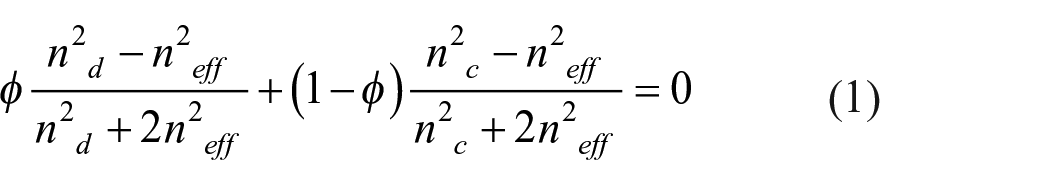Abstract
Microbial biofilm build-up in water distribution systems can pose a risk to human health and pipe material integrity. The impact is more devastating in space stations and to astronauts due to the isolation from necessary replacement parts and medical resources. As a result, there is a need for coatings to be implemented onto the inner region of the pipe to minimize the adherence and growth of biofilms. Lubricant-infused surfaces has been one such interesting material for anti-biofouling applications in which their slippery property promotes repellence to many liquids and thus prevents bacterial adherence. Textured and porous films are suitable substrate candidates to infuse and contain the lubricant. However, there is little investigation in utilizing a nanoparticulate thin film as the substrate material for lubricant infusion. A nanoparticulate film has high porosity within the structure which can promote greater lubricant infusion and retention. The implementation as a thin film structure aids to reduce material consumption and cost. In our study, we utilized a well-studied nanoporous thin film fabricated via layer-by-layer assembly of polycations and colloid silica and then calcination for greater stability. The film was further functionalized to promote fluorinated groups and improve affinity with a fluorinated lubricant. The pristine nanoporous film was characterized to determine its morphology, thickness, wettability, and porosity. The lubricant-infused film was then tested for its lubricant layer stability upon various washing conditions and its performance against bacterial biofilm adherence as a result of its slippery property. Overall, the modified silica nanoparticulate thin film demonstrated potential as a base substrate for lubricant-infused surface fabrication that repelled against ambient aqueous solvents and as an anti-biofouling coating that demonstrated low biofilm coverage and colony forming unit values. Further optimization to improve lubricant retention or incorporation of a secondary function can aid in developing better coatings for biofilm mitigation.
Keywords
Introduction
Microbial biofilms are significant, yet often can be a troublesome aspect of life. The concept of a biofilm was first established in 1930s by Henrici 1 and Zobell and Allen. 2 Biofilms consist of bacterial cells adhered to surfaces and contained in a dense self-produced organic polymer matrix.3,4 The complex extracellular polymer networks provide protection against various environmental stresses and biological and chemical agents.3,5 Biofilm formation can be influenced by the hydrodynamics and surface properties of the surrounding environment. 6 Biofilms are beneficial in the natural environment to promote microbe survival and symbiosis 7 as well as in synthetic systems for applications in agriculture, 8 food fermentation, 8 bioremediation3,9 and corrosion inhibition. 10 However, biofilm formation is detrimental in the healthcare, 11 water distribution 12 and food industries 13 as bacterial biofilm build-up poses a threat to human health and hardware material integrity.
Accumulation of bacterial biofilms onto surfaces causes biofouling, which pollutes tubing and equipment. Biofouling and biofilm growth can result in an increased risk of pathogen transmission and contamination to humans,11,12 where waterborne diseases are responsible for an estimate of over 40,000 hospitalizations at a cost of $970 million per year. 14 In addition, biofouling can cause the malfunction of key equipment such as water distribution units and radiators, where biofilm build-up can impede and eventually block the liquid flow.12,15 Likewise, the metabolic activity of the microbes in the biofilm can result in the formation of organic acids and compounds, which can lead to corrosion and deterioration of equipment and tubing. 16 The impact is more devastating in space stations and to astronauts. Microbial biofilms in space can adapt in new environmental features such as microgravity and elevated radiation to allow for feasible growth.17,18 Due to isolation from necessary replacement parts and medical resources, astronauts and equipment are more vulnerable to the bacterial exposure and microbial corrosion, respectively.16,19 In 2010, biofilms caused a reduced flow rate in the water processor assembly unit on the International Space Station, and a filter replacement of every 3–6 months was recommended but deemed impractical due to limitation in logistics and crew time. 20 As a result, it is critical to implement biofilm mitigation and prevention strategies.
