Abstract
Purpose:
In this study, pore size and porosity distribution of porous Ti-6Al-4V scaffolds (pTi) were controlled by 3D printing. The effects of pore size distribution at a constant porosity, or porosity distribution at a constant pore size pertaining to functions of adhesion, proliferation, and differentiation of the mouse embryonic osteoblast precursor (MC3T3-E1) cells were researched separately.
Methods:
3D printing was used to design five groups of pTi, designated as PS300/HP, PS300/LP, PS500/HP, PS500/LP, and PS800/HP based on pore size and porosity distribution. MC3T3-E1 cells were cultured on pTi, and non-porous Ti-6Al-4V samples (npTi) were prepared as control. The pTi was characterized with the scanning electron microscopy (SEM). MC3T3-E1 cells were stained via AlamarBlue assay and viability and proliferation analyzed. The mRNA levels of alkaline phosphatase (ALP), osteocalcin (OCN), collagentype-1 (Col-1), and runt-related transcription factor 2 (Runx2) in MC3T3-E1 cells were analyzed by real-time PCR analysis.
Results:
The average pore size and porosity of pTi were recorded as (301 ± 9 μm, 58.8 ± 1.8%), (300 ± 9 μm, 43.4 ± 1.3%), (501 ± 11 μm, 58.3 ± 1.2%), (499 ± 12 μm, 42.7 ± 1.1%), and (804 ± 10 μm, 58.9 ± 1.3%), respectively. SEM images confirmed active attachment of cells and oriented with the direction of metal rod after pTi/MC3T3-E1 co-culture for 3 and 7 days. In addition, MC3T3-E1 cells grown on the PS800/HP displayed significantly higher proliferation compared with each group after 3 days incubation (p < 0.05). Moreover, cells showed some degree of proliferation in all groups, with the highest value recorded for PS800/HP after culture for 7 days (p < 0.05). The gene expression pattern of ALP, OCN, Col-1, and Runx2 confirmed that these were down-regulated when pore size increased or porosity decreased of pTi (p < 0.05).
Conclusion:
The pTi facilitated the adhesion and differentiation of osteoblast when pore size decreased or porosity increased. The scaffold model resembles physical modification with porous structures, which has potential application in the surface modifications of Ti implant.
Introduction
The loosening of titanium (Ti) implant is one of the main reasons for postoperative complications of orthopedics. 1 The short-term stability of the Ti implant is subject to the strength of the fixation and the degree of the match, and the long-term stability is determined by the connection between the bone and the implant.2,3 This generation of connection is dependent on a series of cellular activities on the surface of the implant, including cell adhesion, proliferation, and differentiation.4-6 Otherwise, the long-term postoperative results are the loosening and displacement of implant, and eventual treatment failure. 7 Therefore, surface modification has been highlighted in the fixation of Ti implant.
Thus far, various surface modifications have been advanced for modifying Ti implant surfaces.8,9 Various chemical coating modifications, involving the deposition of graphene, calcium phosphate apatite, and hydroxyapatite, and biological modifications such as growth peptide and platelet-rich plasma coatings have been developed to strengthen Ti bioactivity.10-13 Another major factor that directly affects the implant/cell interaction is the physical modification of Ti surface topography, which includes the structure of micro-pores. 14 A wide variety of cell features can be affected by surface topography, including cell adhesion, integration at tissue/implant interface, and osteo-differentiation, as well as cell proliferation. 15
For decades, traditional physical modification has determined Ti implants as the most successful medical instrument and commercial design, based on machining processes. 16 Since then, several physical techniques capable of altering Ti surface topography have been applied to Ti implants to enhance surface features, such as Ti plasma spraying, grift-blasting, or powder sintering. 17 These alter the topography of the Ti surface layer, thereby improving response at the tissue/implant interface. 18 There is a consensus that micro-poring of the implant surface enhances bone integration at the tissue/implant interface, and therefore stability. 19 Similarly, micro-pores of the tissue/implant interface interact with osteoblast populations, which are reactive to pore size, pore shape, as well as interconnectivity. 20 To produce a useful physical modification, elements such as topological design, pore size, porosity, interconnectivity, mechanical properties, and surface characteristics have to be carefully considered. 21 However, using traditional physical modification methods it is difficult to induce the formation of interconnected and precise micron-scale micro-pore structures on Ti surfaces; therefore, the relevance of three-dimensional (3D) printing physical modification has been recently explored.
