Abstract
Background:
Targeted delivery to the Rheumatoid arthritis (RA) which is characterized by destruction and degeneration of bones due to chronic inflammation is of great need. RA being a chronic autoimmune disorder might result in severe disability and morbidity. A targeted delivery system is designed to deliver methotrexate (MTX) for RA.
Methods:
Here, we synthesized folic acid (FA) conjugated hydrophobically modified glycol chitosan (GC) self-assembled nanoparticles (FA-GC-SA) for the targeted delivery of MTX to RA. The FA conjugation and hydrophobic modification of GC by stearic acid (SA) was confirmed by Fourier-transform infrared spectroscopy (FTIR). The FA-GC-SA was exploited for developing targeted nanoparticles encapsulating MTX by the ionic gelation method. The particles were characterized and evaluated for their targeting potential in in vitro cell culture studies. Further their in vivo efficacy in arthritis induced rats using collagen was also evaluated.
Results:
FTIR confirms the successful modification of GC-SA and FA-GC-SA. The FA-GC-SA-MTX of size 153 ± 9 nm were prepared with high encapsulation efficiency of MTX. The FA-GC-SA-MTX size was further confirmed by transmission electron microscopy (TEM). In vitro cell studies revealed the superior efficacy of FA-GC-SA-MTX in cell cytotoxicity. Also, significantly higher cellular uptake of FA functionalized FA-GC-SA-MTX was observed in comparison to non-functionalized GC-SA-MTX attributed to folate receptors (FRs) mediated endocytosis. In vivo results confirms the potential of FA-GC-SA-MTX which reduces reduces the pro-inflammatory cytokines, paw thickness, and arthritis score in collagen induced rats.
Conclusion:
The results shows that FRs targeted FA-GC-SA-MTX has superior efficacy in the treatment of RA.
Introduction
Rheumatoid arthritis (RA) is a disease causing joint dysfunction by inflammation, cartilage, and bone destruction. 1 This chronic autoimmune disease, if not treated well could result in severe disability and morbidity.1,2 RA affects both the genders with a higher ratio to females. Anyone can acquire RA at any stage of life with a higher possibility at the age reaching to 50. 3 The exact mechanism of its pathogenesis is not yet identified, although tumor necrosis factor TNF-α, interleukins and several other inflammatory mediators are known to play an important role in the progression of RA.4,5 The complete cure of RA is still unknown and the quality of life of the affected people are just improved by supporting medications which mostly include anti-inflammatory agents such as non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticosteroids, disease-modifying antirheumatic drugs (DMARDs) and some biologic agents.6,7 In some of the cases, the treatment of RA could also be associated with surgery. 8
Methotrexate (MTX) is highly efficient DMARDs class of drug used for the treatment of RA. 9 The degeneration of joints in RA could be reduced by MTX as it affect the pro-inflammatory cytokines such as interleukins and TNF-α, thus can further reduce the inflammation in joints. 10 However, the rapid clearance of MTX results in lower concentration of drug in the system. Nanoparticles (NPs) which are playing an important role in the field of medicine, with their wide role in the treatment and diagnosis of the disease could also be exploited for controlled and targeted delivery.11,12
The folate receptors (FRs) is a 38 kDa glycosylphosphatidylinositol-anchored protein that binds to the vitamin FA with high affinity (KD < 1 nM) 13 FRs expression on activated macrophages can be exploited to selectively target imaging agents to sites of inflammation in rats with adjuvant-induced arthritis.14 –16 Interestingly, covalent conjugation of small molecules, proteins, and even liposomes to the carboxyl moiety of folic acid does not alter its ability to bind the folate receptor and undergo endocytosis by receptor bearing cells. 17
In the study presented here, we designed a targeted cargo to deliver MTX for RA. The FA was chemically attached to the nano-delivery system for targeting RA affected site. Scheme 1 represents the folate-conjugated hydrophobically modified glycol chitosan nanoparticles for targeted delivery of methotrexate in rheumatoid arthritis. The FA modified self-assembled nanoparticles were loaded with MTX and were thoroughly characterized. The efficacy of the delivery system was validated for biocompatibility assessment through in vitro cell line studies. Anti-RA activity was validated through in vivo experiments for which RA induced animal model was used and pro-inflammatory cytokines was evaluated.
