Abstract
To observe the metabolic changes and antialcoholic effect of Puerarin-PLGA nanoparticles (PUE-NP) in mice. PUE-NP was prepared and characterized by particle size distribution and morphology. The mouse models with acute alcoholism were established to observe their behavioral changes after alcohol poisoning. The expressions of biologically active enzymes such as CRE, BUN, AST, ALT in serum and SOD and TLR4 in liver of mice in each group were detected, and the pathological changes in liver and kidney tissues were observed by HE staining. The PUE-NP metabolism in mice was determined by in vitro release assay and HPLC. PUE-NP nanoparticles had good morphology and structure, and the mouse models with alcohol poisoning were established successfully. Compared with alcohol group, puerarin and PUE-NP increased the disappearance latency time of righting reflex, and the recovery time of righting reflex was significantly shortened. Water maze results showed that Puerarin and PUE-NP had inhibitory effect on impaired memory. HPLC results showed that PUE-NP reached its peak in mice after 1 h, and the content percentage was twice that of puerarin preparation alone, and the distribution time of puerarin concentration in vivo was prolonged, indicating that PLGA nanoparticles had a loading and slow-release effect on puerarin and increased the bioavailability of puerarin in mice. In addition, compared with the alcohol group, Puerarin and PUE-NP improved serum ALT, AST, CRE, and BUN levels in mice, enhanced SOD activity in liver, and inhibited TLR4 expression. The effect was better in the PUE-NP group than in the Puerarin group. PUE-NP delayed the release and metabolism of Puerarin and had better effect in the treatment of the alcoholic liver and kidney injury.
Introduction
Radix Puerariae is the root of leguminous plants in the wild pueraria and kudzu, and it’s one of the commonly used Chinese herbal medicines. 1 Puerarin is one of the main components of pueraria 2 and has a variety of pharmacological effects including antioxidant, improvement of cardiovascular and cerebrovascular diseases, regulation of hypertension, alleviation of atherosclerosis, and alleviation of alcoholic hepatitis.3–6 Moreover, Puerarin has non-toxic, non-sensitive and non-teratogenic effects in animals, and it can resist the liver damage caused by liver toxin. 7 However, the chemical structure characteristics of Puerarin lead to poor water solubility and lipid solubility, which results in poor oral absorption, low bioavailability, and limited clinical application. At present, there are only Puerarin injections and Puerarin eye drops on the market, and they require frequently or high-dose injections or drops due to their short half-life. 8 Therefore, it is necessary to develop an effective drug delivery system to prolong the drug action time and improve the bioavailability of Puerarin.
Solid lipid nanoparticles and polymer nanoparticles are the focus of the clinical drug delivery system research. 9 Biodegradable nanoparticles have the advantages of controlled release, biocompatibility, subcellular size, blood stability, non-inflammation, and non-immunogenicity. It has become the first choice for the nano-encapsulation of such bioactive factors as drugs, proteins, DNA, and imaging agents.10,11 Nanotechnology and nanoparticles are widely used in catalysis, environmental remediation and biomedicine, Buazar 12 reported on the synthesis of biocompatible nano silica and its application in green soil remediation. Seaweed synthesis also has a wide surface area and catalytic capacity. 13 Green nanotechnology has a synergistic antibacterial effect on selected pathogens. 14
Among the common biomaterials, polylactic acid glycolic acid (PLGA) and PLGA composite nanoparticles have been well applied in targeting, imaging and therapy,15–17 and PLGA can be completely degraded in aqueous solution. Studies have reported that PLGA with higher glycolic acid content greatly improves the emulsification performance of protein-loaded solid lipid nanoparticles and the encapsulation efficiency and stability of emulsion have been improved. 18 Multiple studies have proved that PLGA shows low toxicity in cells and the liver and kidney tissues within 7 days. 19 In the past decade, PLGA nanoparticles have been synthesized by the nano precipitation method, 20 emulsification-diffusion method, 21 emulsification-evaporation method 22 and solvent evaporation method 23 respectively. The oral bioavailability of Puerarin nanoparticles is 2.83 times higher than that of raw Puerarin after pure nanoparticles are prepared by the emulsion solvent evaporation and freeze drying method. 23
In this study, PLGA nanoparticles were used as synthetic materials, and the optimal material ratio was obtained to prepare the PUE-NP preparations by the preliminary optimized experiments. 24 The metabolic changes at different time in vivo and in vitro, biological effects of anti-alcohol and liver protection and inflammatory molecular changes of the PUE-NP preparations were observed by HPLC in the mouse models with alcohol poisoning, which provided the theory and basis for the biological effect of Puerarin on the mice with alcohol poisoning and the production of new preparation.