The 3D printing industry emphasizes accuracy and flexibility, which has prompted a rising interest in the area of 3D printing, more commonly known as additive manufacturing. While the essence of conventional manufacturing methods include machine-stripping material from the bulk form (i.e. subtractive manufacturing), 3D printing is based on building up a product layer by layer, adding material merely where necessary. 22 One mainstream direction for 3D printing is for medical applications, specifically orthopedics. 23 This paper focuses on the manufacture of micro-pores for physical surface modification by metallic 3D printing. The process, which in conventional manufacturing is impossible or immensely difficult to achieve for crafting a customized micro-pore structure, becomes relatively easy using 3D printing, through building up a form layer by layer, including internal cross-sections. 24
Recently, many researchers have begun to take advantage of 3D printing to study the effect of micro-pore structure on biological behavior in vitro and in vivo. Taniguchi et al. 25 reported that the porous Ti-6Al-4V scaffolds (pTi) with porosity of 65% and pore size of 600 μm had better fixation ability and greater bone ingrowth than those with pore sizes of 300 and 900 μm. Fuduka et al. 26 studied the osteoinduction of pTi with a channel structure. In this work, significant osteoinduction was observed when pore sizes were 500 and 600 μm. Chang et al. 27 concluded that Ti scaffolds with a porosity of 70% and a pore size of 313 and 390 μm exhibited the best cell proliferation and bone ingrowth capability. Li et al. 28 studied Ti scaffolds with 30–70% porosity and reported that Ti scaffold with a porosity of 60–70% could notably match human cancellous bone. However, an increase in porosity greatly affected pTi mechanical strength. Esen and Bor 29 reported that the increase of 12% (from 43% to 55%) of the porosity caused a decrease of yield strength and Young’s moduli by 60%. In general, the compromise in mechanical properties of the scaffold with increasing porosity sets an upper limit in terms of how much porosity can be tolerated. Imwinkelried 30 has studied the effects of porosity on mechanical properties of Ti scaffolds and concluded that 50–80% porosity of Ti scaffolds was consistent with natural bone in terms of stiffness and strength. Moreover, based on a well-designed experiment, Caravaggi et al. 31 investigated the selective laser melting (SLM) complex of 3D porous lattice CoCr scaffolds such as crossing-rod, trabecular, and circular with pore sizes of approximately 510, 670, and 800 μm, respectively. The research showed favorable biocompatibility and mechanical property of the SLM porous CoCr scaffolds.
Accordingly, these previous studies demonstrated that micro-pore structures with pore sizes of 300–800 μm and porosities of 30–80% exhibited reasonable performances. However, to accurately evaluate the effect of pore structure, other parameters, such as porosity, pore size, interconnectivity, and surface characteristics needed to be kept identical. For example, Ti scaffolds (with pore sizes of 160, 400, and 680 μm and varying porosity) were examined in goats by Li et al. 32 They concluded that increased pore size and porosity induced greater bone formation. Likewise, in Ti scaffolds produced using powder sintering, large pores (500–1500 μm) were reported to initiate greater bone formation than small pores (250–500 μm) in rabbit femur. 33 The Ti scaffolds used in these studies had poorly controlled architecture and wide pore size distribution. Therefore, it is difficult to discern the true effect of pore size or porosity from these studies. To date, no reports have clarified the true effect of micro-pore structure in terms of pore size at a constant porosity, or porosity at a constant pore size.
The aim of this study was to investigate a suitable micro-pore structure of physical modification for osteoblast adhesion, proliferation, and differentiation. In this study, we have made pTi by 3D printing, which allowed separate control of pore structure (pore size and porosity), to determine the optimal porous structure for pTi manufactured by this process; therefore, pTi had three constant pore sizes (300, 500, and 800 μm) with two constant porosities (43.3 and 58.1%). Mouse embryonic osteoblast precursor (MC3T3-E1) cells were cultured on pTi to observe osteoblast adhesion, proliferation, and differentiation properties via scanning electron microscope (SEM), AlamarBlue assay, and real-time polymerase chain reaction (PCR), respectively (Figure 1). We demonstrated that the integration of pore size and porosity distribution on pTi by 3D printing affected initial cell attachment, subsequent cell proliferation, and differentiation positively in a longitudinal study of 3 weeks.
