Abstract
Objectives:
The aim of this study is to evaluate the effect of pH treatment on the formation of hydroxyapatite (HA) coating layer and the biodegradation rate of hydroxyapatite coated AZ31 alloy for applications in biodegradable implants.
Methods:
HA was grown on the surface of AZ31 substrate at three different pH solutions of 7.5, 9.0, and 10.5 respectively by chemical solution treatment method. The coated samples were evaluated their biodegradation behavior by immersion test in the simulated body fluid (SBF) for 14 days. The biodegradation rate of the samples during immersion test was observed by Mg2+ ion quantification method and measuring the increase of pH of the medium.
Results:
HA coatings were successfully grown on the AZ31 substrate at three different pH conditions. The sample coated at pH 7.5 showed its uniform and dense coating layer resulted in highest corrosion resistance. While the highest biodegradation rate was observed for the sample coated at pH 10.5 due to many micro-cracks formed in the HA layer. After 14 days of immersion, Mg(OH)2 and calcium phosphate were corrosion products on the surface of the specimens coated at pH 10.5 and pH 9.0 respectively. While HA almost remained on the surface of sample coated at pH 7.5.
Conclusion:
AZ31 alloy coated with HA at pH 7.5 has lowest biodegradation rate and become suitable for applications in biodegradable implants.
Introduction
In the past decades, magnesium and magnesium alloys have demonstrated great potentials for applications in biodegradable orthopedics implants and cardiovascular devices for their biodegradability and biocompatibility.1–5 The mechanical properties and elastic modulus of Mg alloys are close to those of cortical bone, which may avoid stress shielding effect occurred in other metallic implants. 6 The biodegradability is considered as the biggest advantage of Mg alloys compared to other metallic biomaterials because they have the ability to degrade inside the human body to avoid the secondary surgery.5,6 Although these advantages of Mg alloys make their become potential candidates for use as bioabsorbable and biodegradable materials, their clinical application has been hindered due to their fast biodegradation in vitro and in vivo.7,8 For clinical use of Mg alloys in orthopedics implant or cardiovascular devices, a huge amount of studies has been conducted in order to improve their corrosion resistance. Many methods have been tried including alloying,9,10 heat treatment,11,12 reducing grain size, 13 and surface modification.14–17 Among above approaches, the surface modification such as calcium phosphate coating,14,15 micro-arc oxidation coating, 16 polymer coating, 17 have demonstrated to be the most effective methods that can improve corrosion resistance, biocompatibility, and bioactivity of the substrate. Calcium phosphate coatings including HA have demonstrated their ability to improve biocorrosion resistance of Mg alloys for their thermodynamic stability. 18 Especially, in addition to improve biocorrosion resistance and biocompatibility of the substrate, HA coating was reported to be able to accelerate the healing process during implantation.19,20 One of the most positive results was reported by Hiromoto et al. 14 that HA coating by chemical solution treatment was able to improve significantly biocompatibility and corrosion resistance of AZ31 alloy substrate. In that research, substrates were coated at pH 8.9 and were evaluated both in vitro and in vivo. Salman et al. 21 coated HA on the surface of AZ31 substrate by electrodeposited method in a coating solution with pH 6.0 for implant application. The results suggested that the biocorrosion resistance of the AZ31 substrate was improved with HA coating and secondary deposited HA during immersion was able to improve bioactivity of the materials. And recently, Ngo et al. 22 coated HA layer on the surface of AZ31 alloy at pH 7.5 and 8.6 respectively. The results suggested that the adhesion strength and corrosion resistance of HA coated AZ31 alloy were satisfied for application in biodegradable materials. Although demonstrated the positive effect of HA coating on the improvement of biodegradation resistance of AZ31 substrate, the previous research works only carried out with a narrow range of the pH of coated solution.
The objective of this study is to evaluate the biodegradation behavior of HA coated AZ31 alloy, which coated at wide range of pH conditions. The AZ31 alloy was chosen as a substrate because it contained low content of Al which may avoid the issues related to the toxicity of the released Al3+ ions. The effect of pH on the growth of HA layer on the AZ31 substrate was examined. The effect of the uniformity and crystal structure of HA layer on the degradation rate of the alloy was evaluated. And the effect of corrosion rate on the formation of corrosion products on the surface of the samples after immersion was also studied.