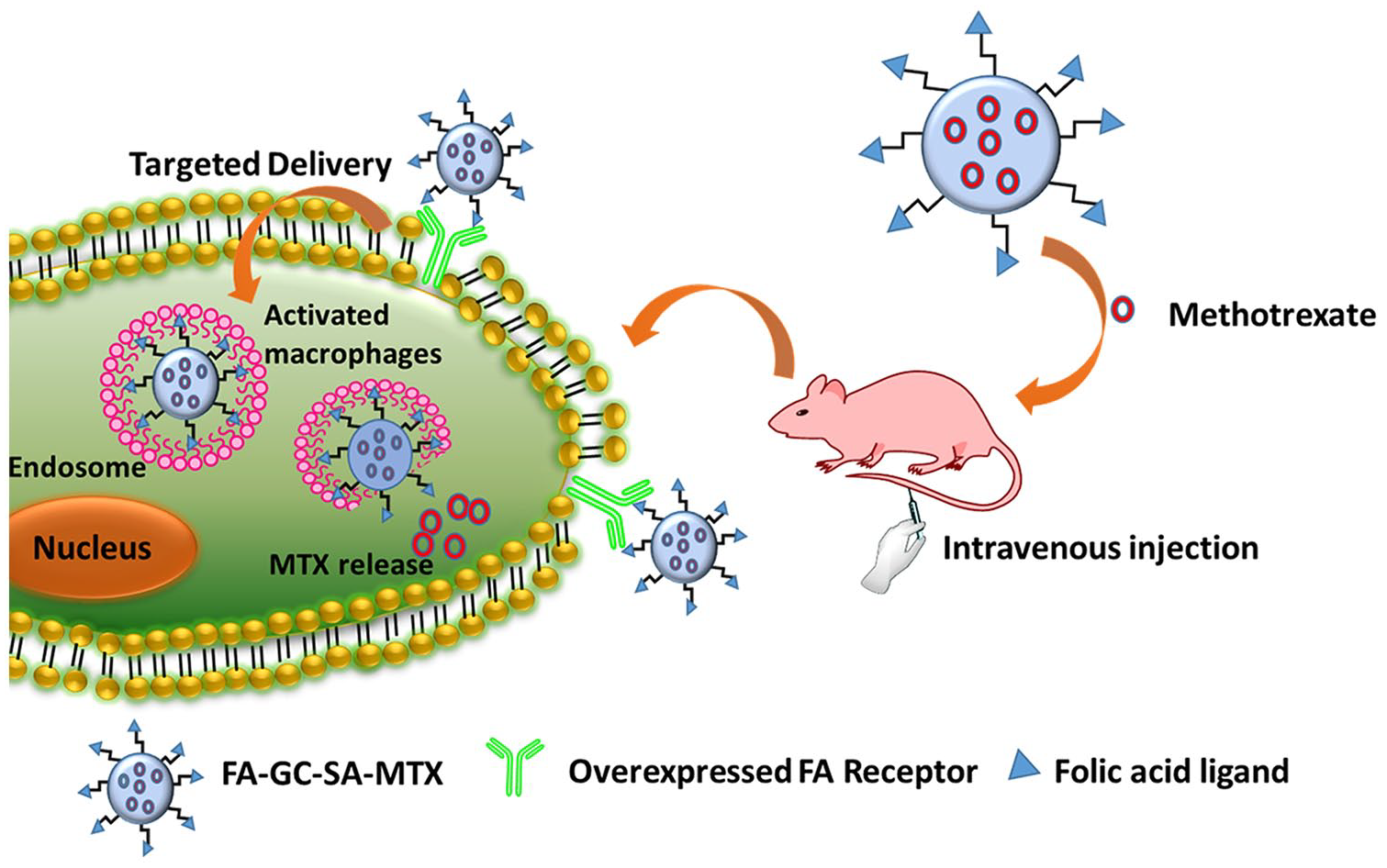
Schematic representation of folate-conjugated hydrophobically modified glycol chitosan nanoparticles for targeted delivery of methotrexate in rheumatoid arthritis.
Material and methods
Glycol Chitosan was bought from Yuhuan Marine Biochemistry Co Ltd, Zhejiang, China). Stearic acid was obtained from Chemical Reagent Co Ltd (Shanghai, China). 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC), 2,4,6-trinitrobenzene sulfonic acid (TNBS), Folic acid was obtained from Sigma (St Louis, MO), Dulbecco’s Modified Eagle’s Medium, and trypsin-ethylenediamine were purchased from Gibco BRL (Gaithersberg, MD). All solvents used were of analytical grade and the distilled water was used wherever required.