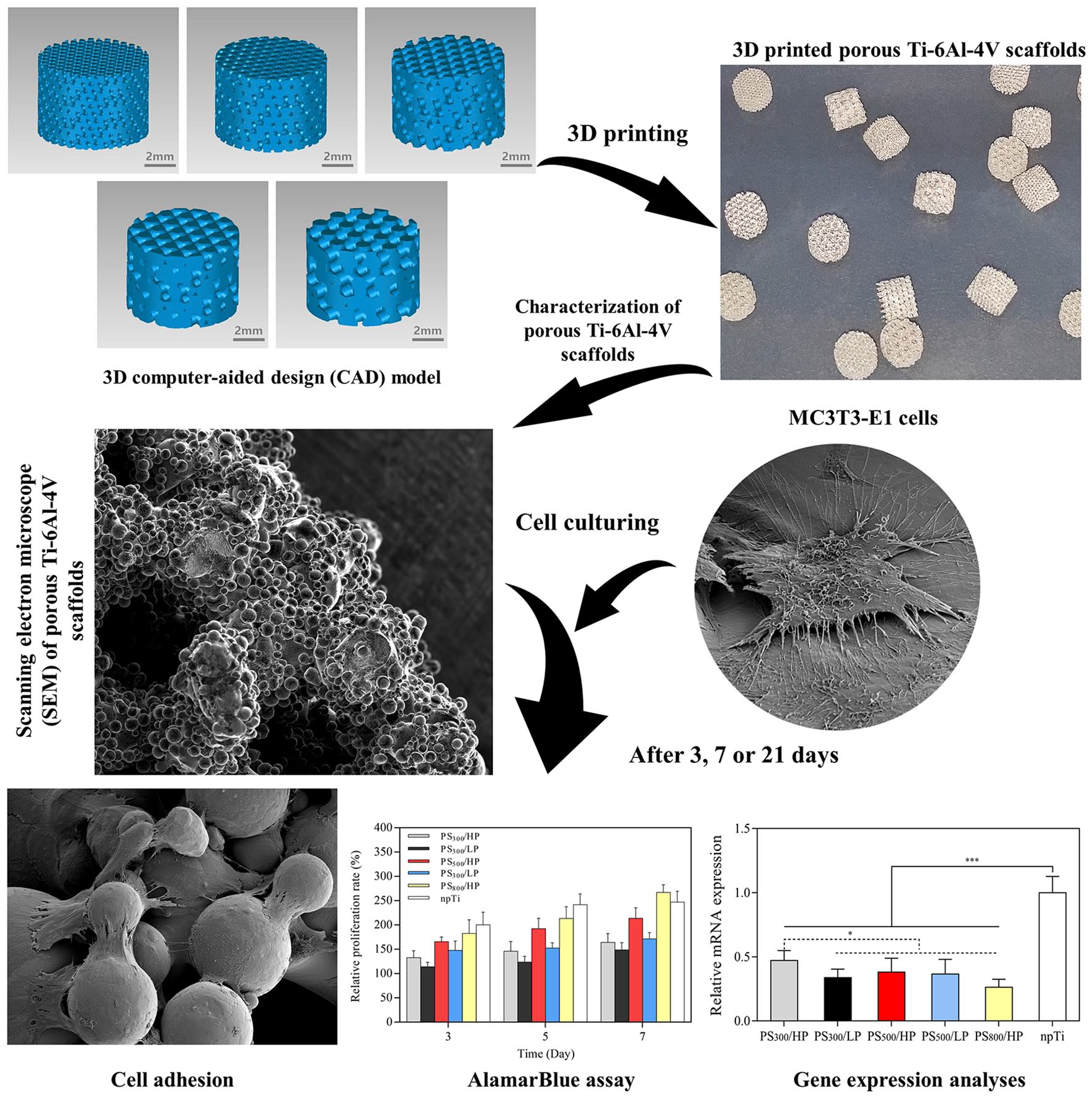
The flowchart of the 3D printed porous Ti-6Al-4V scaffolds preparation protocol and experimental method. 3D computer model technology was used to design five groups of porous Ti-6Al-4V scaffolds (pTi), the computer-aided design (CAD) data were imported into the selective laser melting (SLM) machine for 3D printing. Macroscopic inspection was performed for each and scanning electron microscopy (SEM) images were taken for the characterization analysis. The mouse embryonic osteoblast precursor (MC3T3-E1) cells were cultured on pTi, then tested by SEM, alamarBlue assay and real-time PCR for adhesion, proliferation and differentiation.
