Abstract
A slowly degradable bone scaffold can well maintain the balance between new bone regeneration and scaffold resorption, esp. for seniors or patients suffering from pathological diseases, because too fast degradation can lead to the loss of long-term biological stability and result in scaffold failure. In this present study, calcium phosphate silicate (CPS) and polydimethylsiloxane (PDMS) were blended in different ratios to formulate slurries for scaffold fabrication. The effects of crosslinked PDMS on the CPS material properties were first characterized and the most viable formulation of CPS-PDMS slurry was determined based on the aforementioned results to 3D fabricate scaffolds. The biocompatibility of CPS-PDMS was further evaluated based on the scaffold extract’s cytotoxicity to osteoblast cells. Furthermore, real-time PCR was used to investigate the effects of scaffold extract to increase osteoblast proliferation. It is showed that the crosslinked PDMS interfered with CPS hydration and reduced both setting rate and compressive strength of CPS. In addition, CPS porosity was also found to increase with PDMS due to uneven water distribution as a result of increased hydrophobicity. Degradation and mineralization studies show that CPS-PDMS scaffold was slowly degradable and induced apatite formation. In addition, the in vitro analyses show that the CPS-PDMS scaffold did not exert any cytotoxic effect on osteoblast cells but could improve the cell proliferation via the TGFβ/BMP signaling pathway. In conclusion, CPS-PDMS scaffold is proved to be slowly degradable and biocompatible. Further analyses are therefore needed to demonstrate CPS-PDMS scaffold applications in bone regeneration.
Introduction
Bone fracture management is becoming increasingly important as a result of elevated number of critical fractures and their impacts at the socioeconomic level. 1 Although native bone is capable of self-regeneration, its regenerative capacity is severely impaired when fracture size is greater than the critical value.2,3 In case of critical-size bone fracture, bone graft (autograft, allograft or biomaterial) is usually needed to support regeneration and, among them, biomaterial scaffold has been showed to be superior to autografts or allografts.2,3
Scaffold is supposed to promote osseointegration between implant and native tissue in order to maintain sufficient structural integrity during degradation. 4 However, as a result of poor vascularization and low osteogenesis in seniors or patients who suffering from pathological bone diseases, for example, osteoporosis, new bone formation is unmatched with excessive resorption of bone or scaffold, affecting structural integrity between bone and scaffold and leading to implant failure.4–6 Therefore, slowly degradable scaffold is advantageous to maintain the balance between resorption and remodeling and becomes increasingly important in bone fracture management. 6 Among these biomaterials, self-setting calcium phosphate silicate (CPS) has been proved to be biocompatible, degradable as well as osteogenic and to encourage osseointegration in vivo.7–9
Ideal scaffold should be biocompatible, degradable and of proper macroporous structure to support cell migration, attachment and proliferation. 10 Macroporosity allows the diffusion of oxygen and nutrients in exchange of waste products and facilitates cell migration and attachment. 11 Another important aspect of ideal scaffold is appropriate degradation that can be matched with the rate of new bone growth. 12 Several studies have successfully fabricated CPS-based bone scaffolds and demonstrated promising results in bone regeneration, however, because of macroporosity, scaffold shows too fast degradation and causes severe inflammation and serious osteolysis.13–15 Therefore, one major objective of this present study is to develop macroporous CPS scaffold with slow degradation.
Unlike other studies blending highly hydrolytic polymers, e.g. polylactic acid or polyethylene glycol, to fabricate scaffolds, CPS-based composite was formulated with polydimethylsiloxane (PDMS) in this work to increase scaffold resistance to fast degradation. Medical-grade PDMS is biocompatible and has a long history in prothesis fabrication, e.g. artificial breast, nose etc.16,17 Recent studies have proved that the crosslinked PDMS is slowly degradable in vivo and hydrolyzed into monomeric dimethylsilanediols, which are finally decomposed into CO2 and silica.17–21 Thus far, to our knowledge, there is yet no study attempting to fabricate bone scaffold with PDMS and CPS and to evaluate its in vitro biocompatibility.
In this present work, the crosslinked PDMS was blended with the self-setting CPS to formulate a composite and the effects of PDMS concentrations on CPS setting time, compressive strength, porosity, hydrophilicity and crystallinity were characterized and analyzed. One formulation of CPS-PDMS slurries was selected to fabricate scaffold via the microextrusion-based 3D printing process and scaffold degradation and mineralization were also investigated. Furthermore, scaffold extract medium was prepared to evaluate its cytotoxicity to osteoblast cells and promoting effects on proliferation-specific gene expressions. These results suggest that the slow-degradable scaffold not only demonstrates in vitro biocompatibility but also encourages osteoblast proliferation.
