Abstract
Methylprednisolone sodium phosphate (MP) is an anti-inflammatory corticosteroid which is used in the treatment of spinal cord injury (SCI), however the overdose of MP has toxic effects Therefore it is prerequisite to develop novel approaches to overcome the side effects of MP and enhance its efficacy. In the present work, we have developed alkaline phosphatase (ALP) trigger self-assembly system of oligopeptides to physically entrap and locally deliver MP. The synthesis of Nap-Phe-Phe-Tyr(H2PO3)-OH (1P) was achieved using solid phase peptide synthesis and was characterized using mass spectroscopy. The 1P is a hydrogelator, which in presence of ALP self-assembles to form the hydrogel. During the self-assembly of 1P, MP was physically entrapped without losing the physical strength of hydrogel as revealed in the rheology study. The consistency of this hydrogel and the structure was characterized using circular dichroism. The MP was released from the hydrogel in a sustain manner and 80% of the drug release was observed at 120 h. The MP + 1P were non-toxic to the cells at lower concentration however toxicity increases with the increase in concentration of MP. Further, the in-vivo administration of MP + 1P significantly reduces the pro-inflammatory cytokines and the histological analysis revealed improvement in the SCI. In conclusion, it could be stated that the synthesis of 1P for the delivery of MP provides the novel opportunity in for the treatment of SCI.
Introduction
SCI is an alarming clinical glitches that have severe side effects on health care systems and patients. 1 The traumatic SCI recruits a group of biochemical events that may lead to overwhelming sensory as well as motor functional injury, nerves deficits, and everlasting paralysis, involving axon and blood vessel injuries, disruption of tissue integrity, and cell membrane damage. 2 The primary injury may categories as severe nerves damage due to medical insult. The secondary damage occurs following the primary injury. In secondary damage free radical damage and lipid peroxidation cause deterioration of the affected area. 3 Following SCI a severe neuronal damages occur due to oxidative stress, inflammation and lipid peroxidation. 4 In order to minimize the functional loss after SCI, various strategies have been investigated to observe the biochemical and pathophysiological changes. 2
Since decade the scientist and clinicians have made serious efforts to develop effected treatment methods for SCI, but the recovery efficiency is still a big challenge. Various neuroprotective drugs are in practice to overcome the side effects of primary injury and prevent the secondary insult. However, only methylprednisolone has been shown promising results. 5 The human neurological deficit following SCI could be minimized by administering high-dose of MP. The MP mechanism of action is not clear but it is believed by clinicians that its effect on SCI could be related to exclusion of inflammatory response and lipid peroxidation. 6
The high dosage administration of MP in SCI patients might cause severe side effects, including acute corticosteroid myopathy sepsis, wound infection, gastric bleeding, and pneumonia; these effects could be improved by neurological recovery. 7 These drawbacks might also be associated to toxic effects of MP therapy. Target specific delivery 8 or site-specific delivery can be alternative to avoid the toxicity of the drug. Consequently, the site specific delivery of MP is the key factor for its effective clinical use 6
In recent decades the supramolecular hydrogels have been in practice due to their wide spread biomedical applications such as drug delivery,9–11 wound healing, 12 cell fate control, 13 metal ion absorption, 14 tissue engineering, 15 biomarker sensing, 16 functional materials design, 17 etc. The supramolecular hydrogel obtained by enzyme-instructed self-assembly (EISA) are biocompatible hence, it is an ideal candidate for drug delivery. 18
Inspired by the previous finding, we have co-assemble MP in a supramolecular hydrogelator by employing the EISA system. This system increases the efficiency of MP by enhancing its retention time, increase its anti-inflammation property and minimize the side effect (Figure 1). EISA for the delivery of MP to enhance its anti-inflammation property in in vitro or in vivo has never been reported previously. Nap-Phe-Phe-Tyr (H2PO3)-OH (1P) is a hydrogelator percussor which has been synthesized to achieve this goal. The 1P has property to form Nap-Phe-Phe-Tyr-OH (1) when it is subjected to alkaline phosphatase (ALP). The MP was physically entrapped in the hydrogel during the co-assembly of 1P and its efficacy in SCI rats was evaluated.
