Abstract
Surgical post-operative adhesions can lead to serious clinical complications. Barrier agent is the broad usage for the prevention of post-operative adhesions. This study aimed to evaluate the reducing adhesion efficacy of non-animal hyaluronic acid (HA) hydrogel in pigs undergoing conventional laparotomy pelvic surgery. HA hydrogel was applied to eighteen female pigs who underwent conventional laparotomy. The adhesion degrees and histopathology were evaluated in bilateral uterine horns as well as peritoneal sidewall excision. In the present study, all animals survived and had no complications after the surgery. The histopathological observations were demonstrated that HA obviously improved laparotomy pelvic surgery-induced adhesion in peritoneal sidewall and uterine horn. The anastomotic healing score of injury + HA group was significantly lower than the injury alone group. We conclude HA hydrogel can attenuate the post-operative adhesions in porcine.
Introduction
Adhesion often occurs after common abdominal, gynecological, orthopedic, and cardiovascular surgeries. 1 Surgical post-operative adhesions could lead to serious clinical complications, including small bowel obstruction, abdominal and pelvic pain, infertility, etc.2,3 It was reported that 74% of small bowel obstructions are caused by abdominal adhesions. 4 More than 55% of pelvic adhesions were caused by gynecological surgery. 5 Reducing post-operative adhesions is currently being investigated across surgical disciplines. 6
There are two main methods for the prevention of post-operative adhesions, including the improvement of surgical techniques and the use of barrier agents. Laparoscopic surgery is a common and successful treatment for abdominal adhesions, associated with recurrence rates lower than open surgery. However, complex adhesions may be less favorable, as the complete removal of all adhesions is of high risk and prone to recurrent adhesion formation. 7 Laparoscopic surgery is therefore high risk in acutely obstructed bowels and puncturing the distended bowel, which requires potential conversion to open laparotomies. 8 For the latter, barrier agents are most commonly used.
The commercial adhesion barrier materials are polymers, like polytetrafluoroethylene, polylactic acid, oxidized regenerated cellulose, and hyaluronic acid (HA), but still present some limitations. The unabsorbable polytetrafluoroethylene needs to be removed by a second operation, 9 increasing surgical frequency and risk. Polylactic acid presents poor adhesion and requires suturing and wound-fixture, increasing the surgical processes an d operation time. 10 Oxidized regenerated cellulose is used in close proximity to the blood vessels and might lead to scar contractions, affecting blood flow. 11
HA is present in all vertebrates and ubiquitous uronic acid composed of repeating units of disaccharides of D-glucuronic acid and N-acetylglucosamine connected via β-1,3 and β-1,4 glycosidic bonds. 12 HA forms a distinct tertiary structure in aqueous solution and demonstrates moisturizing capability. 13 However, cockscomb-derived HA is also at risk of disease transmission (avian influenza, etc.). 14 Additionally, its short half-life and easy liquidity limits the application. 15 Various chemically modified forms of HA have been developed, including Seprafilm®, auto cross-linked polymer gel, hylan gel, Intergel®, and photo-cross-linked HA gel, to extend the retention time on bodily application sites. In the human body, the catabolic half-life of pristine HA in the blood was 2–5 min. 16 In contrast, the half-life of intra-articularly injected exogenous HA in the blood of rabbits was 6.2 days. 17 In addition, Xue et al. 15 found that in vitro degradation of cross-linked HA by 1,4-butanediol diglycidyl ether was 12–72 h. Although modified HA products could prolong the rate of degradation, it may also increase the risk of side effects. HA is not immunogenic, toxic and induced inflammation, and thus is used widely in cosmetic, biomedical, and food industries. 18 It can be degraded by endogenous hyaluronidase to fragments and absorbed by cells. 19
Here, we examined the anti-adhesive effects of bacterial-derived HA hydrogel in porcine models, after laparotomy pelvic surgery. A porcine adhesion model was established to better mimic conditions during pelvic surgery. It is used to evaluate the preventive effects of commercial substance for pelvic surgery-induced adhesion by previous characteristics and anastomotic healing score methods. 20
Materials and methods
Anti-adhesion hydrogel
PROTAHERE™ absorbable adhesion barrier hydrogel (SciVision Biotech Inc., Kaohsiung, Taiwan) contains approximately 4.1% HA. The HA of PROTAHERE™ is produced from a bacterium (Streptococcus). The properties of PROTAHERE™ were the ratio to saline osmolality = 0.32; pH = 5.6 and molecular weight = 2 × 1023 Da.
