Abstract
With the rising incidence of cancer, radiotherapy has become an increasingly popular treatment modality. However, radiation-induced skin injuries (RSIs) have emerged as a commonly reported side effect of radiotherapy, thereby presenting a significant challenge in the field of radiotherapy. In this study, we report the successful synthesis of a photosensitive hydrogel via amide reaction for grafting the photosensitive group, methacrylate anhydride (MA), onto chitosan (CHI) and gelatin (GEL), with subsequent physical incorporation of epigallocatechin gallate (EGCG). The resulting composite photosensitive hydrogels exhibited favorable swelling properties, rheological properties, and biocompatibility, which promote angiogenesis and demonstrate notable therapeutic efficacy against RSIs. These findings provide valuable insights into the clinical utility of EGCG composite hydrogels for the effective management of RSIs.
Introduction
Cancer is a major cause of mortality worldwide, with radiotherapy being a prominent localized tumor treatment modality. Approximately half of all cancer patients receive radiation therapy (RT), which can result in specific side effects.1,2
Radiation skin injuries (RSIs), one of the most reported side effects of radiation therapy, affects 95% of patients undergoing RT. 3 RSIs are caused by the direct absorption of radiation energy by crucial cellular structures such as DNA and indirect absorption by highly reactive free radicals. 4 There are many ways to treat RSIs. Commonly used pharmacological treatments include biological agents such as epidermal growth factor, 5 and colony-stimulating factor, 6 synthetic drugs like triethanolamine, 7 corticosteroids, 8 statins, 9 as well as vitamins such as vitamin C, 10 and vitamin B5. 11 However, these treatments have several drawbacks, including high side effects, expensive prices, and inaccurate efficacy. As a result, natural herbal drugs such as chamomile, 12 aloe vera, 13 and tea polyphenols have gained attention. Tea polyphenols, in particular, have attracted the most attention due to their efficient scavenging of free radicals.
Epigallocatechin gallate(EGCG), the most abundant tea polyphenol in tea, 14 has been shown to inhibit radiation damage by scavenging hydroxyl radicals, hydrogen peroxide, and superoxide anions. 15 Topical application of EGCG has resulted in a substantial reduction in pain, burning, and itching in breast cancer patients with RSIs, preventing the development of RSIs. 16 However, current studies on EGCG for RSIs rely on direct spraying of EGCG solution onto the affected area, requiring multiple daily doses and high patient compliance. Local concentrations of the drug are uncertain due to local cut, friction, and evaporation of the topical aqueous solution.
Recent research on macromolecular hydrogels in tissue engineering has provided new and efficient drug delivery routes. Biomacromolecular hydrogel materials are used mainly as hydrogel dressings and injectable hydrogels for wound healing. Injectable hydrogels for skin injuries are mainly used for burns and wounds caused by diabetes, 17 with limited studies on injectable hydrogels for the treatment of RSIs.
In this paper, chitosan (CHI) and gelatin (GEL) were grafted with photosensitive methacrylic anhydride (MA) via amide reaction, and then EGCG was added to the hydrogel by physical mixing, and administered by minimally invasive injection. This approach helps to prevent the decrease or loss of activity caused by the self-oxidation of the phenolic hydroxyl group, which tends to occur in the presence of air. 18 Furthermore, it transforms the hydrogel into a stable drug delivery system that enhances the effective concentration and duration of the drug in the affected area, and promote wound recovery by promoting the expression of angiogenesis-related genes and cytokines, which also provides a new way for the treatment of RSIs and is expected to become an effective means of treating RSIs in the future.
Materials and methods
Experimental cells and materials
Human Umbilical Vein Endothelial Cells (HUVEC; Shanghai Cell Bank, Chinese Academy of Sciences), CHI (Macklin), GEL (Macklin), MA (Macklin).
Fabrication of hydrogel
CHI was dissolved in an acetic acid solution at a concentration of 1.5% (w/v) and mixed with MA to regulate the grafting rate of CHI amino to MA at 33.6%. The mixture was heated to 60°C and kept warm while being uniformly stirred for 6 h to produce CHI-MA. GEL was dissolved in deionized water at 50°C to create a 10% (w/v) aqueous solution. Then, 6% (w/v) MA liquid was added to the GEL solution system. The solution was kept warm at 50°C and stirred uniformly for 3 h to form GEL-MA (Supplemental File).
Subsequently, the reaction mixture was dialyzed in deionized water for 96 h and then freeze-dried to obtain a powder. After the reaction mentioned above, CHI-MA and GEL-MA were mixed according to the experimental requirements to generate CHI-GEL-MA, followed by the addition of EGCG with physical stirring to produce EG-CHI-GEL-MA. The addition of cross-linker MBA improved the mechanical properties, followed by the introduction of LAP. The resulting mixture was irradiated under purple light at 405 nm for 7 s to form a gel.