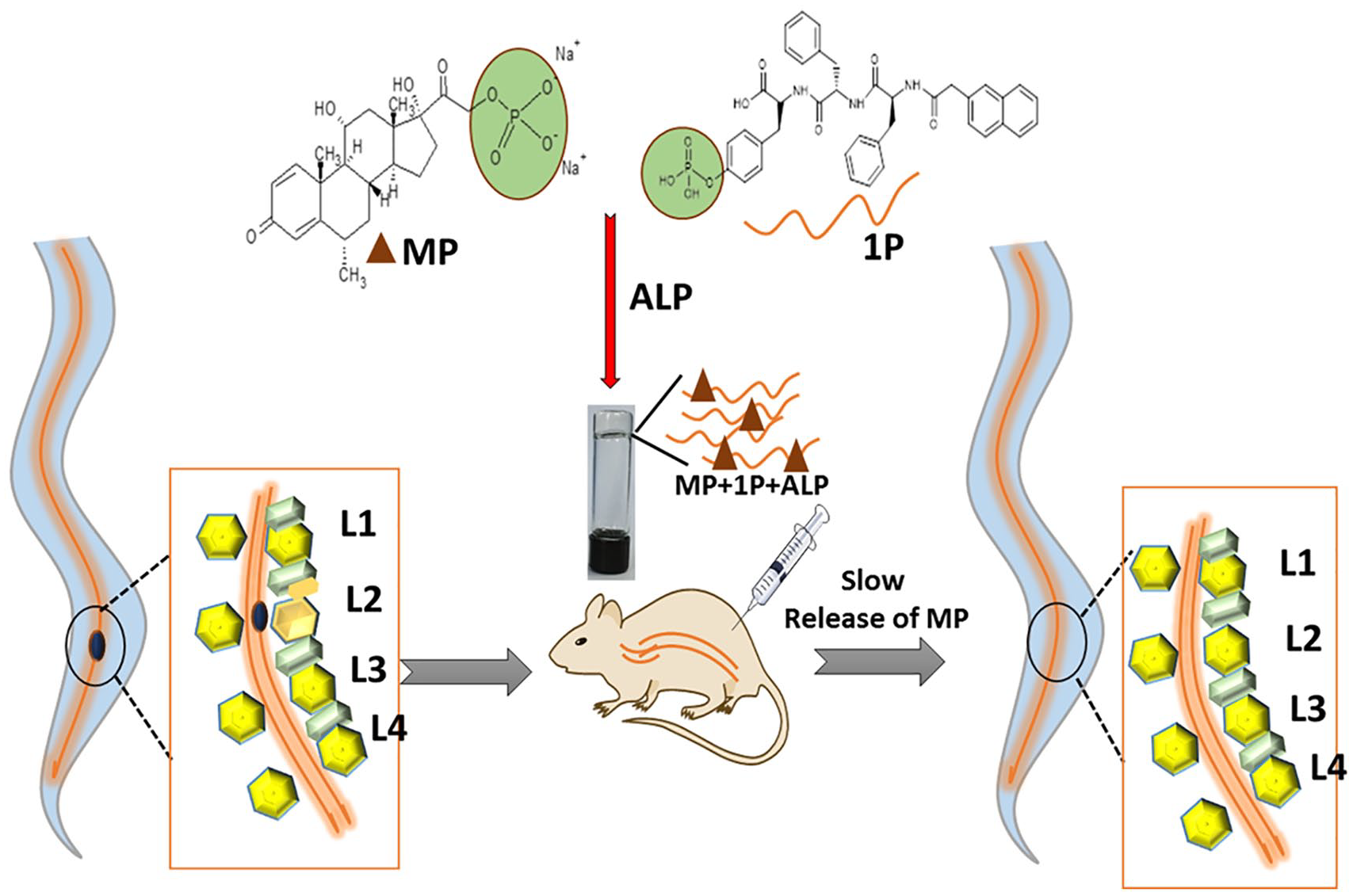
ALP triggered self-assembled hydrogel for the target delivery of methylprednisolone sodium phosphate for the treatment of spinal cord injury.