Materials and methods
CPS-PDMS slurry preparation
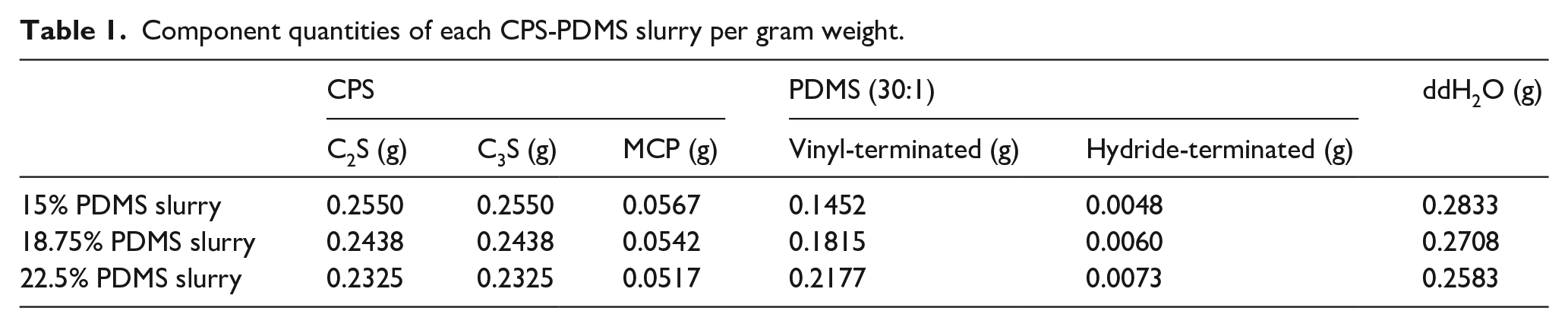
Dicalcium silicate (Ca2SiO4, C2S) and tricalcium silicate (Ca3SiO5, C3S) were synthesized via the sol-gel method as previously described and 10% of monocalcium phosphate (Ca(H2PO4)2, MCP, Sigma-Aldrich, USA) was subsequently combined with 45% C2S and C3S to prepare self-setting CPS precursor. 22 The CPS precursor powder was homogenized by grinding in the planetary ball mill (YXQM-2L, MITR, China) for 4 h. The two-component PDMS composed of vinyl-terminated PDMS (Mw ~25000, Sigma-Aldrich, USA) and hydride-terminated PDMS (Mn ~580, Sigma-Aldrich, USA) at a mass ratio of 30:1, and 0.1 wt% platinum chloride (Sigma-Aldrich, USA) was added as the catalyst to accelerate the crosslinking reaction between these two components (Figure 1). 23 The two-component PDMS was blended with the CPS precursor, and deionized water (ddH2O) was added to initiate the hydration reaction of CPS. The actual compositions of CPS-PDMS slurry are presented in Table 1.
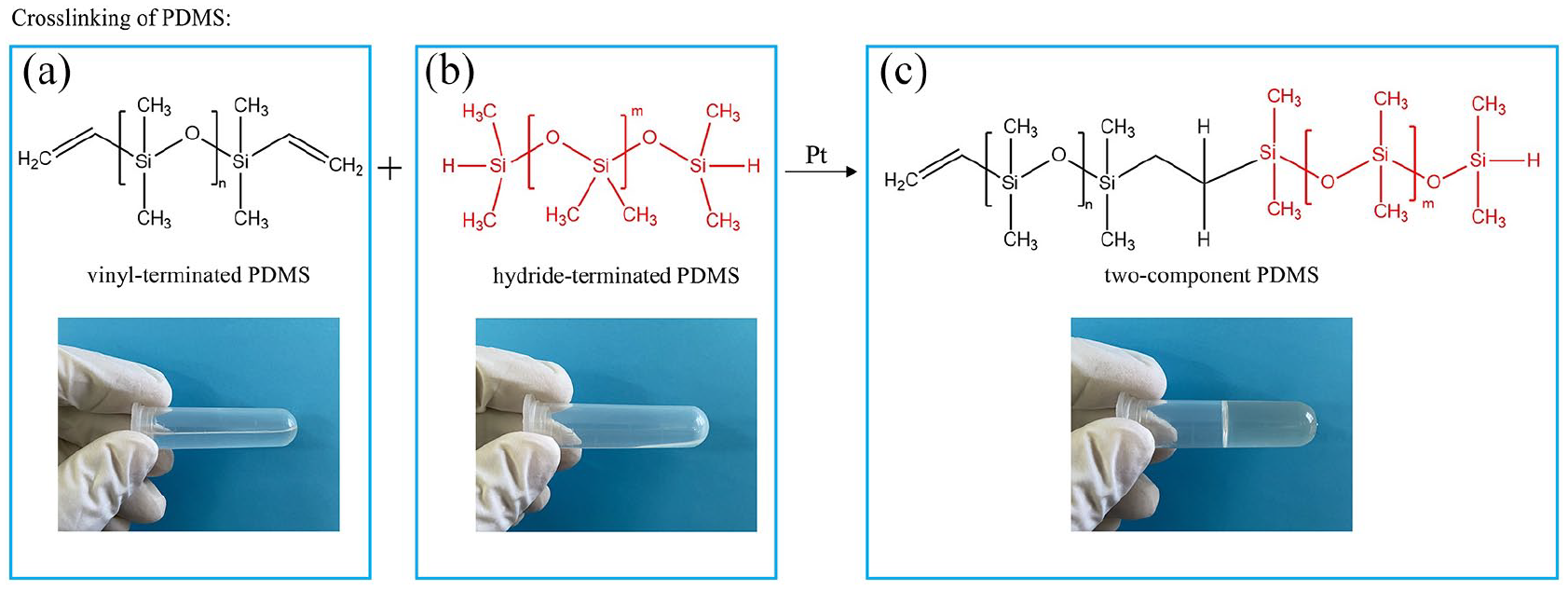
The schematic illustration of the crosslinking reaction between: (a) vinyl-terminated PDMS, (b) hydride-terminated PDMS into the (c) two-component PDMS. It is showed that both vinyl-terminated and hydride-terminated PDMS were in liquid form and the crosslinked two-component PDMS became a gel.