Several techniques are studied and employed to minimize the bacterial biofilm accumulation. Biocides such as iodine, colloidal silver and oxidizing chemicals and ionizing radiation such as ultraviolet (UV) light can promote cellular damage and inhibit repair mechanisms to kill bacteria and minimize growth; however, bacterial cells have demonstrated greater resistance to many biocides and UV, which reduces effectiveness and leads to the need for higher dose concentrations.21 –23 Physical cleaning and washing of pipes are also very costly in detergents and water, especially in payload and spacecrafts.13,22 A passive approach such as the implementation of anti-biofouling surfaces can be more advantageous in biofilm mitigation to reduce the need for replacement or maintenance.17,21 Such surfaces can be implemented onto the inner region of the pipe to minimize bacterial adherence and thus inhibit biofilm formation and growth at the first stage.6,14 The surfaces that are physically and/or chemically modified can exhibit specific surface parameters such as increased surface texture, hydrophobicity, low stiffness, and positive charges, which have been demonstrated to discourage bacterial adhesion and thus deter biofilm formation and growth.24 –27 However, there are variations in biofilm adhesion results due to the differences in bacterial strains and surface characteristics. 27 Thus, there is a need for an anti-biofouling surface that can employ the effect against a wide range of bacterial strains.
Lubricant-infused surfaces (LISs) are one such interesting material for anti-biofouling applications. The concept of LISs were first introduced by Aizenberg and colleagues, who were inspired by the slippery surface property of the Nepenthes pitcher plant. 28 LISs promote excellent repellency to a broad range of liquids,28,29 in which such slippery surface can be translated as an advance at minimizing adhesion of bacteria cells and thus preventing biofouling.30 –34 LISs are fabricated by infusing a lubricant liquid into rough textured or porous film substrates to obtain smooth slippery surfaces. 35 The textured or porous substrates are comprised of micro- or nanostructures such as micropillars and wells which aim to hold the lubricant sturdily within the solid textures via capillary and intermolecular forces.35 –38 In particular, nanoporous nanoparticulate thin films have been demonstrated as the potential substrate for LIS design39,40 and anti-biofouling application. 31
Nanoparticulate thin films are unique substrates in which inorganic nanoparticles (NPs) as structural units are assembled into a highly nanoporous thin film matrix. 41 Silica nanoparticulate thin films are well-suited as a host substrate for a LIS design. Firstly, the surface textures and matrix pores of a silica NP-based film promotes a nano-wicking effect, which is the rapid infiltration of fluids into the film network through the nanopores, and thus exhibits stable superhydrophilic behavior. 42 This is advantageous to the titanium oxide nanoparticulate films where the stability of the superhydrophilic state requires light activation for improvement. 43 In addition, upon using specific material type such as metal and metal oxides in the inorganic NP units, unique thickness-dependent properties are included alongside the wettability behavior42,44,45; for example, silica films have demonstrated low refractive index, antireflection and antifogging. 42 In addition, the surface chemistry of the porous matrix can be modified via coupling and functionalization reactions. For example, silica is versatile in coupling with silane compounds to promote various functional groups on the surface 46 for properties such as wettability,47,48 metal ion extraction 49 and lubricant infusion. 50 The high porosity and the textured top surface of the modified film substrate can express greater lubricant infusion and retention.41,42 Furthermore, implementation as a thin film structure aids to reduce material consumption and cost. 21 Therefore, a silica nanoparticulate thin film is promising to design slippery LISs to minimize bacterial adherence and growth.
In this study, we developed LIS films for anti-biofouling applications with the silica nanoparticulate thin film as the host substrate material. Figure 1 displayed a schematic of the overall fabrication process. The nanoparticulate thin film was fabricated via sequential absorption and layer-by-layer (LbL) assembly based on polycations and silica NPs, followed by calcination of the LbL films to create stable nanoporous films with high porosity. Two particle sizes (22 -nm and 50 -nm) were used in the LbL assembly process to generate thin films with different morphologies and thus properties. The films were functionalized with fluorinated silane compounds to modify the surface chemistry and wettability, and then infused with a fluorinated lubricant. The nanoporous film was characterized to determine its morphology, thickness, surface roughness, wettability, and porosity. The resulting LIS film was characterized in terms of its wettability, slippery property, lubricant stability and finally its performance against bacterial biofilm adherence and growth as a result of its slippery property.

Fabrication schematic for the proposed LIS nanoparticulate film.
Methods
Materials and chemicals
Poly(allylamine hydrochloride) (PAH) (Mw 15000-20000) was obtained from J&K Scientific. Poly(acrylic acid) (PAA) (25 wt.% in partial salt solution, Mw 240000), Ethanol (anhydrous, 99.5 + %), 3 M Fluorinert lubricant (FC-70) and colloidal silica (Ludox TM-40, 40 wt.% suspension, 22 -nm size) were purchased from Sigma-Aldrich. Silica microspheres (5% aqueous dispersion, 50 -nm size) were obtained from Polysciences. Sodium chloride (NaCl, crystalline) was obtained from Fisher Chemicals. Hydrochloric acid (HCl, 12 N, certified ACS plus) and sodium hydroxide (NaOH, pellets) were obtained from Fisher Chemicals, Inc. All aqueous solutions and water rinses were prepared using deionized water further purified from the Thermo Scientific Barnstead Nanopure water purification system under resistivity of around 17 MΩ·cm. Trichloro(1H, 1H, 2H, 2H-perfluorooctyl)silane (TPFS) (97%) were obtained from Alfa Aesar. For the substrates, Fisherbrand glass slides (25 × 75 × 1 mm) were purchased from Thermo Fisher, and silicon wafer (prime, polished one-side) were obtained from Platypus Technologies, Inc.