Methods
Material preparation
Extruded AZ31 magnesium alloy was used as substrate for coating process. The composition of AZ31 alloy was described in Table 1. The alloy bar was machined into round disc samples with the size of Φ15 × 2 (mm). The samples were then ground up to grit of #2000 using SiC papers. After that, the samples were ultrasonically cleaned with ethanol and dried in air. Finally, the samples were immersed in the coating solutions in 2 h at 90°C.
Composition of AZ31 alloy.

The coating solution were made as follows:
- Mixing the same volume of prepared from ethylenediaminetetraacetic acid calcium disodium salt hydrate (C10H12CaN2Na2O8, Ca-EDTA) 0.5 mol/L solution with potassium dihydrogen phosphate (KH2PO4) 0.5 mol/L solution.
- Sodium hydroxide (NaOH) solution was used for pH adjustment to 7.5, 9.0, and 10.5 respectively.
After immersed in the coating solution for 2 h, the samples were retrieved from the solution, cleaned with pure water, and dried for sample characterization and corrosion test.
Material characterization
The coated samples were then characterized by X-ray diffractometry (XRD) and scanning electron microscopy (SEM) to investigate the growth of HA coating layer. Specifically, the composition and crystal structure of the coating layer were characterized by Bruker D8 Advance X-ray diffractometer (XRD) using Cu Kα radiation. The surface morphology of the samples after coating was characterized by field-emission scanning electron microscope (SEM, JEOL 7600F).
Immersion test
The in vitro corrosion tests were carried out in a simulated body fluid (SBF) at 37°C for 14 days. The SBF used in this work was Hanks’s solution with the composition as follows: NaCl 8 g/L, KCl 0.4 g/L, Na2HPO4.H2O 0.06 g/L, KH2PO4 0.06 g/L, MgSO4.7H2O 0.2 g/L, NaHCO3 0.35 g/L, CaCl2 0.14 g/L. The pH of the SBF solution at the beginning was 7.4. The coated samples were immersed in the corrosion solution with the surface area to solution volume ratio of 50 mL:1 cm2. The solution containing samples were kept in the incubator at 37°C to maintain the solution temperature.
The degradation rate of the samples was analyzed by measuring the amount of Mg2+ ions released and the change of solution pH. During immersion period, after every 2 days, the colorimetric method was employed to measure the increase of Mg2+ ions in the medium.23,24 Three samples of each condition treatment were used for the immersion tests and the average values were reported.
After immersed in the medium for 14 days, all the samples were retrieved, cleaned, and dried for surface characterization by optical microscope (OM), scanning electron microscope (SEM), and energy-dispersive X-ray spectroscopy (EDS).
Results
Figure 1 shows the XRD patterns of AZ31 alloy coated at three pH conditions of 7.5, 9.0, and 10.5. All three XRD patterns showed diffraction peaks of HA at 26°, 28.3°, 49.7°, and 53.5° suggesting the formation of HA on the surface of three samples. It could be seen that, the (002)HA peaks on all three samples showed the highest intensity peaks compared to other HA peaks. This result is agreed with results reported in previous studies that (002) plane was preferential orientation of HA on the surface of Mg alloys.15,22 The intensity of (002)HA peak on sample coated at pH 7.5 is much higher than those on two other samples. This indicates that (002)HA formed at pH 7.5 is much more crystalline, while HA layers formed at pH 9.0 and 10.5 may contain amorphous or porous structure.