Component quantities of each CPS-PDMS slurry per gram weight.
CPS-PDMS slurry characterization
Setting times of different CPS-PDMS slurries were determined in accordance with the ASTM standard test method (ASTM ID: C191-08) and only final setting times were recorded. In brief, slurries were casted into 17 mm × 2 mm (diameter × height) polystyrene molds. Every 5 min, slurries were placed under the Vicat needle apparatus. Subsequently, the Vicat needle (ϕ = 1 mm and 8 g) was rested on the surface of slurry sample for 1–2 s and lifted. Slurry was considered set (hardened) when no visible mark of the Vicat needle was impressed on surface and the elapsed time since the initial contact between CPS and ddH2O was recorded as the final setting time. There were four replicates for each slurry formulation (n = 4).
The testing of compressive strength of hardened CPS-PDMS sample was performed on the universal testing system (UTS, Instron 5944, USA). In brief, CPS-PDMS slurries were casted into polystyrene-based cylindrical molds (6 mm × 12 mm, diameter × height) and placed in the 100% relative humidity incubator for 7 days at 37°C. Subsequently, CPS-PDMS cylinders were demolded and examined for their compressive strengths on the UTS equipped with a 2 kN load cell at a crosshead speed of 1 mm/min. There were four replicates for each CPS-PDMS formulation (n = 4). The Archimedes method was used to measure the porosity in hardened CPS-PDMS sample after 7 days. In brief, sample was dried in air and weighted to record the dry weight (Wdry). Subsequently, it was placed in ddH2O under vacuum for 4 h and removed. The wet weight was recorded as Wwet. Finally, it was immersed in ddH2O again and the weight in water was recorded as Wwater. Sample porosity was calculated in the following equation: porosity (%) = (Wwet – Wdry)/(Wwet – Wwater) × 100%. There were four replicates for each CPS-PDMS formulation (n = 4).
To assess the effects of PDMS on the CPS hydrophilicity, the CPS-PDMS samples were hydrated for 7 days and the water contact angles on the CPS-PDMS surfaces were then measured by using static sessile drop technique at room temperature. In brief, a water droplet of approximately 2–3 µL was placed on the sample surface and the water contact angle was captured and measured by the contact angle goniometer (OSA200, NBSI, China) equipped with a high-resolution camera and image processing software. There were four replicates for each CPS-PDMS formulation (n = 4).
Hardened CPS-PDMS samples for 7 days were ground into fine powders (particles size
CPS-PDMS scaffold fabrication
The slurry formulation including 15 wt% of PDMS was used to fabricate scaffolds with different geometries and pore sizes. In brief, the CPS precursor was first blended with the two-component PDMS with catalyst and ddH2O (CPS:ddH2O = 2:1) was mixed to prepare the slurry. Thereafter, a 5-mL plastic syringe was filled with the slurry and placed in the lab-made motor assisted micro-syringe system (3D printer). Solidworks (USA) was used to design the geometries (cubic and cylinder) of scaffold and the spacings (400 and 600 μm) between extruded filaments. Subsequently, the STL files of scaffold were exported into the 3D printer to fabricate designed scaffolds. The extruded filament diameter was 400 μm. After fabrication, scaffolds were placed in the 100% relative humidity incubator at 37°C for 7 days to achieve sufficient mechanical strength before the following characterizations.
CPS-PDMS scaffold characterization
In the degradation study, cps-pdms scaffold was weighted (initial dry weight, Wi) and immersed in the 0.05 M Tris-HCl solution (200 mL/g, pH 7.4) at 37°C for 1, 3, 7, 14, 21, 28 and 35 days. At each endpoint, scaffold was removed from solution and desiccated at 37°C for 24 h. The pH of Tris-HCl solution was recorded for each endpoint. Subsequently, the final dry weight (Wf) of scaffold was recorded and the percentage weight loss (% degradation) was calculated by the following equation: % degradation = (Wi − Wf)/Wi × 100%. Finally, the compressive strength testing of scaffold was performed on the UTS. For each endpoint, there were three replicates (n = 3).
