Abstract
In this study, a detailed physical, chemical, and mechanical investigation of bone substitute (β-TCP/DCPD-PHBV) was carried out. In fact, it is composed of biocompatible materials such as ceramic phosphocalcic, consisting of tricalcium phosphate (β-TCP) and dihydrated dicalcium phosphate (DCPD) and 3-hydroxybutyrate-co-3-hydroxyvalerate (PHBV) polymer having a weight fraction 40%/60%. For these analyses, diverse techniques were used, including SEM-EDS, mercury porosimeter, Fourier Transform Infrared Spectroscopy, and, finally, uniaxial compression test machine. A morphological investigation of biomaterials using MEB revealed uneven particle shape and size, as well as a rough surface with a porous and microcracked structure. In fact, this architecture promotes the development of bone within biomaterials. Compositional studies applying FTIR technology, also, revealed the existence of chemical components, comparable to those found in the mineral phase of bone (Ca2+, PO43−, and HPO42−). The following compounds prove the bioactivity of
Introduction
With reference to the aging population, the pace of life speeding up, and the urban environment deteriorating, millions of people suffer from bone defects, caused by natural diseases, the incidence of road traffic, sports, and work injuries. Thus, its treatment has become a major clinical operation. 1
For decades, autograft has been the gold standard for bone grafts. Indeed, the most significant advantages are the flawless biocompatibility of this orthopedic remedy as well as the absence of virus transmission risk. 2 Nevertheless, this surgical technique has various drawbacks, to mention the need for two simultaneous surgeries such as taking the graft from one location and settling it in another. Furthermore, limited amount of bone graft volume and donor-site morbidity have established the need for the development of alternative methods such as scaffold-based tissue engineering (TE). 3
Actually, the development of biomaterials to imitate the structure and properties of human natural bone is one of the major challenges in tissue engineering. With the introduction of synthetic bone substitutes in orthopedics, implanting biomaterials into bone tissue to restore the functionality of the treated organ, has become a frequent procedure.4,5
Since bone ingrowth into the implant is a very complex phenomenon involving a cascade of cellular and extracellular biological events, the choice of the most suitable bone substitutes requires careful selection. For that, the function and overall success of the biomaterial depend on a number of morphological, chemical, and mechanical parameters (including porosity, shape, size of pores, and compressive stress), affecting the rate of bone ingrowth.
As the bone structure is a composite of organic and inorganic phases, the new generation of synthetic bone substitutes is generally composed by these two material classes.
Tricalcium phosphate (Ca3(PO4)2), sintered hydroxyapatite Ca10(PO4)6(OH), alumina (Al2O3), DCPD-designed dicalcium phosphate dihydrate Ca(HPO)4 2H2O, and CaP cements all bond to living bone spontaneously.6,7 In fact, they have been known as promising biomaterials thanks to their comparable chemical compositions to those of human bone. 8
Copolymers of poly (3-hydroxybutyrate-co-4-hydroxybutyrate), P3HB-4HB, P3HB-3HB, and poly (3-hydroxybutyrate-co-3-hydroxyhexanoate) (PHBV)9,10 are other categories of biomaterials that have been developed as scaffolds for tissue engineering, including cartilage and bone repair.4,8 These biomaterials are applied for their numerous advantages, such as good mechanical properties, durability, resistance to chemical and biological corrosion, as well as low production cost. In addition, they possess specific biological properties like good biocompatibility. 4 Another advantage is that their degradation products are non-toxic. In-vivo, these biomaterials will be transformed into carbon dioxide and water, over a required period of time. However, the hydrophobic property of this polyester limits cell attachment and growth. So, it should be combined with hydrophilic biomaterial to enhance cell attachment. 11
In this research, the CPC; calcium phosphate cement is chosen as the first component of the biomaterial elaborated. It is highly appealing, with many advantages, such as minimally invasive surgery. Additionally, it can be molded to optimally fill irregular bone defects. However, a major drawback of CPC is its poor degradability and brittleness, a fact that necessitates considering additional materials in its applications.12–14 Furthermore, the calcium phosphate ceramic has a basic pH; therefore, it is possible to associate it with a biodegradable polymer in order to neutralize the resultant biomaterial. 15 Accordingly, the second constituent biomaterial used in this research is the PHBV polymer, which is naturally generated by bacteria as intracytoplasmic granules. It has, also, been identified as a promising bone implant material owing to its natural degradability, good biocompatibility, as well as favorable mechanical properties. It does, absolutely, have enough ductility to allow for simple surgical manipulation.16,17
Over the previous 10 years, the incorporation of powdered CPC into the polymer matrix in order to produce biodegradable composite biomaterial (polymer-phosphocalcic ceramics) has been critical in overcoming the aforementioned shortcomings when combining the excellent ductility of biopolymers with the bioactivity of ceramics. Those biocomposites are the most widely used bone substitutes. 18 They generally have great biocompatibility and bioactivity, good mechanical strength, and also an excellent ability to adapt to human organisms.