Nanoparticulate film fabrication
The nanoparticulate thin film was generated via sequential absorption and LbL assembly, using the nanoStrata dipping unit. 42 The dipping time for the polymers and the silica particles was set for 15 min, followed by one 2-minute water rinse and two 1-minute water rinses. An adhesion layer containing five bilayers of PAA and PAH was first created to ensure adequate adhesion of the initial LbL structure to the substrate. The PAA and PAH solutions (0.01 M each, based on the monomer molar mass) were prepared at pH 3–4. Then the body layer containing 16 bilayers of PAH polymer and silica particles was fabricated. The PAH solution (0.01 M) was set at pH 7.5. The silica particle dispersion (0.03 wt.%, with 0.1 M NaCl) was at pH 9 and consisted of either 22 -nm or 50 -nm silica particles to create thin films of different morphologies denoted as 22-NP and 50-NP, respectively. The pH of the solutions was adjusted accordingly. The LbL samples were then calcined in a tube furnace at 400°C for 3 h. The films fabricated on silicon wafer were specifically used for ellipsometry to minimize light backscattering. The films on glass substrates were used for the remaining characterization and tests, especially for the transmittance measurement and bacterial cell visualization due to the transparency of glass.
TPFS functionalization and lubricant infusion
Liquid phase deposition of TPFS 51 was performed to change the surface chemistry of the films. The calcined samples were immersed into a solution containing 1% v/v TPFS in ethanol for 1 h, heating at 60°C–65°C. The samples were removed and washed in the following order: with 100% ethanol, deionized water and finally 70% v/v ethanol-water mixture. The samples were left to dry at room temperature. They were then placed in the vacuum oven at 100°C under vacuum for 4 h to functionalize the TPFS to the surface and pores of the film. Samples after functionalization will be denoted as 22-TPFS and 50-TPFS.
Lubricant infusion was then performed to create the slippery LIS. TPFS-functionalized samples were immersed into FC-70 for 2 min so that the lubricant could cover and fully penetrate through the nanoporous film. The samples were removed and held vertically for 5 min to drain out most of the excess lubricant. The samples were then briefly rinsed in a water bath to remove additional unretained lubricant. Samples after infusion will be denoted as 22-LIS and 50-LIS.
Characterization
The calcined nanoparticulate films were visualized and characterized with the following instruments. Surface morphology of the calcined nanoparticulate films were observed with Zeiss Ultra-55 scanning electron microscope (SEM). Film thickness was measured using the Dektak XT surface profilometer. The porosity of the nanoparticulate films was estimated using an effective medium approximation in terms of the effective refractive index. The effective refractive index (neff) was obtained with the Woollam M2000 mapping variable angle spectroscopic ellipsometer. The spectra were fitted using the CompleteEASE software according to the film thickness and material specifications of the film and substrate. The refractive index at 550 nm was chosen as the value of interest for neff. The porosity was calculated based on the Bruggeman model (equation (1)) which represents a binary aggregate system with no defined homogenization. The term nd is the refractive index of the dispersed phase (silica = 1.47), nc is the refractive index of the continuous phase (air = 1), and ϕ is the fill fraction of the dispersed phase. The porosity was obtained by subtracting ϕ from 1.
The films after surface chemistry modification and processing were further characterized. Static water contact angles for the nanoparticulate films during each processing stage were measured with the CAG100 contact angle goniometer. The slippery property of the LIS films was further visualized by utilizing a goniometer and a specialized stage that provided a tilt angle of 15°. Various liquids such as water, ethanol and vegetable oil were dropped at 3 µL volume onto the lubricated film, and videos of the resulting effect were captured.
Lubricant stability testing
The lubricant stability was analyzed by monitoring the film transmittance in different washing conditions. The optical properties of the proposed nanoparticulate coating derived from the silica intrinsic property and film porosity to provide transparency and anti-reflection effects. This emerged a unique study where the change of the film transmittance could be observed as the lubricant gradually depletes from the coating over time. Various parameters in the washing process involved using water at various temperatures (5°C denoted as cold water, 25°C denoted as r.t. water, 60°C denoted as hot water), an ethanol-water mixture (denoted as EtOH-water), and Dulbecco’s phosphate-buffered saline (DPBS) medium. The transmittance spectra were obtained using the ThermoFisher Evolution 220 UV-visible spectrophotometer. The transmittance value measured at the wavelength of 450 nm was taken. The transmittance of the TPFS-functionalized film would act as the control limit for the comparison.