Swelling ratio
A total of 0.2 g hydrogel (n = 3) was dried for 2 h at 80°C in an oven, weighed, and labeled as m1. 15 The dried sample was immersed in deionized water until equilibrium was reached. The excess water was removed by wiping with filter paper and the sample was re-weighed and labeled as m2. The swelling ratio was calculated using the following equation:
Enzyme hydrolysis stability
A total of 0.3 g dry hydrogel (n = 3) was swelled in 8.8 ml phosphate-buffered saline (PBS) and a blank sample was added with 8.8 ml PBS and 1 ml 95% sulfuric acid. 15 The sample was mixed with 0.2 ml hyaluronidase solution (30 U/ml, dissolved in PBS), and then incubated for 10, 60 and 90 min at 37°C. The mixture was filtered through 6 μm filter paper and the glucuronic acid concentration in the filtrate was determined according to the Bitter-Muir method. 21 The hydrolysis ratio was calculated using the following equation:
Porcine adhesion model
All animal experiments and care-protocols were approved by the Institutional Animal Care and Use Committee of the Agricultural Technology Research Institute (Approval No. IACUC-105083).20,22 Twenty female pigs (weight range: 27–37 kg, mean weight: 32 ± 5 kg) were obtained and acclimatized for a minimum of 5 days prior to surgery. The animals were maintained on a mix of 1–3% isoflurane and oxygen inhalation for anesthesia during surgical procedures. The celiotomy was created via midline abdominal incision from the umbilicus to the pubis. Both uterine horns were transected at the midpoint after incision with surgical scissors, and then reconjugated using two absorbable polyglycolic acid sutures (3-0, EU-TEK, UNIK surgical sutures Mfg. Co., LTD, Taiwan). The peritoneum of the pelvic sidewall opposing each uterine horn was excised from the round ligament to the infundibulopelvic ligament to expose approximately 5 × 4 cm areas on the pelvic sidewall. An intraoperative view of a completed peritoneal injury, just prior to uterine horn transection and anastomosis, is shown in Figure 1(a).

(a) Injury group. Intraoperative view of peritoneal injuries, just prior to uterine horn transection and anastomosis and (b) injury + HA group. The 20 ml HA was applied between peritoneal and uterine horn injury sites.
Application of adhesion hydrogel
Two pigs which formed the sham group were sutured after animal abdominal cavity incision without any injury, and another eighteen pigs received the peritoneal sidewall and uterine horn injury. After injury completion, the left or right sides of the injury sites were assigned to the injury group or injury+HA group, at random. For the injury+HA group, we used PROTAHERE™ hydrogel. For the injury+HA group, 20 mL of hydrogel was applied to the injury sites; the gel covered the surgical area with a thickness of 1–2 mm. For the injury-only group, gel was not applied. Typical applications at the treated sites are illustrated in Figure 1(b).