In addition, scaffolds (n = 3) were placed in the prepared simulated body fluid (SBF, pH ~7.4, 200 mL/g, including 142 mM Na+, 5 mM K+, 1.5 mM Mg2+, 2.5 mM Ca2+, 148.8 mM Cl−, 4.2 mM HCO3−, 1 mM HPO42− and 0.5 mM SO42−) at 37°C for 14 days. Afterward, scaffolds were removed from SBF and rinsed with ddH2O for 1 min. subsequently, the cross-sections of scaffold were sputter coated with gold and observed in the scanning electron microscope (SEM, S-3000N, Hitachi, Japan) to evaluate the in vitro mineralization on CPS-PDMS scaffold.
Osteoblast isolation
Osteoblast cells were isolated from calvaria of neonatal (<2 days old) Sprague-Dawley rats by an enzymatic digestive process as previously described. 24 In brief, rat calvaria fragments were washed three times with phosphate buffer saline (PBS, pH = 7.4) and digested in the 0.25% trypsin–EDTA solution at 37°C for 20 min. Osteoblast cells were released from calvaria in 1 mg/mL collagenase (Gibco, USA) solution at 37°C for 90 min and collected by centrifugation at 1000 rpm for 10 min. Cells were subsequently incubated in T25 culture flasks (ThermoFisher, USA) containing 10 mL of 10% fetal bovine serum (FBS, Gibco, USA)-Dulbecco’s modified eagle’s medium (DMEM, Sigma-Aldrich, USA) culture medium at 37°C and 5% CO2. Culture medium was replaced every 2 days and only cells between the second and fourth passages were used in the following biocompatibility analyses.
CPS-PDMS scaffold biocompatibility study
The cytotoxicity of CPS-PDMS scaffold was evaluated based on the cytotoxicity of scaffold extract to osteoblast cells in accordance with the ISO 10993-5:2009 standard. In brief, scaffold was first placed in 10 mL of PBS at 37°C for 3 days and removed. The leftover PBS solution is called scaffold extract and was sterilized by filtering through a 0.22 µm membrane. Subsequently, three amounts of scaffold extract were combined with 10% FBS-DMEM culture medium to formulate different extract culture medium (extract/total extract culture medium = 10%, 20% and 30%). To evaluate the cytotoxicity of extract culture medium, osteoblast cells were seeded in a 96-well culture plate at a density of 5 × 103 cells/well and 100 μL of extract culture medium was added into each well. Thereafter, cell-seeded plate was incubated at 37°C and 5% CO2 for 1 day and cells incubated with the FBS-DMEM culture medium without extract were served as the control group. The optical density (OD) of viable cells was determined with the help of CCK-8 assay (Dojindo, Japan) by the microplate reader (CLARIOstar, BMG, Germany) at 450 nm and the relative cell viability was calculated as ODscaffold/ODcontrol × 100%. Each group had six replicates (n = 6).
Polymerase chain reaction (PCR) was performed to evaluate CPS-PDMS biocompatibility. In brief, osteoblast cells were incubated with 10% extract culture medium at 37°C for 3 days for total RNA extraction. Total RNA was isolated via an RNA isolation kit (RNeasy Mini Kit, Qiagen, Germany) and reversely transcribed into complementary DNA (cDNA). Cells co-cultured with extract-free FBS-DMEM culture medium were served as the control group. In the first step of PCR analyses, 84 genes related to the TGFβ/BMP signaling pathway were examined based on commercial gene arrays (RT 2 Profiler PCR Array, Qiagen, Germany) in accordance with the manufacturer instruction and each group had three replicates (n = 3).
In the second and last steps of PCR analyses, eight genes (bmp2, bmp5, bmp7, smad2, smad4, smad5, smad6 and runx1) and two genes (runx1 and samd5) found in the TGFβ/BMP signaling pathway were further analyzed by the real-time PCR and droplet digital PCR (ddPCR) methods, respectively. Gene primers were designed and purchased from Takara Biology (Dalian, China) and the detailed information regarding primer sequences was given in Table 2. The β-actin gene was selected as the housekeeping gene. In the real-time PCR analysis, gene amplifications were carried out on the CFX Connect PCR machine (Bio-Rad, USA) in the following sequence: 30 s of 95°C initialization, 5 s of 95°C amplification, 30 s of data collection at 60°C for 40 cycles and each gene was repeated in four wells (n = 4). In the ddPCR study, droplets (90 µL) of cDNA templates were generated by the QX200 Droplet Generator (Bio-Rad, USA) and pipetted into the 96-well ddPCR plate (Bio-Rad, USA). The plate was amplified by a thermal cycler (T100 Thermal Cycle, ThermoFisher, USA) and then read on the QX200 Droplet Reader (Bio-Rad, USA). In the data analysis of real-time PCR results, fold changes of gene were calculated based on the 2ΔΔCt method and the relative fold change was calculated as fold changesample/fold changecontrol.