Characterization of CHI-GEL-MA hydrogels
Subgroups
Group A
The concentration of CHI-GEL-MA was 6%, and the ratios of CHI-MA and GEL-MA concentrations were 1:4, 1:2, and 1:1, respectively, which were recorded as CHI-GEL-MA (1:4), CHI-GEL-MA (1:2), and CHI-GEL-MA (1:1).
Group B
The concentration ratio of CHI-MA and GEL-MA was 1:4, and the concentration of CHI-GEL-MA was 8%, 6%, and 4%, respectively, which were recorded as CHI-GEL-MA (8%), CHI-GEL-MA (6%), and CHI-GEL-MA (4%).
Group C
The concentration of CHI-GEL-MA was 6%, the concentration ratio of CHI-MA and GEL-MA was 1:4, and the amount of MBA added was 0.1%, 0.2%, and 0.3%, respectively, which were recorded as CHI-GEL-MA (0.1% MBA), CHI-GEL-MA (0.2% MBA), and CHI-GEL-MA (0.3% MBA).
Porosity, infrared absorption spectra, and X-ray photoelectron spectroscopy of CHI-GEL-MA hydrogels
The hydrogel was freeze-dried to obtain lyophilized powder, and an appropriate amount of lyophilized powder was taken to observe the internal structure and morphology of the hydrogel using a scanning electron microscope (Regulus-8100, JEOL), and 10 random areas were selected to calculate the pore size with the average value.
X-ray photoelectron spectrometry (XPS; K-Alpha, ThermoFisher) was used to analyze lyophilized powder by spot sweeping with an Ar cluster ion gun to determine the binding energy and elemental composition of the hydrogels.
To conduct absorption spectroscopy by Fourier Infrared Spectroscopy (Excalibur3100, Varian, USA), the lyophilized powder was mixed with KBr, ground, pressed, and measured using a Fourier infrared spectrometer within the range of 400–4000 cm−1.
Solubilization and rheological properties of CHI-GEL-MA hydrogels
The rheological properties of each group were tested using a rotational rheometer (MCR-702, Anton Paar, Germany) by utilizing a pair of 25 mm diameter plates with a plate spacing of 20 μm and a plate temperature of 40°C. Specifically, each group’s storage modulus, loss modulus, and viscosity were assessed.
Square molds of 1 cm³ were used to create hydrogels which were then placed in a PBS solution at 37°C. At predetermined intervals, excess water was aspirated, and the hydrogels were weighed after being gently removed with forceps.
Biocompatibility assessment of EG-CHI-GEL-MA hydrogels
Hydrogel cell adhesion, cytoskeleton staining, live-dead cell staining, and proliferation experiment
The half effective concentration (EC50) of EGCG for HUVEC proliferation was 0.02 μmol/ml, and the maximum effective concentration was 0.04 μmol/ml. Because the concentration of EGCG added to the hydrogel and released into the culture medium may change, in order to select the most suitable concentration of EGCG added to the hydrogel, 0.02, 0.04, and 0.06 μmol/ml of EGCG added to the hydrogel were selected. They were named as EG-CHI-GEL-MA (0.02), (0.04), and (0.06), respectively.
Cell adhesion assay
A 3 × 105 cells were inoculated on each group of hydrogels, and the cell adhesion was observed by applying DAPI staining solution at 1, 2, and 4 h after inoculation. Five fields of view were randomly selected for cell counting by fluorescence microscope.
Cytoskeleton staining
A 105 cells were inoculated on the surface of each group of hydrogels, and FITC-phalloidin/DAPI staining was applied 24 h after inoculation to make the cytoskeleton proteins fluorescent in yellow-green color. Five randomly selected fields of view were observed for cell morphology by fluorescence microscope.
Live/dead cell staining
A 105, 8 × 104, and 6 × 104 cells were inoculated on the surface of the hydrogel for 1, 2, and 3 days for live/dead cell staining, respectively. Cell viability was calculated by randomly choosing 10 fields of view on a fluorescence microscope.
Cell proliferation assays
HUVEC was inoculated on the surface of the hydrogel at a cell density of 50,000 cells/ml, and CCK-8 was applied to count the cells at 1, 2, and 3 days, respectively, to observe its effect on cell proliferation.
Evaluation of the antioxidant effect of EG-CHI-GEL-MA
The fluorescent probe DCFH-DA was loaded into the cells and then inoculated into an EG-CHI-GEL-MA hydrogel, and the medium mixed with Rosup was added and detected using a fluorescent zymography after 2 h.
Evaluation of the effect of EG-CHI-GEL-MA on the function of vascular endothelial cells
The effect of EG-CHI-GEL-MA hydrogel on the expression of cellular angiogenesis-related genes was investigated using RT-PCR. The impact of EG-CHI-GEL-MA hydrogel cytokines that play a crucial role in cellular angiogenesis was assessed via ELISA.
Hydrogel for the treatment of radioactive skin damage
Twenty-eight rats (purchased from the Experimental Animal Center of Shanxi Medical University) were divided into four groups, A: Normal control group, B: RSIs group, C: RSIs + CHI-GEL-MA hydrogel group, D: RSIs + EG-CHI-GEL-MA hydrogel group.