Bacterial biofilm growth studies
Preliminary testing for the anti-biofouling property was first conducted with biofilm growth in liquid media under no-shear flow conditions. Staphylococcus aureus (S. aureus, MRSA USA300) and Escherichia coli (E. coli, BL21) were used to test the films against a Gram-positive and Gram-negative bacterial species, respectively. The film samples on glass underwent UV sterilization for 3 min prior adding them to a sterile 12-well plate. were each grown in Luria Broth medium overnight at 37 °C in loosely capped tubes on an orbital shaker. The inoculums were prepared and diluted to obtain a stock suspension with 5–6 × 108 cell concentration. The 12-well plate was seeded at 1% cell concentration in the following: 3% w/v tryptic soy broth (TSB) medium with 1.5% w/v NaCl for S. aureus or M9 minimal medium for E. coli. The bacterial cultures were incubated at 37 °C for 24 h.
Another test protocol was conducted based on the ASTM E2562 standard test method for the quantification of bacterial biofilm grown with shear and continuous flow using the CDC biofilm reactor and Pseudomonas aeruginosa (P. aeruginosa, 10145). Figure 2 shows a visual image of the CDC biofilm reactor setup. Two 20-L carboys were sterilized with ethanol and sterile water. The reactor components and the modified carboy lids with connected ports and bacterial vents were cleaned and then autoclaved for sterilization. 20 L of 100 mg/L sterile TSB media were added to the carboy for the continuous input flow. The film samples on glass were cut into 1 × 1 cm square coupons, sterilized with ethanol and UV exposure, and installed into the rod holders. The reactor was then assembled, the tubing was connected, and the flow lines were clamped. The biofilm reactor was then activated. 500 mL of 300 mg/L sterile TSB media was aseptically added into the reactor vessel through the inoculation port, and then 1 mL of P. aeruginosa preculture (1 × 108 cells/mL) was inoculated into the vessel. The inoculation port was rinsed with sterile water. The reactor was stirred at 125 ± 60 rpm at 21°C ± 2 °C for 24 h to allow the culture to grow in the vessel. Then the flow lines were unclamped, and a continuous flow of TSB media was pumped into the reactor at a flow rate of 12 ± 0.2 mL/min for 24 h, with excess media being flowed into the waste carboy.

CDC biofilm reactor setup based on the ASTM E2562 test method.
Biofilm coverage analysis and CFU determination
For imaging and coverage quantification of the attached biofilm, the film samples in the bacterial studies were removed from the liquid culture and then carefully dipped into DPBS medium three times to remove the unattached planktonic bacteria. The samples were immersed in 0.1% w/v crystal violet aqueous solution for 20 min. The samples were removed and gently rinsed with deionized water to remove excess stain solution and then left to air dry. Images of the stained samples were obtained on the Zeiss Axioscope 5 at 50× objective magnification in dark field mode. Three different areas on each sample were imaged for triplicate measurements. The images were processed on the ImageJ software to obtain the binary images via intensity thresholding and then the biofilm area percentage by measuring the area fraction of the dark pixels.
Colony forming unit (CFU) values were determined for the film samples from the biofilm reactor study. The coupons were vortexed in 2 mL of phosphate buffer saline (PBS) media for 30 s to detach the biofilm from the coupons. 180 µL of PBS was added to the wells of a 96-well plate, and 20 µL of the suspensions was pipetted across the first lettered row. Then an eight-fold serial dilution was performed by transferring 20 µL of the suspensions stepwise from one well to the next. A spot plating technique was employed by transferring 5 µL onto a R2A agar plate, spotting the plate with three rows for triplicate results. The plates were left briefly on the workbench to allow for solvent evaporation and then were incubated at 35°C overnight. The colonies were counted using a manual colony counter, and the CFU was determined by multiplying the number of colonies to the dilution factor and then dividing over the volume used for spotting. The CFU among the samples were compared in terms of log values, and two trial runs for CFU determination were conducted and averaged for the final CFU values.
Results
Nanoparticulate film characterization
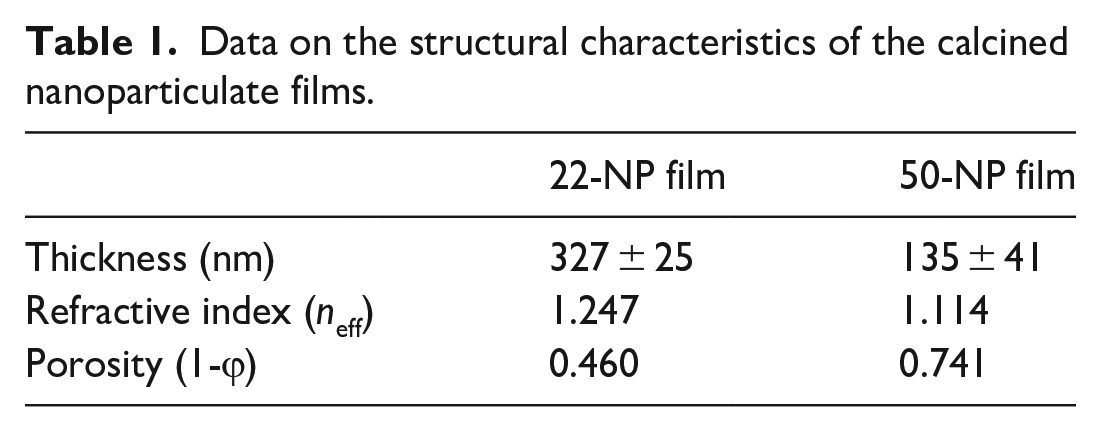
The structure of the nanoparticulate film after calcination was examined by a scanning electron microscope (SEM). Figure 3 displays the top view of the films generated with the two studied particle sizes and on glass and silicon substrates. The substrate was thoroughly covered with particles which assembled into densely packed films, with greater number of large cracks and pores observed on the glass samples. Table 1 lists the thickness, refractive index, and porosity for the calcined film structures. The thicknesses for the 22-NP and 50-NP films were 327 ± 25 and 135 ± 41 nm, respectively. The 22-NP film has a refractive index of 1.247 and a calculated porosity of 0.46. The 50-NP film has a refractive index of 1.114 and a porosity of 0.741.

