Abstract
Background:
We aim to coat a novel polyvinyl benzyl chloride-D-glucaro-1, 4-lactonate polymer-coated bile duct stent for anti-biliary mud deposition and investigate its in vivo and in vitro impacts. Biliary mud deposition is a leading cause of plastic biliary stent obstruction after its placement. Orally administrated D-glucaro-1, 4-lactonate is a specific competitive inhibitor of β-glucuronidase that causes biliary mud deposition.
Methods:
In this study, the stents coated with polyvinyl benzyl chloride-D-glucaro-1,4-lactonate polymer (PVBC-DGL) were placed in an ex vivo bile duct model perfused with porcine bile and observed every week until completely blocked. Post establishing the model of bile duct stenosis in piglets, stents are observed through endoscopy, hematology, patency time, and pathological changes within 6 months.
Results:
The 70% PVBC-DGL stents achieved the highest percentage of the inhibitory effect when the drugs were completely released in vitro. Results were obtained from 14 pigs (5: no coating (original), 4: 0% coating, and 5: 70% coating). The overall patency time of the stents was prolonged in all groups; however, the group with 70% coated stents showed a significantly prolonged patency time as compared to no coating (original) and the 0% coating groups in pigs (23.4 ± 1.8 vs 11.2 ± 2.1 w (p = 0.05); 23.4 ± 1.8 vs 10.5 ± 2.5 w (p = 0.05), respectively).
Conclusion:
The stents with 70% PVBC-DGL better prevent and control the deposition of bile mud and prolong the patency time of stent placement in the subject animals and may be proposed for further clinical trials in patients.
Graphical Abstract

Introduction
Benign and malignant biliary stenosis1,2 is a common clinical disease. When surgical intervention is impossible, endoscopic biliary stent placement3–5 becomes an effective method to relieve biliary obstruction and improve life quality. Metal stent implantation has a large internal diameter, good drainage effect, and resistance to restenosis, but at the same time, these stents are expensive and difficult to remove after implantation. These are commonly used for malignant biliary stenosis.6–8 In contrast, plastic stents are cheap, convenient to be replaced repeatedly and easy to remove after implantation. Moreover, plastic stents could be used for both benign and malignant stenosis. Although the diameter of plastic stents is small, it is easy to get restenosis due to biliary mud deposition in a very short time.9,10 Thus, plastic stents often need to be replaced repeatedly under endoscopy, which is not only painful but also expensive and risky.11,12 To deal with such issues, it is crucial to prevent the deposition of biliary mud in stents and prolong the patency time of plastic stents as much as possible. 7
Current studies have found that biliary mud deposition13,14 caused by bacterial infection is the leading cause of restenosis after plastic stent implantation. β-glucuronidase plays an important role in stricture and gallstone formation after biliary stent implantation.15,16 To date, any study about prolonging the patency time of biliary stents by inhibiting β-glucuronidase activity and reducing biliary mud deposition has not been reported. Therefore, we aim to prove our speculation that inhibiting β-glucuronidase may prevent biliary mud deposition and prolong biliary stents’ patency time. Sacco et al. 17 reported that polyvinyl benzyl chloride-D-glucaro-1,4-lactonate (PVBC-DGL) polymer is a highly effective and non-toxic inhibitor of β-glucuronidase that does not get absorbed in the intestine. 18
In this study, PVBC-DGL is coated on the biliary stent to explore its preventive effect on biliary sludge deposition in an ex vivo bile duct model perfused with porcine bile. A double stenosis model of the bile duct is used in piglets to investigate the safety and efficacy of this novel stent, along with its anti-deposition effect.
Methods
Preparation of PVBC-DGL-coated biliary stent
Polyvinylbenzyl-D-Glucaro-(1,4) lactonate (PVBC-DGL) polymer was synthesized by a modified chemical method described elsewhere 17 (Institute of Pharmacy, Shanghai Changhai Hospital of Naval Medical University, China). D-Glucaro (1, 4) lactone (5 g, 26 mmol) was dissolved in a mixture of methanol and water (100:1) at 4°C and neutralized by adding 20% Cesium bicarbonate. From the air-dried mixture, Cesium (Cs) salt was obtained at 80°C under vacuum. Then PVBC (1.8 g, 12.3 mmol in N, N-dimethyl-formamide (DMF)) was added to Cesium salt of D-Glucaro (1, 4) lactone (4 g, 12.3 mmol) in the presence of a catalyst, CsI (300 mg) to obtain an orange colored solution, further stirred/incubated for 7 days at 50°C. The final product was air-dried, washed with a sequence of 1 M KOH, 1 M HCl, water and methanol (100 mL each, 4°C), and vacuum dried at 80°C (Figure 1(a) and (b)).