The main parameters of catalysts, such as texture, porosity, and chemical surface, are determinants to relate the catalytic activity to the property of the scaffold. The purpose of this study is to present the morphological, chemical, and mechanical properties of the bioresorbable bone implant
Materials and methods
Materials
Phosphocalcic ceramic cement consisting of tricalcium phosphate (β-TCP) and dehydrated dicalcium phosphate (DCPD) and 3-hydroxybutyrate-co-3-hydroxyvalerate (PHBV) are basic components of implant material used in this research. This composite’s weight fraction is 40/60.
The

Its characteristics were determined using several techniques and experimental conditions, including scanning electron microscopy (SEM), mercury porosymetry, infrared spectroscopy, and, uniaxial compression testing was conducted to determine the specimen’s properties under compressive loading.
Methods
SEM analysis
The microstructure of β-TCP/DCPD-PHBV biomaterial was analyzed using a scanning electron microscope (SEM) in the Center for Scanning Electron Microscopy and Microanalysis (CMEBA) of the University of Rennes 1, obtaining thereby magnified three-dimensional-like images of its surface.
Mercury porosimetry measurements
Mercury porosimetry experiments were performed using a PoreMaster mercury porosimeter (Quantachrome Instruments®, France). It is an extremely useful characterization technique for porous materials. Moreover, it provides a wide range of information, such as pore size distribution, total pore volume or porosity as well as the specific surface area of a sample. The instrument is automated for pore measurements that range from 0.0036 µm to more than 950 µm.
The measurements of the intruded volume of mercury versus applied pressure were obtained, and the pressures were converted into pore sizes using the Washburn equation (1), where

FTIR analysis
The samples were followed by Fourier Transform Infrared Absorption Spectroscopy (FTIR). It is a technique that analyzes chemical functional groups present in the material, through the detection of chemical bond vibration characteristics. Technically, this approach is consisted to expose a sample to infrared radiation and then quantify the wavelengths and intensities absorbed by the substance. To apply this chemical analysis, the materials are compressed into pellets using a pelletizer of 3 MPa pressure. Each pellet includes 30 mg of potassium bromide (KBr) and 3 mg of biocomposite.
Compression test
Compression testing determines the behavior of materials under a crushing load. The findings generally reveal the ultimate compressive strength, yield strength, elastic modulus, and toughness of a sample. To realize this analysis, a uniaxial compression test was carried out according to International Standard ISO 604. The compression machine used is of type QTEST/25, manufactured by MTS System Corporation. The specimen, which has a disk shape (13.1 mm in diameter and 5.1 mm in thickness), is placed between the loading platens and compressed at a constant rate of 0.2 mm/min until the sample is destroyed. The applied force and crosshead displacement data are collected in real-time. A diagram “Compressive stress-strain” is plotted and exploited to determine the Young modulus and max compressive strength.
Results
SEM analysis of β-TCP/DCPD-PHBV (40/60)
The micrographs of

Apparent microcracks on the surface of

Porosity structure in

SEM micrograph of an aggregate of composite β-TCP/DCPD-PHBV (40/60).