Material and methods
Methylprednisolone sodium phosphate (MP) was obtained from Shanghai Yuanye Co. Ltd. Alkaline phosphatase (ALP) is purchased from sigma Aldrich (Shanghai, China). High performance liquid chromatography (HPLC) analyses were performed on a Shimadzu system, with CH3CN (0.1% of TFA) and ultrapure water (0.1% of TFA) as the eluent. Electrospray ionization (ESI) mass spectra were obtained on a Finnigan LCQ Advantage ion trap mass spectrometer (Thermo Fisher Corporation). Cryo transmission electron microscopy (cryo-TEM) images were obtained on a Tecnai F20 transmission electron microscope at 120 kV. MTT was analyzed using IEMS Analyzer (Lab-system, Type 1401).
Synthesis of 1P and 1
Synthesis of 1P: In order to synthesize the 1P percussor, we have used the standard solid phase peptide synthesis (SPPS) method. Initially 0.5 mmol/g of 2-chlorotrityl chloride resin were taken and activated, afterward the first amino acid was loaded. The capping regent (DCM:MeOH:DIPEA = 17:2:1) was used to protect the active sites of resins. The Fmoc group was removed by using a solution of 20% piperidine in DMF afterward the second Fmoc-protected amino acid was coupled to the free amino group using HBTU as the coupling reagent. By following the same procedure, the third amino acid was coupled and finally the amino acid chain was separated from the resin and purified using HPLC system. The HPLC system was consisted a Shimadzu PRC-ODS column with couple of LC-20AP pumps having SPD-20A UV/vis detector. The mobile phase used was acetonitrile and water with 0.1% of TFA. Supplemental Table S1 describes the HPLC condition used for the purification of 1P and 1, which were further characterized using ESI-MS.
Nap-Phe-Phe-Tyr-OH (1) was also synthesized using SPPS method as described above. The obtained peptide chain was purified and analyzed as described above.
Preparation of Gel I and Gel II
To prepare the hydrogel (Gel I), 4 mg of 1P was dissolved in 400 µL of phosphate buffer saline (PBS, 0.2 M, pH 7.4) in a glass vial and stirred at room temperature for 1 min to form a clear solution. Afterward 40 units of ALP were carefully mixed in 1P solution and incubate for 5 h at 37°C. As a result, the solution changed into clear, transparent Gel (Gel I, without MP). In order to evaluate the co-assembly of MP in 1P under ALP catalysis, we dissolve 2 mg of MP and 4 mg of 1P in PBS buffer. Subsequently, 40 units of ALP was added and the solution was incubated for 5 h at 37°C to obtain another Gel (Gel II, with MP). The Gel I and Gel II has been shown in the top right inserted image of Figure 2 respectively.

Nap-Phe-Phe-Tyr(H2PO3)-OH (1P) was synthesized with solid phase peptide synthesis (SPPS).
Rheological measurement
In order to evaluate the viscoelastic properties of both Gel I and Gel II, an AR 1500ex rheometer (TA Company, America) was used. Initially, we studied the dynamic strain sweep by using rheometer to select the condition for the dynamic frequency sweep. We observed that, both storage modulus (G′) and the loss modulus (G″) of Gel I and Gel II exhibit a weak dependence from 0.01% to 10.0%, hence we selected strain amplitude at 1.0% to conduct the rheology study. The whole study was performed at room temperature. The dynamic frequency sweep analysis was carried out at range of 0.1–100 rad/s at 1.0% strain.
Drug release profile of MP from Gel II
In order to obtain the drug release profile of MP a dialysis bag was used. Initially, an equal volume of 0.25 mL of Gel II (1.0 wt %, 6 h after the addition of ALP) loaded with 1.25 mg of MP was placed in dialysis bag in PBS buffer solutions (pH = 7.4). At specific time interval (0, 2, 4, 8, 16, 24, 36, 48, 56, 75, 98, and 120 h), 1 mL of aliquot was taken from the bulk and replaced with fresh PBS of same volume in order to maintain the sink condition. The amount of MP was quantified using UV spectroscopy at 247 nm. The experiment was conducted in triplicate and the results were evaluated as mean value.