The gene sequences of forward and reverse primers.

Statistical analysis
The software SPSS® Statistics 19.0 (SPSS Inc., USA) was used in the statistical analyses of our data. In the comparison of multi-groups (n ⩾ 3), one-way ANOVA was performed to compare values among individual groups (p < 0.05); in the comparison between two groups, unpaired two-sample t-test was performed to investigate the significant level (p < 0.05).
Results
CPS-PDMS slurries characterization
Figure 2 shows the setting time, compressive strength and porosity of each CPS-PDMS slurry sample. It is observed that the setting rate, inversely relating to the final setting time, and compressive strength were decreased with PDMS concentration; the overall porosity of composite was shown to be the highest in the 22.5% CPS-PDMS sample. These results clearly indicate that the presence of the crosslinked PDMS could interfere with the hydration of CPS and reduced both the setting rate and compressive strength. Figure 3 shows the water contact angles of CPS and CPS-PDMS slurries after 7 days of hydration. It is showed that the water contact angle significantly increased with PDMS (50.2° for CPS vs. 138.4° for 22.5% PDMS in CPS).

The setting time (black, square), compressive strength (red, hexagon) and porosity (blue, circle) of the CPS-PDMS slurry samples (15%, 18.75% and 22.5% PDMS in CPS). In both compressive strength and porosity testing, CPS–PDMS slurries were hardened for 7 days.

The water contact angles of CPS and CPS–PDMS slurries (15%, 18.75% and 22.5% PDMS in CPS) after 7 days of hydration.
The XRD patterns of CPS-PDMS slurry samples are depicted in Figure 4. It is clearly revealed that, in all composite samples, C2S was not completely reacted and calcite (CaCO3) was formed after hydration of CPS. The results obtained from Raman spectra (Figure 5) strongly agreed with the XRD analysis. The band at 491 cm−1 was attributed to the Si-O-Si stretching vibration, which was resulted from the hydration of CPS and crosslinking of PDMS. The rocking modes of CH3 in PDMS could be found at 790 and 857 cm−1. Bands near 708 and 1410 cm−1 were assigned to the Si-C symmetric stretching and CH3 asymmetric bending modes, respectively. The v1 symmetric stretching of P-O bond was near 963 cm−1 and the v3 asymmetric stretching was found between 1020 and 1100 cm−1, which coincided with the v1 symmetric stretching mode of carbonate ion (CO32−) at 1085 cm−1.
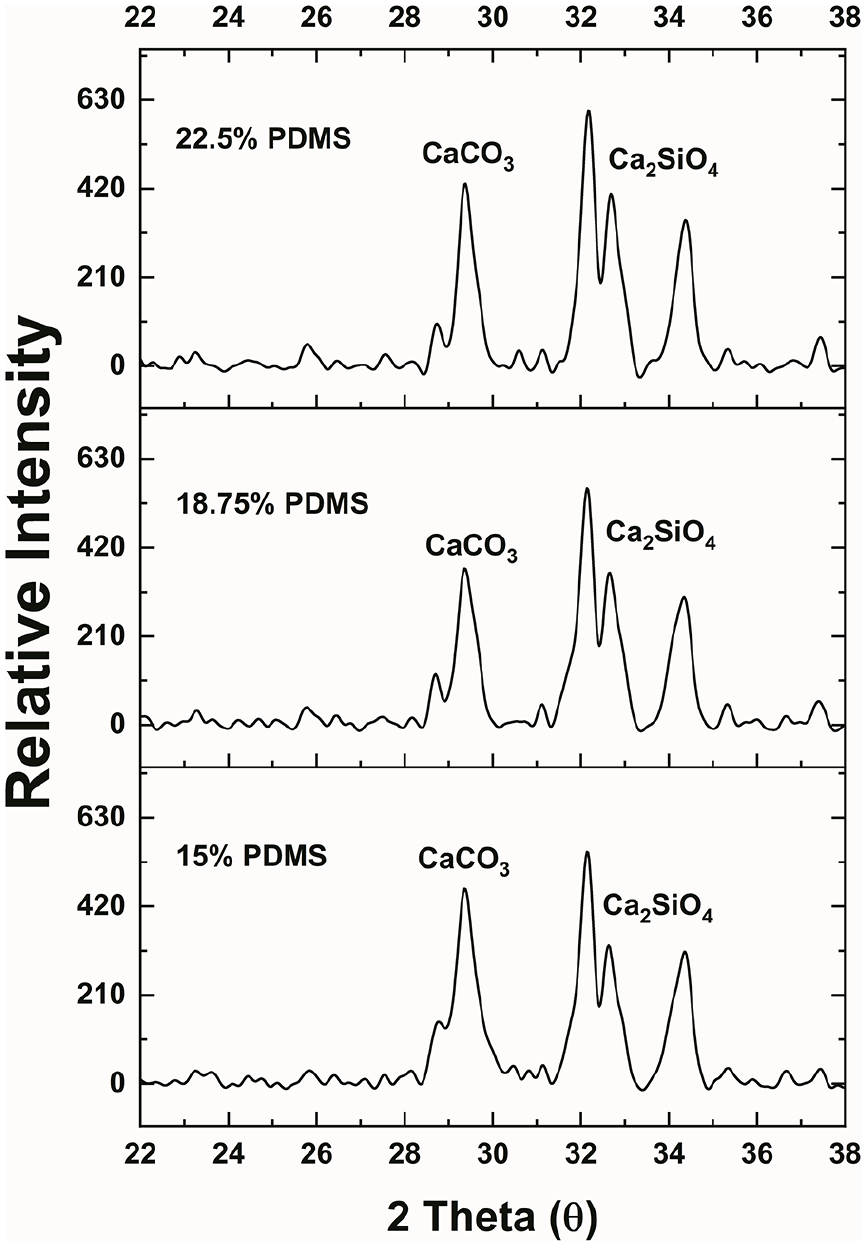
The XRD patterns of CPS–PDMS slurries (15%, 18.75% and 22.5% PDMS in CPS) after 7 days of hydration.

The Raman spectra of CPS–PDMS slurries (15%, 18.75% and 22.5% PDMS in CPS) after 7 days of hydration. Si–O–Si stretching vibration: 491 cm−1; Si–C symmetric stretching mode: 708 cm−1; CH3 rocking modes: 790 and 857 cm−1; v1 symmetric stretching of P–O: 963 cm−1; v3 asymmetric stretching of P–O: 1020 and 1100 cm−1; v1 symmetric stretching mode of CO32−: 1085 cm−1; CH3 asymmetric bending mode: 1410 cm−1.
CPS-PDMS scaffold characterization