Synthesis of hydrophobic glycol chitosan
The SA was used to hydrophobically modify GC to develop self-assembled nanoparticles. The modification was carried out through EDC coupling method. 18 Briefly, the carboxylic group of SA was activated using EDC by dissolving SA and EDC in alcohol (10 mL) at a molar ratio of 1:1.2 respectively using vortex to obtain clear solution A. The aqueous solution of GC was prepared at a concentration of 200 mg/mL in a separate beaker to obtain solution B. Finally, the GC-SA was obtained by adding solution A drop wise to the solution B with constant stirring (150 rpm) at ambient temperature for 4 h. GC-SA thus prepared was purified using dichloromethane and lyophilized. The conjugation of GC-SA was further characterized by using FTIR and the characteristic peaks were compared with GC and SA.
Synthesis of folic acid modified hydrophobic glycol chitosan
The FA modified GC-SA was obtained by conjugating free amine group of GC-SA to carboxylic group of FA using the EDC coupling reaction. The EDC and FA solution was first prepared in dimethyl sulfoxide at ambient temperature with stirring at a molar ratio of 10:1 respectively. Subsequently, the solution having activated carboxylic group of FA was added slowly to the solution of GC-SA and the solution was kept under constant stirring for overnight. The FA conjugated GC-SA (FA-GC-SA) was finally obtained by several washing and the finally lyophilized. The successful synthesis of FA-GC-SA was further confirmed by using FTIR.
Synthesis of methotrexate loaded nanoparticles
The MTX was loaded in the FA-GC-SA-MTX and GC-SA-MTX during the process of nanoparticles fabrication by using ionic gelation method. In brief, sodium alginate and calcium chloride solution containing MTX was added to FA-GC-SA or GC-SA solution at pH 4.5 with continuous stirring up to 1 h followed by sonication for 5 min. Thus prepared FA-GC-SA-MTX and GC-SA-MTX were collected after overnight incubation by centrifugation. We also prepared blank particles without MTX, FA-GC-SA, and GC-SA. However for fluorescent labeled FA-GC-SA-MTX and GC-SA-MTX, the particles were incubated with Nile red solution.
In vitro characterization
The developed FA-GC-SA-MTX and GC-SA-MTX were characterized for their surface morphology and hydrodynamic diameter using Transmission electron microscopy (TEM, CM30, Philips, CA, USA), Scanning Electron Microscopy (SEM, Carl Zeiss Microscopy Gmbh 73447 Oberkochen, Germany) and Malvern zeta sizer (Nano ZS, Malvern, UK) respectively. The surface charge on the particles was also evaluated using the Malvern zeta sizer. The hydrodynamic diameter of the particles were measured after their dilution using distilled water. The TEM analysis was carried out by placing a drop of dispersed particles on a copper grid, which was negatively stained with 5% uranyl acetate.
Further we evaluated the amount of MTX entrapped in FA-GC-SA-MTX and GC-SA-MTX and also the loading efficiency was evaluated. The samples were centrifuged using ultracentrifuge at 30,000 rpm for 30 mins and the supernatant was analyzed using UV-Vis spectrophotometer for calculating the free MTX. The entrapment and loading efficiency was calculated using the following equations 1 and 2 respectively.
Where, W1 = weight of MTX taken initially, W2 = weight of free MTX, W3 = weight of MTX in FA-GC-SA-MTX and W4 = weight of FA-GC-SA-MTX
In vitro release studies
Next we evaluate the drug release profile of FA-GC-SA-MTX and GC-SA-MTX in comparison with free MTX at the physiological pH 7.4. FA-GC-SA-MTX, GC-SA-MTX and free MTX having equivalent concentration of MTX were dispersed in 5 mL of PBS having pH 7.4 and were placed in a dialysis bag. Then these dialysis bags were placed in 200 mL of medium in a beaker maintained at a temperature of 37°C with a slight stirring. The samples were collected at different time points and the amount of MTX released was analyzed using UV-Vis spectrophotometer at 303 nm.
Cell culture
We used macrophages cell lines RAW264.7 which were cultured using DMEM medium flourished with 10% of FBS and 1% of antibiotic solution. The cells were maintained in an incubator at 37°C in a 5% CO2 environment. The cells were trypsinized and plated for further experiments according to the experimental design.