In vitro cytotoxicity
Briefly MTT (3-(4, 5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazolium bromide) assay was used to evaluate the cytotoxicity of MP. BV-2 microglial cells lines were used to examine the cytotoxicity of MP. Dulbecco’s modified Eagle’s medium (DMEM + 10% fetal bovine serum) was used for the cultivation of cell line. The cells were plated in a 96 well plate with the density of 3000 cells per well. The cells were divided into three groups (PBS, MP, and MP + 1P) and treated with different concentration of samples (0.1%, 1%, 5%, and 10% of MP in solution or in hydrogel sol) for 24 h. Later on, 4 mg/mL of MTT solution (10 µL) were added into each well and placed for 4 h. Afterwards, the supernatant was carefully removed and 100 µL of DMSO was added and analyzed by using microplate reader. The experiments were repeated in triplicate.
In vivo implantation of MP + 1P
The effect of MP and MP + 1P formulation on SCI model was evaluated on 4-week-old Sprague-Dawley male rats (Shanghai China). Initially the rats were randomly divided into three groups (n = 6) and anesthetized by using 10% chloral hydrate given by intraperitoneal injection. The vertebral column faces upside and surgical microscope was used for T-9 laminectomy. In order to obtain SCI model, approximately 2 mm section of spinal cord was removed after opening the dura mater. The SCI rats without treatment was assigned as group I (control). However, solutions of MP and MP + 1P were applied to the group II and group III rats with SCI respectively. Finally, the rats were sutured by 10–0 sterilized suture (Ethicon, Johnson and Johnson) and skin was closed. The animal’s behavior was observed every other day till the end of the study. The study was conducted for 4 weeks and at the end of the study, the inflammatory cytokines level and histological changes of spinal cord and vital organs were analyzed.
Estimation of inflammatory cytokines
To estimate the inflammatory cytokines level three rats were randomly selected from each group at the end of the study and analyzed by using TNF-α and IL-1β specific ELISA kits. ~1 mm of caudal and rostral slice of injured spinal cord was harvested. These analyses were carried out to quantify the level of TNF-α and IL-1β inflammatory mediators in each group. The harvested tissues were homogenized in PBS buffer and sonicated for 20 s in ice, the sample were kept at 4°C for 2 h for further studies. Finally, the concentration of proteins was analyzed by using ELISA protein assay kit.
Histopathological analysis
At the end of study, the rats were sacrificed by an overdose of ketamine 100 mg/kg and xylazine 20 mg/kg. The vertebral column was opened and perfused with 4% paraformaldehyde in PBS. 20 µm thick tissue sections were cryosectioned. Moreover, other vital organs (heart, liver, spleen, and kidney) were removed and fixed by fixative at 4°C for 6 h. The paraformaldehyde solution (10%) was used for sample storage. The samples were placed at cutting temperature and 10 µm sections of each sample were cut with the help of cryostat. The obtained section was stained by H&E dye and analyzed under microscope. The results were analyzed on the basis of hemorrhage, neuronal vacuolation and inflammatory cell infiltration
Statistical analysis
PRISM (GraphPad Software, CA, USA) was used to perform the statistical analysis. One-way or two-way ANOVA with post hoc Bonferroni multiple comparison tests were performed and the p-values, ***p < 0.001, **p < 0.01, *p < 0.05 was set as significant. The error bars represent the standard deviation in each analysis.
Results
The synthesis of hydrogelators (1 and 1P) was carried out as following the method of solid phase peptide synthesis (SPPS) according to the scheme presented in Figures 2 and 3. These 1P and 1 were first purified using HPLC. The purified 1 and 1P was further characterized using mass spectroscopy. The results of mass spectroscopy analysis were provided in the supporting information as Supplemental Figures S1 and S2 for 1P and 1 respectively. Also, we observed that when 1P was treated with ALP, 73.6% of 1P gets dephosphorylated and converts into 1 as illustrated in Supplemental Figure S3. The synthesized hydrogelators were evaluated for their ability to form the hydrogel when treated with ALP in the presence or absence of MP. The results revealed that the 4.0 mg of 1P is dissolved in 400 µL phosphate buffer and 1P along with MP in a ratio of 1:2 could form a clear gel in the presence of 40-unit ALP at 37°C. Figure 4(a) and (b) represents the morphology of nanofibers obtained using cryo-TEM of 1P + ALP as Gel I and MP + 1P + ALP as Gel II respectively. The nanofibers of Gel II were observed to be thicker than Gel I with the diameter of 4.7 ± 0.5 nm and 3.4 ± 0.2 nm respectively. The difference between the diameter of nanofibers in Gel I and Gel II (i.e., nanofiber 1 is slightly weaker than nanofiber 2) well explained the mechanical difference between these two hydrogels (i.e., Gel I was mechanically weaker than Gel II). The clear gel obtained for Gel I and Gel II in the glass vial has been inserted on the left corner of the Figure 4(a) and (b) respectively. However, we observed that the MP alone could not be transformed into hydrogel in presence of ALP. However, in presence of ALP the prodrug MP is dephosphorylated into its active parent drug.