Experimental section
Chemical reagents and instrumentation
Puerarin standard and Poly (lactic-co-glycolic acid, 1000) (PLGA75/25) was purchased from Chengdu Institute of Organic Chemistry, Chinese Academy of Sciences. The ALT, AST, BUN, CRE, and SOD kits were purchased from Nanjing Jiancheng Biotech. Histone extraction kits were purchased from Shanghai Yase Biotechnology Biotech. The male KM mice were purchased from Dashuo Experimental Animal Center (Sichuan, China, SCXK-2020-030).
Animals and experimental design
Twenty-four male mice aged from 6 to 8 weeks and weighing (25 ± 5) g were fed with adequate food and water in a dust-free room with the humidity of 50–60% at the room temperature of 18°C–22°C. The mice were randomly divided into four groups with six mice in each group, and the mice in the same group were placed in a cage (545 × 395 × 200 mm). After 7 days of adaptive feeding, they were given intragastric administration for 14 consecutive days, once a day. The mice in the control group were received normal saline by intragastric administration. In the alcohol group, the mice were given normal saline and alcohol (50%v/v, 0.15 mL/10 g body weight). In the puerarin group, the mice in the puerarin group were treated with puerarin (0.3 mg/10 g body weight) and alcohol (50%v/v, 0.15 mL/10 g body weight). In the PUE-NP group, the mice were treated with PUE-NP (0.5 mg/10 g body weight) and alcohol (50%v/v, 0.15 mL/10 g body weight) by intragastric administration. The treatments (normal saline/puerarin/PUE-NP) were given 30 min before administration of alcohol. The experimental protocol was approved by the Animal Ethics Committee of Chengdu Medical College (IACUC-20-068).
Preparation of Pue-PLGA-NPs
PUE-NP was prepared according to previous experiments: 0.1 g PLGA was dissolved in 10 mL acetone to prepare organic phase, 0.05 g Tween 80 was dissolved in 10 mL distilled water to prepare water phase, and 0.01 g Puerarin was dissolved in 100 mL distilled water to prepare 100 μg/mL Puerarin solution for later use. Organic phase, aqueous phase and Puerarin solution were mixed and stirred with magnetic force for 13 min with the ratio of 1:2:1.4. Then the mixture was added with 40 mL deionized water for magnetic stirring for 1.5 h after ultrasonic shock for 10 min. The 30-min water bath at the constant temperature of 65°C was used to centrifuge and discard the supernatant. The PUE-NP suspension was prepared by ultrasonic shock for 5 min and the PUE-NP freeze-dried powder was obtained by freeze-drying suspension.
Characterization of puerarin nanoparticles
The prepared PUE-NP freeze-dried powder was added into deionized water to prepare the PUE-NP solution. The particle size and Zeta potential distribution were collected and recorded by the particle size analyzer. The morphology and particle size distribution of PUE-NP were observed under the transmission electron microscopy by negative phosphotungstic acid staining. 25
Preparation of Puerarin standard curve
About 100 μg/mL standard Puerarin stock solution was prepared by dissolving 0.01 g standard Puerarin in methanol and diluted into 0.5, 1, 5, 10, 15, 20,and 25 μg/ mL standard solution respectively. Two hundred microliters of blank plasma were collected from seven mice and 10 µL standard solution was added into the plasma respectively for direct injection analysis by high performance liquid chromatography (HPLC) with the chromatographic column of C18 column (4.6 mm × 250 mm, 5 μm), the flow rate of 1.0 ml/min detector, the UV wavelength of 250 nm, the column temperature of 25°C, the injection volume of 20 µL, the mobile phase with the methanol and acetonitrile ratio of 90:10 (V/V). Peak height, peak area and retention time were recorded and Puerarin standard curve 1 was drawn.