In vitro cell cytotoxicity studies
The cell cytotoxic effect of FA-GC-SA-MTX and GC-SA-MTX was evaluated in the RAW264.7 cell lines and the results were compared with free MTX. Briefly, 3000 cells per well were plated in a 96 cell culture well plate and incubated for 24 h at 37°C. Next day the cells were treated with FA-GC-SA-MTX, GC-SA-MTX and free MTX at different concentrations (5, 10 and 20 µg/mL). Following 24 h incubation the cells were treated with 10 µL of 5mg/mL MTT solution. The cells were incubated again for 4 h and subsequently the formazon crystals formed were dissolved using 100 µL of DMSO. The optical density was evaluated using Elisa plate reader at 520 nm.
In vitro cellular uptake studies
The efficacy of FA-GC-SA-MTX over GC-SA-MTX was evaluated in RAW 264.7 cells. The endocytosis mediated by FRs was confirmed in LPS treated RAW 264.7 cells following treatment with Nile red labeled FA-GC-SA-MTX and GC-SA-MTX. After 4 h of incubation the internalization of particles was monitored using confocal fluorescent microscope (Nikon, Japan).
In vivo experiments in collagen induced rats
To further evaluate the potential of FA-GC-SA-MTX and GC-SA-MTX in animal model, the ethical approval to conduct the experiment was obtained from the local ethical committee of The First People’s Hospital of Huzhou. The experiments were conducted under the protocol number: FPHAA-2019-081. The Sprague Dawley rats of 5 weeks were obtained from the Shanghai Institute, Shanghai and were kept in a standard condition. After 2 days, the arthritis was induced in the rats by injecting Complete Freund’s adjuvant (CFA) (100 μL) intradermal to the lower legs. 19 The arthritis was developed within 5 weeks in four groups, following which the rats were divided into Group II, Group III, Group IV, and Group V. Group I was control whereas group 2 to group 5 were administered intravenous with PBS, free MTX, FA-GC-SA-MTX, and GC-SA-MTX respectively every 3 day. The equivalent dose of MTX (0.5 mg/kg) was used in the experiment. The effect of the treatment was evaluated based on the paw thickness and arthritis score during the whole study. The level of cytokines of IL-1β and TNF-α in the blood of the rats were also measured at the end of the study
Statistical analysis
The results of the studies were compared and evaluated for their significant difference using ANOVA. p < 0.05 was taken as statistically significant.
Results
The present study describes the synthesis of targeted FA-GC-SA-MTX. We started with the hydrophobic modification of GC using SA and the resulting GC-SA was further modified using FA by simple carbodimide reaction. The detailed scheme has been illustrated in Figure 1. 4 The successful synthesis of GC-SA and FA-GC-SA was confirmed by FTIR spectra. Figure 2 shows the characteristic peaks of GC, SA, GC-SA, and FA-GC-SA. As shown in Figure 2, a broad peak corresponding to -OH group of GC was observed at 3390 cm−1 whereas the peak of primary amine might be overlapped in it. The strong peak of –CH stretching was observed for SA at 2907 cm−1. The GC-SA spectrum shows the peak of –OH with slight reduced intensity at 3340 cm−1 with strong peak at 2910 cm−1 of –CH stretching. However the peak of FA-GC-SA shows the –OH peak at 3412 cm−1 with strong peak at 2890 cm−1. The other peaks of C=O of amide was present at 1690 cm−1. The results clearly demonstrated the successful modification of GC with SA and their further functionalization with FA.

Schematic illustration of preparation of glycol chitosan stearate (GC-SA) and further functionalizing it with folic acid (FA-GC-SA).

FTIR spectra showing the characteristic peaks of GC-SA and FA-GC-SA in comparison with glycol chitosan and stearic acid.
Formulation and characterization of MTX-GC-SA
The FA-GC-SA-MTX and GC-SA-MTX thus prepared were characterized for their physicochemical properties including surface morphology, particle size, size distribution. The hydrodynamic evaluation of the FA-GC-SA-MTX and GC-SA-MTX evaluated by DLS revealed the size of 153 ± 9 nm and 135 ± 7 nm which was further confirmed by TEM. As shown in Figure 3, the size obtained by SEM and TEM results were similar to the results obtained from DLS. The surface charge on FA-GC-SA-MTX and GC-SA-MTX were found to be −25.6 ± 1.28 mV and −21.2 ± 1.37 mV respectively.

(a) The dynamic light spectroscopy image of FA-GC-SA-MTX. (b) The transmission electron microscopic image of FA-GC-SA-MTX. (C) Scanning Electron Microscopic image of FA-GC-SA-MTX.
The evaluation of entrapment efficiency of FA-GC-SA-MTX and GC-SA-MTX was observed to be 72% and 79% respectively whereas the loading efficiency observed for FA-GC-SA-MTX and GC-SA-MTX were 4.8% and 5.6% respectively.