Porosity
Mass, sample volume, total porosity (Table 1), and pore size distribution (Figure 5) have been determined using the Mercury porosimetry analyzes. Based on these findings, the porous structure of this biomaterial is characterized by the formation of two types of pores: micropores (1–100 μm), which are the most prevalent, and macropores (
Some properties of β-TCP/DCPD-PHBV testing samples.

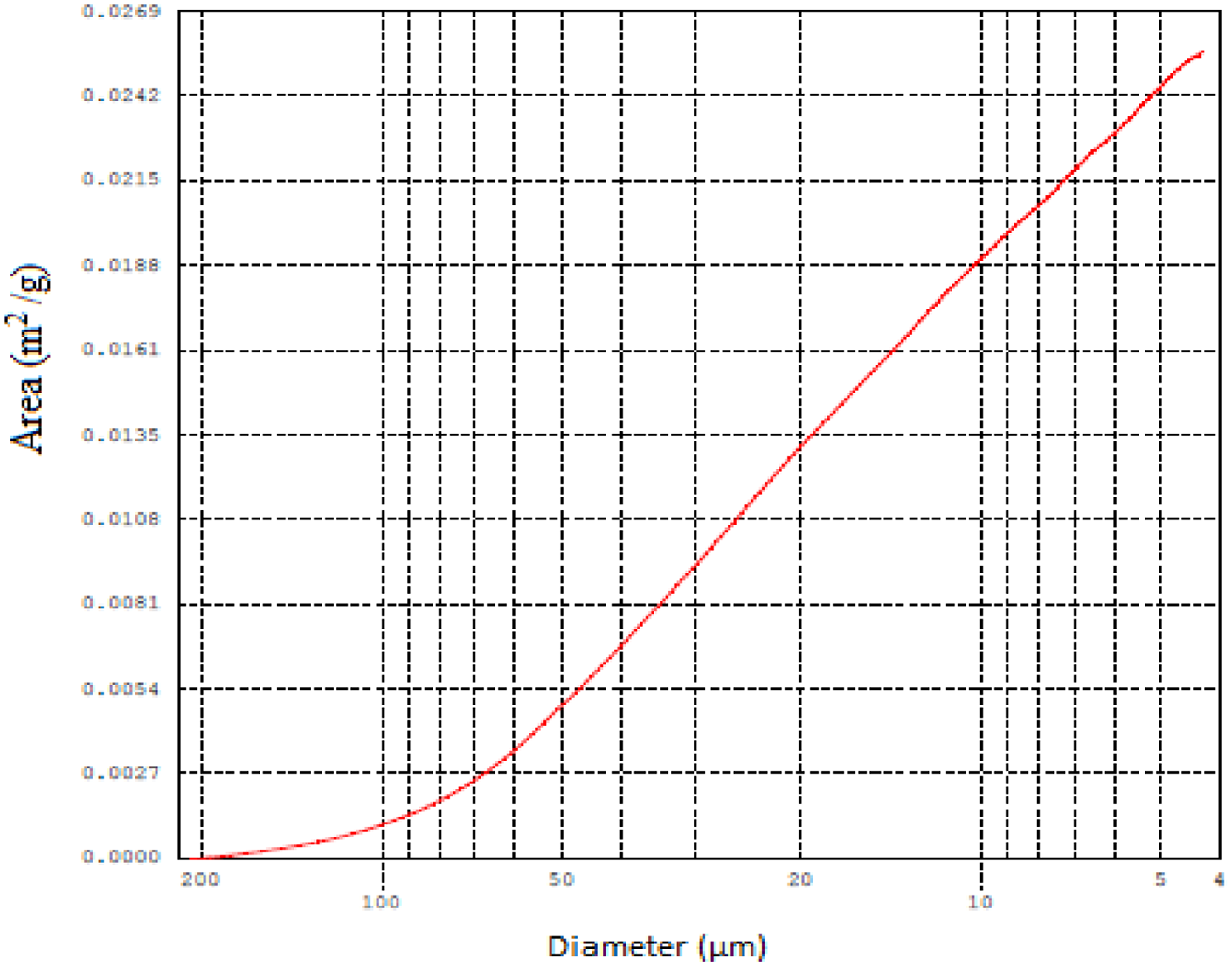
Pore size distribution.