Gross examination and adhesion evaluation
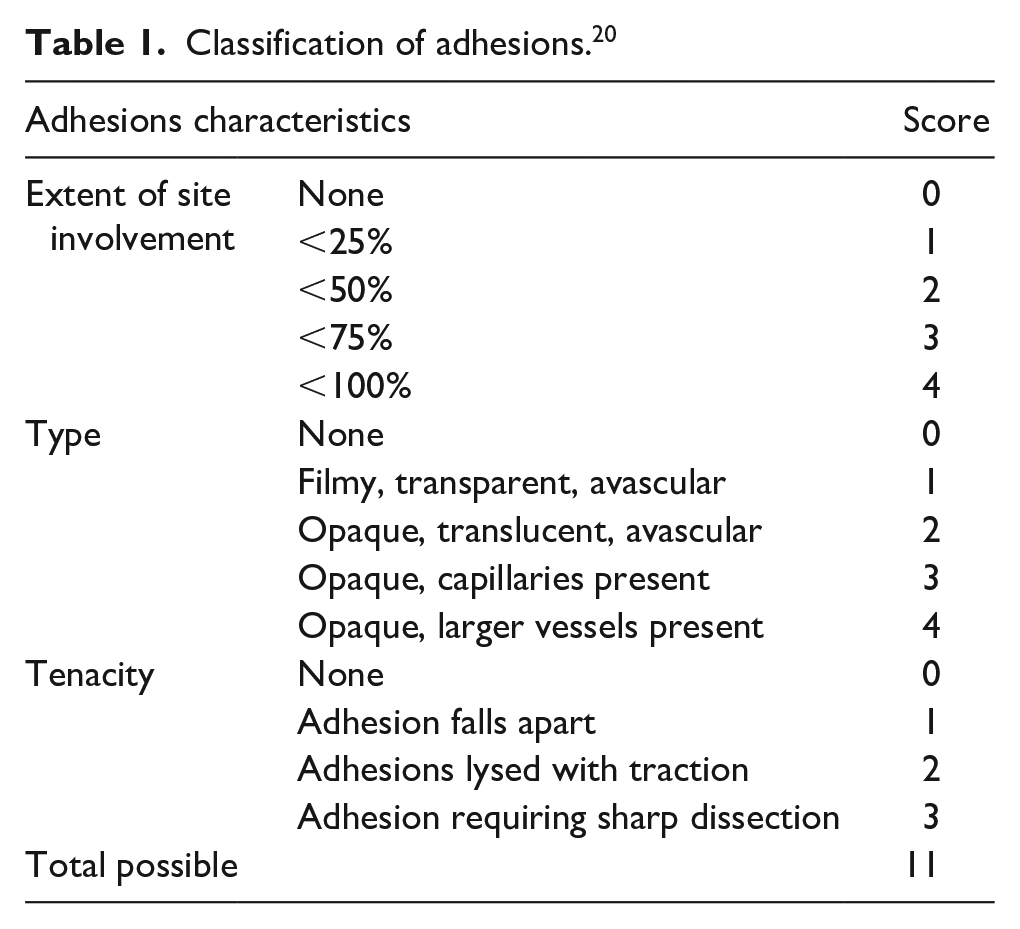
The pigs were weighed and then euthanized by exsanguination while under anesthesia, at 14 days after the initial operation. Autopsies were carefully performed to ensure the integrity of present intraabdominal adhesions from the initial abdominal incision. Clinicians performing the autopsies were blinded concerning the animal group. The grades of abdominal adhesions were described as in Table 1. 20 Both injury and treatment sides of all pigs were evaluated. The scores of each pig were evaluated by three different observers. The total scores were the sum of each items and the lower score represented better anti-adhesive and healing effects.
Classification of adhesions. 20
Histopathology
The tissues were fixed in 10% buffered formalin, embedded in paraffin, sliced on slides for hematoxylin and eosin (HE) staining, and further observed by microscopy. The evaluation of anastomotic healing was modified and based on the presence of anastomotic ulcer, reepithelization, granulation tissue, inflammation, eosinophilic infiltration, serosal inflammation, and the presence of microscopic adhesions (Table 2). 22 Both injury and treatment sides of all pigs were evaluated. The scores of each pig were evaluated by three individual observers. Total scores were calculated as the sum of scores of these parameters. A lower score represented better anti-adhesive and healing effects.
Histological parameters of anastomotic healing score. 22

Statistics
All data are presented as the mean ± standard error of the mean. Adhesion score evaluation was performed using a one-way analysis of variance, followed by Duncan’s method (Systat Software, San Jose, CA, USA). Differences were considered significant at p < 0.01.
