Abstract
In recent years, more and more metal or non-metal materials have been used in the treatment of cardiovascular diseases, but the vascular complications after transplantation are still the main factors restricting the clinical application of most grafts, such as acute thrombosis and graft restenosis. Implant materials have been extensively designed and surface optimized by researchers, but it is still too difficult to avoid complications. Natural vascular endodermis has excellent function, anti-coagulant and anti-intimal hyperplasia, and it is also the key to maintaining the homeostasis of normal vascular microenvironment. Therefore, how to promote the adhesion of endothelial cells (ECs) on the surface of cardiovascular materials to achieve endothelialization of the surface is the key to overcoming the complications after implant materialization. At present, the surface endothelialization design of materials based on materials surface science, bioactive molecules, and biological function intervention and feedback has attracted much attention. In this review, we summarize the related research on the surface modification of materials by endothelialization in recent years, and analyze the advantages and challenges of current endothelialization design ideas, explain the relationship between materials, cells, and vascular remodeling in order to find a more ideal endothelialization surface modification strategy for future researchers to meet the requirements of clinical biocompatibility of cardiovascular materials.
Introduction
As one of the most deadly diseases in the world, the number of deaths due to cardiovascular disease (CVD) has accounted for 31% of all deaths in the world. 1 Atherosclerosis is a chronic inflammatory disease caused through imbalance of lipid metabolism and poor immune response.2,3 Coronary atherosclerotic heart disease (CADs), which originates from the peripheral coronary arteries of the heart, is a typical type of cardiovascular disease. This is a disease in which atherosclerotic plaques on the vascular wall cause vascular stenosis, which interferes with the continuous blood supply of the coronary arteries, resulting in insufficient blood supply or ischemia to the heart, and ultimately causing cardiac dysfunction.
With the continuous development of biomedical materials, implant materials such as artificial blood vessels,4–6 tissue-engineered blood vessels,7–9 and cardiovascular stents10–13 have become important choices for the clinical treatment of CADs.
However, the risk of restenosis14–16 and late thrombosis17–19 in the lesion site after implantation is increased, which severely limits the application of implant materials. First, the adsorption of plasma proteins on the surface of the material occurs within seconds of contact between the material and blood. The platelets and other blood cells in the plasma are interact with the protein adsorption layer, the platelets are activated, and the fibrinogen converts to fibrin, which stimulates a large number of activated platelets to aggregate and lead to the formation of acute thrombosis. 20 Then the implantation of the material induces damage to the vascular endothelial layer and activates the surrounding ECs. These activated ECs and platelets release a large number of chemokines and inflammatory factors, which induce inflammatory cells to aggregate toward the lesion and trigger an inflammatory response,20,21 which peaks within 2 days. 22 In addition, clinical research data show that the peak of intimal hyperplasia is within half a year after implantation. 23 However, about 2 weeks after implantation, the rapid proliferation of smooth muscle cells (SMCs) will also occur, and the cell proliferation tends to be stable after 4 weeks.24,25 and this early rapid proliferation of SMCs is also closely related to the occurrence of restenosis in late stages. In order to overcome these complications after material implantation, people have been exploring more effective solutions.
The natural vascular endothelial layer has excellent anti-coagulation and anti-intimal hyperplasia abilities, which is the key to maintaining the steady state of the environment between the blood vessel wall and the blood.26–28 ECs are a type of multilateral flat cells with jagged edges, located between the blood and the media of the blood vessel wall, and provide a barrier for the media of the blood vessel wall 29 (Figure 1). ECs prevent coagulation and thrombosis by secreting or expressing biological factors (such as Nitric Oxide (NO), 30 Prostaglandin I2 (PGI2), 31 tissue factor pathway inhibitor (TFPI) 32 ). On the other hand, ECs can shield the stimulating contact of growth factors in the blood to SMCs, and avoid the transformation of SMCs phenotype from contraction type to synthesis type. Synthetic SMCs secrete a large amount of extracellular matrix and invade the endometrial layer to form excessive endometrial hyperplasia, at the same time, the biological factors (such as NO) secreted by ECs can prevent the abnormal proliferation of SMCs, thereby preventing vascular stenosis. 33 It is precisely these abilities that ECs possess to maintain the homeostasis of the vascular wall microenvironment.

Endothelial cells maintain the homeostasis of vascular wall environment.
Based on the above studies, the formation of a complete endothelial layer on the surface of cardiovascular materials is widely regarded as a more ideal treatment method. In early studies, researchers directly planted ECs on the material surface to form a complete endodermis layer to achieve the endothelialization of the material surface. This method is intuitive, simple, and operable in vitro, so it has achieved certain success. However, direct planting of cells has more stringent requirements for materials and cells, which led to a higher failure rate and longer planting time, so this idea was gradually abandoned. With extensive development and in-depth exploration of research, people’s understanding about endothelialization of implant materials has been further improved, and the surface modification of materials for promoting endothelialization has gradually attracted attention. Through the biofunctional design of the material surface, the ability of surface to promote ECs adhesion and proliferation was strengthened, and the process of surface endothelialization was accelerated. In addition, stem cells with endothelial differentiation ability such as endothelial progenitor cells (EPCs) 34 and mesenchymal stem cell (MSCs) 35 have gradually entered the field of researchers in recent years, providing new modification ideas for materials based on in situ induced endothelialization in vivo.