The standard reserve solution with a concentration of 1 mg/mL was prepared by dissolving 50 mg Puerarin in 50 mL methanol and then diluted with methanol into standard series of Puerarin with a mass concentration of 50, 100, 200, 400, and 800 μg/mL respectively. The Puerarin samples were determined by Ultraviolet spectrophotometer at 260 nm, and then Puerarin standard curve 2 was drawn.
Evaluation of drug release in vitro
In the study of Zhang et al. 25 the release of PUE-NP was investigated by dialysis. PUE-NP dry powder was dissolved and put into a pre-treated dialysis bag. The dialysis bag was completely immersed in PBS solution with pH 7.4 at 0.083, 0.167, 0.333, 0.667, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, and 15 days respectively, and 3 mL dialysate was collected to measure the spectrophotometric value (at the same time, 3 mL dialysate was added), and the in vitro release curve was prepared to observe the in vitro release of PUE-NP.
Behavioral experiment
The righting reflex test was used to evaluate the inebriation of mice. After the mice were lavage alcohol and drugs, they were gently placed in the animal cage with their backs down. If the supine position was maintained for more than 30 s, the righting reflex was considered to have disappeared, and the mice were recorded from the time of drinking to the disappearance of the righting reflex. The latency time (the time from the modeling to the disappearance of the righting reflex) and the recovery time of the righting reflex (the time from the modeling to the recovery of the righting reflex). Morris water maze was used to test the memory and cognitive impairment of alcohol in mice. The mice were tested on the water maze after four times of swimming training, and the incubation period and path of disembarking were recorded. The incubation period of disembarking was the time required for the mice to successfully find the platform for the first time after entering the water. 26
Biochemical analysis
After 14 days of intragastric administration, the mice were fasted for 12 h and sacrificed after being anesthetized. The blood samples were collected and the supernatant was obtained by centrifugation. The liver and kidney tissues were cryo-preserved in the refrigerator with the ultra-low temperature of −80°C for later analysis. The contents of urine creatinine (CRE), blood urea nitrogen (BUN), alanine transaminase (ALT) and aspartate transaminase (AST) in mouse serum and the contents of superoxide dismutase (SOD) in liver were determined according to the instructions of the kit.
Determination of PUE-NP metabolism in vivo
After 3 days of adaptive feeding, 114 mice were randomly divided into puerarin group (n = 54), PUE-NP group (n = 54) and blank group (n = 6). Each group was divided into nine subgroups, including three mice in puerarin group and PUE-NP group and one mouse in blank group. Before the experiment, fasting and water treatment for 6 h. Puerarin group and PUE-NP group were given puerarin (0.3 mg/10 g bw) and PUE-NP orally (0.5 mg/10 g bw), and blank group was given the same amount of normal saline intragastric. Mice in each group were sacrificed at 10 min, 30 min, 1, 2, 3, 4, 6, 12, and 24 h after intragastric administration, and blood samples and liver and kidney tissues were taken. The percentage of the Puerarin concentration was set as the ordinate and the time of gavage was set as the abscissa. The scatter plot was drawn to obtain the curve of Puerarin metabolism in plasma, liver, and kidney.