Preparation of PVBC-DGL-coated biliary stent: (a) molecular formulas of PVBC-DGL. D-Glucaro 1,4-lactone (DGL) is combined with polyvinyl benzyl chloride (PVBC) via a carboxyl bond using a bimolecular nucleophilic substitution reaction, (b) synthetic sample of PVBC-DGL, and (c) coaxial electrospinning device diagram.
We used 8.5 Fr. original straight plastic stents (Nanjing Microtech Medical Industry Co, Ltd, Nanjing, China) that were 3.0 mm wide and 60 mm long. Mixed uniform solution carrier (polylactic acid: lactic acid glycolic acid copolymer at 3:4 molar ratio) as the “shell” layer and the PVBC-DGL drug as the “core” layer in a coaxial electrospinning device (Figure 1) to obtain the “shell-core” structure of the drug-carrying fiber membrane, which was coated on the sterilized bare bile duct stents. Six drug-coated biliary stents (coating thickness 0.20 ± 0.01 mm) with different drug contents of 0%, 10%, 30%, 50%, 70%, and 90% of PVBC-DGL were prepared (Figure 2(a)).

Coated stents and the degradation test in vitro biliary perfusion system model: (a) original, 0%, 10%, 30%, 50%, 70%, and 90% PVBC-DSL coating from left to right and (b) the stents in vitro biliary perfusion system model.
Stents test in an ex vivo bile duct model perfused with porcine bile
The stents (original, 0%, 10%, 30%, 50%, 70%, and 90% PVBC-DGL coating) were perfused with porcine bile and placed into the ex vivo bile duct model as previously described. 19 The pig bile sterilized with ultraviolet light in the liquid box was added to E. coli and cultured at 37°C for at least 2 h until its content was 2–6 × 106. Then the bile was allowed to flow through the valve and enter the sample experimental area for the bile perfusion test (Figure 2(b)). The stents were taken out and observed every week until they were completely blocked.
Establishment of the porcine biliary stenosis model and the stents placement
All experimental procedures were approved by the Animal Care and Use Committee of Shanghai General Hospital, affiliated to Shanghai Jiao Tong University. A total of 19 pigs aged 3–4 months and weighing 15.9–27.3 kg were included in the study. The pigs were randomly divided into three stent groups: six with no coating, six with 0% coating, and seven with 70% coating. The porcine biliary stenosis model was established as previously described 20 (Figure 3(a)). Endoscopic retrograde cholangiopancreatography (ERCP) was used to find the common bile duct, and a stent was placed into the porcine biliary stenosis model under the endoscope. An X-ray examination was performed after each implantation, and the stent position was determined (Figure 3(b)).

ERCP stent placement and X-ray examination: (a) the X-ray angiography before stent placement under endoscopy, the arrow shows the narrow part of the porcine common bile duct and (b) shows the stent placement during X-ray examination, the arrow indicates the placement of stents.
The inhibitory effect of PVBC-DGL on the β-glucuronidase activity in bile
Measurement of PVBC-DGL concentration in bile: To configure the standard DGL aqueous and methanol solution, scan the DGL solution repeatedly with a full wavelength of an ultraviolet spectrophotometer, and get an approximate DGL absorption spectrum. The concentration of PVBC-DGL was obtained by measuring the optical density of bile under this spectrum and comparing it with the standard DGL solution.
Measurement of β-glucuronidase activity: According to the Fishman method, 21 phenolphthalein glucoglycane glycyrrhizic acid was used as the substrate, kept warm under pH 7.0 (bacterial) and hydrolyzed by β-glucuronidase. Phenolphthalein was colored in an alkaline solution. The extinction of phenolphthalein, determined by a SPECORD 200 PLUS double-beam UV/Vis absorption spectrophotometer, shows a wave number of 17,670 cm-1 equivalents to 565.93 nm. This method could eliminate the interference of bile pigments. The results were expressed in the Fishman unit; 1 μg of phenolphthalein released per hour is considered 1 unit.
Within a specific concentration range, the concentration of p-nitrophenol was proportional to the absorbance value. The enzyme activity was converted by measuring the standard curve of p-nitrophenol concentration and absorbance value and checking the concentration of p-nitrophenol in the standard curve from the absorbance value. The standard curve equation is y = 0.0059x−0.0124, where y is absorbance (Figure S1). Absorption of p-nitrophenol is correlated to the solution pH. The higher the pH, the darker the solution color. Therefore, when determining the enzyme activity, the pH of the solution to be measured must be adjusted to 7.0 to eliminate the color error caused by the influence of pH.