Results
HA hydrogel reacted with hyaluronidase to confirm hydrolysis stability of HA hydrogel. It shows that the degradation rate of HA hydrogel were 42.86% ± 2.75%, 87.20 ± 1.33% and 100.07% ± 1.18% after 10, 60 and 90 min, respectively. HA hydrogel can swell in water without dissolving. The swelling rate of HA hydrogel was 38.90 ± 1.04%.
All pigs (n = 20) survived the procedure without complications. Animals were sacrificed 14 days postoperatively and no adhesion of the uterine horn with surrounding organs or with the peritoneal sidewall was observed in the two animals belonging to the sham group. This shows that if the uterine horn and the peritoneal sidewall are not injured in an open surgery performed on pigs, no adhesion would result, as shown in Figure 2(a). In the injury group, adhesion was observed around the suture site, and in some animals, more severe adhesion was observed at the excision site of the peritoneal sidewall, the uterine horn, the intestinal tissue, or the bladder, as shown in Figure 2(b). No hydrogel residue persisted in the pelvic cavity of the animals in the injury+HA group, indicating that the hydrogel was absorbed. In the injury+HA group, adhesions mostly occurred around the suture site and were comparably mild, as shown in Figure 2(c). Adhesions were scored based on the adhesion evaluation standard shown in Table 1. Of the 18 animals, 13 animals had better scores in the injury+HA group compared to the injury group. Statistics show that the score of the injury+HA group (2.20 ± 1.80) was significantly lower than that of the injury group (5.18 ± 3.10) as shown in Figure 2(d).

(a) Gross appearance of the sham group, (b) injury group, (c) injury+HA group, 14 days after surgery, and (d) quantification of adhesion scores according to the classification of adhesions from Table 1. The injury sites of the injury group exhibited severely adhesive presentations with tissue swelling. The peritoneal injury site and the uterine horn sutures in the injury + HA group were separated without adhesion and swelling. Adhesion scores corresponded with the experiment photographs. The hydrogel obviously prevented surgery-induced tissue adhesion.
All animal tissues were stained pathologically by HE staining to assess the formation of adhesion tissues as well as tissue inflammation at the excision site of the peritoneal wall and around the uterine horn. The peritoneal sidewall stains of all groups are shown in Figure 3(a) to (c) and the histological sections were scored based on the anastomotic healing score standard shown in Table 2. As shown in Figure 3(a), in the sham group, spindle-like muscle cells were neatly arranged, and no infiltration of inflammatory cells or formation of adhesion tissue was observed. In the injury group (Figure 3(b)), peritoneal serosa damage, fibroblastic proliferation, and collagen fiber formation were observed and contributed to the formation of densely arranged adhesion tissues. Inflammatory cell infiltration in adhesion tissues was found. Moderate diffuse necrosis was observed in muscle tissues and the damaged cell membrane of muscle tissues resulted in larger gaps between cells. In addition, muscle tissues exhibited moderate inflammatory cell infiltration as well as macrophage phagocytosis of muscle cells. In the injury+HA group, as shown in Figure 3(c), loose adhesion tissues can be observed on peritoneal sidewall tissues, together with the formation of collagen fiber and inflammatory cell infiltration, though not as severe as the condition in the injury group. Muscle tissues exhibited mild diffuse necrosis and inflammatory cell infiltration. The anastomotic healing score of the injury group at 7.26 ± 0.30 significantly differed from that of the injury+HA group at 5.06 ± 0.28, as shown in Figure 3(d).

Histological analysis of the peritoneal sites of the: (a) sham group, (b) injury group, (c) injury + HA group, and (d) quantification of adhesion scores according to the anastomotic healing scores from Table 2. The proliferation of fibrous tissue in the injury group was thicker than in the injury + HA group. The moderate diffuse necrosis and inflammatory cell infiltration of muscle tissue was exhibited in the injury group and the hydrogel obviously extenuated this phenomenon.