Western blot analysis
The protein samples were extracted from liver tissue homogenate by using a protein extraction kit and quantified by using the kit. The isolated proteins were transferred to PVDF membranes by SDS-PAGE gel electrophoresis. It was sealed with a blocking solution (TBS, 20% Tween 20, containing 5% skimmed milk) and then incubated overnight with primary antibody (TLR4, 1:1000, Bioss, Beijing, China) at the temperature of 4°C. After being washed with TBST (TBS, 20% Tween 20) buffer for three times, HRP coupled secondary antibody was incubated for 1 h. Then the enhanced chemiluminescence (ECL) assay kit (BioSharp, Shanghai, China) was used for development and Quantity One (Bio-RAD, Hercules, CA, USA) was used to analyze the protein bands.
Histopathological observation
The liver and kidney of mice were fixed in paraformaldehyde solution and dehydrated and embedded in paraffin. Then the liver and kidney were sectionalized in 5 μm coronal section. After HE staining, the liver and kidney tissues were sectionalized under the light microscope to observe their morphological changes.
Statistical analysis
IBMSPSS21.0 software was used to analyze the results. The measurement data were expressed as mean ± standard deviation (
Results
Nanoparticle diameter characterization
The particle size measurement and zeta potential measurement results of the prepared PUE-NP are shown in Figure 1(a). The average particle size was 172.7 nm, which has been well evaluated to pass through the human mucosa and various barriers. As can be seen from Figure 1(b), the PUE-NP dry powder particles were full under the mirror with regular shape and uniform size.

Nanoparticle diameter characterization: (a) the particle size measurement results of PUE-NP, (b) TEM of PUE-NP dry powder.
Standard curve of Puerarin
Considering the difference of PUE-NP content in the dialysate of mice in vivo and in vitro, two standard curves were developed to obtain an appropriate linear range. Standard curve one obtained by HPLC is shown in Figure 2(a). The results of R2 = 0.9992, the relative standard deviation of retention time, RSD = 1.65% and a good linear relationship between Puerarin concentration and absorbance were used to determine the Puerarin content in PLGA nanometers in each tissue.

Standard curve of Puerarin. (a) Puerarin standard curve 1 (HPLC). (b) Puerarin standard curve 2 (ultraviolet spectrophotometry).
The standard curve two made by S. spectrophotometer is shown in Figure 2(b), the standard curve equations were y = 0.0016× + 0.0264 and R2 = 0.9995. The linear relationship was well correlated and were used for the calculation of the Puerarin concentration in PLGA nanometers in in vitro release experiments.
In vitro release experiments
The in vitro release curve of Puerarin in PLGA materials is shown in Figure 3. The Puerarin loaded in PUE-NP was not released within 0.0083, 0.167, 0.333, and 0.667 day respectively, but it was gradually released from day 1, and the release peaked at day 5 and then remained a relatively stable state.

In vitro release curve of PUE-NP.
Behavioral experiment and analysis of liquor effect
Righting reflex experiments
The results based on the recorded incubation period after drinking alcohol are shown in Figure 4. Under the same dose of drugs, the disappearance latency of righting reflex in PUE-NP group was significantly increased (p < 0.05) and recovery time was significantly shortened (p < 0.01) compared with alcohol group. In addition, compared with alcohol group, the disappearance latency of righting reflex was increased and recovery time was shortened in puerarin group, but the therapeutic effect was significantly lower than that in PUE-NP group.