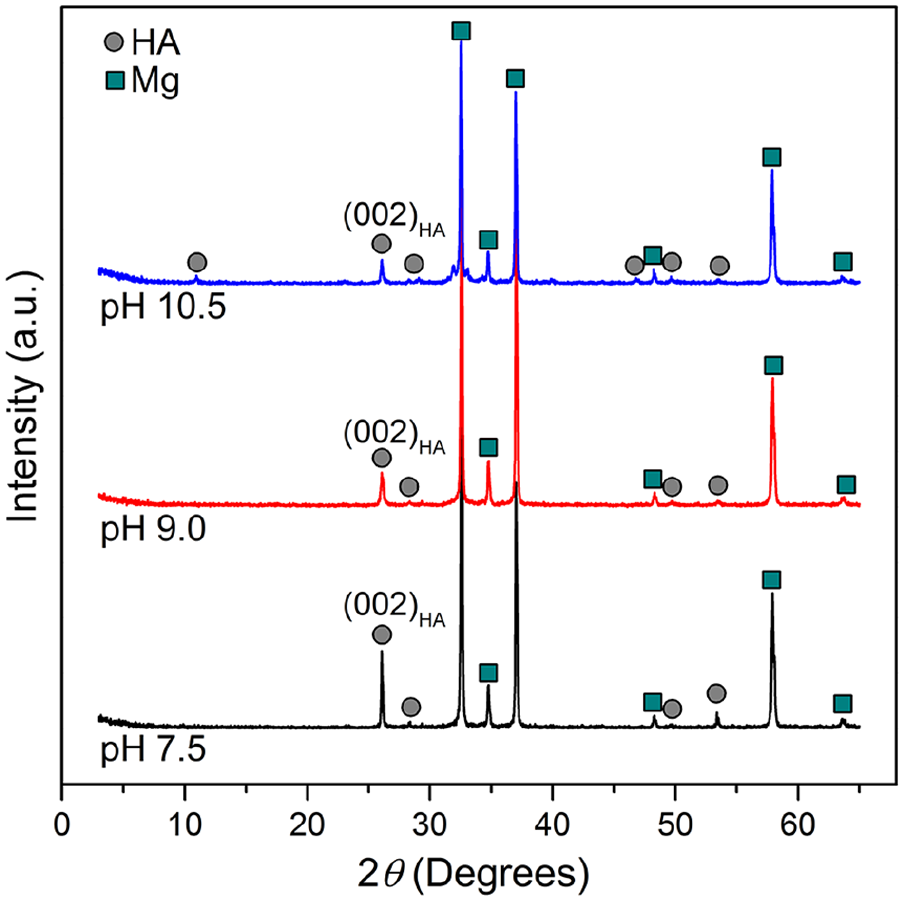
XRD patterns of the AZ31 alloy coated at pH 7.5, 9.0, and 10.5.
Figure 2 shows SEM images at different magnifications of HA coated samples, uncoated AZ31 alloy substrate, and the cross-sectional SEM images of the samples coated at pH 7.5 and pH 9.0. The cross-sectional SEM image of sample coated at pH 10.5 could not be observed due to the brittle coating layer. Figure 2(a) and (e) show the surface morphology of AZ31 substrate without coating layer. It can be seen from Figure 2(b) that a dense and uniform HA layer was formed at pH 7.5 on the surface of AZ31 substrate. There is almost no micro-crack appeared on the HA layer formed at pH 7.5. On the other hand, micro-cracks appeared on HA layer formed at pH 9.0 (Figure 2(c)) and pH 10.5 (Figure 2(d)). The density of micro-cracks on HA layer formed at pH 10.5 is much higher than that on HA layer formed at pH 9.0. Higher magnification SEM images of as coated samples were shown in Figure 2(f) to (h). As shown in Figure 2(f), HA layer formed at pH 7.5 has plate-like structure with high crystallinity. Figure 2(g) shows that HA layer formed at pH 9.0 has needle-like structure and less crystalline than HA layer formed at pH 7.5. The micro-cracks on the HA layer formed at pH 9.0 can be seen more clearly on this Figure. On the other hand, HA layer formed at pH 10.5 almost showed porous structure with very low crystallinity (Figure 2(h)). This result agreed with XRD patterns of three samples because the sample coated at pH 7.5 has high crystalline HA showing highest intensity HA peaks, while samples coated at pH 9.0 and 10.5 has low crystalline HA showing lower intensity HA peaks.

SEM micrographs of the uncoated substrate and HA coated AZ31 alloy: (a) and (e) uncoated substrate, (b) and (f) coated at pH 7.5, (c) and (g) coated at pH 9.0, (d) and (h) coated at pH 10.5, (i) and (j) cross-sectional images of samples coated at pH 7.5 and 9.0.