Staining results of uterine horn tissues are presented in Figure 4(a) to (c) and the anastomotic healing scores are shown in Figure 4(d). As shown in Figure 4(a), uterine tissues of the sham group consisted of the innermost lumen, endometrium, myometrium, and the outermost perimetrium. The perimetrium is a membrane tissue composed of a complete and continuous layer of mesothelial cells. No inflammation or adhesion was observed in uterine tissues of the sham group. The perimetrium structure was lost in the injury group (Figure 4(b)), and myometrium tissues were connected to adhesion tissues. Most adhesion tissues in histological stains demonstrated formation of diffuse capillaries, minor fibroblastic proliferation, collagen fiber formation, and mononuclear cell infiltration. Figure 4(c) shows that adhesion persisted in the injury+HA group, but fragments of the perimetrium can be observed with continuously arranged mesothelial cells that visibly served as a boundary between myometrium tissues and adhesion tissues. Fibroblastic proliferation can be observed in adhesion tissues, combined with collagen fiber formation and mononuclear cell infiltration, though not as severe as the condition in the injury group. The anastomotic healing score of the injury group and the injury+HA group were 5.09 ± 0.36 and 3.19 ± 0.34 respectively, with significant difference.

(a) Histological analysis of the uterine horn of the sham group, (b) injury group, (c) injury + HA group, and (d) Quantification of adhesion scores according to the classification of adhesions from Table 2.
Discussion
Laparotomy pelvic surgery was conducted to evaluate the anti-adhesion property of HA hydrogel. Animals were dissected 14 days postoperatively. In the injury group, adhesion tissues proliferated, and severe adhesion was noted between the peritoneal wall and the adjacent small intestine as well as with the uterine horn. On the other hand, with the use of HA hydrogel, no hydrogel residue was observed within the body, and adhesion was milder compared to the injury group. Staining of the uterine horn and peritoneal sidewall tissues revealed dense adhesion tissues in the injury group, together with muscle cell necrosis and inflammatory cell infiltration. HA hydrogel mitigated the above conditions demonstrating the anti-adhesion effect of the hydrogel.
Post-surgical abdominal trauma induces inflammatory reactions, fibrinous exudate, and fibrin formation. 23 Permanent adhesion may result from the imbalance of coagulation and fibrinolysis. During coagulation, temporarily formed fibrin matrix deposits on the surface of damaged peritoneum, possibly causing the peritoneum to adhere to adjacent organs. However, fibrinolysis occurs within 72 h after injury as matrix metalloprotease begins to degrade the fibrin matrix and mesothelial cells proliferate to prevent the contact of adjacent surfaces. In addition, macrophages recruit mesenchymal cells and fibroblasts to migrate to the site of peritoneal damage to repair the damaged peritoneal surface. 24 Therefore, adhesion tissues form within 3–5 days postoperatively and clinical anti-adhesives must be effective during this period. 6 The ideal anti-adhesion barrier should not only be resistant to adhesion, but also demonstrate biocompatibility, bioabsorbability, safety, ease of use and immunogenicity while not eliciting an inflammatory response or interfere with healing. 25
Types of physical barriers available currently can be categorized into solid sheets, viscous polymer solutions with fluid properties, and hydrogels. 26 Raw materials commonly used for solid sheets include cellulose, HA, and PTFE. 26 The advantage of solid sheets is that they can be trimmed flexibly into the size and shape required for the surgery to isolate the wound from adjacent organs. However, several drawbacks of solid sheets 26 include (1) the need of a second procedure to remove the non-absorbable solid sheet, (2) the requirement of skilled operation techniques to secure the solid sheet to the correct position to achieve complete hemostasis, (3) the possibility of adhesion at the suture site due to slow degradation and prolonged exposure of the material to the wound, 27 and (4) difficulty in usage because absorbable solid sheets are generally thin and attach easily to wet gloves or equipment, preventing repositioning when needed. HA, polyethylene glycol berberine liquid, and icodextrin are anti-adhesion materials commonly used to prepare fluid polymer solutions or gels. Polymer solutions remain for a shorter period on the injured site 28 and thus demonstrate subpar anti-adhesion effects. Furthermore, due to the poor adhesive performance and low mechanical property of fluid materials, combined with factors such as post-operative patient activities, polymer solutions may not property cover the wound, resulting in more severe adhesion. 29 Nevertheless, the above shortcomings have been gradually improved in research studies and products via chemical modifications.