Endothelialization-promoting modification on the surface of materials induces ECs proliferation, migration and adhesion, or promotes the homing and directional differentiation of stem cells. The strategies are mainly divided into three categories: Surface design based on material surface science method, Surface design based on bioactive molecules, and Surface design based on biological function intervention and feedback (Figure 2). Based on the reports of vascular implants in recent years, this paper reviews the research on endothelialization of materials by surface modification, and discusses the advantages and challenges of current surface modification methods, in order to provide new reference ideas for endothelialization design of vascular implants.

Design strategy for material surface endothelialization.
Surface design based on material surface science method
In the normal vascular wall, the main function of ECs is to maintain vascular homeostasis and avoid atherosclerosis caused by abnormal changes in blood environment.36–39 Therefore, ECs are very sensitive to the abnormal hemodynamic changes in the blood environment, and the changes in the physicochemical properties of the material surface can also affect the function of ECs. The endothelialization of the surface can be effectively promoted by controlling the changes in the physicochemical properties or microstructure of the material surface.
Surface microstructure
In the normal physiological environment, the function of ECs depends largely on their interaction with extracellular matrix, while extracellular matrix (ECM) exhibits micron or nano-scale microstructure characteristics in physical structure, and cells are very sensitive to this structure. Thus microstructure can effectively regulate the morphology of ECs on the surface of the material, and promote the secretion of extracellular matrix, thereby endowing the surface with better biocompatibility. 40 Periodic pattern with a certain interval distance is a common modification method of material surface microstructure, which can effectively promote the migration and adhesion of ECs.41–43 Some grooved structures can effectively promote ECs to arrange along the direction parallel to the pattern, and has stronger migration ability.
Different roughness has distinct effects on cell activity and behavior.44,45 Due to the difference in the average size of ECs and SMCs, their adaptability to the roughness is significantly different. Comparing the ratio of cells adhered to the same roughness range, it was found that the proportion of ECs increased greatly as the roughness increased. 46 Thus the microstructure size is also an important factor affecting ECs behavior. 47 The microstructure of suitable size (15 μm) can keep the ECs in the form of a fused monolayer on the surface, which can effectively promote the ordered arrangement of ECs on the surface of the material. 48 When the size of the microstructure is in the micron level, the large size of the pattern (7656 μm2) makes the cells “free expansion” across the region, and the smaller (1781 μm2) makes the cells “limited mobility.” This shows that when the area of the micropattern is less than the maximum area of the cell spreading, the cell shape is related to the size and shape of the pattern. 49 Of course, in addition to regular structures, Random structures have the ability to regulate cell behavior at micro-nano scale. Wu et al. 50 enhanced the adhesion of ECs on the material surface by constructing a random pattern with controllable size on the nonmetallic material surface, and avoided the interference of wall shear stress on the attached ECs. Metal materials such as titanium can use physical vapor deposition-thermochemical or anodic oxidation methods to construct microstructures with certain roughness on the surface.51,52 By adjusting the roughness, the protein adsorption, blood compatibility, and cell behavior on the material surface were affected to selectively promote the growth of EC and avoid restenosis after implantation.
Due to the close interaction between cells and materials, cells can make corresponding stress responses to the surface morphology of materials through actin polymerization, and this reaction can not only be reflected in ECs and SMCs, but also the differentiation and migration of stem cells.53,54 The multi-layer structure of the vascular implant uses the directional microgrooves between the biomimetic layers to promote the directional growth and migration of ECs, and better mimic the arrangement of natural vascular cells. 55 Vascular implant materials containing microchannel structures both highly interconnected and circumferentially oriented can not only effectively affect the morphology, arrangement, and phenotypic changes of SMCs, but also improve the directional deposition of ECM on the surface of the material and the enrichment and polarization of macrophages, promote cell infiltration and accelerate the process of endothelialization. 56 By manipulating the micro-nano scale morphology of the material surface, the anchoring of MSCs membrane protein can be effectively regulated, and the process of differentiation into SMCs can be induced by the traction of F-actin, which is further used for the repair of vascular microenvironment. 57
Physicochemical properties