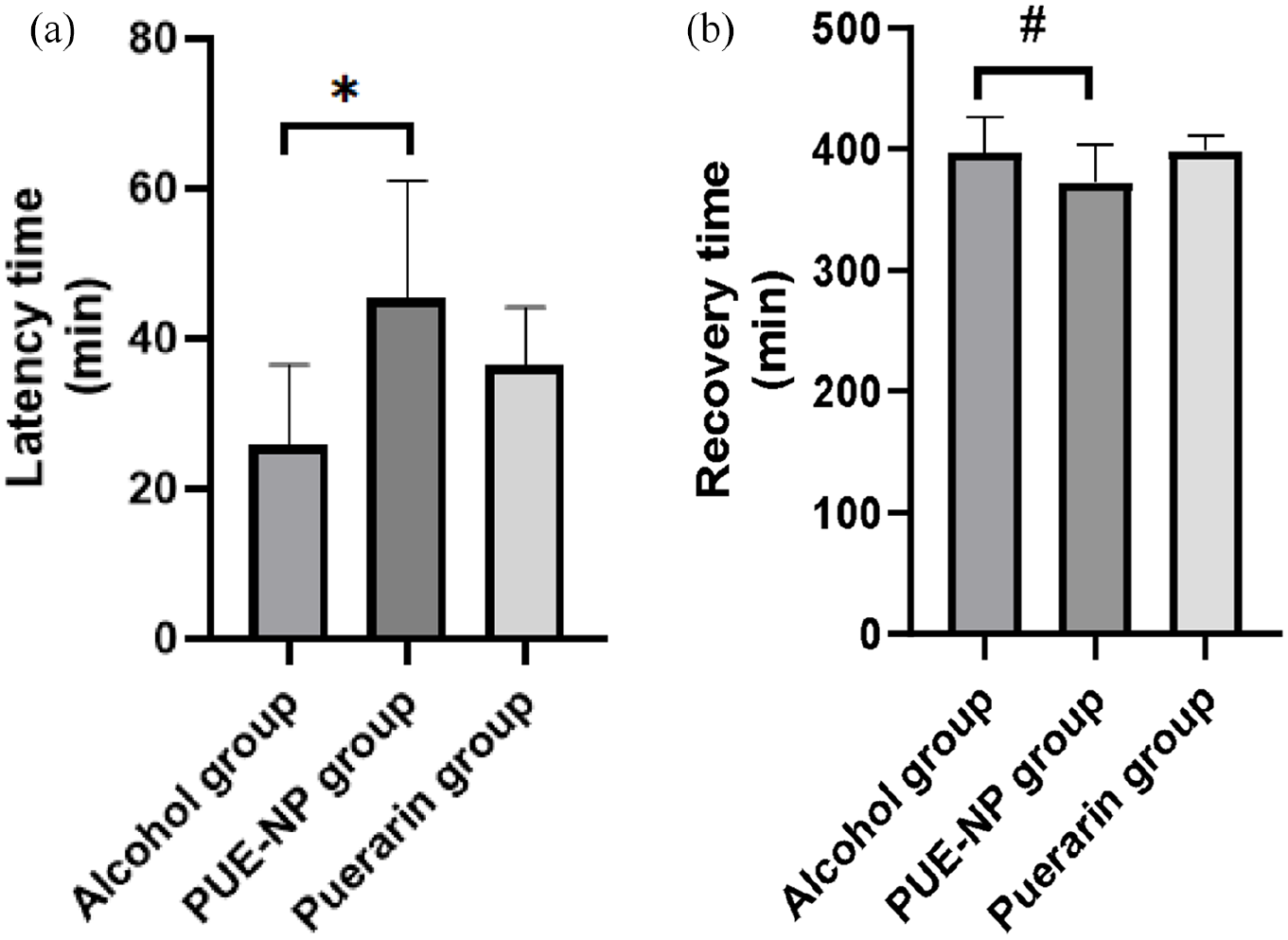
Sobering up test in mice: (a) latency time (min) and (b) recovery time (min). Values are presented as
Water maze experiments
Morris water maze results showed that it was showed that the exploration strategy was linear in the blank group, random in the alcohol group and of trend type in the Puerarin and PUE-NP groups, which indicated that puerarin and PUE-NP had better memory behaviors than the alcohol group (Figure 5(a)). The escape latency of the alcohol group was significantly longer than that of the blank group (Figure 5(b)). (p < 0.01). Compared with the alcohol group, the escape latency of the PUE-NP group and puerarin group was significantly shorter (p < 0.01), and the escape latency of the PUE-NP group was significantly shorter than that of the puerarin group (p < 0.01). The results showed that puerarin and PUE-NP improved the spatial learning and memory ability of mice under the influence of alcohol, while PUE-NP had better effect than puerarin.