Figure 6(a)and (b) display 3D printed CPS-PDMS scaffolds of different geometries. In both geometries, two filament-spacings were imparted in scaffold fabrication. It is clearly showed that the current formulation of CPS-PDMS slurry could be successfully fabricated into scaffold and the designed shape as well as microstructure of scaffold were well maintained without scaffold being collapsed. On the other hand, Figure 6(c) and (d) demonstrate the microstructure changes of scaffold before and after the in vitro mineralization study. In Figure 6(c), it is showed that the actual diameter of extruded filament was larger than the expected value (400 μm) and, as a result, the square space enclosed by any four filaments appeared to be smaller than expected. As compared to Figure 6(c), Figure 6(d) clearly indicates that the size or diameter of filament increased further after the mineralization study, implying that apatite-like minerals had been precipitated on surfaces of filament.

The optical images of cubic (a) and cylindrical (b) scaffolds with two printing patterns (400 and 600 µm spacing). One scale bar represents 1 cm. The SEM images of CPS-PDMS (15%) scaffold before (c) and after (d) immersing in SBF for 14 days.
The degradation of CPS-PDMS scaffold with time is depicted in Figure 7. In Figure 7(a), it is showed that the pH of Tris-HCl solution was continuously increasing over 35 days, implying that there was some degradation in scaffold over time. The weight loss of scaffold was also evidenced (Figure 7(b)), it is showed that the percentage weight loss of scaffold was increasing with time and, at the 35th day of degradation, there was approximately 29% of total weight loss occurred in scaffold. Furthermore, the reduction in compressive strength of scaffold was also remarkable, that is, the scaffold strength changed from 18 MPa at the first day of degradation study to only 6 MPa at the end of this study.

The degradation of 15% CPS–PDMS scaffolds in Tris-HCl solution for 35 days: (a) represents the change of pH in Tris-HCl solution over the study course and (b) demonstrates the weight and compressive strength loss of scaffolds.
CPS-PDMS scaffold biocompatibility
Figure 8 shows the relative viability of osteoblast cells co-cultured in both extract-free (control) and extract-formulated culture medium (samples). Since all the cell viabilities were normalized with respect to the value of the control group, the relative viability of the control is equal to 100%. It is clearly demonstrated that, regardless of the concentration of extract (10%, 20% or 30%) in FBS-DMEM medium, cells incubated with extract culture medium were significantly viable than those cultured with extract-free culture medium, implying that CPS-PDMS could improve osteoblast proliferation.

The relative viability of osteoblast cells cultured in extract-formulated culture medium (extract/total culture medium = 10%, 20% and 30%) for 1 day. Cells cultured in the extract-free culture medium was served as the control group. The relative viability was calculated as the ODsample/ODcontrol at the wavelength of 450 nm. Each group was repeated in six technical replicates (n = 6).