The adhesion and migration of cells on the surface of the material depend on the adsorption of plasma protein and cell secretory protein on the surface of the material, and the physical and chemical properties of the surface of the material affect the type, conformation, and adsorption amount of the surface protein. 58 Grafting hydrophilic substances or UV irradiation can enhance the hydrophilicity of the material surface and promote the adhesion and proliferation of cells on the surface.59–61 Plasma treatment can significantly improve the hydrophilicity of metal surface. After plasma activation, the concentration of C-O group on the material surface increases, and the proliferation and migration of ECs are enhanced. 62 It is noteworthy that due to the water barrier effect, the binding form of water molecules on the hydrophilic surface has a significant effect on the adhesion of cells. Chemically bound water molecules cannot provide the required binding site density for ECs, but physically bound water molecules can provide it. 63 In addition, the surface charge can control protein (such as Fibronectin) adsorption, thereby regulating cell behavior, thereby enhancing cell adhesion and migration. 58
The stiffness and the magnetic of the surface can directly or indirectly affect the adhesion and proliferation of ECs on the material.64,65 Being anchorage-dependent, the adhesion and proliferation of ECs were inhibited on soft surface, while promoted on higher stiffness surface. But the ECs monolayer on the soft surface showed higher endothelial function than that on the hard surface. 66 Chang et al. 64 used this discovery to construct a Vascular endothelial growth factor (VEGF)-loaded poly (l-lysine)/hyaluronic acid (PLL/HA) material surface, and then control the stiffness of the material surface by changing the crosslinking degree. The results showed that VEGF could effectively promote ECs adhesion on the material surface. The gene expression of ECs on soft surface (200 kPa) was significantly increased compared with that on hard surface (430 kPa), and the cell activity was significantly enhanced. The magnetic properties of materials can also significantly affect cell behavior. Under the action of 300 mT external magnetic field, the magnetized material surface can achieve high adsorption of magnetically labeled endothelial progenitor cells within 3 h. At the same time, due to the weakening edge effect, cells can be uniformly covered on the surface of the material. 67 And Zhang et al.’s 65 study shows that cell adhesion is also significantly related to the oscillation frequency of the magnetic field, and can effectively promote cell growth under low frequency magnetic field (0.1 Hz), and has long-term stability.
Whether microstructure or surface physicochemical properties, the surface modification methods based on material surface science focus on the interaction between materials and cell behavior, which makes the related modification methods can effectively promote cell adhesion and directional migration on the material surface. Cell adhesion and directional migration can rapidly form an endothelial layer on the surface of the material, which is the basis for rapid endothelialization and long-term stability of the vascular graft after implantation.
Surface design based on bioactive molecules
The excellent endothelial cell compatibility on the surface of vascular implant materials is the basis of vascular regeneration. The surface can create a good regeneration micro-environment for the migration and adhesion of cells, which is conducive to the process of endothelialization of the material. In order to improve the cytocompatibility of the material surface, the surface of vascular implant material is usually modified based on bionics methods., that is, immobilize natural or synthetic biological functional molecules on the surface of biological materials to construct natural physiological microenvironment, such as heparin, 68 fucoidan, 69 hyaluronic acid, 70 ECM protein, 71 and peptide. 72 Based on the characteristics and advantages of heparin and hyaluronic acid, many researchers have carried out a large number of related studies and have made important progress. This article will also describe them separately.
Heparin
Heparin is a commonly used anticoagulant in clinic. It is often used to modify the surface of vascular implants and has anti-inflammatory function.73,74 Heparin also has the ability to protect ECs and resist intimal hyperplasia.75,76
Covalent binding is a common modification method. Heparin can be introduced into the surface of materials by EDC/NHS crosslinking reaction. 68 Negatively charged heparin can also be introduced into the surface of the material by electrostatic interaction in the form of layer-by-layer self-assembly.77,78 However, compared with covalent modification methods, biomolecules assembled by electrostatics are often less stable, and uncontrolled molecular bursts occur in dynamic environments. Therefore, the characteristics of heparin with negative charge can be used to form nanoparticles with positively charged biological molecules to control its stability. 79 For example, Song et al. 80 used the specific binding of nerve growth factor (NGF) and heparin and the electrostatic interaction between heparin and chitosan to design a heparin/chitosan nanoparticle loaded with NGF to accelerate the endothelialization of the material surface. In addition to chitosan, poly-L-lysine is also a biomolecule that can be electrostatically bound to heparin to form nanoparticles. 81 It is worth noting that too high density of heparin can also reduce ECs growth and enhance SMCs proliferation. 82 Therefore, heparin content on the surface of the material may be the primary consideration in the process of surface endothelialization.