The cumulative percent release of MTX from FA-GC-SA-MTX and GC-SA-MTX at physiological pH 7.4 was evaluated and compared with free MTX as shown in Figure 4. The release of MTX from FA-GC-SA-MTX and GC-SA-MTX was observed to be sustained when compared with free MTX which was completely released within 8 h. There was no initial burst release, revealing the proper encapsulation of MTX in FA-GC-SA-MTX and GC-SA-MTX. We also did not observed any significant difference in the release pattern of FA-GC-SA-MTX and GC-SA-MTX. However, only slight decrease in release of MTX from FA-GC-SA-MTX was observed in comparison with GC-SA-MTX with 65% and 72% of MTX release respectively.

Cumulative percent release of MTX from MTX solution, GC-SA-MTX and FA-GC-SA-MTX in PBS pH 7.4.
Cytotoxicity studies and in vitro cellular uptake studies
Next, we evaluated the cytotoxicity effect of FA-GC-SA-MTX and GC-SA-MTX on RAW264.7 cell lines using MTT assay. The cells when treated with equivalent concentration of MTX in free MTX, FA-GC-SA-MTX and GC-SA-MTX and their efficacy were evaluated after 24 h of treatment. The results were illustrated in Figure 5. The free MTX induced lowest cytotoxicity at all the concentrations whereas highest cytotoxicity was induced by FA-GC-SA-MTX at all concentrations. The results might be attributed to the higher internalization of MTX from FA-GC-SA-MTX and GC-SA-MTX due to the receptor mediated opsonization in comparison with free MTX. 20

Percent cell viability of free MTX, GC-SA-MTX and FA-GC-SA-MTX after the 24 h of treatment with different concentration of MTX. FA-GC-SA-MTX resulted in significantly high cytotoxicity (*p < 0.05) in comparison with free MTX and (#p < 0.05) in comparison with GC-SA-MTX at the equivalent concentration of 20 µg/mL.
The in vitro cellular uptake studies were conducted to evaluate any superiority of functionalized FA-GC-SA-MTX over non-functionalized GC-SA-MTX in the RAW264.7 cell lines. The Nile red labeled FA-GC-SA-MTX and Nile red labeled GC-SA-MTX at an equivalent concentration of Nile red were incubated with RAW264.7 cell lines. The red fluorescence was emerged from internalization of Nile red labeled particles and the blue fluorescence was due to staining of nucleus using Hoechst as shown in Figure 6. The images obtained by the confocal fluorescence microscope revealed significantly higher red fluorescence around the nucleus of the cells treated with FA-GC-SA-MTX in comparison to GC-SA-MTX. The results might be attributed to the receptor (FRs) mediated internalization in addition to the regular endocytosis process.21,22 However, the week fluorescence of GC-SA-MTX in cells could be attributed to the only mechanism of endocytosis involved in the internalization of these particles.

Confocal fluorescent Images of cellular uptake after 4 h of incubation of cells with equivalent concentration of Nile red.
Arthritis score and the paw thickness
The therapeutic efficacy of developed formulations was compared with the control and free MTX treated group rats. Figure 7(a) represents the effect on paw edema after the treatment with control, PBS, free MTX, GC-SA-MTX, and FA-GC-SA-MTX in collagen induced arthritis rats. The arthritis score and the paw thickness were continuously monitored after the treatment in the rats of different groups. As shown in Figure 7(b), the results demonstrated that the PBS group rats have continuous increase in arthritis score and paw thickness. However, all the other group rats showed improvement after the treatment. The FA-GC-SA-MTX group of rats revealed the significant improvement in arthritis score compare to other groups corresponding to its targeting ability due to the presence of FA which could be recognized by the FRs and the particles could be accumulated at the affected site resulting in higher MTX concentration and improved efficacy. The significant reduction in paw thickness was also observed in FA-GC-SA-MTX treated rats which could be attributed to selective targeting. The results might be attributed to the accumulation of the MTX-GC-SA in the affected region of arthritis.