SEM images of calcined nanoparticulate films based on various silica particle sizes and on glass and silicon substrates.
Data on the structural characteristics of the calcined nanoparticulate films.
Wettability and slippery property
The surface wettability of the films at each processing stage was investigated using a goniometer. The 22-NP and 50-NP films had contact angle values of 4.9° and 2.4°, respectively (Figure 4(a)). The contact angles increased to 114.9°–115.8° for 22-TPFS and 138.9° for 50-TPFS (Figure 4(b)), indicating that addition of the fluorinated silane compounds aided to decrease the surface energy of the films. The inset displayed a water droplet pinning effect when inverting 22-TPFS film. The contact angles changed slightly to 119.7°–119.6° for 22-LIS and 120.3°–120.2° for 50-LIS (Figure 4(c)). Upon closer inspection, a visible lubricant ridge can be seen on the 50-LIS film (Figure 4(d)).

Images of water droplets on 22 -nm and 50 -nm NP films on glass at various processing stages: (a) after calcination (22-NP, 50-NP), (b) after TPFS functionalization (22-TPFS, 50-TPFS) and (c) after lubricant infusion (22-LIS, 50-LIS). (d) Close-up images of droplets on LIS films.
The slippery property of the LIS films was tested by observing the droplet motion at a tilt angle of 15°. Water droplets slid off the surface quickly within 1 s for 22-LIS film and 2 s for 50-LIS film. (Figure 5(a)). Vegetable oil droplets slid at a slower pace than water, with minimal difference in the sliding rate between the two LIS films (Figure 5(b)). With ethanol on the 22-LIS film, the droplets slid for 1 s before stopping and slowly wetting the surface (Figure 5(c)). On the 50-LIS film, the ethanol droplet was pinned in place, indicating the loss of the slippery property.

Time-lapse images of droplets on LIS films tilted at 15° (22-LIS outlined blue, 50-LIS outlined green): (a) with water, (b) vegetable oil, and (c) anhydrous ethanol.
Lubricant retention and depletion
The lubricant stability in the LIS films was tested by monitoring the film transmittance over wash cycles under various conditions. The transmittance of the TPFS-functionalized film was found at 87% and would act as the control limit, marked by the magenta dotted lines in the graphs. When the lubricant is depleted from the film matrix, the transmittance would decrease toward the control limit. Thus, minimal transmittance change during the washing process indicates sufficient lubricant stability and retention. Firstly, transmittance of the 22-LIS film (Figure 6(a)) showed little change when the samples were washed with cold water (94%–96%) and with DPBS (92%–95%). With the room temperature water washes, the film transmittance remained at a plateau (94%–96%) until 80 washes, indicating an eventual lubricant loss. Transmittance greatly decreased after five washes in warm water. The transmittance change was more gradual with the ethanol-water mixture washes, decreasing from ~94% to 91% within 15 washes, plateauing before dropping toward the control limit after 50 washes. In comparison, the transmittance of the 50-LIS films (Figure 6(b)) was maintained at 94%–96% when the samples were washed with cold water, room temperature water and DPBS. Transmittance decreased from 95% to 87% within 15 warm water washes. With the ethanol-water mixture washes, the film transmittance plateaued within five washes until it gradually dropped within the next 25 washes.

Film transmittance monitored during 100-wash cycles with various solvent parameters and types: (a) for 22-LIS and (b) 50-LIS film. Magenta dotted line displayed the transmittance value of the TPFS-functionalized films which acted as the control limit.