Gene expressions were compared between cells cultured in the extract culture medium (15% CPS-PDMS scaffold) and cells cultured in the extract-free culture medium (control) and the results from PCR analyses are given in Figure 9. In Figure 9(a), it is showed that the expressions of most genes from the TGFβ/BMP signaling pathway were found statistically unchanged and only nodal gene was significantly upregulated (p < 0.05), implying that this part of result was inconclusive and further analyses were required. In the real-time PCR analysis (Figure 9(b)), eight genes were compared and six of them (smad2, smad4, smad5, smad6, bmp7 and runx1) were found to be significantly (p < 0.05) upregulated. In the last ddPCR analysis (Figure 9(c)), genes, runx1 and smad5, were further compared between the sample and control groups and it is showed that both genes were significantly upregulated (p < 0.05), indicating that CPS-PDMS can improve osteoblast proliferation via the TGFβ/BMP signaling pathway.

The gene expression of osteoblast cells cultured in the extract medium as compared to the control group. There were 84 TGFβ-BMP signaling pathway-related genes were examined by the pre-prepared PCR array and gene expressions are depicted in the volcano plot (a). Each group had three biological replicates (n = 3). The volcano plot of real-time PCR results illustrated in (b). Each group was repeated in four technical replicates (n = 4). The bar chart of gene expressions found in ddPCR is showed in (c). Each group was repeated in three technical replicates (n = 3).
Discussion
Impacts of PDMS on CPS hydration
One of the primary goals in this present study is formulate a printable CPS-PDMS slurry, which can ensure the scaffold, after printed, to maintain the shape and microstructure as designed. Therefore, it is important to understand the mechanisms of crosslinked PDMS impacts on CPS material properties, for example, setting time, strength, crystallinity etc. In our study, CPS precursor was made of C2S and C3S in equal amount and 10% of MCP. The hydration reaction between C2S or C3S and water produces calcium silicate hydrate (C-S-H) and calcium hydroxide (Ca(OH)2).25,26 C-S-H is polymerized into C-S-H gel with time and becomes responsible for CPS slurry hardening and compressive strength. As showed in Figure 2, it is found that both setting rate and compressive strength of CPS were affected by the crosslinked PDMS. This may be attributed to the hydrophobic nature of PDMS. 27 In Figure 3, it is showed that the water contact angle significantly increased with PDMS in CPS, implying that the CPS-PDMS slurries became hydrophobic. Because water is repelled by PDMS, C2S or C3S cannot access water freely and hydration is impeded. As a result of deficient formation of C-S-H gel, both the setting rate and mechanical strength are reduced. Furthermore, the uneven distribution of water in CPS-PDMS slurry can also lead to the formation of macroscopic pores and increases the overall porosity, which is inversely related to the compressive strength.
XRD and Raman were also performed to investigate the interference of the crosslinked PDMS on CPS hydration. In Figure 4, it is showed that only C2S was detected on the XRD patterns of all CPS-PDMS slurry samples. Because C3S hydrates at a much faster rate than C2S, in CPS-PDMS samples after 7 days of hydration, C3S was completely consumed and only a fraction of C2S was hydrated. The formation of CaCO3 can be attributed to the reaction between Ca(OH)2 and carbonate acid as a result of carbon dioxide dissolution in water. 26 The formation of CaCO3 was also confirmed on the Raman spectra (Figure 5). The material characterization study strongly indicates that the crosslinked PDMS can result in uneven distribution of hydration and affect CPS setting time and mechanical strength in a concentration-dependent manner.
Degradation and mineralization of CPS-PDMS scaffold
In order to achieve the maximum mechanical property of printed scaffold, the slurry formulation of 15% was used to fabricate scaffolds via 3D printing. However, in our study, rheology of CPS-PDMS slurry was not performed because the hydration of C2S and C3S is a continuous process. As a result, hardened CPS particle can easily scratch the plates in rheometer and damage equipment. In fact, the 15% CPS-PDMS slurry was fabricated in the 3D printer immediately after prepared to avoid blockage in the printer nozzle.
As showed in Figure 6(a) and (b), this formulation of slurry can be fabricated into two different geometries with different inter-filament spacings. In the SEM picture of scaffold cross-section before mineralization (Figure 6(c)), the filament diameter is larger than the expected value because the layers above compress those below, resulting in some deformation of filament. In the SEM picture of scaffold cross-section after mineralization (Figure 6(d)), the further increase in filament diameter can be attributed to the precipitation of apatite on filaments.