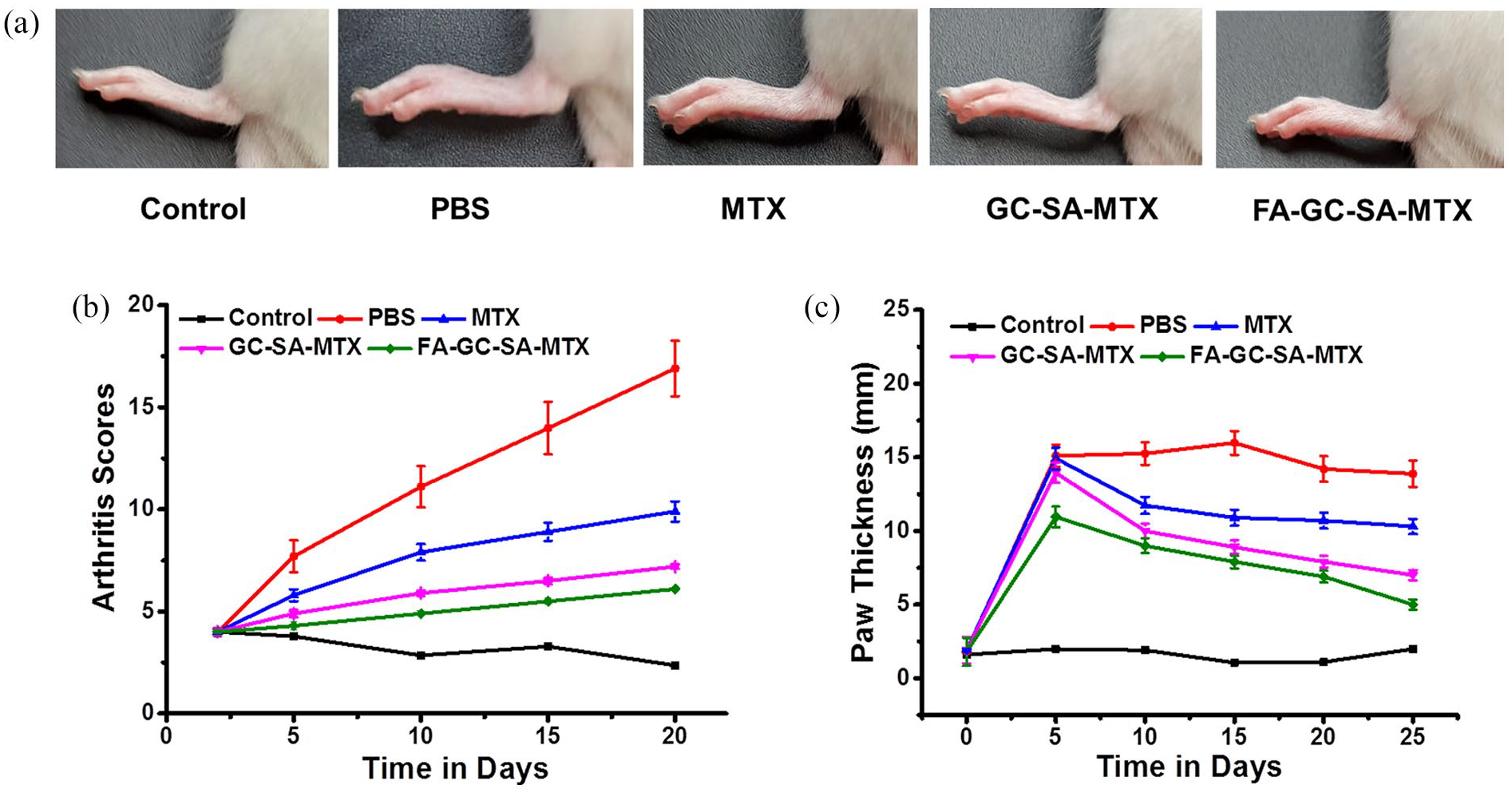
(a) The images represents the effect on paw edema after the treatment with control, PBS, free MTX, GC-SA-MTX, and FA-GC-SA-MTX in collagen induced arthritis rats. (b) The data represents the arthritis score. (c) The data represents of the Paw thickness after the treatment.
The pro-inflammatory cytokine TNF-α and IL-1β levels were reduced in rats after the treatment with FA-GC-SA-MTX, GC-SA-MTX, and free MTX, corresponding to the reduction in the RA progression. 23 However, the significant (p < 0.05) reduction were observed in the rats treated with FA-GC-SA-MTX in comparison to free MTX and GC-SA-MTX. The level of significance of FA-GC-SA-MTX was much higher with the rats administered with PBS and was comparable to control. The results obtained for TNF-α and IL-1β have been illustrated in Figure 8(a) and (b) respectively. The results were comparable with the results of arthritis score and paw thickness. The significant effect of FA-GC-SA-MTX could be attribute to the FRs mediated recognition of FA conjugated particles as also observed in cell uptake studies.