Synthesis of 1: Nap-Phe-Phe-Tyr-OH (1) was synthesized after deprotection of the protecting group from 1-Nap-Phe-Phe-Tyr(tBu)-OH.

Cryo-TEM images of the nanofibers in Gel I (a) and Gel II (b).
Next, Gel I and Gel II were analyzed for their viscoelastic properties. It was observed that when the frequency was increased from 0.1 Hz to 10 Hz to define the gel dynamic frequency sweeps of Gel I and Gel II, loss modulus G″ was almost six times smaller than storage modulus G′ for both. The results shown in Figure 5(a) and (b) clearly suggests that both gels have sufficient strength to tolerate the external force. The Gel II was almost two times higher G′G″ values in comparison with Gel I revealing high strength of Gel II against external force. The mechanical strength of the gels might be attributed to the π−π stacking of the naphthalene groups in Gel I. However, the insertion of MP improved mechanical strength of Gel II which might be attributed to the π−π stacking of phenyl groups.

Dynamic frequency of storage modulus (G′, black) and the loss modulus (G″, red) of Gel I (a) and Gel II (b) at the strain of 1.0% and 25°C.
The circular dichroism (CD) studies were also conducted to verify the molecular packing of prepared gels (Supplemental Figure S4). The results suggest that the Gel I have positive cotton effect at 215 nm which might adopt β-sheet-like secondary structures. However, in Gel II also we observed the positive cotton effect but the peak at 305 nm gets flattened in comparison with Gel I revealing a different tertiary structure. The results might be attributed to the π−π stacking of Phe groups of MP and 1P.
The in vitro drug release studies revealed that the release of MP from MP + 1P + ALP could be significantly sustained in comparison with free MP and MP + ALP (Figure 6). We observed that free MP was completely released within 12 h in the release media. However, MP + ALP was release in sustained release pattern and 96% of MP released in 72 h this sustain release is due to dephosphorylation of MP due to presence of ALP. In the presence of ALP, the MP is converted into its active parent drug. Interestingly, the drug release pattern of MP + 1P + ALP was 98% in 120 h. The sustain release of MP might be attributed to the formation of hydrogel in presence of 1P which results in physical entrapment of MP in 1P nanofibers.

In vitro drug release profile of free MP, MP + ALP, and MP + 1P + ALP in PBS at pH 7.4.
Effect of MP on BV-2 microglial cells
MTT assay was used to study the toxic effect of different concentrations of MP and MP + 1P on BV-2 microglial cells viability. There is no significant toxicity was observed in the control group. Moreover, the cells viability was also high at concentration of 0.1% and 1% of MP and MP + 1P. While a significant proliferation inhibition was observed at concentration of 5%. With increase in concentration up to 10%, the viability was observed as 59 ± 4.7% and 72 ± 4.9% for MP and MP + 1P groups respectively. These finding shows that MP alone has more toxic effect as compared to MP + 1P at same concentration, this fact might attribute the slow release of MP entangles in 1P nanofiber. There is no significant change in cell viability was observed in control group (Figure 7).