Before irradiation, all rats were dehaired in their buttocks and anesthetized with an intraperitoneal injection of 0.6% sodium pentobarbital solution, except for group A. The rats received local irradiation with a single dose of 300 cGy/min from a medical electron linear accelerator at a source skin distance of 100 cm. In the affected area of the rats, different gels were injected once every three days until day 30, when they were sacrificed. The experiments complied with the Regulations on the Administration of Laboratory Animals and met ethical requirements, Ethical Review No. SYDL2023026.
Investigation of pathological recovery using hydrogel for treating radiation-induced skin injuries
To evaluate the thickness of granulation tissue, inflammatory response, collagen level, and vascular neoplasia in affected tissues of rats, histological techniques, including HE staining, Masson staining, and immunohistochemistry, were utilized.
Investigation of functional recovery using hydrogel for treating radiation-induced skin injuries
Gene expression for inflammation as well as angiogenesis was detected using RT-PCR, and protein expression for angiogenesis as well as autophagy was detected using ELISA.
Results and discussion
Structural characterization of functional hydrogels
Porosity of hydrogels, infrared spectroscopy analysis, and XPS analysis
Under the electron microscope (Figure 1(a)–(f)), it can be observed that the hydrogel materials with different component ratios all showed a pore-layer structure, and the pore diameters of CHI-GEL-MA (1:4; 1:2; 1:1) hydrogel were (130.75 ± 16.61) µm, (139.46 ± 41.74) µm, and (284.93 ± 41.05) µm, respectively (p < 0.05). The pore diameters of CHI-GEL-MA (0.1% MBA; 0.2% MBA; 0.3% MBA) hydrogel were (313.99 ± 35.65) µm, (251.12 ± 52.51) µm, and (181.79 ± 4.68) µm (p < 0.05), respectively (Supplemental File). It can be seen that the larger the concentration ratio of CHI-MA and GEL-MA, the larger the average pore diameter of the hydrogel. The higher the MBA addition, the smaller the hydrogel voids. Polymer type, preparation method, crosslinking agent type and concentration can affect the pore size of hydrogels. The smaller the concentration ratio of CHI-MA and GEL-MA or the more crosslinking agent added, the denser the structure of the hydrogel is, the pore size decreases. Han et al. 19 found better cell proliferation and differentiation in hydrogels with 100 and 200 µm pores. Oxygen and nutrients could not be transported in 50 µm pore space, while 400 µm pore space had poor cellular differentiation. It has been suggested that the optimal pore size for HUVEC migration and tissue vascularization in hydrogels is 200–250 µm, 20 and a hydrogel with a pore size of about 200 µm is more appropriate for subsequent experiments.

Scanning electron microscope picture of composite hydrogel, Bar: 200 μm (a–f), infrared absorption spectra of composite hydrogels (g), XPS general spectrum of composite hydrogels (h), and high resolution XPS spectra of composite hydrogels (i–k).
The infrared spectral data were normalized and processed to obtain the results shown in the figure (Figure 1(g)). The absorption peaks of CHI-MA at 1617 and 1541 cm−1 are the amide I band and amide II band, respectively; the absorption peak of GEL-MA at 1541 cm−1 is amide II band, which indicates the amide reaction between MA and CHI and GEL. CHI-GEL-MA group, The absorption peaks at 1653 and 1541 cm−1 are generated by C=C stretching vibration and N-H bending vibration, indicating the existence of hydrogen bonding between CHI-MA and GEL-MA. The absorption peak of the CHI-GEL-MA + MBA group at 1034 cm−1 is caused by C-O stretching vibration, indicating the cross-linking of MBA with the material. The broad peak at 3265 cm−1 in the EG-CHI-GEL-MA group is the -OH stretching vibration peak; the absorption peak at 2887 cm−1 is the C-H stretching vibration; the absorption peaks at 1374, 699, and 591 cm−1 are caused by C-H bending vibration. It indicates that the introduction of EGCG has a hydrogen bonding interaction with the hydrogel material and achieves physical cross-linking.
From the full spectrum (Figure 1(h)), it can be seen that there are mainly C 1 s peaks, O 1 s peaks, and N 1 s peaks in the polymer. High-resolution scans of the above elements yielded binding energies, as shown in the figure (Figure 1(i–k)). Three peaks on the C 1 s high resolution spectrum of were attributed to C–C/C=C, C–O, and C=O, respectively. The C content of CHI-GEL-MA hydrogel was 64.54%, and the C content of EG-CHI-GEL-MA hydrogel was elevated to 79.04%. Three peaks on the O 1 s high resolution spectrum of were attributed to C–C/C=C, C–O, and C=O, respectively. The O content of CHI-GEL-MA hydrogels was 14.02%, and the O content of EG-CHI-GEL-MA hydrogels decreased to 5.36%. A peak on the N 1 s high-resolution spectrum of CHI-GEL-MA hydrogels with a binding energy of 399.53 eV was attributed to C-NH2. A new peak on the EG-CHI-GEL-MA hydrogels with a binding energy of 398.62 eV was attributed to C-NH2. The N content of CHI-GEL-MA hydrogels was 21.44%, and that of EG-CHI-GEL-MA hydrogels was reduced to 15.59%. This indicates that EGCG has been successfully introduced into CHI-GEL-MA hydrogels.