Methods
Fabrication of porous Ti-6Al-4V scaffolds by 3D printing
The 3D computer-aided design (CAD) model of pTi was created using Geomagic Studio 2013 software, and distributed pore size and porosity with 300, 500, and 800 μm (PS300/PS500/PS800) and low/high porosity (LP/HP), respectively (Table 1). The samples were designated as PS300/HP-pTi, PS300/LP-pTi, PS500/HP-pTi, PS500/LP-pTi, and PS800/HP-pTi based on their pore size and porosity values. The CAD data were then transferred to the SLM machine (M280, EOS GmbH Company, Germany). Laser melting was used to produce a cross-section layer by melting and fusing the medical-grade Ti-6Al-4V powder (TLS Company, Germany, 15–53 μm powder size). The chemical composition of Ti-6Al-4V powders is shown in Table 2. Subsequently, continuous medical-grade Ti-6Al-4V powder layers were applied until the entire pTi had been manufactured into a disk shape (diameter 7 mm; height 5 mm). Non-porous Ti-6Al-4V samples (npTi) were also fabricated with the same method as above. The external shape was disk (diameter 7 mm; height 5 mm) and the internal architecture was non-porous structure.
Pore size and porosity distribution of the 3D model.

The chemical composition of Ti-6Al-4V powder.

Scanning electron microscopy of 3D printed Ti-6Al-4V scaffolds
The morphology of 3D printed pTi was studied using SEM (TeneoVS FEI Company, America). Gold palladium was used to coat and cover the pTi surface before the morphology analysis. Image J software was used to analyze the pTi morphology.
Cell culture studies using MC3T3-E1 cells
MC3T3-E1 cells (Shanghai Biotechnology Company, China) were cultured in Minimum Eagle’s Medium (MEM-α, Thermo Company, America); MEM-α was supplemented with 10% fetal bovine serum (FBS, Thermo Company, America) and 1% penicillin/streptomycin (PS, Beyotime Biotechnology Company, China). MC3T3-E1 cells were plated at cell dish and cultured at 37°C, 5%CO2. MEM-α was changed every 3 days.
Cell adhesion and morphology analysis
The cell adhesion and morphology of MC3T3-E1 cells on pTi were observed by SEM. Before cell seeding, all pTi were autoclaved, followed by wetting in MEM-α for 24 h, then transferred into 24-well plates. MC3T3-E1 cells were seeded on pTi (1×104 cells/pTi) and cultured for 3 and 7 days in vitro. After incubation for 3 and 7 days, pTi were taken out from the 24-well plate and washed twice with phosphate buffer solution (PBS, Thermo Company, America), creating the SEM sample.
alamarBlue assay
The alamarBlue assay kit (Invitrogen Company, America) was used to evaluate MC3T3-E1 cell proliferation after cells were seeded onto samples (pTi and npTi) and cultured for 1,3, 5, and 7 days in vitro. This assay is a growth indicator adopted fluorometric and colorimetric method that transforms from an oxidized (nonfluorescent, blue) form to a reduced (fluorescent, red) form when reduced by mitochondrial respiration. At a concentration of 1×104 cells/pTi, the MC3T3-E1 cells were cultured in 24-well plates using MEM-α. The alamarBlue indicator was added at predetermined time points, and cells were incubated with 10% alamarBlue indicator for 4 h. The absorbance was measured at 570 and 600 nm using a fluorescence plate reader (Tecan Company, Austria).
Quantitative real-time PCR
The mRNA levels of collagentype-1 (Col-1), runt-related transcription factor 2 (Runx2), alkaline phosphatase (ALP), and osteocalcin (OCN) in MC3T3-E1 cells were analyzed using real-time PCR (Table 3). The mRNA of MC3T3-E1 cells was extracted using Trizol reagent (Invitrogen Company, America) and Total RNA Extraction kit (Roche Company, Switzerland) after 7 days and 3 weeks. According to the transcriptor first-strand cDNA synthesis kit (Roche Comapany, Switzerland) processes, the mRNA was reversed-transcribed into first-strand complementary DNA (cDNA). β-actin was used as a housekeeping gene. The process was conducted in PTC-100 real-time PCR instrument (Applied Biosystems Company, America). The relative mRNA levels of multiple cytokine genes in each sample were displayed as 2-ΔΔCt values and were representative of at least three independent experiments.
Primer sequences of ALP, OCN, Col-1, Runx2 and β-actin.

Statistical analysis
Statistical analysis was performed using SPSS 22.0 statistical software, and the image was produced using GraphPad Prism 5.0 software. The data are expressed as the average ± standard (¯x ± s ) deviation. One-way analysis of variance (ANOVA), followed by LSD test were performed for comparison between groups; p < 0.05 was considered statistically significant.