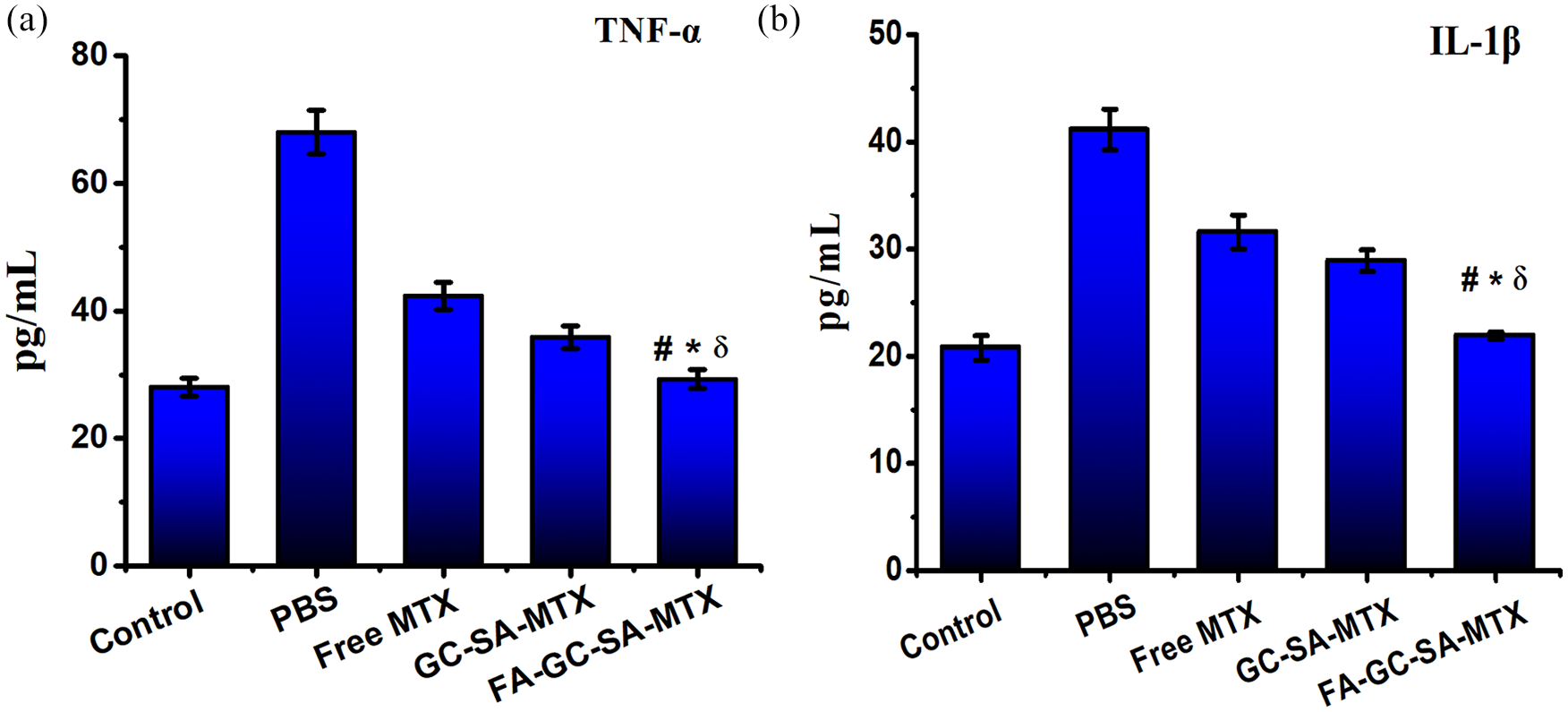
(a) The TNF-α and (b) IL-1β cytokines levels in blood after the treatment with PBS, free MTX, GC-SA-MTX and FA-GC-SA-MTX in comparison with healthy rat designated as control. The cytokine levels were significantly (ᵟp < 0.05, *p < 0.05, #p < 0.05) decreased in the rats treated with FA-GC-SA-MTX in comparison with PBS, free MTX and GC-SA-MTX treated groups respectively at the end of the study.