General conditions, serum laboratory samples, and X-ray examination
Each day during the first and second weeks after the model-making operation and each weekend after the stent placement under the endoscope, the appetite, the amount and color of the stool, the color of the sclera and urine were observed to see if there were any signs of depression and agitation in animals.
Animals resumed their usual diet 24 h after stent placement. At weeks 0 (pre-operation), 1, 2, and at the end of weeks 4, 8, 12, 16, 20, and 24, the levels of serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), total bilirubin, gamma-glutamyl transpeptidase, amylase, creatinine, and leukocytes were determined. At the end of weeks 4, 8, 12, 16, 20, and 24, after the stent placement, the piglets were observed for the stent position by X-ray examination under anesthesia. If the general condition of piglets was abnormal, or the total bilirubin levels were between 34.2 and 171.2 μM, additional hematological and X-ray examinations were conducted every 2 weeks.
Execution and specimen collection of pigs
When the total bilirubin level of piglets was ⩾171.2 µM, the piglets were sacrificed to observe the pathological changes in the stent and bile duct. The final follow-up time was 6 months, and all piglets were sacrificed in 6 months.
Stent patency evaluation
It was difficult to evaluate when the stent was blocked. Thus, we established a new evaluation system: (1) Time point of stent patency in the ex vivo bile duct model: To simulate the pressure of the human bile duct, a pressure of 50 cm was maintained by hydrostatic pressure height, and bile was allowed to drip slowly from one end of the stent. The stent was considered not completely blocked if bile could flow out from the other end. In other cases, the stent was considered blocked if bile could not flow through. The follow-up time was the blockade time of all stents. (2) The time point of stent patency in piglets: if the total bilirubin of pigs was >171.4 μM, the stent was completely blocked; when the piglets died due to stent blockage, the time of death was the time point of stent patency. If the stent was not in the position or the pig died due to operation, postoperative infection, or anesthesia, it is excluded as “0.”
Histological analysis
Hematoxylin and eosin (HE) stain was performed 22 to observe any inflammatory response of local tissue against stent placement. For this purpose, stents were obtained from sacrificed animals, embedded in poly (methyl methacrylate), sectioned and stained before observing them under a microscope. 23
Statistical analyses
All statistical analyses were performed using SPSS version 13.0 for Windows (SAS Institute, Cary, NC). Data are presented as a mean with a standard deviation. Student t-tests were used to compare continuous quantitative data of the weight increase of bile mud before and after treatment. A 2-tailed Wilcoxon signed-rank test was used to compare discrete variables. Because multiple comparisons were performed on some data, it is noted that statistical significance would be removed by correction using the Bonferroni method. p Values <0.05 were considered statistically significant.
Results
The concentration of PVBC-DGL and its inhibitory effect on the β-glucuronidase activity
In vitro experiment results indicate that the concentration of PVBC-DGL in bile decreased with time. The concentration fluctuations of all coated stents were divided into two stages. In the first stage, the curves were gentle, linear, and slowly varied. These curves had a focal point of about 5–8 weeks, which might be the critical point for molecular disintegration of the coating. When the drug contents were 0%, 10%, 30%, 50%, 70%, and 90%, the concentration of PVBC-DGL could be kept above 0.007, 0.027, 0.045, 0.080, and 0.125 mM, respectively. In the second stage, the concentration curves were steep, non-linear, and varied rapidly. The concentration of PVBC-DGL in bile dropped rapidly to 0, specifically in the last several weeks. Thus, the higher the content of PVBC-DGL in the coating, the faster the coating degradation process and quicker the drug release with its higher concentration in the bile (Figure 4(a)).

(a) Concentration curve of PVBC-DGL in bile and (b) inhibition of β-Glucuronidase activity in the presence of PVBC-DGL.
When the concentration of PVBC-DGL in bile reached about 0.03, 0.05, and 0.08 mM, the inhibition rate of β-glucuronidase activity crossed the range of 30%, 60%, and 95%. The inhibition curve showed that the higher the concentration of PVBC-DGL, the stronger the inhibition effect on the activity of β-glucuronidase. It shows that the inhibition rate is positively related to the concentration of PVBC-DGL. Beyond 0.08 mM concentration of PVBC-DGL, the inhibition rate does not change further with increasing PVBC-DGL, which confirms the saturation phenomenon (Figure 4(b)).