Specter IR of β-TCP/DCPD-PHBV (40/60)
The absorption bands observed on the IR spectrum (Figure 6) showed the presence of the groups O-H positions at 948, 1278, and 3500 cm−1 respectively, as well as the C-H bands were observed at 2981 cm−1.
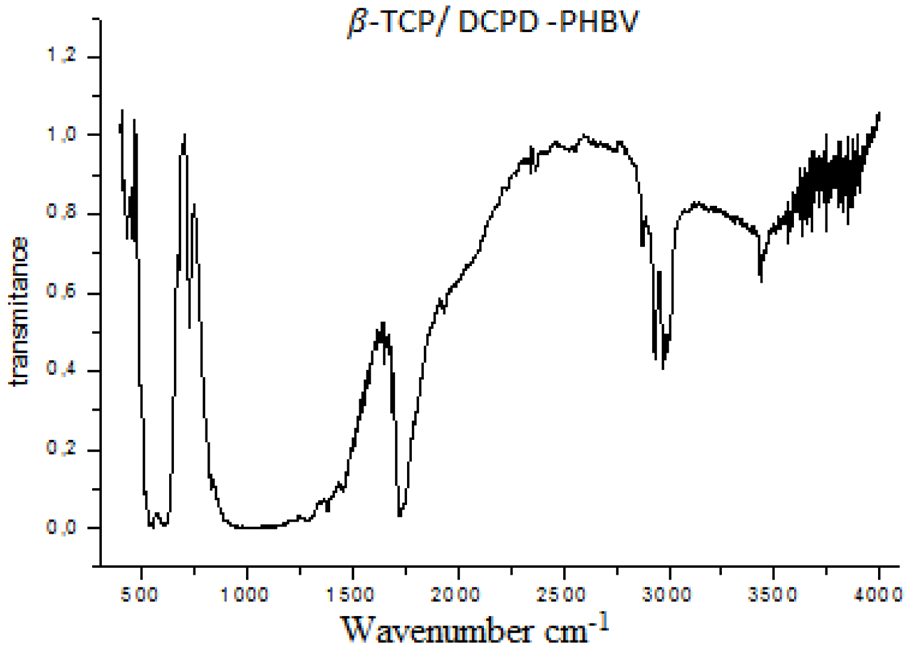
IR spectrum of
The C = O group band is detected at 1734 cm−1 and the PO43− group band is present at 458 cm−1, as well as the group HPO42− is at 544 cm−1.
Compression test results
The results of the compression tests are shown in Figure 7. Absolutely, these curves reflect the evolution of stress-strain given by five specimens. The average maximum stress in this test is 195.21 MPa, and the average Young modulus is 2050.68 MPa.