Discussion
RA has been characterized by inflammation in synovial joints which further leads to the destruction of bones. 24 Although several strategies have been applied for the treatment of RA, but most of them are not safe and has severe drawbacks. Basically the treatment approaches consist of disease-modifying anti-rheumatic drugs (DMARDs) aided by corticosteroids or non-steroidal anti-inflammatory drugs. The combination therapy reduce the inflammation and pain of joints, enhance the working and function of joint and overcome deformity of joints. The TNF-α targeting agents are main biological DMARDs. 25 The clinical RA therapies reduce the inflammation and joint damage but they have high risk of adverse effects. Hence, it is an utmost need to modify the disease control measures and methods that directly target the tissues of desired site. The macrophage activation of synovial is an ideal approach to overcome local inflammation, systematic inflammation as well as irreversible damage of joints.
GC is a biocompatible polymer soluble in aqueous medium at all pH. GC could be hydrophobically modified with hydrophobic compounds including SA by using a simple coupling reaction. 26 The amine group of CS reacts with carboxylic acid of hydrophobic compound to result GC-SA. In addition, the GC-SA were functionalized with FA using the EDC-mediated coupling. Further, the drug could be encapsulated in the modified polymer during the self-assembly of the nanoparticles. The developed FA-GC-SA-MTX could actively target FRs. Interestingly, the FA has great affinity for the FR present in biological environment even after carboxyl groups are derivatized. With the help of receptor-mediated endocytosis the internalization of folate conjugates occur via FRs.27,28 Therefore, the folate conjugation is an ideal approach for delivery of therapeutic mediators to FRs cells. These therapies attack only pathologic cells and keep themselves away from healthy macrophages. Hydroxychloroquine, sulfasalazine, leflunomide and MTX are the first line drugs used in the therapy of RA. We have chosen MTX as it is also a folate analog and have its physiochemical properties and structure similar to folate. MTX is capable of inhibiting the pyrimidine and purine synthesis which further inhibits the synovial cells proliferation. 29
The nanoparticles obtained were optimum in size for targeted delivery of therapeutics. The surface charge on FA-GC-SA-MTX and GC-SA-MTX was negative which might be attributed to the FA and SA present in the anionic polymer. The zeta potential obtained was optimum for the stability of particles in drug delivery. 30 As nanotechnology revolutionized, the drug circulation time, long time retention, bioavailability and targeting of numerous therapeutic agents could be improved by using novel drug delivery system. The drug loaded in nanoparticles has diverse advantages as compared to free drug administration. 31 The targeting efficiency of drugs is greatly improved with the help of nanoparticles due to their altering composition property. The developed FA-GC-SA-MTX could improve FRβ macrophages targeting and the efficiency of MTX in arthritic rats. 32
The nanoparticles could be easily get engulfed by the cells mediated by the mechanism known as phagocytosis. We observed that FA-GC-SA-MTX was significantly engulfed by the cells in vitro. The FA present on the FA-GC-SA-MTX could be recognized by the FRs present on the surface of the cells. These FRs up-regulates the transport of FA-GC-SA-MTX into the cells through caveolae-mediated endocytosis as these folate carriers could associate with FRs present on the surface of cells. 33 The MTX loaded FA nanoparticles could overcome various parameters in arthritis patients such as paw volume, bone resorption and decrease in body weight. Exciting results were obtained in the rats treated with FA-GC-SA-MTX as we observed significant reduction in the inflamed joints in comparison to the non-targeted GC-SA-MTX. However, both FA-GC-SA-MTX and GC-SA-MTX could accumulate at the diseased site and reduced the arthritis score, FA-GC-SA-MTX could effectively reduce the inflamed joints owing to active targeting through FRs in comparison to passive targeting of GC-SA-MTX. The pro-inflammatory cytokines including TNF-α and IL-1β, secreted by activated macrophages could enhance the progression of RA. The significant reduction in the production of these cytokines by FA-GC-SA-MTX clearly reveals the potential of active targeting. FA-GC-SA-MTX effectively cured collagen induced RA in rats. The results significantly revealed the possibility of prepared FA-GC-SA-MTX to treat RA.
Conclusion
The FA functionalized FA-GC-SA-MTX was developed as a delivery system of MTX to treat RA. The FRs mediated endocytosis was employed for better efficacy of the developed FA-GC-SA-MTX. The particles were well characterized and their efficacy was evaluated in in vitro and in vivo experiments which were also compared with free MTX and non-functionalized nanoparticles. The results shown superior efficacy of the developed FA-GC-SA-MTX in the treatment of RA induced rats corresponding to the FRs mediated delivery. Hence, the present work clearly demonstrates the therapeutic applicability of FA-GC-SA-MTX in RA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Project of Administration of traditional Chinese Medicine of Zhejiang Province of China (No: 2018ZA121).