The relationship between the drug content and patency time
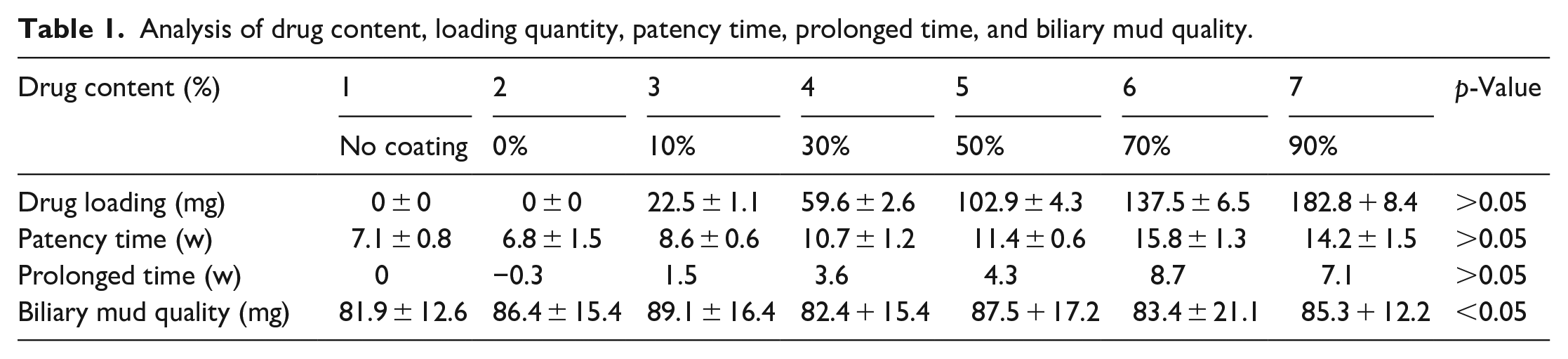
When the drug content is 0%, 10%, 30%, 50%, 70%, and 90%, the patency time is about 7.1, 7.4, 8.6, 10.7, 11.4, 15.8, and 14.2 weeks, respectively; patency time of coated biliary stent was prolonged by 0, 0.3, 1.5, 3.6, 4.3, 8.7, and 7.1 weeks, respectively. Therefore, the stronger the ability of the stent to prevent the deposition of bile sludge, the longer the patency time. However, when the concentration of PVBC-DGL in bile reached a maximum level, its inhibitory effect on the activity of β-glucuronidase halted.
Quality changes of biliary mud with stents
The less the drug content, the shorter the time required for the stent to be blocked by bile sludge. However, we noticed almost no difference in the amount of bile sludge deposited during obstruction; the total amount was about 85 mg.
Follow-up changes in laboratory parameters and X-ray examination after stent placement
Mild to moderate abnormal levels (<1.5–3.5 times normal value) of serum ALT, AST, and amylase (Table S1) and slightly varied (<1.5 times normal value) leukocyte counts are observed. However, those get restored to almost normal levels after 4 weeks. Such changes are speculated to be because of surgery or biliary stenosis.
During the experimental setup, five pigs were excluded due to deep anesthesia (n = 1; in the 0% coating group in X-ray examination at week 4), stent migration (n = 4; the one in the no coating group at week 2, the one in the 0% coating group at week 8; the other two in the 70% coating group at week 12 and 16). The remaining 14 pigs were distributed among the groups: 5 in the original group, 4 in the 0% coating group, and 5 in the 70% coating group. At the end of the follow-up, the final weight of piglets increased to 38.4–56.4 kg.
Patency time and bile mud analysis
The patency time of the stents gets prolonged for all groups; however, the 70% coated stents group showed markedly prolonged patency time as compared to no coating and the 0% coating groups of pigs (23.4 ± 1.8 vs 11.2 ± 2.1 w (p = 0.05); 23.4 ± 1.8 vs 10.5 ± 2.5 w (p = 0.05), respectively). We observed almost no difference in the deposition of bile mud among the three groups (87.5 ± 18.4 vs 86.0 ± 25.3 vs 84.8 ± 21.4 mg (p = 0.05), respectively) (Table 1).
Analysis of drug content, loading quantity, patency time, prolonged time, and biliary mud quality.