Morris water maze results: (a) the swim tracks of the mice, (b) escape latency(s). Values are presented as
Detection of CRE, BUN, ALT, AST and SOD
The mice were sacrificed after being anesthetized 14 days after gavage, and the serum of the mice in each group was collected and tested according to the instructions of the kit. The results are shown in Figure 6. The levels of kidney markers CRE and BUN and liver markers ALT and AST in the alcohol group were significantly higher than those in the blank group (p < 0.01), indicating that the liver and kidney of the mice was severely damaged under the action of alcohol. Compared with the alcohol group, the CRE, BUN, ALT, and AST levels of the mice in the PUE-NP group and the puerarin group were decreased (p < 0.05), and the PUE-NP group was significantly lower than the puerarin group. In addition, under the effect of alcohol, the level of SOD in mice was significantly decreased by alcohol (p < 0.01). SOD level in PUE-NP group and puerarin group was significantly increased (p < 0.05), and PUE-NP group was higher than puerarin group. Our results suggest that puerarin and PUE-NP can reduce oxidative stress state, while PUE-NP is more effective.

Determination of CRE, BUN, ALT, AST, and SOD: (a) CRE, creatinine, (b) BUN, blood urea nitrogen, (c) ALT, alanine transaminase, (d) AST, aspartate transaminase and, (e)SOD, superoxide dismutase. Values are presented as
Determination of the PUE-NP metabolism in vivo by HPLC
As shown in Figure 7, the peak of Puerarin appeared at 7–8 min. The concentration of Puerarin in the mouse plasma, liver and kidney in the Puerarin and PUE-NP groups were recorded at 10 min, 30 min, 1, 2, 3, 4, 6, 12, and 24 h after 14 days of gavage. The metabolic changes of Puerarin in plasma (A), liver (B), and kidney(C) are shown in Figure 8 respectively. The results showed that the content of Puerarin was significantly higher in the PUE-NP group than in Puerarin group (p < 0.05).

HPLC diagram of PUE-NP.