Results
Scaffold morphology analysis
The pTi and CAD model of diamond molecular structure are similar in shape. SEM images showed that the size and shape of the pTi micro-pores were uniform. Porosity evaluation by the density method revealed the accuracy of the pTi. The pore size and porosity distribution of pTi is shown in Figure 2.

The SEM images and pore size distribution plots of 3D printed porous Ti-6Al-4V scaffolds. Scale bars are the same for all SEM images. SEM showed that the size and shape of the micro-pores of the pTi were uniform, and unsintered Ti-6Al-4V powders not attached to the surface of pTi. In addition, a large number of partially melted Ti-6Al-4V powders bonded to the pTi during the SLM process. Partially melted Ti6Al4V powders on the surface changed the surface topography of pTi, which increased the surface roughness.
Cell adhesion and morphology analysis
In this study, the cell growth was shown on the SEM images after 3 and 7 days (Figure 3(a)). At both time points, MC3T3-E1 cells spread, penetrated, and oriented themselves along the direction of the pTi. This suggested that the guidance of cell morphology and cell cytoskeleton extension had been conspicuously influenced by structure orientation. According to the SEM images, the PS300/HP and PS300/LP showed more active cell attachment morphology, when compared with other groups after 3 days. The images show stretched out filamentous cells and slab-like pseudopods anchored to the surface of the pTi in PS300/HP and PS300/LP; the cell protrusions were fewer and shorter in PS500/HP, PS500/LP, and PS800/HP. Also, after 7 days, MC3T3-E1 presumably completely covered the pTi. The number of cell protrusions increased, and the connections between the protrusions became nets.

The MC3T3-E1 cells culture on the 3D printed porous Ti-6Al-4V scaffolds (in vitro assessment and gene expression). (a) SEM images of MC3T3-E1 cells adhesion on pTi after 3 and 7 days, where dashed lines and square frames highlight high magnification cells; (b) cell viability results of all groups tested via alamarBlue assay after 3, 5, and 7 days (* p < 0.05); (c) the relative mRNA expression of ALP, Col-1, OCN, and Runx2 after 7 days and 3 weeks (* indicates statistical significance, * p < 0.05; ** p < 0.01; *** p < 0.001).
Cell proliferation
After evaluation of cell proliferation by alamarBlue assay, it was determined that cell proliferation increased as incubation time increased in both pTi and npTi (Figure 3(b)). It was demonstrated that after 3 days, cell proliferation was significantly greater in the PS800/HP compared with each group (p < 0.05). After 7 days, the cells proliferated actively in all groups. The PS800/HP showed a higher level of cell viability compared with each of the other groups (p < 0.05). These findings suggested that the micro-porous effect of pTi on cell proliferation was positive.
Quantitative real-time PCR
The gene expression pattern of the osteoblast differentiation markers ALP, OCN, Col-1, and Runx2 confirmed that the micro-pores of the pTi affected the osteoblast differentiation (Figure 3(c)). The markers—ALP, OCN, Col-1, and Runx2—were down-regulated in the presence of increased pore size or reduced porosity (p < 0.05). Cells cultivated in the presence of PS800/HP revealed the lowest expression, and highest in the presence of PS300/HP. The differences between the micro-pore modifications became significant for ALP, Runx2, and Col-1 after 7 days (p < 0.05). The gene expression of OCN was comparably up-regulated after 3 weeks. In combination with PS300/HP, the osteoblast marker OCN showed the highest expression (p < 0.05).
Discussion
3D printing has become a new method for physical modification in recent years. The physical modification of a Ti implant is usually governed by pore structure, which further depends upon the extent of porosity and pore size range. 3D printing is suited to the manufacture of pTi with a precisely controlled pore size, porosity, and interconnectivity. However, no reports have compared pTi of different pore sizes with a constant porosity, or of different porosities with a constant pore size, when manufactured using 3D printing. In this regard, our study demonstrated the independent effect of pore size with a constant porosity and porosity with a constant pore size; i.e. the superiority of PS300/HP in osteo-differentation and adhesion, and superiority of PS800/HP in proliferation was evident.