Hyaluronic acid
Hyaluronic acid (HA) is a linear polysaccharide composed of D-glucuronic acid and N-acetylglucosamine distributed in extracellular matrix, which has high biocompatibility with cells. 83 As the main component of ECM, HA has excellent functions of promoting endothelial cell proliferation and anticoagulation, so it is often used for the modification of vascular materials and bone materials.84,85
Some polymeric materials usually do not have good endothelial cell compatibility, 86 so HA can be introduced into polymer materials by simple electrospinning and chemical crosslinking, which can improve the blood compatibility of the surface and promote the endothelialization of the material. 70 For example, Dimitrievska et al. 87 covalently grafted thiol-modified sodium hyaluronate onto the surface of decellularized scaffold materials with thiol-reactive properties, and cross-linked through the disulfide bond between HA to form a uniform thin layer on the surface of the material. In addition, the deposition of dopamine on the surface of the material can also graft HA onto the metal material, which can promote the endothelialization of the material surface and improve the corrosion resistance of the metal material. 88
In addition, some studies have found that the biological characteristics of hyaluronic acid are related to its relative molecular weight. Low molecular weight HA promotes inflammatory response and SMCs proliferation.89,90 In Jiang et al.’s 91 study, they found that HA with a molecular weight of 100 kDa confers anti-inflammatory, anti-proliferative, and reendothelializing properties to the scaffold. Although high molecular weight HA can be grafted on the surface, high molecular weight HA can be decomposed into low molecular weight hyaluronic acid in vivo, which is not conducive to long-term implantation of materials. At the same time, too high molecular weight HA can also inhibit the proliferation of ECs. 92 Therefore, HA is often used in conjunction with other biomolecules to overcome the adverse effects of HA. For example, ECs-affinity peptides (RGD and YIGSR) were grafted onto HA hydrogel (100–150 kDa) by click chemistry with single or orthogonal density gradients, the modified material surface showed synergistic effect on cell adhesion and demonstrated the specific competitive adhesion of ECs. 93 Low molecular weight HA (5 kDa) promoted cells adhesion and proliferation through EDC/NHS cross-linking Type I collagen. 94 HA (4 kDa) hydrogel scaffold formed by calcium ion crosslinking of sodium alginate (SA) can provide excellent microenvironment for stem cells. 95 This scaffold has a high ratios of water content and a slow degradation rate, exhibits a porous structure suitable for stem cell loading and good rheological behavior, which are helpful for stem cell differentiation. In vivo experiments further demonstrated that this modified material could protect human umbilical cord mesenchymal stem cells (hUC-MSCs) and maintain their cellular viability.
ECM protein
The adhesion, migration, and proliferation of ECs largely depend on the interaction between cells and the surrounding ECM proteins, so ECM proteins are also widely used in the surface modification of vascular materials to promote endothelialization. 58
Fn is a common ECM protein, which can effectively improve the adhesion ability of ECs and EPCs as a protein coating on the surface of materials.96,97 Plasma can be used to treat the material surface to introduce fibronectin, but the differences in parameters will lead to great changes in the affinity, conformation, and direction of adsorbed Fn, which in turn significantly affects endothelial cell-material and cell-cell interactions.98,99 Surface deposition of dopamine can also effectively graft Fn onto the material to promote surface endothelialization.100,101 In addition to fibronectin, other proteins such as serum human laminin (Ln) 71 and collagen 102 have also been gradually applied to promote endothelialization on the surface of materials.
There are three kinds of collagen commonly used: type I collagen and type III collagen distributed in the media of blood vessel, type IV collagen distributed in the intima of blood vessels. Type I collagen can provide a good microenvironment for cell migration and differentiation. 102 Compared with simple physical coating, the type I collagen structure on the material surface introduced by covalent crosslinking is more stable. 103 Considering that type III collagen had platelet binding sites, Yang et al. 104 prepared a new recombinant human type III collagen (hCOLIII), which retained the fragment that promoted cells adhesion and bypassed the platelet activation sequence containing hydroxyproline. Type IV collagen was fixed on the surface of the polymer material to construct a biomimetic vascular basement membrane.105,106 The results of cell experiments showed that the modified material surface promoted the proliferation of ECs and inhibited platelet adhesion.
Peptide
Specific amino acid sequences can mediate cell adhesion, proliferation, and differentiation by binding to transmembrane protein integrin family receptors or other receptors. Therefore, in recent years, more and more studies have focused on the extraction or synthesis of amino acid sequences that play a key role, and the application of these polypeptide molecules in the surface modification of vascular implants to promote the endothelialization of such materials (Table 1).
Endothelial cells affinity peptides and their modification methods.

ECs are surrounded by an ECM composed of type IV collagen, Ln, and heparin sulfate polysaccharide (HSP), which is filled with collagen fibers and glycoproteins. ECM triggers intracellular signaling pathways through the interaction between its receptors and integrins on ECs, thereby regulating the differentiation, proliferation, and migration of ECs. Therefore, biomimetic-based surface modification is intended to provide a good growth microenvironment for cells. The physiological behavior of ECs was regulated by constructing biomimetic ECM microenvironment or specializing a binding site, so as to achieve the effect of material surface endothelialization.
Surface design based on biological function intervention and feedback
After the material is implanted into the blood vessel, the induction of endothelial regeneration in situ on the surface of the material is called in situ endothelialization. In situ endothelialization is mainly aimed at two types of cells: ECs and EPCs. For the former, on the one hand, it promotes the migration and proliferation of cells in the contact area between the material and the blood vessel to the material surface, and on the other hand, it attracts and induces the adhesion of circulating ECs (cECs) in the blood. 136 The latter is a precursor cell derived from bone marrow, distributed in the blood, and has the potential to differentiate into mature ECs. EPCs are specifically captured on the surface of the material and induced to differentiate to achieve surface endothelialization in situ. 137 Therefore, different researchers intervene the biological function of the two types of cells by functional factors, antibodies and gases on the surface of the implant materials, respectively/jointly. Using specific positive feedback, ECs/EPCs in vivo are induced to proliferate and differentiate on the surface of the material to achieve in situ endothelialization of the material in vivo.