Gross and microscopic findings
The bile duct was dilated with a diameter between 0.9 and 2.0 cm. The inner wall of the duct was smooth, with no adhered sediments and dilated lumen cavity. Micro-scopically, the mucosa from bile duct walls did not show apparent changes in exfoliation and proliferation, no obvious infiltration of inflammatory cells, and ulceration or necrosis formation under the mucosa. There was no obvious infiltration of neutrophils, monocytes, lymphocytes, and plasma cells in the whole layer. Moreover, there was no proliferation of fibroblasts and nerve fibers. The vascular endothelium of the bile duct remained good, with no proliferation and thickening of the vascular wall and without thrombosis in the blood vessel. It indicates that there is no obvious pathological damage to the bile duct caused by the litholytic biliary stent (Figure 5).

Histological findings of a common bile duct: (a) macroscopic appearance of the common bile duct and (b and c) microscopic appearance of the common bile duct, showing no obvious inflammation and invasion. Hematoxylin and eosin stain; ×200 (b), ×400 (c).
Discussion
Current research emphasizes that biliary mud deposition caused by bacterial infection is a leading cause of restenosis after plastic stent implantation. Skar et al.18,24 suggested that the pathogenic bacteria in the biliary tract, especially E. coli and P. perfringens, produce β-glucuronidase, which could decompose bilirubin-glucuronide di-ester, producing bilirubin and bind with ionic calcium, resulting in the deposition of calcium bilirubin, thus form biliary mud and bile duct stones. Leung et al.25,26 found that the surface of the plastic stent was covered by fibronectin, collagen, fibrin, and immunoglobulin A immediately after implantation. Under the action of β-glucuronidase produced by bacteria, calcium bilirubin crystallized continuously and accumulated, resulting in the deposition of biliary mud in the stent, eventually leading to the stent’s occlusion. It could be seen that β-glucuronidase played a vital role in stricture and gallstone formation after biliary stent surgery. Therefore, we speculated that inhibiting β-glucuronidase might inhibit bacterial biofilm formation and biliary sludge deposition, thus prolonging the patency time of biliary stent.
Macfadyen and Ho established during animal experiments that the activity of β-glucuronidase in bile could be inhibited by 75% when the average concentration of D-glucaric acid-1,4-lactone (DGL) in bile reaches 0.06 μmol/mL after oral administration of different doses (50–2600 μmol).27,28 Sacco et al. 17 linked DGL to polyvinyl benzyl chloride (PVBC) via a carboxyl bond through a bimolecular nucleophilic substitution reaction. In animal experiments, PVBC-DGL could also significantly inhibit the activity of β-glucuronidase that is insoluble in water and could not be absorbed by the intestine, thus alleviating the effect of endogenous β-glucuronidase on the liver and kidney. The study also confirmed that PVBC-DGL had excellent biological safety. All the above results indicated that PVBC-DGL could be used as a highly effective and non-toxic inhibitor of β-glucuronidase.
We synthesized PVBC-DGL by a chemical method, mixed PLA with PLGA, and loaded it into a biliary stent by coaxial electrospinning.27,29 The carrier degraded in the bile faster than in saline or natural conditions. It might because the bile is weakly alkaline, and bacteria could also play a role, which leads to the acceleration of self-degradation of the carrier.30–32
We report a positive linear relationship between the concentration of PVBC-DGL in bile and the inhibition rate of β-glucuronidase activity. When the levels of PVBC-DGL reached 0.03, 0.05, and 0.08 mM, the inhibition rate of D-glucucaro-1,4-lactone on the activity of β-glucuronidase reached 30%, 60%, and 95%. Our findings are consistent with those of Macfadyen and Ho 28 and Sacco et al. 17 When the concentration of PVBC-DGL continued to increase, the inhibition rate of the activity of β-glucuronidase did not change much beyond 0.08 mM (Figure 4). We speculate it as a maximum limit or saturation of PVBC-DGL for the inhibition rate of the activity of β-glucuronidase. A constant increase in the concentration of PVBC-DGL could not escalate the inhibition rate of β-glucuronidase activity. Hence, only elevated self-degradation time of the carrier could prolong the inhibition time of β-glucuronidase activity, which ultimately helps to prolong the patency time of stents.