Anti-biofouling in no-shear conditions
The anti-biofouling property of the LIS films was tested firstly in no-shear flow conditions. Figure 7 showed the area coverage percentages for the films at the different processing stages after bacterial growth and brief washing. Firstly, in the E. coli study (Figure 7(a)), the coverage on untreated glass showed 60.0 ± 18.8%. The 22 -nm NP and 50 -nm NP films contained 86.3 ± 8.0% and 70.6 ± 14.8% E. coli coverage, respectively. The 22-TPFS and 50-TPFS films were covered at 56.8 ± 12.2% and 61.4 ± 4.3%. The LIS films demonstrated the lowest E.coli coverage values, 9.1 ± 5.6% for 22-LIS and 10.0 ± 8.5% for 50-LIS. In the S. aureus study (Figure 7(b)), there was 88.3 ± 6.4% of S. aureus on the glass substrate. The 22-NP and 50-NP films had coverages of 56.8 ± 9.7% and 90.5 ± 7.3% respectively. The 22-TPFS and 50-TPFS films were 79.0 ± 9.6% and 40.4 ± 2.6% covered. The LIS films were low in S. aureus coverage with 6.9 ± 2.9% for the 22-LIS and 5.5 ± 1.9% for the 50-LIS.

Biofilm coverage percentages on films after a 24-h no-shear incubation period with: (a) E. coli and (b) S. aureus.
Performance under shear and continuous flow
The anti-biofouling property was further tested by installing the samples into a biofilm reactor system with shear and continuous flow, using P. aeruginosa as the bacterial species. The dark-field images (Figure 8(a)) showed that the bacterial biofilm (shown in violet) covered the surface of glass substrate, calcined films, and TPFS-functionalized films, while the coverage was scarcer on the LIS films. The 50-LIS film had a greater amount of biofilm present on the surface compared to the 22-LIS films. The area coverage percentage values are compared in Figure 8(b). The glass substrate had a coverage of 99.6 ± 0.3%. The 22-NP, 50-NP, 22-TPFS and 50-TPFS films were also high, ranging within 99.6–99.9 ± 0.1%-0.3%. The LIS films had the lower coverages, with 37.4 ± 19.4% for the 22-LIS and 70.0 ± 27.7% for the 50-LIS. Figure 8(c) showed the log CFU values to determine the biofilm amount on the films. The glass substrate contained a log CFU of 8.56 ± 0.21. The log CFU for the 22-NP and 50-NP films was 8.79 ± 0.11 and 8.73 ± 0.12, respectively. A log CFU value of 8.74 ± 0.21 and 8.88 ± 0.07 were determined for the 22-TPFS and 50-TPFS films. The LIS films showed lower values at 8.48 ± 0.05 and 8.49 ± 0.19 for the 22- LIS and 50-LIS, respectively.
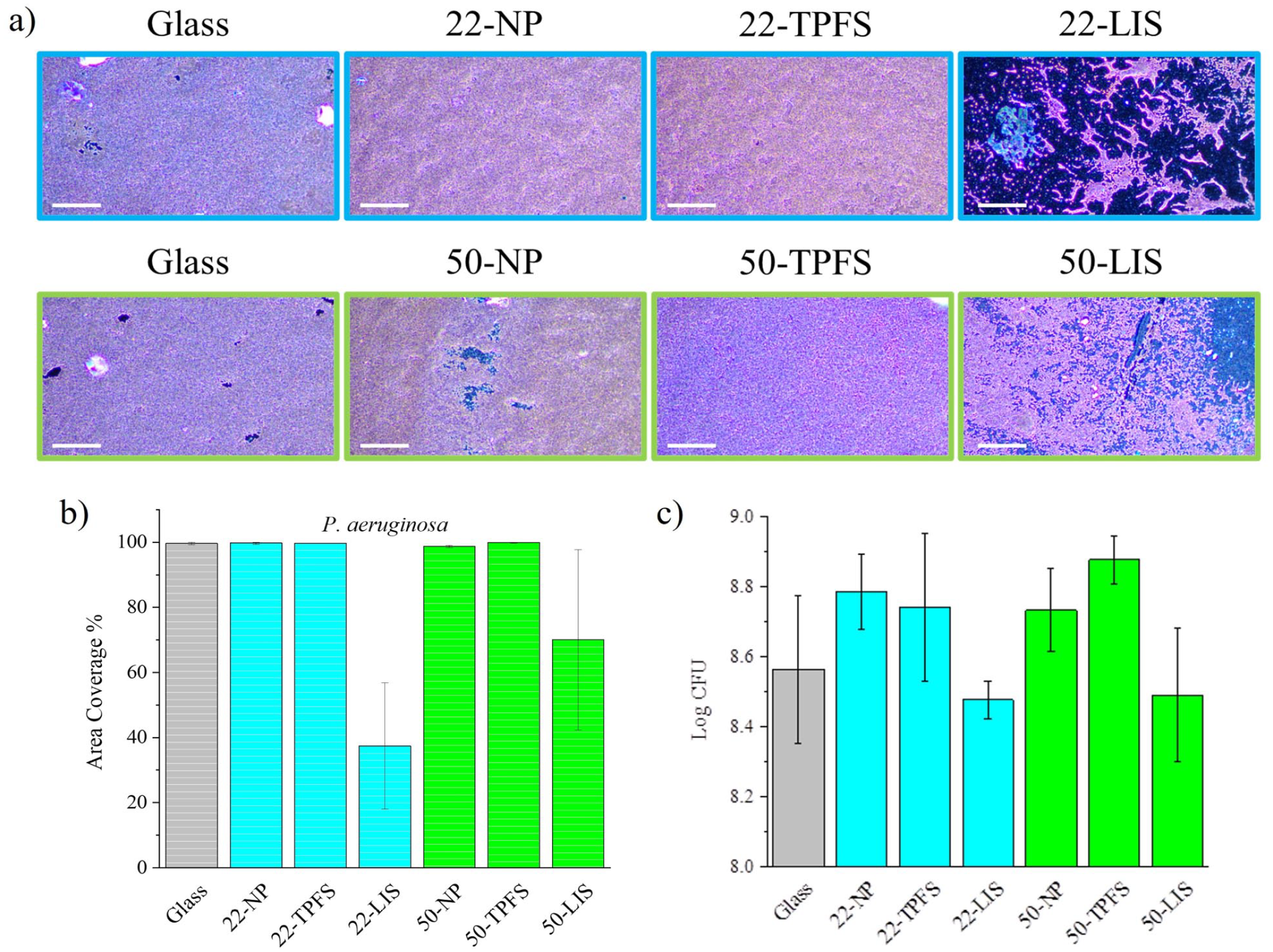
P. aeruginosa biofilm coverages and biofilm amounts on films after a 2-day growth in the biofilm reactor system under shear and continuous flow: (a) dark-field images of the film surface (the 22 -nn NP films outlined blue, 50 -nm NP films outlined green) containing crystal violet-stained biofilms, (b) area coverage percentages, and (c) log CFU values.
Discussion
The investigation on the morphology and properties of a silica nanoparticulate thin film is essential to determine if such film is a suitable base substrate for LIS fabrication. Three criteria for a stable LIS design were proposed by Wong et al. 28 (1) The lubricant liquid must be able to wick, wet and stably adhere within the substrate, which can be achieved by using a textured, rough and/or porous substrate to promote van der Waals and capillary forces. (2) The solid substrate and lubricant liquid must have stable preferential interactions and affinity with each other, which can be attained by modifying the surface chemistry and physical properties. (3) The lubricant liquid and working fluids must be immiscible. These criteria should be considered to determine if a stable LIS can be generated with the proposed substrate.