Functional factor
VEGF is a key factor in angiogenesis, which can promote the migration and proliferation of ECs and the proliferation of EPCs.138,139 The surface of the material is loaded with VEGF in the form of physical coating or electrostatic deposition for orderly release in the body, which can significantly improve the in-situ endothelialization ability of the vascular implant material.140,141 Min et al. 142 used collagen to construct biomimetic renal vascular 3D stent, and then VEGF is introduced into the surface of the material by grafting heparin on the stent to prepare a vascular implant material with VEGF controlled release ability. Due to the short half-life of VEGF in the blood environment, early burst release can increase the content of VEGF in the environment, so as to achieve rapid endothelialization. Although early burst release can increase the content of VEGF in the environment to achieve rapid endothelialization, studies have shown that high concentrations of VEGF may inhibit the migration of ECs. 143
The introduction of VEGF on the surface by covalent immobilization can effectively inhibit the endocytosis of VEGF by cells, prolong the action time of VEGF, and control the local VEGF concentration, so as to better promote cell adhesion, proliferation, and directional differentiation on the surface of the material. By depositing dopamine on the surface, VEGF is grafted onto the material by covalent binding of dopamine to VEGF. 144 By the interaction of dopamine-heparin and heparin-VEGF, VEGF was immobilized on the surface of the material to prepare a bio-functional coating with specific capture of EPCs and enhanced ECs activity to achieve in situ endothelialization of the material surface.145–147 Since Fc fusion protein can promote the expression of VEGF function at low concentration, VEGF-Fc was synthesized by genetic engineering, and then VEGF was introduced into the material surface by using the high binding force between Fc and hydrophobic surface. 148 VEGF on the material surface enhanced the expression of PI3K and MAPK by activating vascular endothelial growth factor receptor-2 (VEGFR2), and promoted the adhesion and proliferation of ECs, thus achieving rapid vascularization in vascular implant materials.
Because stromal cell-derived factor-1 (SDF-1) and its receptor CXCR4 play an important role in EPC mobilization, migration and homing, they can be used to mediate EPC mobilization and homing of endogenous EPCs to implant materials to participate in the endothelialization of materials.149–151
SDF-1 was introduced into the surface of small-diameter implant materials to promote EPCs recruitment, migration, and proliferation of mature endothelial cells and ECs. 152 Shafiq et al. 153 constructed SDF-1 release surface on PCL-collagen implant material, which enhanced the recruitment of surface stem cells, accelerated the surface endothelialization and promoted angiogenesis through SDF-1 release. Liu et al. 154 constructed a novel SDF-1 α/laminin-loaded nanoparticle coating on the surface of 316L SS to improve the endothelialization ability of the material surface. The modified material can effectively inhibit the adhesion and activation of platelets on the material surface, and induce the migration of ECs and the aggregation of EPCs. Gao et al. 155 increased the free radicals on the surface of expanded polytetrafluoroethylene (ePTFE) by plasma immersion ion implantation, and realized the grafting of heparin, SDF-1α, and CD47 onto the material surface. Immobilized SDF-1α could effectively capture EPCs under flow conditions and enhance EPCs activity on the surface of materials.
Other functional factors that affect endothelialization are listed in Table 2.
Other factors that promote endothelialization and their modification methods.

Antibody
EPCs and ECs express some specific markers, such as CD31, VEGFR-2, CD133, and CD34. The surface of the material can increase the adhesion and proliferation of ECs or EPCs by introducing these common or specific antibodies to promote in situ endothelialization of the material.164,165
Anti-CD31 antibodies can specifically induce ECs binding, so that anti-CD31 modified materials can effectively promote the adhesion of ECs on the surface and enhance their activity. 166 Through EDC/NHS cross-linking reaction, antibodies such as CD31 or VEGFR-2 can be grafted onto the surface of the material, specifically capturing circulating EPCs and ECs to promote in situ endothelialization of the material surface. 165 VEGFR-2 is expressed on the surface of ECs, cECs, and EPCs, but its effect on cECs is about 20 times higher than that on EPCs. 167 Therefore, the recombinant anti-VEGFR-2 antibody fragment was grafted onto the aminated surface. 168 In vivo experiments proved that the modified material has the potential to accelerate the surface endothelialization in situ. CD133 and CD34 are proteins specifically expressed by EPCs, but the non-specific binding of CD34 to other cells will interfere with their specific binding to EPCs, so CD133 is a more specific EPCs marker. 146 After sulfhydrylation of the material surface, the anti-CD-133 antibody was fixed by disulfide bond formed by sulfhydryl and cysteine. 169 After oxidation treatment of anti-CD133 antibody, it can also introduce thiol or amino functionalized 316 L SS surface. 170 The modified material surface can effectively capture EPCs and reduce the growth of SMCs.