Dissolution properties of composite hydrogels and rheological mechanical properties
The swelling properties of hydrogels have many effects on the structural integrity of their scaffolds as well as morphological and mechanical properties and even drug delivery efficiency.21,22 Our study showed that the composite hydrogels exhibited good swelling properties. When the concentration ratio of CHI-MA and GEL-MA was decreased from 1:1 to 1:4, the swelling degree increased from 105% to 130% at 300 min, and when the concentration of CHI-GEL-MA was decreased from 8% to 4%, the swelling degree increased from 110% to 140% at 300 min. The hydrogel with 0.1% cross-linking agent was the most swollen, reaching 125% (Figure 2(a)–(c)). The swelling performance of hydrogels is related to many factors including the molecular structure of the polymer, cross-linking density, intermolecular interaction forces, etc. GEL-MA has more intermolecular hydrophilic groups than CHI-MA, and its structure is more loosely packed, so the smaller the ratio of the concentration of CHI-MA and GEL-MA, the larger the swelling degree. The lower the concentration of the polymer, the weaker the interaction force between its molecular chains, the looser the network, and the greater the degree of swelling. The smaller the amount of cross-linking agent added, the lower the cross-linking density, the looser the network, the hydrogel can accommodate more water molecules, the greater the swelling.
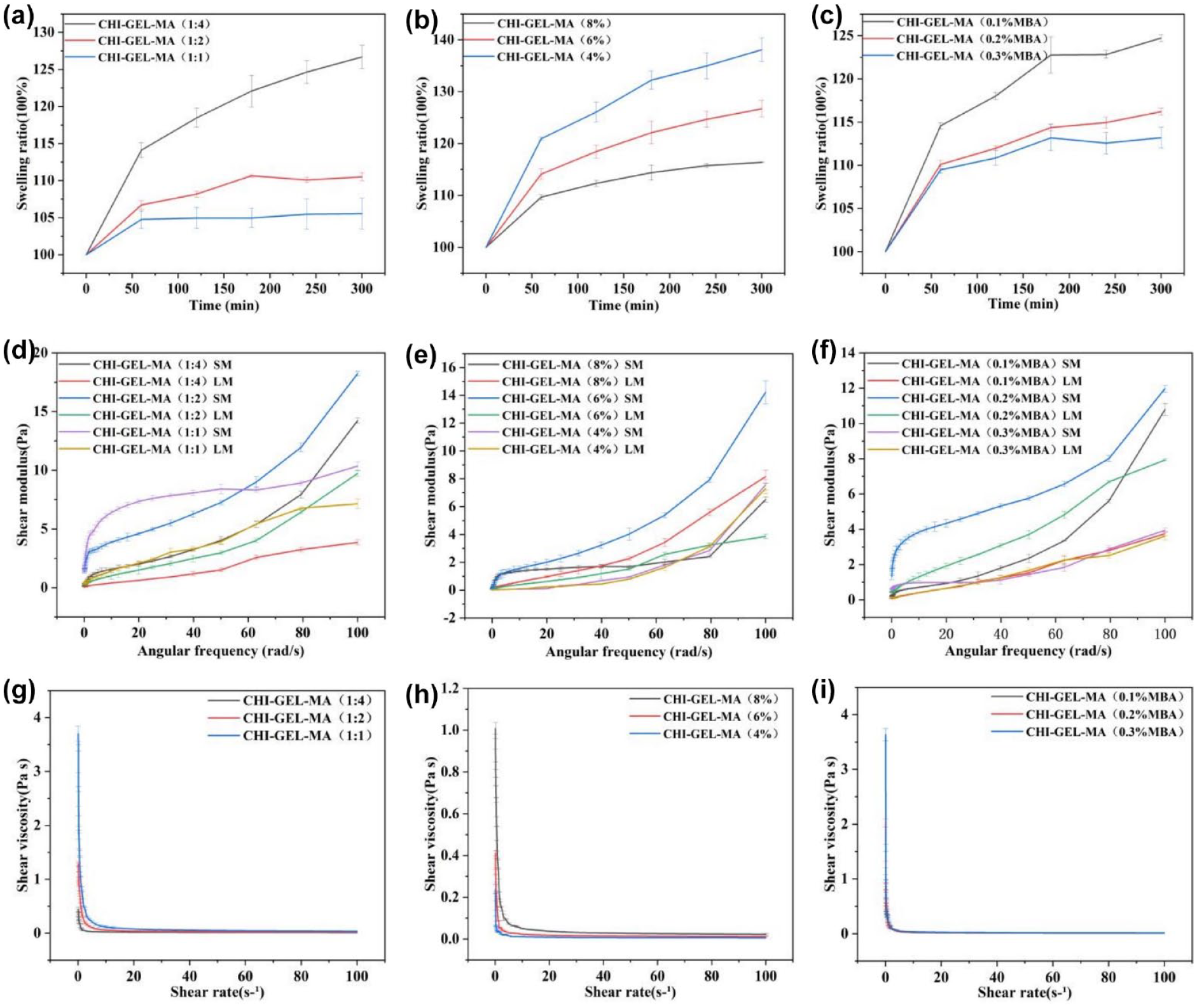
(a–c) Swelling properties of composite hydrogels, (d–f) storage modulus and loss modulus of composite hydrogels, and (g–i) viscosity of composite hydrogel.
