rich resources, and low toxicity.6,17 BBR and CUR have broad pharmacological effects and are safe, but their instability, extremely low water solubility, quick metabolism, and limited oral bioavailability make it difficult to use them in clinical settings. Numerous studies have focused on a wide range of noteworthy and prospective nanocarriers for effective and prolonged BBR and CUR distribution, including micelles, conjugates, liposomes, nanoparticles, solid dispersions, and tween-based nanovesicles.45-47 For the encapsulation of hydrophobic pharmaceuticals, tween-based nanovesicles are preferable to phospholipid vesicles due to their greater encapsulation effectiveness, biodegradability, biocompatibility, less expensive manufacture, acceptable stability, simple storage, and lack of organic solvents.18,48 In order to increase the efficacy of A549 human lung cancer cells, the combination delivery of CUR and BBR, enclosed in novel tween-based nanovesicles, was investigated in this study. On BBR/CUR-loaded TNV formulations, the impact of several tween types with differing hydrophobic tails was assessed. In Table 1 was showed that the efficacy of curcumin and berberine encapsulation was increased with increment the hydrophobic chain of the used tween. Due to the poor interaction between the hydrophobic acyl chain of tween 20 and BBR/CUR, the tween 20-acyl chain (F1 vs F2) had a minor impact on the entrapment efficiency of BBR/CUR. The plausible assumption for this higher entrapment was the length of the C-H chain, which is 18 in tween 60versus 12 in tween 20. Longer hydrophobic chains made it possible to trap the hydrophobic BBR and CUR more effectively than when they were combined with TNV formulations. 24 In contrast to tween 60, tween 80 had a poorer encapsulation efficiency despite having the same alkyl chain. The addition of double bonds makes the oleate alkyl chain more pliable to rotation and bending, which is the cause. By rotating in this way, the steric barrier between the hydrophobic chains would inevitably rise, making it harder for BBR and CUR molecules to cling tightly to the oleate chain. Additionally, the hydrophobic obstruction makes the self-assembled tween-based nanovesicles leakier, which also explains the reduced encapsulation effectiveness of the tween 80 formulations.24,49 Consequently, the formulas using tween 60 had the highest encapsulation efficiency (equation (2)) with encapsulation efficiencies of 85.9% and 87.50% for CUR and BBR, respectively.
PEGylation in the TNV formulations results in better stability, higher drug encapsulation, a smaller mean diameter, and lower drug release.55,56 Therefore, it was thought to modify the F2 formula by 5% PEG. According to the findings, the F4 tween-based nanovesicles formula displayed more drug entrapment, a smaller diameter, lower drug release, and reduced PDI when compared to the F2 formula.
To create the best system with the desired drug-release profile, the drug release rate from a delivery system must be analyzed. The function of a delivery system in the ideal situation was predicted using in vitro release analysis, presumably presenting some signs of its in vivo effectiveness. The cumulative release profile of CUR and BBR appeared to be biphasic, with an early fast release period followed by a later release phase, according to in-vitro drug release. The released CUR and BBR were easily emptied out of the dialysis bag because CUR and BBR are tiny molecules and the permeability cut-off was 8–14 kDa. As a result, neither the dialysis bag nor the drug’s size limited their release. The diffuse mechanism (concentration gradient of BBR/CUR between buffer and tween-based nanovesicles) controlled the initial fast-release rate.18,28,31,50,51 The cumulative drug release was fitted into mathematical release models that are frequently used for understanding release kinetics and comparing release patterns in order to assess the in vitro release of CUR and BBR from the TNV formulation. The BBR/CUR-TNV formulations were developed in response to the Higuchi model. These findings demonstrated that CUR and BBR molecules were dispersed inside the matrix of tween-based nanovesicles. Additionally, no potential interactions between the medicines and the tween-based nanovesicles components were discovered.52,53
In the current study, the impact of CUR and BBR combination therapy on A549 cells was examined in both free and tween-based nanovesicle forms (Tables 4 and 5). The combined anti-cancer effects of CUR and BBR dramatically reduced the proliferation of A549 cells. The cytotoxic effects became more obvious when the free medicines were administered in TNV formulations.
Maiti et al. and colleagues assessed the co-treatment effects of the solid lipid curcumin particles (SLCPs) and berberine on cell viability’s glioblastoma cell lines (U-87MG and U-251MG). They found that SLCP + BBR induced ~50% cell death after 24 h in U-87MG cells and 42% in U-251MG cells. 17
Lee et al. formulated curcumin (Cur) and quercetin (Que) nanoparticles and investigated their synergistic effect on lung carcinoma (A549) cells. The IC50 values for Cur-NPs alone, Que-NPs alone, and “Que-NPs
In contrast, in our study, 1.2-fold and threefold reductions in concentration were noted when BBR and CUR were administered in “BBR-TNV+ CUR-TNV” form, respectively, compared to BBR-TNV alone and CUR-TNV alone. Therefore, Lee formula was more effective in reducing the concentration of curcumin and quercetin than the formula developed by our team.