Moreover, our study establishes that when the drug content was 0%, 10%, 30%, 50%, 70%, and 90%, the patency time was about 7.1, 7.4, 8.6, 10.7, 11.4, 15.8, and 14.2 weeks, respectively. The patency time of coated biliary stent was prolonged by about 0, 0.3, 1.5, 3.6, 4.3, 8.7, and 7.1 weeks, respectively. When the concentration of PVBC-DGL reached the saturation level, the inhibition of β-glucuronidase by PVBC-DGL was halted, and the inhibition rate of β-glucuronidase activity remained unchanged. Therefore, when the optimum drug loading content of the coating was 70%, the patency time could be 15.8 weeks in vitro and 23.4 weeks in pigs (Table 1). The excessive concentration of PVBC-DGL could not prolong the patency time of the stent, which might be the reason that the stents with 70% PVBC-DGL coating had a longer patency time than that of 90% coating.
In vivo stents showed prolonged patency compared to in vitro stents (Table 1); we consider it due to the relatively uninfected environment of the stent that slows down the biliary mud deposition. On the other hand, it could be related to bile hydrodynamics in vivo and in vitro. In vitro, the bile flow is uniform, while in piglets, the bile secretion occurs mainly at the feeding time. At this time, the bile secretion is fast, the pressure is high, and the bile flows quickly through the stent. During the non-feeding time, the bile secretion is less, and the flow rate is slower. At this time, it is easy to cause bile sludge deposition, but the volume is significantly reduced.
We also report that there was no significant difference in the quality of biliary mud deposition among all stents, about 85 mg (Table 1), which might be related to the length and diameter of the stent itself. It could be because the bile duct secretes fibronectin, collagen, fibrin, immunoglobulin A, and other inflammatory mediators. These substances may have some effect on the deposition and cross-linking of bile mud. Our research shows that the drug carrier and PVBC-DGL did not affect the liver function enzyme levels, and there was no obvious pathological damage to the bile duct was observed by histological studies. It could be well compatible with pig tissues, neither caused bile duct hyperplasia or stenosis nor produced aberration or canceration. No changes in chronic inflammation, fibrosis, granuloma, and necrosis were locally observed after stent placement (Figure 5). Therefore, we believe that the coated stents have good histocompatibility, safety, and reliability, with no toxic side effects.
This preliminary study has some limitations as well. Firstly, the patency time of the coating in the pig’s model, possibly with a more inflammatory reaction and secreted mucin, was longer than in the in vitro model, probably with a bacterial infection environment. We couldn’t get it determined and suggest mucin quantification in future studies. Secondly, the cross-sectional area of the common bile duct with the increase of piglet weight increased correspondingly, whereas the human bile duct may not change over 1 year. It is really hard to distinguish whether the stent itself was not blocked or it is somehow blockaded the bile flow through the enlarged space around the stent.
Moreover, we noticed stent migration in four piglets. However, it is related to the self-contraction of the common bile duct and the movement of the piglet; on the other hand, the weight of the piglet kept increasing (some piglets doubled the weight), the bile duct expanded with the weight increase and cross-sectional area of the stenosis as well. We suggest a comprehensive study with large sample size, answering to questions and limitations of this study.
Conclusions
The 70% PVBC-DGL stents may effectively prevent and control the bile mud deposition and prolong the patency time of stent placement in subject animals. These stents can be proposed for further studies in human patients and clinical trials to explore their potential to prevent bile mud deposition.
Supplemental Material
sj-docx-1-jbf-10.1177_22808000221134988 – Supplemental material for Studying in vivo and in vitro effects of a novel polyvinyl benzyl chloride-D-glucaro-1, 4-lactonate polymer-coated bile duct stent for anti-biliary mud deposition
Supplemental material, sj-docx-1-jbf-10.1177_22808000221134988 for Studying in vivo and in vitro effects of a novel polyvinyl benzyl chloride-D-glucaro-1, 4-lactonate polymer-coated bile duct stent for anti-biliary mud deposition by Weixing Zhang, Fariha Kanwal, Aima Iram Batool, Muhammad Mustaqeem, Muhammad Fayyaz ur Rehman and Xinjian Wan in Journal of Applied Biomaterials & Functional Materials
Footnotes
Author contributions
Conceptualization, WXZ and FK; methodology, WXZ; software, MFR; validation, MFR, XJW; formal analysis, XJW; investigation, WXZ, MS, and FK; resources, XJW; data curation, MFR; writing—original draft preparation, AIB, WXZ, MFR; writing—review and editing, MFR, FK; supervision, XJW; project administration, WXZ, MS, and FK; funding acquisition, XJW. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the National Natural Science Foundation of China (No. 81870452) and the Shanghai Natural Science and Technology Fund (No. 19441906700). Authors are also thankful to the Science and Technology Commission of Shanghai Municipality (No.20JC1419302) and Shanghai Clinical Research Center for Digestive Diseases (19MC1910200) for providing funding.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