Rheology is also an important property of hydrogels. When the concentration ratio of CHI-MA and GEL-MA is reduced from 1:1 to 1:4 at a corner frequency of 100 rad/s, the energy storage modulus increases from 7 Pa to about 19 Pa, and the loss modulus decreases from 7 Pa to about 3 Pa. When the concentration of CHI-GEL-MA is increased from 4% to 8%, the energy storage modulus increases from 8 to 15 Pa, and the change of loss modulus increases from 2 to around 8 Pa. The energy storage modulus is 13 Pa and loss modulus is about 8 Pa at 0.02% crosslinker addition, which is higher than other groups. When the energy storage modulus is much larger than the loss modulus, the material mainly undergoes elastic deformation and is in the solid state. By contrast, the loss modulus is always larger than the energy storage modulus, the material mainly undergoes viscous deformation and is in the liquid state, and the energy storage modulus for each group of hydrogel is always slightly larger than the loss modulus, but basically in the same state, which indicates that the material is semi-solid, in accordance with the typical hydrogel (Figure 2(d)–(f)).
When the concentration ratio of CHI-MA and GEL-MA was reduced from 1:1 to 1:4, the viscosity decreased from 4 to about 0.4 Pa · s. When the concentration of CHI-GEL-MA grew from 4% to 8%, its viscosity grew from 0.2 Pa · s to about 1 Pa s. There was a small increase in viscosity with the increase in MBA addition (Figure 2(g)–(i)). During the process of hydrogel injection, the hydrogel will be extruded from the syringe needle and will be subjected to shear force, so appropriately reducing the gap between the storage modulus and loss modulus of the hydrogel as well as decreasing the viscosity is conducive to the realization of the injectability of the hydrogel.