HA is present in all vertebrates. Hyaluronidases can degrade different molecular weight HAs and resulting fragments are distributed to tissues via the lymphatic system 30 or absorbed by adjacent cells after being degraded by lysosomes. 19 During tissue damage and wound healing, the synthesis of HA increases. 31 The molecular weight of HA determines how it influences the wound healing mechanism. The decomposition product of low molecular HA can promote inflammatory reactions and lead to more severe tissue damage. In vitro, medium molecular weight HA upregulates the expression of tight junction protein 1, thereby promoting cell migration and accelerating wound closure. High molecular weight HA demonstrates immunosuppressive effect and angiogenesis inhibition. 31 It prevents epithelial cell proliferation via inhibiting the formation of microvascular networks. 32 Therefore, medium and high molecular weight HAs are used postoperatively to provide physical barrier and prevent cell adhesion, thereby achieving anti-adhesion efficacy. Degradation products from HA medical materials can accelerate wound healing based on the above functions.
Currently, commercial adhesion barrier materials, such as polytetrafluoroethylene, polylactic acid, and oxidized regenerated cellulose, continue to present adverse effects. For example, polytetrafluoroethylene cannot be absorbed and therefore must be removed by a second operation, 9 which increases surgical-related risks. In addition, polylactic acid has poor adhesion and requires suturing and wound-fixture, further adding to the necessary surgical processes. 10 Finally, because oxidized regenerated cellulose is used in close proximity to blood vessels, it can lead to scar contractions that affect blood flow. 11 HA is synthesized endogenously in vertebrates without immunogenic, toxic, and proinflammatory properties. Furthermore, it can be degraded to fragments by endogenous hyaluronidase and then absorbed by cells. 19 HA is already used widely in the cosmetic, biomedical, and food industries. 18 Insoluble HA hydrogel in water can act as a barrier with low surface tension and low-level mechanical stimulation in animals. Furthermore, good diffusion abilities of HA facilitate nutrient transport and removal of cellular waste. 15 Given these properties, HA hydrogel is suitable for development into an anti-adhesion medical material. In the present study, a one-time administration of HA hydrogel produced anti-adhesion effects and attenuated infiltration of inflammatory cells into a pelvic surgery-induced injury site. Moreover, residual HA hydrogel was not observed within the body. Staining of the uterine horn and peritoneal sidewall tissues revealed dense adhesion tissues in the injury group, together with muscle cell necrosis and inflammatory cell infiltration. An anti-adhesion barrier material such as HA hydrogel should not only be resistant to adhesion but should also be biocompatible, bioabsorbable, safe, easy to use, and immunogenic without eliciting an inflammatory response.