Figure 2(i) and (j) show the cross-sectional SEM images of samples coated at pH 7.5 and pH 9.0. The HA layer formed on both pH 7.5 and pH 9.0 had two sub-layers. The inner dense sub-layer and the outer sub-layer. The thickness of both HA layers formed at pH 7.5 and 9.0 was almost the same at around 4 µm. There were many cracks appeared at the cross-section of HA layer formed at pH 9.0. This is consistent with the micro-cracks on the surface of this sample in Figure 2(c) and (g). While several cracks at the cross-section of HA formed at 7.5 may be due to the cross-section preparation process.
Figure 3 shows the Mg2+ ions release rate of three HA coated samples immersed in Hank’s solution in 14 days. It could be seen that the corrosion rate of the three samples increased with the increase of treatment pH value. Specifically, the corrosion rate of sample coated at pH 10.5 showed very fast degradation rate. Especially, the amount of Mg2+ ion release from this sample indicated that rapid corrosion occurred at early stage of the immersion. At the same time, the rapid evolution of hydrogen gas bubbles was observed also. After 4 days of immersion, the corrosion rate gradually decreased. For the samples coated at pH 7.5 and pH 9.0, no rapid corrosion occurred. The corrosion rate almost kept constant throughout the immersion period. The sample coated at pH 7.5 demonstrated its lower degradation rate than the sample coated at pH 9.0.
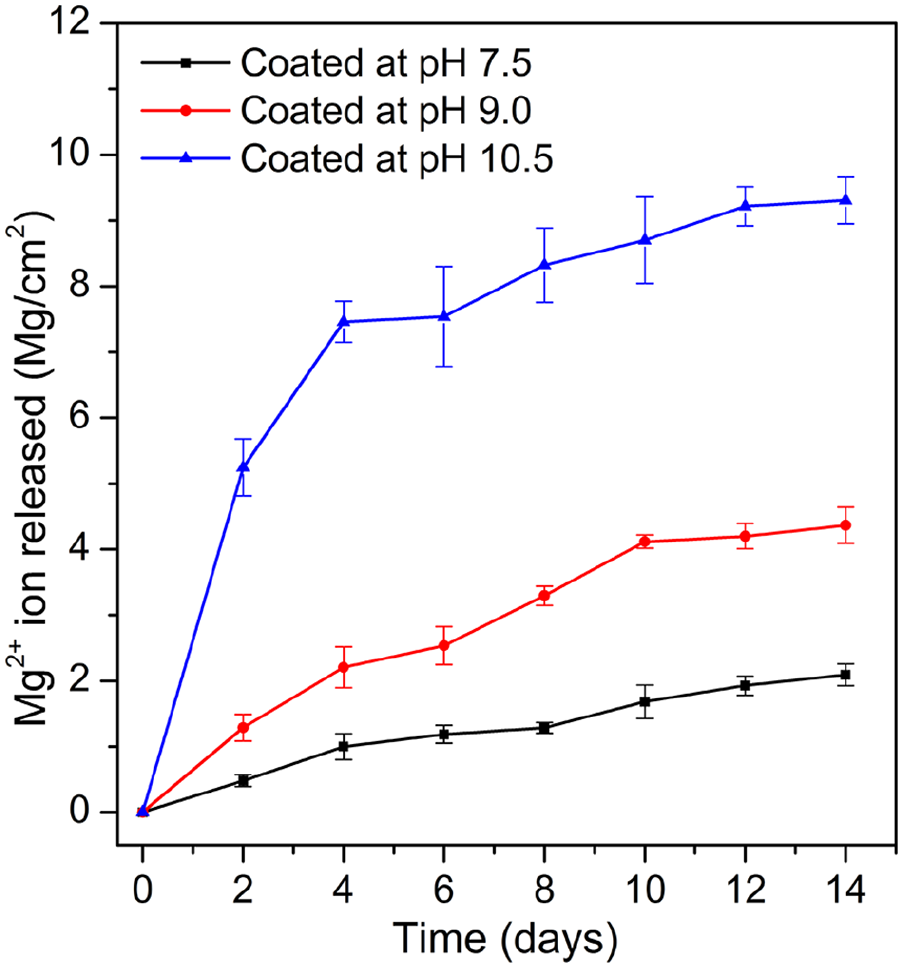
Amount of Mg2+ ion release in SBF during immersion period.