CD34 antibody is the most widely studied antibody in the field of endothelialization. Fixation of CD34 antibody on the surface of biological materials can promote the adhesion of EPCs and the formation of endothelium. 171 CD34 monoclonal antibody was introduced into the material surface by EDC/NHS cross-linking with the surface-encapsulated ECM. 172 Anti-CD34 antibody stimulated the adhesion and proliferation of EPCs on the material surface, and improved the endothelialization ability and long-term patency rate of graft.Anti-CD34 antibodies can also be covalently grafted onto a hydroxyl-terminated e ePTFE surface with a lubricant injection layer by silanization, and the introduction of antibody enhanced the ability of the material surface to capture ECs specifically. 173 At present, the CD34 antibody stent (COMBO) has been clinically approved, and the clinical results have shown a relatively good target disease vascular recanalization rate.174,175 But there are still certain risks, it may be related to the tendency of the captured EPCs to differentiate into SMCs and precursor inflammatory cells, thereby causing the proliferation of the neointima.176,177
NO
As a gas signal molecule, NO is an important factor in maintaining the steady state of vascular microenvironment.178,179 NO promotes the growth of ECs while inhibiting platelet activation, leukocyte-endothelial adhesion, and SMCs proliferation, thereby reducing intimal proliferation and inflammation to maintain vascular homeostasis. 30 The research results in recent years are shown in Table 3.
Different ways of NO and and their modification methods.

Conclusion
Table 4 is a summary of different material surface endothelialization designs in recent years, including key design ideas, the advantages of different design strategies, and their disadvantages.
The key, advantages, and disadvantages of different surface modification methods.

Since interventional surgery has been used to treat cardiovascular disease, vascular grafts have been key to the success of this treatment modality. In previous treatments, either abnormal proliferation after metal stent implantation or late restenosis caused by drug-eluting stents can cause serious complications for long-term treatment of vascular grafts. Tissue engineered blood vessels can cause adverse cardiac events due to insufficient compliance or strength. Therefore, it is generally recognized that most vascular grafts can only “restore” the blood supply function of blood vessels. But to “repair” damaged blood vessels requires the participation of cells, especially endothelial cells.
Endothelialization is a key factor for the long-term effect of implant materials. The delay of endothelialization on the surface of the material is the main cause of restenosis and advanced thrombosis after implantation. When the implant material is implanted, the incomplete endothelialization of the material surface at the early stage of implantation will activate the platelets in the blood, trigger the related coagulation mechanism, and form acute thrombosis in the short term. Inflammatory cells infiltrate into the vascular wall, and the growth factors in the blood environment stimulate the excessive proliferation of SMCs to cause vascular stenosis. These factors in turn affect the adhesion and proliferation of ECs on the surface of the material. With the prolongation of implantation time, the delayed endothelialization of the implant surface further accelerates this trend, eventually leading to restenosis and late thrombosis.
Based on this point of view, most researchers have done a lot of research on vascular grafts to achieve the regeneration of endothelial cells on the material surface through different surface modification methods (microstructure, physicochemical property, bionics and biological factors, etc.). These studies based on the interaction of materials with cells have achieved surface endothelialization in vitro or in vivo to varying degrees, but the repair of blood vessels is a complex process, which consists of three stages: In the early stage of implantation, the vascular implant may not provide an ideal platform for endothelial cells to homing, adhere and proliferate due to the coverage of platelets and coagulation proteins; Good cell viability is the basis for the rapid proliferation of implanted mid-stage ECs; In long-term implantation, good histocompatibility is central to maintaining the homeostasis of vascular graft endothelialization.
Endothelialization is the most ideal design idea and final effect of vascular implant materials, it is undeniable that the endothelialization process of vascular implants after implantation is not necessarily or completely around ECs. The complete remodeling of vascular wall structure is affected by many factors, such as the mechanical properties of materials, hemodynamics, plasma protein adhesion, and drug effects.98,201–203 Therefore, it is determined that the endothelialization design strategy should be a holistic consideration of the post-implantation pathological environment rather than surface modification under the influence of a single factor. This means that not only the effectiveness, feasibility, and time-sequence of the modification method need to be considered, but also the impact of the material surface on the entire vascular microenvironment, including cells, blood, and tissues (Figure 3):
Implant material-Blood: After implantation, the material first faces the blood environment. Therefore, vascular graft must have good blood compatibility. This requires vascular implant materials to inhibit the activation of platelets and thrombin on the one hand and avoid acute thrombosis caused by the generation and aggregation of plasma fibrinogen. On the other hand, it is to avoid further local vascular inflammation caused by monocyte chemotactic factors and adhesion molecules. 204 Inflammation will aggravate endothelial dysfunction, which is not conducive to endothelialization of the material surface. 205 Therefore, the material surface endothelialization design through a variety of means, such as surface physicochemical properties change, 206 heparinization, 207 and NO controlled release, 208 which can effectively improve the blood compatibility of the material.