The scientific literature has shown that the effect of pore size on cell adhesion is determined by the density of the pTi rod. The smaller pore size increases the density of the pTi rod, thereby increasing adsorption to promote cell adhesion. 34 In this study, the morphology of cells in each group was observed by SEM, and it was discovered that osteoblasts adhered better to small-aperture pTi, in accordance with the above findings. Previous studies have found that porosity also affects protein adsorption; the higher the porosity, the more it promotes cell adhesion ability. 35 As porosity increases, surface area increases, thereby providing space for cell adhesion. 36
Previous studies have also suggested that larger pore size and porosity are more conducive to cell proliferation. This is similar to the results of Mygind et al., 37 who used hydroxyapatite scaffolds for cell experiments and discovered that large pore sizes promote cell proliferation capacity. In addition, higher surface areas in porous structures and pore connections through transportation of oxygen and nutrients to cells were proved to promote tissue adhesion, growth, and vascularization. Moreover, the porous structures combined with interconnected pores promoted nutrient diffusion, which may be involved in the conjugation between new bone and pTi. In our study, when cell proliferation was tested by alamarBlue assay, it was discovered that when porosity was similar, cell proliferation increased according to increased pore size. When pore size was similar, cell proliferation of high porosity pTi was higher. Although the npTi showed a higher cell proliferation rate at most times, cell proliferation of npTi was gradually lower than PS800HP, after being cultured for 7 days.
Runx2 is an osteogenic marker gene that regulates differentiation of osteoblasts and synthesizes Col-1 and matrix-associated proteins, marking the beginning of osteogenic differentiation. 38 ALP is associated with the reconstruction of bone matrix and is one of the signs of early osteogenic differentiation. OCN is considered a late marker of osteoblast differentiation and maturation. In this study, Runx2 and Col-1 were expressed with a similar trend, indicating their correlation. This study found that pore size and porosity have a tendency to influence cell differentiation. Therefore, when pore size is constant, increased porosity can enhance osteoblast differentiation, and when porosity is constant, reduced pore size can enhance osteoblast differentiation. Although there were no statistically significant differences between the individual groups in this study, the above trends were still present in these groups. The reason for this analysis is the small sample size or experimental error. Cheng et al. 39 produced titanium scaffolds that simulated human cancellous bone, and found that high porosity was more conducive to cell differentiation. Teixeira et al. 40 studied the effect of titanium scaffold on osteoblast differentiation. Although the pore size design was different, the result was that the smaller pore size boosted differentiation of osteoblasts and, therefore, was consistent with the results of our study.
Some limitations of this study should be acknowledged. The first limitation relates to manufacturing restrictions. Several factors restrict the pore size of SLM-manufactured objects, including laser spot size, laser power, scanning speed, particle size of Ti metal, and effect of thermal conductivity around the material. Therefore, we are currently unable to investigate the effects of a pore size smaller than 300 μm. In the future, with equipment improvements, it may be possible to manufacture and examine smaller-sized or more precise pores. Second, this study did not detect cell adhesion by quantitative methods, resulting in excessive cells on the pTi that affected SEM differences in cell adhesion after 7 days, which is another deficiency. Third, animal and human in vivo experiments are needed for further investigation; the optimal pore size and porosity for animals and humans under in vivo conditions may be slightly different, thus additional study is warranted.
Conclusions
In conclusion, a pore size and porosity distribution model of pTi controlled by 3D printing was successfully constructed. We have found that the pore size and porosity distribution of pTi influence the proliferation, adhesion, and differentiation of osteoblasts. Notably, pTi facilitates the adhesion and differentiation of osteoblasts when pore size is reduced or porosity is increased. The scaffold model resembles physical modifications with porous structures, which has potential application in the surface modifications of Ti implants.
Footnotes
Acknowledgements
We wish to thank the anonymous reviewers for their helpful remarks, Guo-dong Sun, Wei Li, Kun-xiang Peng and Peng-yu Chen for critical comments, and Ms. Charron of Yale University for revising the manuscript.
Contributorship
WJ (Jin Wo) and HSS (Shi-shu Huang) contributed equally. WJ (Jin Wo) and HSS (Shi-shu Huang) developed the hypothesis, designed experiments, performed the experiments, and contributed to editing the manuscript. WDY (Dong-ying Wu) and ZJ (Jun Zhu) provided administrative, technical, and material support. YF (Feng Yuan) and LZZ (Zhi-zhong Li) supervised the study and helped the manuscript editing and discussions. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of Conflicting Interests
The authors declare no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science Foundation for Key Research and Development Program of Jiangsu (BE2016647), Six Talent Peaks Project of Jiangsu (53031106), Science and Technology Plan Project of Guangzhou (201803010001).
Guarantor
YF (Feng Yuan).