Similarity our study, the synergistic chemopreventive effects of berberine and doxorubicin have been reported by Gupta et al. on MDA-MB-231 and T47D. Gupta et al. studied the co-delivery berberine and doxorubicin through poly (lactic-co-glycolic acid) (PLGA) nanoparticles (PDBNP; Berberine loaded PLGA-doxorubicin nanoparticles) against MDA-MB-231 and T47D breast cancer cell lines. PDBNP showed significant anti-proliferative with IC50 of 1.94 and 1.02 μ M, for MDA-MB-231 and T47D respectively, which was significantly better than both the berberine and doxorubicin solutions. 45
The combined chemopreventive effects of BBR and CUR on human breast cancer cells were also reported by Wang et al. 6 Additionally, Balakrishna and Kumar discovered that CUR and BBR had a synergistic anti-cancer action on a variety of cancer cells, including liver, lung, breast, glioma, bone, and leukemia cell lines. 55
The tween-based nanovesicle formulas were effectively swallowed in A549 cancer cells, as shown by the qualitative cellular uptake study (Figure 5). Tween-based nanovesicles appear to carry the payload to subcellular locations within the cell effectively. Additionally, in A549 non-small cell lung cancer, the accumulation of berberine and curcumin in the nucleus could successfully limit cell proliferation and promote apoptosis, as obvious from the presence of green dots in nucleus of cells. 56 This event is attributed to uptake mechanism of BBR/CUR -TNV via endocytosis that sustained the continuous bioavailability of BBR/CUR -TNV formulations in A549 cell. 57 This result has been confirmed in a study by Rohanizadeh et al., whereby the curcumin micellar nanoparticles were fabricated and cellular uptake was evaluated against A549 cell.
This result has been confirmed in a study by Rohanizadeh et al., whereby the curcumin micellar nanoparticles (Cur-NP) were fabricated and cellular uptake was evaluated against A549 cell. The Cur-NP was efficiently internalized into A549 cell and were localized in membrane and cytoplasm. 57 Numerous findings have showed that BBR / CUR nanoparticles improved cellular uptake in many cancers cell lines. The improved intracellular uptake of BBR/CUR-TNV formulations in our study might be due to their nanosized vesicles. Nanoparticles with a small size (<100 nm) possess the potential to easily get to the cell and even nuclear membrane, thereby accumulate in the nucleus of the cell. Therefore, through improved permeation and retention effects, small nanoparticles are useful for the passive targeting of cancer tissue.57,58
Additionally, the antioxidant properties of berberine and curcumin allow them to combat free radicals in cancer cells.59,60 When curcumin and berberine were enclosed in TNV formulations as opposed to the medications’ free forms, A549 cells showed a higher rate of cellular death (p 0.05). In addition, compared to single-agent chemotherapy, the combination medications BBR-TNV and CUR-TNV showed the highest rate of apoptosis in lung cancer cells. There have been numerous attempts to reestablish the chemosensitivity of the chemotherapeutic drugs. Following that, Rudy Juliano et al. (1976) investigated P-gp, a membrane glycoprotein that alters membrane permeability to chemotherapeutics. 61 P-gp has been studied extensively over the past 40 years, interacting with over 300 different substances. P-gp function can be inhibited by a variety of P-gp inhibitors, which have been made public. However, undesirable pharmacokinetic alterations and immunosuppressive side effects prevent their continued use in clinical settings. 62 New P-gp inhibitors, especially naturally occurring molecules with the ability to interact with many targets, have received increasing attention. 63 Additionally, it was discovered that curcumin and berberine are strong natural chemopreventive substances for a variety of malignancies. 17 The synergistic effects of curcumin and berberine on MDR1 activity were examined using the fluorometric MDR test. As a result of the increased intracellular intensity of fluorescent P-gp substrate in the A549 cell in comparison to the control group, both free curcumin and free berberine blocked the activity of MDR1 pumps (Figure 7). Additionally, compared to single-agent chemotherapy, combination medication therapy using CUR and BBR was more successful in reducing MDR1 activity. Additionally, compared to the free drug solution, this combination medication therapy trend was significantly more effective (****p-value less than 0.0001) when curcumin and berberine were delivered in TNV formulations. P-glycoprotein (P-gp) downregulation and NF-B pathway inhibition by CUR and BBR were credited for the combination medication therapy’s increased therapeutic activity. More critically, because CUR inhibits NF-B signaling pathways, cancer cell proliferation and apoptosis induction are suppressed. 64
Conclusion
We created the tween-based nanovesicles formulation for curcumin and berberine combined medication therapy in A549 lung cancer cells. The findings demonstrated that the TNV formula’s entrapment efficiency was affected by the type of tween. The TNV mix made from tween 60, cholesterol, and PEG led to greater entrapment efficiency for BBR and CUR. When compared to single-agent chemotherapy, the integration of CUR and BBR increased the rate of apoptosis, especially when given in TNV formulations. The results show that free curcumin and free berberine increased the intracellular intensity of the fluorescent P-gp substrate by inhibiting MDR1 activity. Furthermore, when curcumin and berberine were delivered in TNV formulations as opposed to free drug solutions, combination pharmacological therapy of CUR and BBR was significantly more effective. The current study clearly shows that the berberine and curcumin nanovesicles formulation for combined medication therapy may be a promising lung cancer treatment.
Footnotes
Authors’ contributions
Data curation: AA, MHK, MS, SAH; Formal analysis: SAH, AV, SHK; Funding acquisition: AV, SHK, MF; Investigation: AA, MHK, SAH, AV; Project administration: MHK, MS, SAH; Resources: MS, SAH, AV, MF; Software: AV, SHK, MF, MS; Supervision: AA, MS, SAH; Validation: AA, AV, SHK; Visualization: MF, AA, MS; Writing – original draft: MF, AA, AV, MHK, MS, SAH; Writing – review & editing: AA, AV, SHK, MS. All authors read and approved the final manuscript.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Behbahan Faculty of Medical Sciences, Behbahan, Iran (Grant No.9926).
Ethical approval
This work was approved by the Ethics Committee of the Behbahan Faculty, Medical Sciences, Behbahan, Iran (IR.BHN.REC.1399.006)
Consent for publication
Not applicable