Biocompatibility evaluation of EG-CHI-GEL-MA hydrogels
Survival of vascular endothelial cells cultured on the surface of EG-CHI-GEL-MA hydrogel
Biocompatibility is an important indicator for testing hydrogels (Supplemental File). Cells were observed 1, 2, and 4 h after inoculation using DAPI staining (Figure 3(a)) to show that the number of endothelial cells adhering to the hydrogel surface gradually increased with the increase of time, and there was no significant difference in the number of cells adhering to the hydrogel when compared with that of the control group (p > 0.05). It indicates that the cells can adhere well to the hydrogel surface. After 24 h of cell inoculation, FITC-phalloidin/DAPI staining showed that the cells were spindle-shaped, similar to the morphology of the control cells, which indicated that the endothelial cells unfolded well on the surface of the hydrogel (Figure 3(b)).
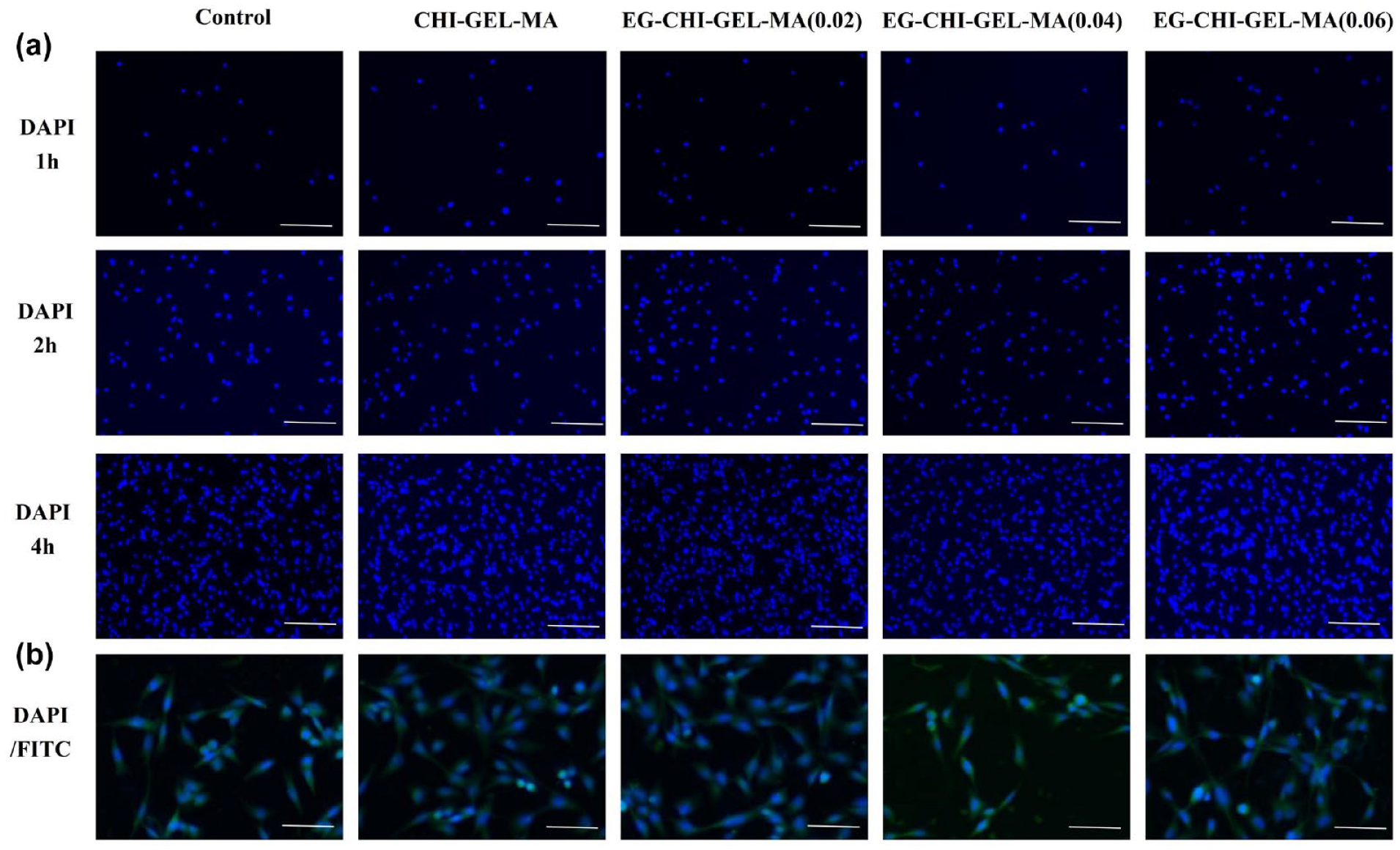
(a) Cell adhesion (DAPI) staining, bar: 100 μm and (b) cytoskeleton (DAPI/FITC) staining, bar: 200 μm.
After 1, 2, and 3 days of incubation in the incubator, the results of live-dead staining (Figure 4) were shown, the cell survival rate was above 98% of each group, and there was no significant difference in the cell survival rate between the EG-CHI-GEL-MA hydrogel and the control group (p > 0.05). Compared with the control group, EG-CHI-GEL-MA hydrogel had a significant promotion effect on cell proliferation, with EG-CHI-GEL-MA (0.02) and EG-CHI-GEL-MA (0.04) having a more significant promotion effect (Figure 5(a)).
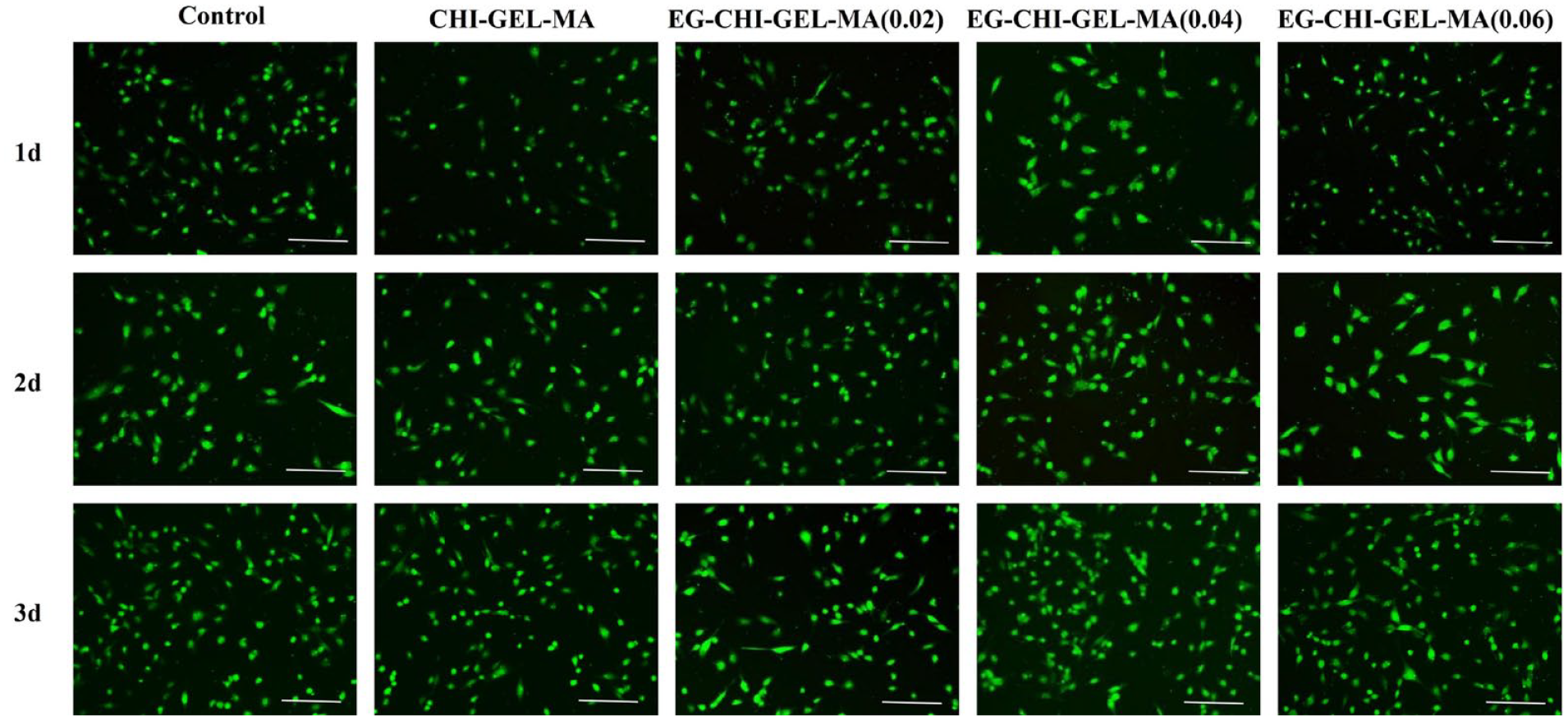
Cell live/dead staining, bar: 100 μm.