Because the crosslinked PDMS is a slow-degradable polymer, the degradation of scaffold can be mainly attributed to the following sources. As revealed in the XRD patterns and Raman spectra, there are unreacted C2S and newly-formed CaCO3 in the CPS-PDMS scaffold. In addition, MCP reacts with Ca(OH)2 to form apatite and water. Because hydrophobic PDMS prevents water from dissolving unreacted C2S and both CaCO3 and apatite are considered slowly degradable, CPS-PDMS scaffold demonstrates slower degradation than scaffolds fabricated of calcium phosphate or calcium silicate (CPS-PDMS vs. calcium silicate: 29% vs. 50%).13,28 Meanwhile, the degradation of scaffold in terms of weight loss has a great impact on the scaffold mechanical strength. The compressive strength of scaffold after 35 days of degradation is only one-third of the initial strength; however, this strength is still much higher than that of native cancellous bone tissue (~ 2 MPa). 29
CPS-PDMS scaffold biocompatibility
In this work, the biocompatibility of CPS-PDMS scaffold was first evaluated based on scaffold cytotoxicity to osteoblast cells. According to the ISO 10993-5:2009 standard, cytotoxicity can be evaluated by extract, indirect- or direct-contact method. 30 Because the extract method is more suitable to evaluate the degradation products of CPS-PDMS and yields results consistent with in vivo results, this method is used in our present study. In practice, material elution, also called extract, should be diluted with culture medium in different concentrations to evaluate concentration-dependent cytotoxicity. In our study, the scaffold extract was combined with FBS-DMEM culture medium in 10%, 20% and 30% (v:v) to formulate the extract culture medium and osteoblast cells were incubated in the extract culture medium for 1 day to test the cytotoxicity. As showed in Figure 8, the CPS-PDMS scaffold showed no cytotoxicity to osteoblast cells at all dilution levels and the cell viability was significantly increased after the incubation with extract culture medium. Moreover, no statistical difference in cell viability was detected among three dilution levels, imply that the cytotoxicity of CPS-PDMS scaffold to osteoblast cells is not concentration-dependent.
Since the scaffold extract can improve the viability of osteoblast cells, a series of PCR analyses were performed to verify the effects of CPS-PDMS on osteoblast proliferation. TGFβ/BMP signaling pathway plays an important role in skeletal development and bone homeostasis, and can regulate the proliferation of osteoblasts.31,32 Hence, the first PCR analysis is PCR array study, in which 84 genes relating to the TGFβ/BMP signaling pathway were analyzed together on the same array plate, and it is found that only the nodal gene was significantly upregulated. Ligands, like nodal and bmp (bone morphogenic proteins), can bind with the specific type-II receptors and mediate the downstreaming signal transduction. 33 Therefore, the PCR array result suggests that more genes involved in this signaling pathway may participate in osteoblast proliferation.
The PCR array analysis is robust and can test multiple genes at the same time; however, the accuracy of result is not as good as real-time PCR. Therefore, eight genes closely related to the nodal gene were further analyzed by the real-time PCR. As showed in Figure 9(b), the bmp7 gene was the most significantly upregulated. The protein encoded by bmp7 gene can induce the phosphorylation of smad5, whose gene expression was also significantly upregulated, and lead to the complex formation with the smad4 protein. Subsequently, this complex is translocated into the nucleus and upregulate other gene expressions, for example, runx1. In addition, the protein encoded by bmp7 forms a heterodimer with the protein encoded by the nodal gene and prevents the subsequent phosphorylation of smad2. Therefore, the insignificant upregulation of smad2 gene is attributed to the significant upregulation of bmp7 gene.
Although smad5 and runx1 genes were found to be statistically upregulated by the real-time PCR analysis, the fold regulations of two genes were much lower than that of bmp7. Therefore, the ddPCR analysis was performed to evaluate the expressions of these two genes. In this analysis, two genes were again showed to be statistically upregulated in terms of the number of exact copies of target gene per unit volume of cDNA templates. The above in vitro analyses suggest CPS-PDMS scaffold is not cytotoxic to osteoblast cells and improve cell proliferation via the bmp7-samd5/smad4-runx1 signaling transduction.
Conclusions
In this work, we developed different formulations of CPS-PDMS slurries and evaluated the impacts of the crosslinked PDMS on CPS material properties. Among these formulations, one formulation containing 15% the crosslinked PDMS in CPS was further fabricated into scaffold via 3D printing. This CPS-PDMS scaffold is showed to be slowly degradable and induce mineralization in SBF. Subsequently, biocompatibility tests were performed and demonstrates that CPS-PDMS scaffold has not cytotoxicity to osteoblast cells and improve cell in vitro proliferation. However, there are some limitations in this present study. For example, the osteogenic effects and bioactivity of CPS-PDMS scaffold were not analyzed. Moreover, animal studies are also required to investigate scaffold in vivo effects on bone regeneration. Therefore, the findings from this present work show that CPS-PDMS scaffold can be a good candidate graft in bone regeneration and these positive results warrant further evaluations and studies in the future.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was sponsored by the Project 51502034 supported by National Natural Science Foundation of China; Project 51872042 supported by National Natural Science Foundation of China; and Project N181908006 supported by Fundamental Research Funds for Northeastern University.