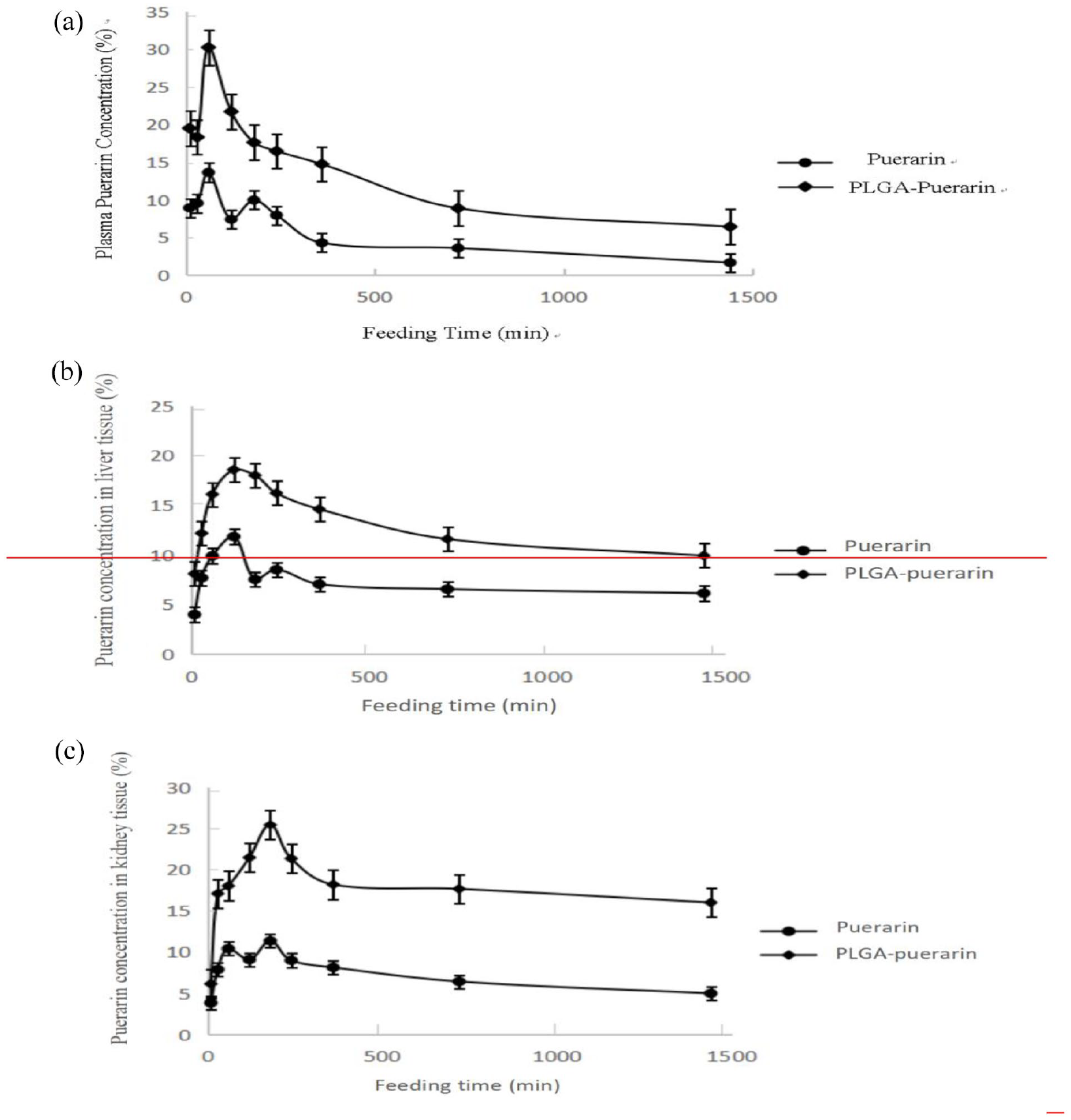
The concentration-time curves of Puerarin in plasma (a), liver (b), and kidney (c).
Western blot
The TLR4 and β-catenin expression levels in the liver tissue of mice in each group were shown in Figure 9. The results showed that the TLR4 and β-catenin expression levels were higher in the alcohol, Puerarin and PUE-NP groups than in the blank group, and they were lower in the alcohol group than the Puerarin and PUE-NP groups. The TLR4 and β-catenin expression level was the lowest in the PUE-NP group.

Western blot analysis of TLR4. Values are presented as
Histopathological alterations
The liver and kidney tissues of mice were stained with HE, and the HE results were shown in Figure 10 The liver(A) and kidney(B) fibrosis and eosinophilia were observed in the alcohol, Puerarin and PUE-NP groups, and they were the most obvious with the accompany of the increase of vacuoles in the alcohol group. The texture of the liver tissue in the PUE-NP group was more uniform than that in the alcohol group. The endothelial cell and mesangial cell proliferation with neutrophil infiltration were observed in the alcohol, Puerarin and PUE-NP groups and the most obvious in the alcohol group, and they were more obvious in the Puerarin group than in the PUE-NP group.

The results of HE staining from the mouse tissues (40×): (a) liver, (b) kidney. (The arrow shows the site of tissue change: As shown in (a), compared with the blank group, vacuoles in the alcohol group increased, liver connective tissue fibrosis and eosinophils increased in the other groups, and the degree of puerarin group was higher than that in the PUE-NP group. As shown in (b), compared with the blank group, the alcohol group had thickened glomerular balloon wall and fibrosis, neutrophil infiltration accompanied by cell proliferation in glomerular capillaries, while the other groups had neutrophil infiltration, and the degree of puerarin group was higher than that of PUE-NP group).