Effect of EG-CHI-GEL-MA hydrogel on cell proliferation (a), antioxidant capacity of EG-CHI-GEL-MA hydrogel (b), effects of EG-CHI-GEL-MA hydrogel on angiogenesis-related gene expression (c), and cytokines secretion (d).
This result suggests that EG-CHI-GEL-MA hydrogel has good biocompatibility. CHI-MA has poor cell adhesion, and it is generally used to improve its adhesion by modifying CHI or adding collagen or GEL.23,24 In this paper, we added GEL-MA, which retains the natural cell-binding sites of gelatin, so that it can well improve the adhesion of CHI-MA. EGCG can stabilize the microtubule protein family to promote the cells to form a better cytoskeleton shape. 25 Therefore, cells can adhere and unfold well on EG-CHI-GEL-MA. Its cell proliferation-promoting effects are related to the activation of cell proliferation-related factors, antioxidant, and anti-apoptotic effects of EGCG. 26
Evaluation of the antioxidant effect of EG-CHI-GEL-MA hydrogels
Rosup is a substance that substantially enhances cellular generation of reactive oxygen species (ROS). As shown, EG-CHI-GEL-MA hydrogel substantially scavenges ROS, scavenging 15% of free radicals within 2 h (Figure 5(b)). The antioxidant effect of EGCG is related to its polyphenolic structure that has the ability to burst reactive free radicals and chelate metal ions in order to prevent the formation of ROS, 27 and it is recognized as a powerful antioxidant.
Evaluation of the effect of EG-CHI-GEL-MA hydrogel on the function of vascular endothelial cells
RT-PCR results (Figure 5(c)) showed that compared with the control group, the EG-CHI-GEL-MA (0.04) hydrogel group had a more significant promotion of the expression of vascular endothelial growth factor receptor (VEGFR), proliferation cell nuclear antigen (PCNA), and 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase-3 (PFKFB3; the expression was 1.61, 1.63, and 1.66, respectively; p < .05).
ELISA results (Figure 5(d)) showed that compared with the control group, the EG-CHI-GEL-MA (0.04) hydrogel group had a significant promotion effect for the VEGF, Ang-II, platelet derived growth factor-BB (PDGF-BB; the expression was 277.89, 29.92, and 50.51 pg/ml, respectively; p < 0.05).
The above findings suggest that EG-CHI-GEL-MA hydrogel promotes HUVEC function well. It is well documented that chitosan as well as its hydrolysates can stimulate endothelial cell growth and induce vascular regeneration and granulation, 28 whereas EGCG plays an important role in endothelial cell metabolism, growth, proliferation, transcription, and protein synthesis 29 by decreasing the expression of endothelial peroxidative factors, inflammatory factors, and decreasing their permeability. 30
Therapeutic effect of hydrogel for the treatment of radiation-induced skin injuries
Hydrogel on the progression of wounds in rats with radiation-induced skin injuries
By recording the skin changes from 1 to 4 weeks, it was found that EG-CHI-GEL-MA hydrogel significantly reduced the wound area of RSIs and inhibited the extent of inflammatory invasion relative to the rats in the RSIs group (Figure 6(a)).

Progress of compound hydrogel in the treatment of wounds in RSIs rats (a), HE staining, Massson staining and CD31 antibody staining of rat skin tissue, Bar: 200 μm (b), effect of EG-CHI-GEL-MA hydrogel on the expression of injury recovery-related genes (c), and proteins (d) in RSIs rats.
