Abstract
An experiment was performed on oral bacteria removal using the design variables, which included the three-segment rotor speed of the testing device and three types of stainless steel meshes (with different layers). The overall hygienic results showed an effect of up to 95% bacteria removal, and some combinations had 100% hygienic effect. The study proposed that the use of nanobubble generated by a high-density stainless-steel mesh-manufactured nozzle removes dental bacteria. In addition, the device could also be used for auxiliary oral hygiene to decrease the frequency of future medical visits due to periodontal diseases or to enable the device to assist patients with severe periodontal disease more conveniently for oral hygiene.
Introduction
Han and Wang 10 and Pedersen 27 proposed that oral bacteria affect diseases, such as cardiovascular diseases, rheumatic arthritis, stroke, inflammatory bowel diseases, colorectal cancer, respiratory tract infection, pneumonia, and diabetes, as well as adverse pregnancy outcomes 5 . A comparison investigative study on 102 randomly selected community subjects and 100 patients with acute myocardial infarction by Mattila et al. 18 showed that, after eliminating interfering factors, chronic periodontitis could be a risk factor for myocardial infarction. Related studies have revealed that microbiome cultured from the oral cavities of elderly individuals include those related to Enterobacter, Pseudomonas, and Staphylococcus, and these pathogens may weaken the body’s immunity and deteriorate the health status of elderly individuals3,11. On the basis of the studies conducted by Ikeura et al. 12 and Sirsi and Borden 29 , we used nanobubbles as carriers to remove oral bacteria through delivery into the oral cavity. Therefore, the commercially available device MD20 was modified and used as a nanobubble testing device. For this nanobubble generator device, water was transported into the outlet using a motor, and a high-density stainless-steel mesh-manufactured nozzle was used to generate and deliver nanobubbles into a dental tray model mimicking a human oral cavity and teeth (Figure 1). The dental tray was fabricated with soft medical silicone, which could fit the shape of the teeth and prevent bubble water from escaping. The study aimed to reveal the effects of oral bacteria removal using a dental tray-microbubble-generating device with a high-density stainless-steel-manufactured nozzle. The bacteria removal scenarios with different combinations were analyzed to determine the combination that produced the best clearing effects.
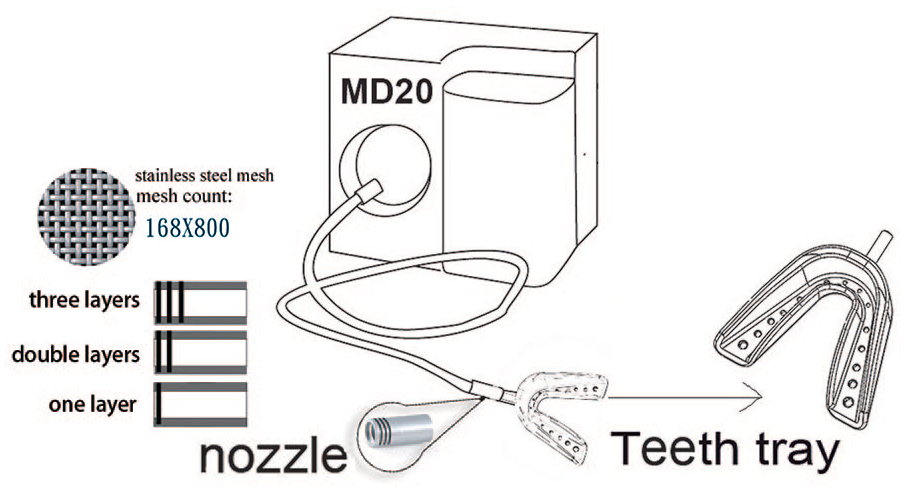
Anterior water outlet measurement of test device M20.