Figure 4 shows the pH value of the solution during immersion period. The tendency of pH change was consistent with the tendency of Mg2+ ion release. As for the sample coated at pH 10.5, at the early stage of immersion, the sample was corroded severely by the alkaline environment. Therefore, rapid formation of OH− and Mg2+ ions resulted in significant increase of the pH value of the solution. After 4 days of immersion, the change of pH value became slower and got the highest value of 10.2 at the 14th day of immersion. For the samples coated at pH 7.5 and pH 9.0, the pH value of the solution increase gradually and reached the highest values at 7.9 and 8.7 respectively.
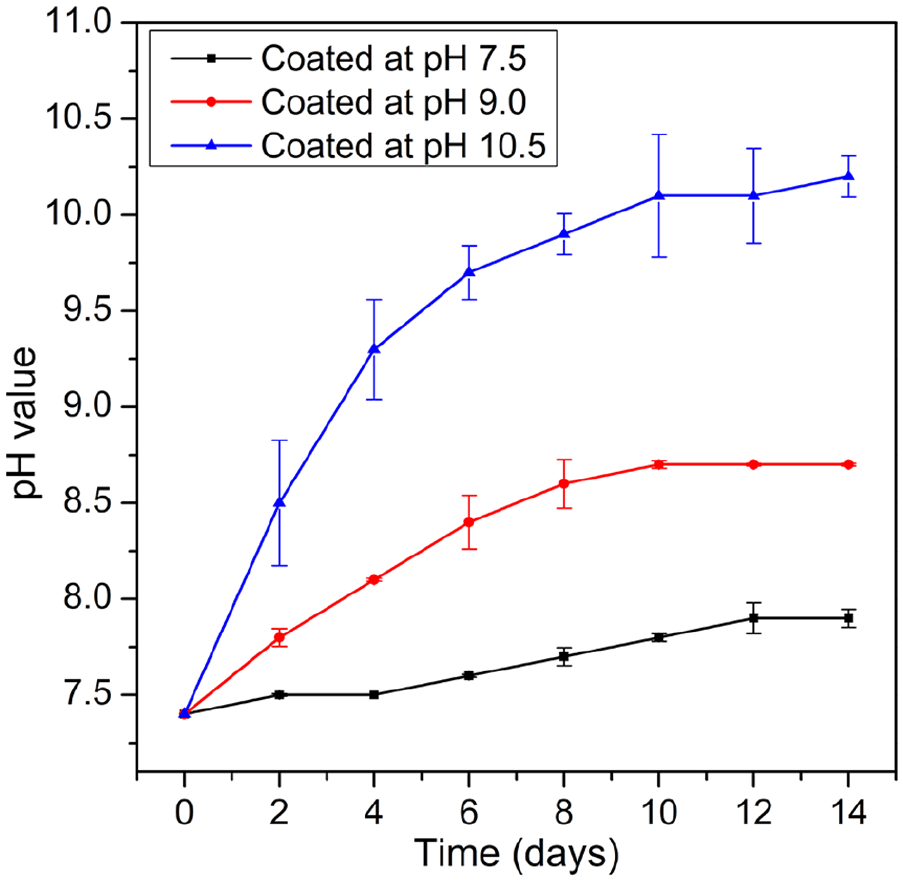
The change of pH of the solution during immersion period.
Figure 5 shows the surface morphology of the HA coated samples before and after immersion. Figure 5(a) to (c) show the surface morphology of samples before immersion, and Figure 5(d) to (f) show the surface morphology of the samples retrieved from the solution after 14 days of immersion. It could be seen that sample coated at pH of 10.5 was corroded severely. The corrosion product formed on whole sample surface and the coating layer was almost corroded completely. As for the sample coated at pH 9.0, there were some local corrosion occurred and some corrosion product formed on a part of sample surface. Some areas on the surface of this sample still remained original morphology and structure of HA. While the sample coated at pH 7.5 has very little corrosion product depositing on its surface. The surface morphology of this sample indicates the remaining of HA layer on almost surface area. These results are consistent with results of Mg2+ ion quantification and pH change in the solution during immersion period.