Firstly, structural morphology could help to determine the wicking capability of silica nanoparticulate film. The fabricated films were densely packed with thicknesses of less than 350 nm and porosity values of ~0.5 or above (Figure 3, Table 1). A thinner film was generated using the 50 -nm sized NPs. The ratio between attractive and gravitational forces would decrease with increasing particle size, in which the larger and heavier particles would be pulled down more easily and deposited less on the substrate. The porosity of 50-NP (1-ϕ = 0.741) was higher than 22-NP (1-ϕ = 0.460) due to looser packing and the increase in pore size during LbL assembly. A more porous film can then promote increased capillarity. 52 This was observed when comparing the water contact angle measurements (Figure 4(a), 22-NP = 4.9°, 50-NP = 2.4°). The silica nanoporous thin film could promote enhanced wicking via capillary force; however, the surface energies of the film and the wicking liquid must be compatible.
Low surface energy of the film is required to provide enough hydrophobic interactions to wick a low-surface energy lubricant liquid. FC-70 has a similar structure to polytetrafluoroethylene and can be assumed to have a similar surface energy (~18.5 mJ/m2). 53 Silica intrinsically has a high surface energy (340 mJ/m2) 54 but can undergo surface functionalization to modify the surface property. Silanes are commonly used as surface functional groups for silica because they readily react through hydroxyl groups on the target surface and introduce desired functional groups. In the study, a fluorosilane compound TPFS was used to react with silica films to introduce fluorinated groups and decrease the surface energy as shown in the water contact angle measurements (Figure 4(b), 22-TPFS = ~115.4°, 50-TPFS = 138.9°). Infusion with the fluorinated lubricant FC-70 slightly decreased the water contact angle by reducing the surface roughness (Figure 4(c), 22-LIS = ~119.7°, 50-LIS = 120.3°), but a slippery effect was present that repelled water and a hydrocarbon oil (Figure 5). This was on par with the work from You et al., 55 who tested FC-70 infused surfaces against various polar and nonpolar liquids. Upon TPFS functionalization, the films were thus more compatible to allow the fluorinated lubricant to wick and retain in the matrix.
Sufficient lubricant stability in the films is crucial to ensure the LIS functionality and longevity. In the wash cycle tests and film transmittance monitoring (Figure 6), 100-wash cycles with water at 5°C and 25°C and DPBS media showed minimal transmittance change and thus minimal lubricant loss. Thus, the LIS films are immiscible to water and salt solutions at ambient conditions. However, the LIS films were negatively affected under certain conditions. The film transmittance and thus lubricant stability greatly decreased in water at 60°C, which would be due to the decrease in lubricant viscosity upon exposure to higher temperatures. Additionally, ethanol and ethanol-water mixtures caused lubricant loss, reduced sliding effect, and eventual wetting on surface (Figures 5(c) and 6). Ethanol may be displacing the lubricant at the top surface as FC-70 has a low kinematic viscosity (12 cSt) 56 and thus could form strong polar interactions with the silica substrate. 57 Nevertheless, the LIS films demonstrated potential to employ a slippery property in ambient aqueous conditions.