Implant material-Cells: The endothelialization of vascular implant materials is based on ECs. The normal ECs on the surface of the material can not only stabilize the interaction between the material and the blood environment, but also regulate the phenotypic changes of SMCs and inhibit restenosis caused by abnormal proliferation. 209 It is worth noting that although the inhibition of SMCs activity has become an important consensus in endothelialization design, it is undeniable that the normal growth of SMCs is also of positive significance for the improvement of vascular structure in vascular remodeling. 57 Therefore, in the process of material surface endothelialization, SMCs should focus on “inhibiting abnormal proliferation” rather than “inhibiting proliferation.”
Implant material-Tissue: Cell-ECM interaction is the basis of cell function regulation. 210 After anticoagulation and promotion of cell adhesion in the early stage of implantation, whether a complete layer of ECs with normal function can be formed on the surface of the material, the key point is whether the material surface can continuously provide an in vivo microenvironment suitable for cell growth. Although ECs will release ECM outside after adhering/migrating to the surface of the material, the pre-built microenvironment of the material through antibodies, peptides, or (continuous and stable release) functional factors are easier for cells to activate in the early stage of adhesion and to rapidly proliferate and stabilize after long-term implantation. It is worth noting that the exposure of extracellular matrix can stimulate the initiation of coagulation pathway in vivo, so when the material surface has good histocompatibility, it should also actively avoid the possible decline in blood compatibility.
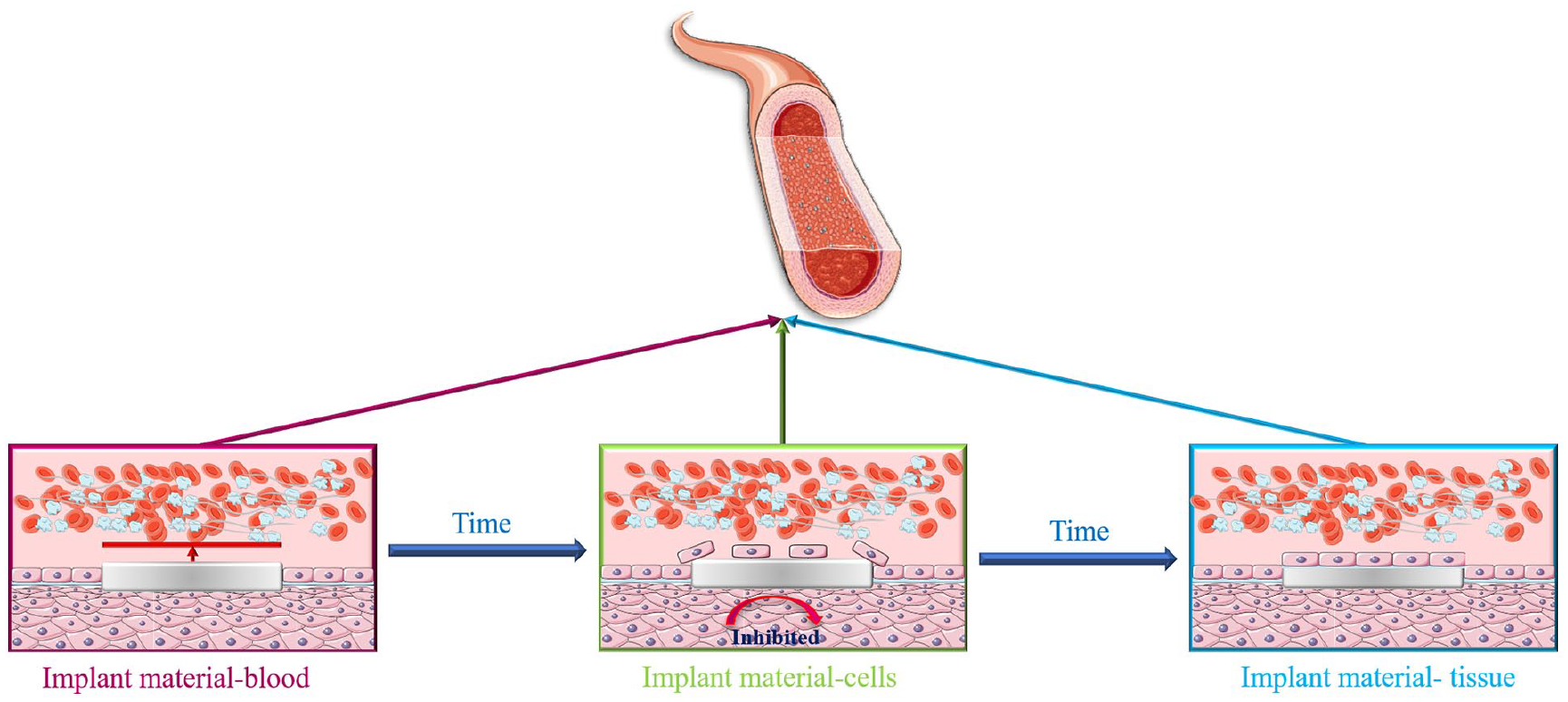
The relationship between materials and blood, cells, and tissues varied with implantation time.