HA is a common biomaterial of anti-adhesion barriers. Sprafilm® (Genzyme corporation, cambridge, MA, USA) is another anti-adhesion material approved by the U.S. FDA for use in open surgeries. The disadvantage of Sprafilm® includes material brittleness which increases the chance of cracking during operation. In rat models of incisional hernia and cecal wall abrasion, Seprafilm has been proven to safely and effectively reduce post-operative adhesion and fully degrade after 14 days. 29 Clinically, Sprafilm® can effectively reduce the likelihood and severity of adhesions after abdominal and pelvic laparotomy. 33 Various modified HA gels such as ACP gel and hylan gel are used in recent years to prevent adhesion.34,35 Comprehensive analysis on randomized clinical trials revealed that these gel reduced adhesion after laparoscopy and uterine cavity surgery. 36 In clinic study, female treated with the same resorbable HA hydrogel PROTAHERE™ have a statistically attenuated intrauterine adhesion than non-treated patient. 37
There are two main industrial production methods of HA, including extraction from animal tissues and microbial fermentation. High molecular weight HAs are produced on a large scale via the extraction of rooster crowns with a yield of 7.5 mg/g. 38 However, the disadvantage is the low recovery rate due to the presence of hyaluronidase in animal tissues that hydrolyzes HA and the harsh extraction condition that destroy HA polymers. In additional, proteins in animal tissues and latent viruses increase the risk of infection or allergic inflammatory reaction. 14 Therefore, production via microbial fermentation has replaced the extraction of HA from animal tissues. Nowadays, fermentative production of HA using Streptococcus sp. is a mature technology. HA production can reach 6–7 g/L 39 and the extraction from the fermentation solution is a relatively simple process with high recovery rate. The disadvantage of bacterial-derived HA includes the risks of contamination by bacterial endotoxins, proteins, nucleic acids, or heavy metal. However, modern biotechnology can be used to screen pathogenic Streptococcus species or non-pathogenic recombinant strands with good safety profile and high yield.
HA hydrogel used in this study was produced by Streptococcus sp. and chemically modified to improve its properties and enhance its anti-adhesion effect. After absorbing water, the hydrogel volume expands. However, the hydrogel is insoluble in water and acts as a barrier. Due to its low surface tension and low mechanical stimulation, the hydrogel does not cause discomfort in animals. Furthermore, it demonstrates good diffusion ability, facilitating nutrient transport and removal of cellular wastes. 15 HA hydrogel, due to these properties, is suited to be developed into an anti-adhesion medical material.
In this study, a porcine model was used to simulate the adoption of a hydrogel barrier after human pelvic surgery. We evaluated the adhesion formation 14 days post-surgery in untreated injury group and HA-treated injury+HA group. After the animals were dissected, the severity of adhesion was observed visually, and adhesion scores were assigned based on appearance. Of the 18 animals, the score in the injury+HA group was higher than the score of the injury group for 13 animals, the degree of adhesion was higher in the injury+HA group than the injury group for three animals, and the score in the injury+HA group was identical to the score in the injury group for two animals. This result shows that HA hydrogel mediated adhesion. Furthermore, no hydrogel residue was observed within the animals in the injury+HA group, indicating that the hydrogel degraded in the body and was either distributed to other tissues or was absorbed by cells.
In the peritoneal sidewall sections of the injury group, dense adhesion tissues can be observed together with evident fibroblastic proliferation and collagen fiber formation. Severe inflammatory cell infiltration can be observed in the muscle and proliferative tissues, and inflammatory cells attacked muscle tissues which resulted in muscle cell necrosis. While the same observation was made in the injury+HA group, adhesion structures appeared more scattered, and inflammatory cell infiltration was milder. In the uterine horn sections of the injury group, mononuclear cell infiltration in adhesion tissues can be observed. While adhesion tissues can also be observed in the injury+HA group, the condition was less severe than the injury group. In addition, fragments of the perimetrium can be observed in selected regions, with mesothelial cells forming a distinct boundary to separate myometrium tissues and adhesion tissues, thereby serving as a protective barrier to separate the uterine horn and the peritoneal sidewall. Tissue sections were graded by the anastomotic healing score standard, and the scores of the injury+HA group were significantly lower than those of the injury group. Overall, the use of HA hydrogel after surgery reduced the formation of adhesion tissues as well as the severity of adhesion.
Conclusions
This preliminary study confirmed the efficacy of HA hydrogel in preventing adhesion formation. The present results of study can be conducted in the future to compare hydrogel with other forms of HA barrier with the target of understanding the relationship between structure and efficacy. Further clinical trials still need to be conducted subsequently to confirm and compare the safety and efficacy of various hydrogel barriers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