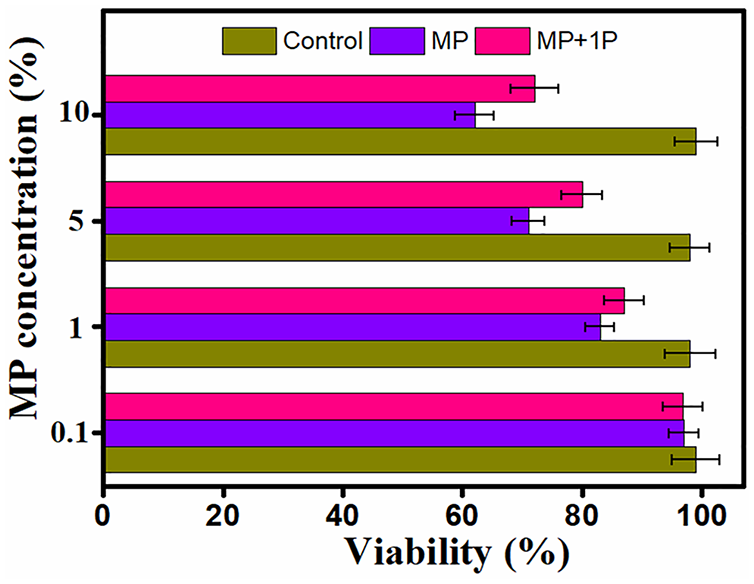
In vitro cytotoxicity of PBS, MP, and MP + 1P against BV-2 cells. These experiments were performed in triplicate. Errors bars represent standard error of mean (SEM).
Level of cytokines in different groups of animals
The ELISA based analysis depicted that as compared to SCI control group a significant alleviation in the level of TNF-α and IL-1β was observed in both MP and MP + 1P group (Figure 8). The alleviation might be due to effective anti-inflammatory response of drug. Moreover, further analysis was performed for MP group and MP + 1P group and result suggested an alleviation in the level of cytokines were more significant for MP + 1P group due to its slow and sustain release from hydrogel. These values are non-significantly differing when compared to that of SCI control.
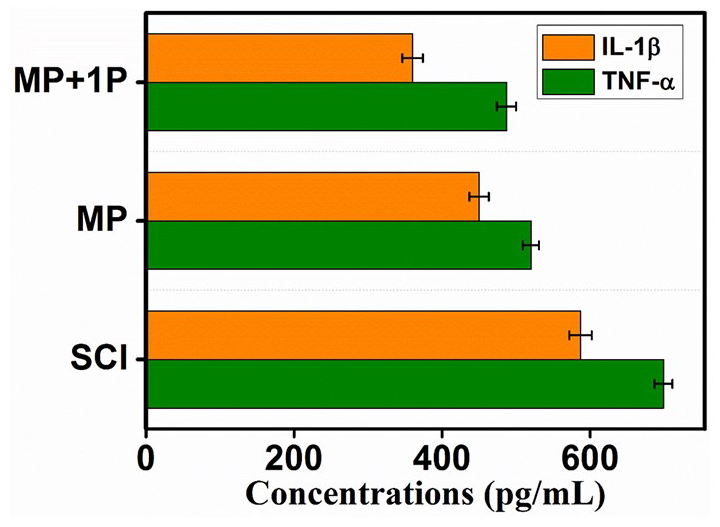
Level of cytokines in SCI rats treated with free MP and MP + 1P.
Histopathological modifications
All the rats were sacrificed at the end of study by following ethical committee protocol (protocol number: SMUG-2019-0172). Following the histopathological analysis, it was observed that the SCI group (without treatment) shows intense hemorrhage, inflammatory cell infiltration and demyelination of axons, still erythrocytes, and lymphocytes could be observed in the white matter. Interestingly, as compared to MP group, which have lymphocytes and edema, MP + 1P treated group revealed reestablishment of spinal cord as shown in Figure 9. This might be due to rheological characteristic of MP + 1P group which allow the formulation to stay at injured site for long time and slowly release the drug. A clear reduction in hemorrhage, inflammatory cell infiltration and pseudocyst volume was observed. These findings show that as compared to control and MP group, MP + 1P show more efficiency in SCI rat model by decreasing the degeneration of neurons and enhancing the development of neurons. Moreover, the histopathological analysis of other vital organs (heart, kidney, liver, and spleen) didn’t show any significant toxicity in all the groups.

H&E staining of section of spinal cord and vital organs of rat at the end of study including heart, liver, spleen and kidney.