Trauma pathological recovery of hydrogel for the treatment of radiological skin injury
HE staining showed significant differences between the control and RSIs groups, with the latter showing disappearance of skin folds, missing epidermis, inflammation, disordered structure, and necrotic tissue. Masson staining did not show a significant difference in collagen between the groups (p ⩾ 0.05). Immunohistochemical results showed a significant increase in CD31 expression after hydrogel treatment compared to the RSIs group (p < 0.05; Figure 6(b)).
Some scholars have observed an increase in skin macrophages, NLRP3 inflammatory vesicles, and fibrotic tissue replacement of skin appendages and adipose tissue in irradiated rats. They also found a non-significant decrease in collagen fibers after 4 weeks of irradiation but a significant increase after 12 weeks, 31 consistent with our findings. Our study showed that EG-CHI-GEL-MA hydrogel was effective in reducing inflammation and promoting neovascularisation. However, it had no effect on collagen fibres, which may be related to the longer process of skin fibrosis.
Functional recovery study of hydrogel for the treatment of radioactive skin injuries
Compared with the RSIs group (Figure 6(c)), the RSIs + EG-CHI-GEL-MA hydrogel group had a significant inhibitory effect on the expression of pro-inflammatory-related genes nuclear factor kappa (NF-κB), MCP-1, and pentraxin 3 (PTX-3; the expression was 0.84, 6.11, and 0.76, respectively; p < 0.05), which indicated that the EG-CHI-GEL-MA hydrogel was able to inhibit the inflammation level. It has been shown that EGCG can reduce the levels of NF-κB, TNF-α, IL-8, IL-1β, and IL-6 in wound tissues to inhibit inflammation thereby promoting skin wound repair,32,33 which is consistent with our study.
PCNA is a gene associated with cell proliferation (Figure 6(c)), and a decrease in the PCNA level in the RSIs + EG-CHI-GEL-MA hydrogel group (the expression was 0.65) compared to the RSIs group (p < 0.05) probably due to the lower level of proliferation in the treatment group because of the less severe injury.
Basic fibroblast growth factor (bFGF), epidermal growth factor-like domain protein 7 (EGFL7) and VEGF are pro-angiogenic genes or cytokines (Figure 6(d)). Compared with the RSIs group, the RSIs + EG-CHI-GEL-MA hydrogel group had a significant promotion of bFGF expression (the expression was 2.72; p < 0.05), and also of EGFL7 and VEGF (the expression was 7908.31 and 566.53 pg/ml, respectively). A study showed that the levels of VEGF in the blood of rats were down-regulated 3.49 fold after 10 Gy irradiation, which implies that the inhibition of angiogenesis hinders wound healing. 34 The ability of EG-CHI-GEL-MA hydrogel to promote the expression of these genes and cytokines suggests that it has a good pro-vascular repair function in the skin.
Ang-II is an effective substance to promote wound repair 35 (Figure 6(d)), compared with the RSIs group, the RSIs + EG-CHI-GEL-MA hydrogel group had a significant promotion of Ang-II expression (expression of 877.44 pg/ml; p < 0.05), indicating its efficacy for wound recovery.
Autophagy favors the recovery of skin wounds, whereas radiation disrupts the cellular autophagy system 36 (Figure 6(d)). Compared to the RSIs group, the RSIs + EG-CHI-GEL-MA hydrogel group had a more pronounced promotion of autophagy-related proteins beclin 1(BECN1) and Light chain protein 3(LC3; the expression was 8543.5 and 2186.11 pg/ml, respectively; p < 0.05), and some study shows that EGCG promotes cellular autophagy usually by inactivating the PI3K/Akt/ mTOR pathway, thus prompting cellular self-repair. 37
Conclusion
In this study, we developed a hydrogel called EG-CHI-GEL-MA by grafting the photosensitizing agent MA onto CHI and GEL through an amide reaction, and incorporating the drug EGCG. This hydrogel exhibits favorable swelling properties, rheological properties, and biocompatibility. It enhances the proliferation of HUVEC, scavenges ROS, and upregulates the expression of angiogenesis-related cytokines and genes. In animal models of RSIs, EG-CHI-GEL-MA hydrogel demonstrates significant anti-inflammatory effects and promotes angiogenesis, thereby representing a viable therapeutic option. Furthermore, it holds promise for future clinical applications, potentially benefitting a larger population of patients.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000231218996 – Supplemental material for Study of EGCG composite hydrogel for the treatment of radiation-induced skin injuries
Supplemental material, sj-docx-1-jbf-10.1177_22808000231218996 for Study of EGCG composite hydrogel for the treatment of radiation-induced skin injuries by Jingying Wang, Lin Gao, Jianbo Song and Sijin Li in Journal of Applied Biomaterials & Functional Materials
Footnotes
Author contributions
Conceptualization: J.S. and S.L.; methodology: J.S.; investigation: J.W.; resources: L.G.; data curation: J.W.; writing: J.W. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received financial support from Open Research Fund Program of Collaborative Innovation Center for Molecular Imaging of Precision Medicine (No. 2020-ZD02).
Guarantor
SL.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