Discussion
WHO pointed out the rising trend of alcoholic liver disease (ALD) in China and ALD has become the second leading disease of liver damage after viral hepatitis in Global Status Report on Alcohol and Health (2018). 27 Puerarin is an isoflavone derivative and has definite curative effect on liver diseases. However, its structure and short half-life limit its pharmacological action. In addition, puerarin has been shown to have anti-alcohol, liver-protecting and anti-inflammatory effects in previous studies.29–31 In our experiment, this results were confirmed that PUE-NP had better anti-alcohol, liver-protecting and anti-inflammatory effects on mice with alcoholic liver poisoning. The possible reason is that PLGA nanoparticles delayed the action time of drugs in the target organs, which indirectly enhances the pharmacological effect of Puerarin, Puerarin and PUE-NP also have certain protective effect on the kidney tissue damage. Dong et al. 28 reduced the toxicity of acetone by the solvent evaporation method during the process of the preparation of PUE-NP in their study. After intravenous injection, Dong prepared puerarin solid lipid nanoparticles by adhesion solvent evaporation method (RGD sequence), the bioavailability and content of nano-puerarin were improved obviously under the condition of lower pH value and faster release rate, the Dong’s puerarin nanoparticles, which can obviously improve the water solubility, biological activity and half-life of puerarin, and improve the ability of liver targeted drug delivery. In vitro release results showed that PUE-NP began to be released on day 1 and peaked on day 5, which confirmed the sustained release effect of PLGA. Figure 6 suggested that PUE-NP could reduce the time of intoxication, inhibit the damage to the liver and kidney tissues by alcohol and prolong the residence time of the drug in plasma and tissues, which proved that PLGA nanoparticles improved the bioavailability of Puerarin.
We found that the mixing sequence of water phase, organic phase and Puerarin had great effect on the accuracy of the experiment. In the organic phase, acetone was used as solvent and reacted easily with tween 80 emulsifier in water phase to form flocculent precipitate, which had wrapped puerarin and not easily dissolved with puerarin. We also found that a small amount of the mixture of water and puerarin was slowly poured into the organic phase and stirred to disperse the nanoparticles evenly. The PUE-NP prepared by this method was characterized and the results showed that the PUE-NP particles were moderate in size and uniform. PLGA was not detected in mice by HPLC, and PLGA was analyzed to decompose into water and CO2 in vivo, which also confirmed that PLGA was safe and effective as a nano-synthetic material.18,28
At present, studies on puerarin and puerarin antialcoholic preparations have emerged one after another, daidzein and glucuronic acid ester is the main metabolites of Puerarin, mainly through the urine and feces. These saponins have protective effect on liver tissue immune damage. Puerarin can protect liver injury through gastric absorption and induce apoptosis of activated hepatic stellate cells,29,30 it could also reverse the chemically-induced liver fibrosis effectively. Studies have shown that when endotoxin from intestinal tract reaches the liver via portal vein, the reticuloendothelial system in liver (mainly Kupffer cells) can recognize and phagocytosis endotoxin through Toll-like receptor 4 (TLR-4) on the cell surface, which was digested and removed by lysosomes. Knockout of TLR 4 gene can significantly reduce the degree of alcoholic steatohepatitis in the mouse model, 31 and this conclusion is also consistent with the TLR-4 molecular experiment.
Conclusion
Puerarin and PUE-NP both have the effect of antialcoholism, and PUE-NP is better than puerarin. PUE-NP delays the release and metabolism of puerarin, and has a better effect on the treatment of alcoholic liver and kidney injury. Taken together, PUE-NP is a new type of effective anti-alcohol and liver protection preparation. We can improve the alcohol-induced liver and kidney damage through the effect of nano-coated Puerarin, which provides ideas for the targeted treatment of the liver and kidney tissue damage in future and a theoretical basis for exploring the efficacy of Puerarin.
Footnotes
Author note
All authors have read the manuscript and agreed to submit it in its current form for consideration for publication in the Journal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2019 National and Provincial College Students’ Innovation and Entrepreneurship Training Program of Chengdu Medical College (201913705010), and Chengdu Science and Technology Bureau focuses on research and development support plan (2019-YF09-00097-SN).