Surface morphology of samples before and after immersion for 14 days in SBF: (a) and (d) coated at pH 7.5, (b) and (e) coated at pH 9.0, and (c) and (f) coated at pH 10.5.
Discussion
The results from Mg2+ ion quantification, pH change, and surface morphology of the samples after immersion indicate that HA formed at pH 7.5 has the most positive effect in improving corrosion resistance of AZ31 alloy, while HA coated at pH 10.5 demonstrated the least effective in protecting substrate from corrosion. This is because the HA coating layer formed at pH 7.5 has uniform-crystalline structure as shown in Figure 2(b) and (f). The uniform-crystalline structure of the HA coating grown at pH 7.5 created a wall to protect the substrate from the solution. However, as reported previously15,22 that there are always nano-pores existed in HA layer formed on the surface of Mg alloys. Therefore, the corrosion still occurred for this sample with a very low rate. On another hand, the HA layer formed at pH 10.5 contained micro-cracks with very high density. Therefore, the solution easily attacked and caused severe corrosion. This explains that corrosion occurred quickly at the early stage of immersion leading to high amount of Mg2+ released as shown in Figure 3 and remarkable change in pH as shown in Figure 4. As for the sample coated at pH 9.0, due to existence of both nano-pores and micro-cracks with lower density in the coating layer, the corrosion rate occurred with a moderate rate.
It should be noted that corrosion products deposited on the surface of all three samples. Figure 6 shows SEM images and surface composition of the yellow marked areas on three samples after immersion analyzed by EDS. The surface of sample coated at pH 10.5 consists of mainly O and Mg with the atomic ratio of O:Mg ≈ 2:1 (Figure 6(c)) suggesting the formation of Mg(OH)2. The formation of Mg(OH)2 on the surface of sample coated at pH 10.5 was consistent with results of previous studies.15,25 Cao et al. and Lei et al. reported that, at a high corrosion rate of Mg alloys, the corrosion product deposited on the surface of the sample was mainly Mg(OH)2. This Mg(OH)2 layer has porous structure as shown. Therefore it is unable to create a strong protective layer to avoid the severe corrosion occurred. As for sample coated at pH 9.0, the surface of this sample after immersion consisted of mainly Ca, P, and O (Figure 6(b)). The atomic ratio of Ca/P was 1.33 suggesting the formation of calcium phosphate on the surface of this sample and there was some HA remained. Cao et al.15,26 also reported that, at a moderate corrosion rate, the corrosion process of Mg alloy and HA coated Mg alloy formed calcium phosphate and deposited on the surface of the sample. This calcium phosphate layer showed its benefit in both improving corrosion resistance and biocompatibility of the materials.19,20 As for the sample coated at pH 7.0, because of its highest corrosion resistance, the HA layer almost remained on the surface (Figure 6(a)). The composition on the surface of this sample indicates that Ca, P, and O are main components of the surface. The atomic ratio of Ca/P was 1.67 indicating the remaining of HA coating layer.

Surface morphology and composition on the surface of samples after immersion test: (a) coated at pH 7.5, (b) coated at pH 9.0, and (c) coated at pH 10.5.
Conclusions
HA coating layers were successfully grown on the surface of AZ31 alloy substrate at different pH values. The HA layer formed at pH 7.5 had dense structure, high uniformity without micro-crack resulted in low degradation rate in vitro. The HA layers formed at pH 9.0 and 10.5 showed their low crystallinity and many micro-cracks leading to fast degradation rate in vitro. Mg(OH)2 formed on HA coating grown at pH 10.5, while calcium phosphate formed on the HA coating grown at pH 9.0 as corrosion product. The result suggested that AZ31 alloy coating with HA at pH 7.5 is most suitable for applications as biodegradable material.
Footnotes
Author contributions
LH, LVH, and DTHH designed and conducted the experiments. NTH, LMH, and NVN analyzed and evaluated the data. All authors involved in manuscript preparation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 103.02-2018.56.
Guarantor
LMH and NVN.