Therefore, a reasonable endothelialization design strategy needs to meet the following requirements (Figure 4):
In the early stage of implantation, the surface of the material should inhibit coagulation and inflammation, while promoting the migration of ECs to the surface of the material to construct a biomimetic microenvironment of normal blood vessels.
In the middle stage of implantation, the proliferation of surface ECs should be enhanced to accelerate surface endothelial regeneration, so as to inhibit the abnormal proliferation of SMCs.
Long-term implantation should provide a good microenvironment for cell growth.
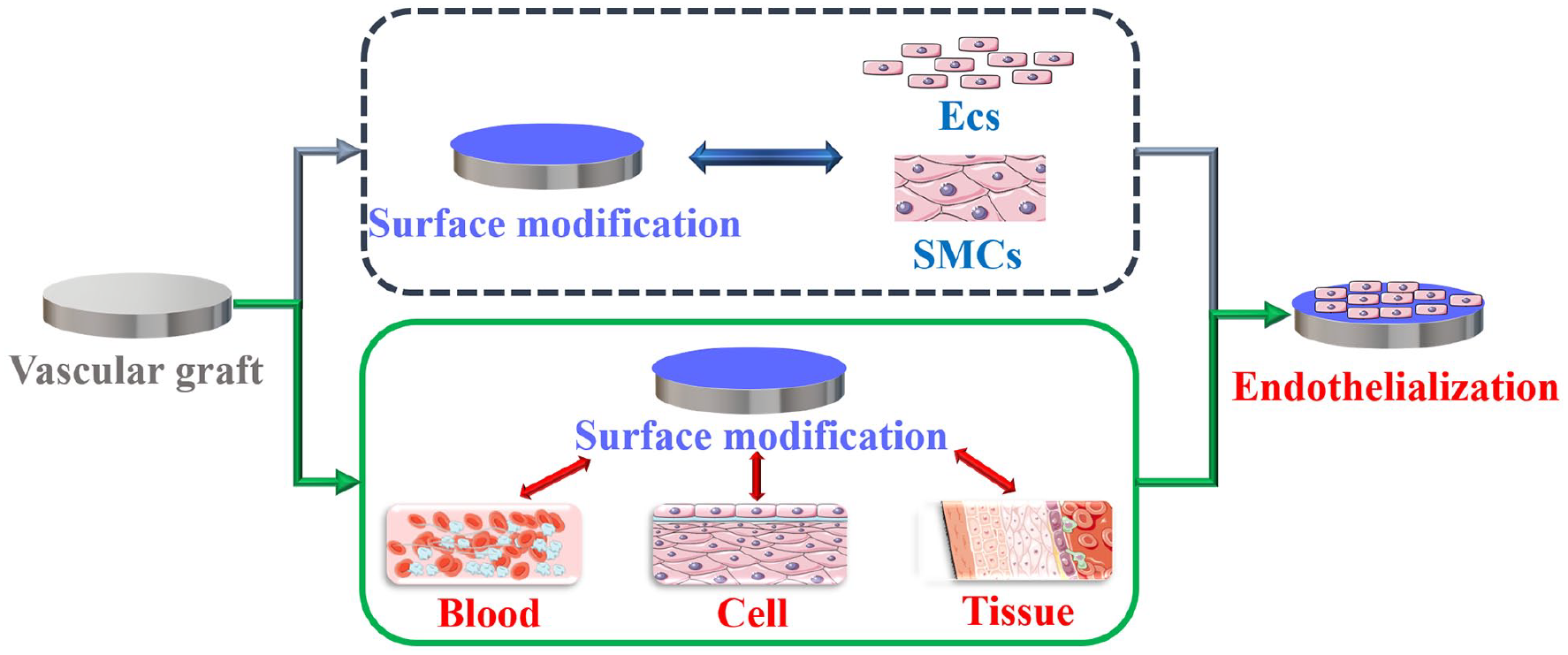
The traditional endothelialization design focuses on the interaction between materials and cells, aiming to enhance the rapid adhesion and proliferation of cells on the surface of materials, thereby realizing the formation of an endothelial cell layer on the surface. However, the reconstruction of blood vessels through endothelialization is a complex process, which requires the surface design of endothelialization not only to consider the interaction between materials and cells, but also to reflect the interconnection between materials and the entire vascular microenvironment, including: blood and vascular wall tissue.
How to construct a reasonable and selective surface that can induce the migration and proliferation of vascular endothelial cells will still be an important research direction for the surface endothelialization of vascular implants in the future. Researchers should take into account the temporal interaction between the material and the vascular environment during implantation, and design a modified method for three stages. Therefore, the ideal scheme to achieve rapid endothelialization of the material surface should not only be based on a single cell factor for material design, but also consider the correct construction of vascular microenvironment on the material surface, so as to guide the healthy repair of vascular tissue.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China, Grant/Award Numbers: 31870955#; National Key Research and Development of China, Grant/Award Number: SQ2020YFC1107303.