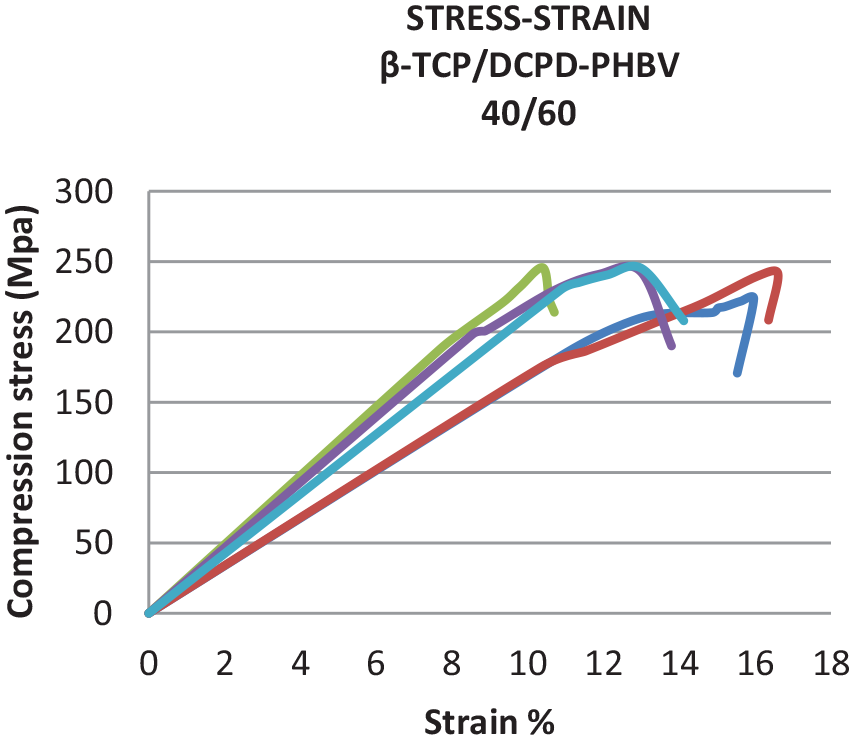
Curves Stress-Strain of
Discussion
In clinical practice, bone substitute materials are being used for reconstructive purposes, bone stock augmentation, and bone repair. Composites of ceramic phosphocalcic and various polymers, such as poly (lactic acid) (PLA), 20 gelatin, 21 poly-hydroxybutyrate (PHB), 22 and collagen, 23 have been successfully fabricated in recent years. They have also showed enhanced bone formation in vitro and/or in vivo. These bone substitute materials diversified in their topography, chemical composition, mechanical strength, and biological mechanism. Each one of them has its own specific advantages and disadvantages. 24
It is known that the intrinsic material properties of bone substitutes have a serious effect on their mechanical behavior and impact cellular responses,25,26 and therefore, they determine their effectiveness.
In order to discover certain intrinsic properties of the biomaterial
A further parameter determining the morphology of the bone growing into the biomaterial is the structure porousness. This physical property is likely to play a fundamental role at several levels: increasing the specific surface area of materials, creating a microenvironment for cell differentiation inside, and boosting the roughness and resorbability of the scaffold. 30 These various parameters, together with the chemical reactivity of the biomaterial, can influence the bioactivity of the material and its absorption capacity. 1
In this research, the
Micropores have a very small size, ranging between 1 and 100 μm in diameter. They are the most dominant type of pores in
FTIR analysis (Figure 6) showed that
Since their emergence, mechanical properties of bone substitutes have stimulated the interest of orthopedic researchers and producers of these types of biomaterials. Compressive strength is the most commonly used characteristic for assessing the mechanical properties of the bone scaffold. Since these materials will directly interact with the bone, their compressive strength must be comparable to that of human cortical. In addition, this mechanical precondition is necessary to prevent the substitute from crumbling or fracture during the surgical operation or after incorporation.
The mechanical strength of the
The mechanical properties are impacted not only by their base materials but also by other parameters, in particular the porosity. These two physical characteristics are fundamental for the biomaterial. As a result, a certain equilibration between porosity and mechanical properties must be considered to ensure a microenvironment favorable to colonization and cell multiplication. 33 Many studies have focused on the effect of porosity and pore size on biomaterial compressive strength; Barralet et al. 40 investigated the effect of porosity reduction by compaction on compressive strength and concluded that a reduction in porosity from 50% to 31% resulted in an increase in wet compressive strength from 4 to 37 MPa. Experimental analysis of the porous HAp given by Liu 41 revealed that for a given porosity volume, the compressive strength behaves roughly linearly with macropore size; a smaller macropore corresponds to a higher compressive strength.
Conclusion
As a conclusion for those experimental analyses, the porosity of this biomaterial is 58.5%, and it varies between micropores and macropores, giving to
Footnotes
Acknowledgements
The author acknowledges Professor RIDHA BEN CHIKH and Professor HICHEM SMAOUI for theirs scientific contributions. The author is grateful to Dr. SIWAR MOSBAHI and Mr. Bertrand Lefeuvre, for their technical assistance.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