Discussion
SCI could severely affect the life of its victim. 19 The anti-inflammatory MP drug has a high oral bioavailability and finds its application in the treatment of SCI. 20 However several factors including nonspecific delivery, high protein binding after systemic administration, and severe toxicity related to its high dose (30 mg/kg) limits its wide use. 21 Therefore there is a wide scope in improving the potency of MP while decreasing its toxicity. 22 The local delivery provides the high concentration of drug at the desired site. 23 Hydrogels are consisted of hydrophilic 3D networks which are comprised of peptides or polymers assembled together through ionic bond, covalent bond, or hydrogen bonding.11,24 The physical entrapment of MP in hydrogels could sustain the drug release and thus could reduce the toxicity associated with its high dose. 1P was successfully synthesized which could dephosphorylate in presence of ALP and converts to 1. 1P + MP were developed for the site-specific delivery in SCI which has the ability to sustain the drug release. The self-assembly of 1P was triggered in presence of ALP present in the cells and its surrounding environment. 25 During the process of self-assembly, MP gets physically entrapped and was released over the period of time. The ability of 1P to adsorb the physical stress was found to be higher than MP + 1P, which might be the result of π−π stacking of Phe group of MP and 1P. The developed hydrogelators could retain enormous quantity of water in their structure and form elastic gels which slowly biodegrades into smaller molecules due to proteolysis or hydrolysis. 26
The drug release study results revealed the initial burst release of MP which might be due to physical entrapments of MP, followed by sustain release profile. 27 The results could be attributed to the initial release of MP from the solution of 1P + MP which later self-assembled in presence of ALP entrapping the drug in hydrogel. The drug then slowly defuses from the hydrogel giving a sustain release profile. However, free MP was released instantly within 12 h. MP + ALP could slightly sustain the release in comparison to the free MP suggesting the dephosphorylation property of MP is presence of ALP The delivery of 1P + MP at the target site prolonged the availability of MP, which diffused in a consistent manner from the hydrogelators. The hydrogelators at high concentration were completely non-toxic to the cells revealing their bio-compatibility and could be used effectively in-vivo. However, the presence of MP could affect the cell viability in a dose dependent manner.
After in-vivo administration of 1P + MP solution in a SCI rat model, the MP + 1P changes to the gel due to dephosphorylation of the MP + 1P in presence of ALP present in the cells. 11 The gel then slowly releases the drug at the site, consequently a significant reduction in the pro-inflammatory cytokines (TNF-α and IL-1β) level was observed as comparison to the other groups including the free MP and control group rats. These results could be clearly attributed to the availability of MP at the specific site. 28 Also, the histopathological analysis clearly revealed the reduction in the volume of lesions with improved activity of treated rats. Further, the study also revealed the non-toxic effect of MP as no sign of toxicity was observed in the vital organs isolated after the completion of study. Thus, it could be summarized that the local delivery of 1P + MP has significant enhanced the efficacy of MP on the SCI rats.
Conclusion
In the present study, we have successfully developed the hydrogelators for the local delivery of MP which could self-assembly in the presence of ALP and form a hydrogel. The local delivery of MP could overcome the drawbacks associated with the systemic delivery of MP including high dose toxicity. The MP + 1P + ALP hydrogel could resist the mechanical stress and have thick nanofibers as revealed by the rheology studies and cryo-TEM analysis respectively suggestion the successful entrapment of MP in hydrogel. The hydrogel could physically entrap MP and release the drug in a sustain manner to maintain the therapeutic level of drug. Also, these hydrogelators were capable of significantly reducing the pro-inflammatory cytokines levels in the blood without causing any toxicity to the vital organs of the rats. Therefore, it could be concluded that this novel hydrogelators could self-assemble in presence of intracellular ALP after local delivery in SCI rats model boosting the therapeutic potential of MP and alleviating its side effects.
Supplemental Material
sj-pdf-1-jbf-10.1177_2280800020978505 – Supplemental material for Alkaline-phosphatase triggered self-assemblies enhances the anti-inflammatory property of methylprednisolone in spinal cord injury
Supplemental material, sj-pdf-1-jbf-10.1177_2280800020978505 for Alkaline-phosphatase triggered self-assemblies enhances the anti-inflammatory property of methylprednisolone in spinal cord injury by Haotao Yu, Ping Zhang, Wei Zhou, Zhihong Zhong and Dongbin Qu in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Science and technology development special fund of Guangdong Province (No. 2017ZC0255) and General guidance project of Guangzhou Health Committee (No. 20181A011084).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