Upon observing the slippery performance and lubricant stability, the LIS films were tested for anti-biofouling application. Biofilm formation begins at the point when the bacteria adhere to the surface where it will grow and build up the colonies. LIS films can prevent the adhesion of the bacteria to surfaces due to its slippery property, which was observed in the low biofilm coverage percentages and log CFU values in the no-shear bacterial and biofilm reactor studies (Figures 7 and 8). To note, reduced bacterial coverage can be achieved by generally employing substrates with hydrophobicity, low stiffness, increased roughness, and positively charged surfaces. 27 However, bacterial adhesion and growth is complex and depends on the bacterial characteristics and strains and a combination of substrate surface properties. Therefore, results for bacterial adhesion can vary greatly. LIS implementation, however, uses a lubricant top layer to separate the substrate surface from the impinging liquids, 35 which then prevents initial bacterial adhesion. Microbial growth was still evident on the LIS films since the FC-70 lubricant is non-toxic, and the bacteria can settle via gravitational sedimentation, but upon a brief rinse, the biofilm was easily removed. Therefore, the slippery property was the sole contributor for the low area coverages in no-shear conditions (~9%–10% for E. coli and ~5%–7% for S. aureus after 24-h incubation) and under shear continuous flow (37 and 70% for P. aeruginosa after 2 days in reactor). In comparison, Xiao et al. 30 found marginal performance from a FC-70 infused porous polymer substrate against marine zoospores and B. Amphitrite cyprids without shear flow compared to the uncoated substrate. There is potential to use our LIS design for no-shear conditions, but optimization would be necessary to employ it for water processing and distribution systems.
As such, the proposed LIS design has its limitations as observed throughout the work. Ethanol and ethanol-water mixtures could cause displacement of the lubricant layer, which reduced lubricant content and slippery effect. Increased temperatures decrease the FC-70 viscosity and cause lubricant loss as well. In addition, the films during the biofilm reactor study had low biofilm coverage and amount, but the error ranges were large over a 2-day trial. To improve the LIS design, a lubricant liquid with a higher kinematic viscosity could be used instead for a more stable lubricant containment within the matrix. 58 Xiao et al. 30 observed excellent results from the LISs containing Krytox 103 and Krytox 100 (<5% cyprids on surface) compared to FC-70 LIS (10%). Also, Sunny et al. 31 infused Krytox 100 into silica NP thin films, which showed little to no visible stained area and minimal protein absorption after flowing water over the substrates at 10 mL/min for 24 h. In addition, the LIS design can be optimized by increasing the film thickness and surface roughness 40 to contain the lubricant more efficiently. Furthermore, a component to kill the bacteria such as metal particles59,60 and antimicrobial drugs 61 could be implemented as an alternative when the LIS property becomes compromised.
Conclusion
In summary, the silica nanoparticulate film demonstrated potential as a base substrate to fabricate LISs for anti-biofouling applications. Stable films with nanoscale thickness (~135–325 nm) and high nanoporosity (0.46–0.74) were generated via LbL assembly and calcination. Upon modifying the surface chemistry with fluorinated silane compounds and infusing a fluorinated lubricant, stable LIS films were generated, in which water and hydrocarbons could be repelled from the surface. The transmittance of the LIS films has minimal changes when washed in cold water, room-temperature water and DPBS media demonstrating that lubricant within the matrix was retained well. Lubricant loss was evident in warm water and ethanol-water mixture. Nevertheless, the anti-biofouling effect of the LIS films was demonstrated, showing low biofilm coverage (5-10%) against various bacteria species under no-shear flow as well as low coverages (37 for 22-LIS and 70% for 50-LIS) and low log CFU values (~8.5) under shear and continuous flow. Further modification to improve lubricant retention or counter for the loss can aid in developing better coatings for biofilm mitigation.
Footnotes
Acknowledgements
Film characterization was performed at the Nanoscience Technology Center, Material Characterization Facility and CREOL cleanroom at the University of Central Florida. Manuscript was presented at the International Astronautical Congress, IAC 2022, Paris, France, 18-22 September 2022. Copyright by IAF.
Contributorship
YYLS was responsible for overall conceptualization, investigation and project administration of the work as well as the writing of the first and following drafts. AJ and AM helped in sample preparation, investigation, and data analysis. MS and PK helped with the ellipsometry measurement and concept. HR provided the work area and resources at the Biomolecular Research Annex to conduct the no-shear bacterial growth study. LBR and MEH provided the resources and mentorship to perform the biofilm reactor experiments at Kennedy Space Center. LZ and LBR were involved in the funding acquisition and supervision of the overall work. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported through the NASA Space Technology Research Fellowship [grant number GR103203].
Guarantor
YYLS, LZ