Literature review
There are two methods of gas and liquid kinematics, which affect the formation of bubbles. The first method connects a moving liquid to a gas outlet and forming smaller bubbles through shearing forces7,22. The other method involves a moving liquid that separates bubbles from the gas injection, thus lowering the possibility for bubble coalescence, increasing the bubble size, and influencing the distribution of bubble sizes 24 . The cavitation is further applied to a nozzle design, wherein the speed and volume of the water jet can be modified. The application of this device is pervasive, and its key factor is the water flow outside the nozzle’s water hole outlet13, 14, 33. Miyamoto et al. 20 performed a study on using micro-bubbles with an average diameter of 70 micrometers to remove oil residues on surfaces, which had higher cleaning action than normal bubbles; if combined with detergent with micro-bubbles, this can make absorption of detergent around bubbles more effective, and, therefore, the cleaning effect is more significant. For these applications, a higher separation frequency generates smaller microbubbles and the cross-flow of fluid injected into the gas results in a higher separation speed and generates smaller bubbles6, 16. Ghaemi et al. 8 examined the effect of nozzle length on fluid bubble characteristics and studied the effects of different nozzle lengths on bubble size, shape, location, and speed with the flow velocities of different gases and liquids. The results showed that bubble size, shape, location, and speed significantly varied with nozzle lengths. Therefore, in the present study, three different shapes of nozzles (Figure 1) were manufactured using a high-density stainless-steel mesh to reveal the differences in the bubbles generated.
Several parameters are associated with the characteristics of bubble dynamics, including bubble size, separation frequency, speed, trajectory, and mode of formation. Many calculations and experimental research results are well established, especially regarding the use of imaging techniques for bubble visualization analysis4, 8, 15, 23, 28. The same analysis method was used in the present study to evaluate flow volume, flow velocity, and microbubble size.
Morris et al. 21 and Oliver et al. 25 showed a fairly high incidence of periodontal diseases in adults and indicated that 50% of adults develop gingivitis. In clinical cases, diabetic patients often experience complications related to bacteria that cause periodontal diseases, and therefore, the underlying relationship between diabetes and periodontal diseases is relatively influential. If oral hygiene is not maintained in a diabetic patient, periodontal diseases may occur, and the risk for destructive periodontitis will be relatively higher 23 . The harmful effects of dental plaques on the teeth increase with deposition time. Oral anaerobes are major pathogens of infective oral diseases, such as periodontal diseases, endodontic diseases, and deep oral abscesses 30 . With the advancements in microbiological technology in recent years, several novel bacteria that are yet to be cultured from the oral cavity were found to be related to periodontal diseases, thus demonstrating the complexity and diversity of microorganisms in the oral cavity31, 32.
Albandar and Rams 1 proposed that maintaining oral hygiene decreases the incidence of periodontal diseases. This concept not only requires promotion by related institutions but also translation into common sense education, and awareness regarding oral cavity. For the healthy population, a regular habit of mouth cleaning can maintain the balance between the different oral bacteria, and can result in coexistence with the bacteria in consideration of the antibodies present in the saliva. However, for immunocompromised individuals, oral cavity pathogens may be life threatening. Fever of unknown origins in numerous hospitalized patients is ultimately confirmed to have a great association with oral cavity pathogens. Thus, regular maintenance of oral hygiene is the basis of health in diabetic patients.
Experiment design
The experiment was designed to reveal the effects of oral bacteria removal with nanobubble generated by a nozzle made from a high-density stainless-steel mesh, and to identify the optimal combination using major design variables and intermediate variables of nanobubble. With controlled rotor speed and number of layers of high-density stainless-steel mesh (Figure 1), the present study aimed to identify the combination that can produce the best hygienic effects of oral bacteria removal from false teeth (dentures) using a dental tray model. The removal process was based on the resulting flow volume, flow velocity, and nanobubble size. The experiment for oral bacteria removal was conducted on a laminar flow working bench, with the maxillary portion of denture used for testing. For the experimental procedure, several studies were referenced, including those by Baba et al. 2 and Paranhos et al. 26 . The bacteria collected from the experimental group were smeared onto the culture medium and evaluated 48 hours later at 37°C, and the quantity of bacteria removed after cleaning was calculated. The procedure is based on the process of bacteria sampling and soaking. The bacterial colonies on the tooth surface were sampled with sterilized cotton swabs for analysis.
Test device and variable control
The commercially available bubble oral care device MD20 of which the water path was modified, was used as a test device in the experiment. It created a thin gap between the water supply pipes to mix the air and water that pass through this gap. Due to different specific gravities, air and water generated a flow velocity gradient, and the fluid and gas were ejected through the high-density stainless-steel mesh, which generated very small nanobubbles 9 . In the present study, the number of layers of stainless-steel mesh on the nozzle was modified (Figure 1) using the studies of Legner 17 and Merkle and Deutsch 19 as reference, who conducted different applied research on nanobubbles. All these scholars believed that the diameter of the nozzle was a key factor affecting nanobubbles, and therefore, the number of layers of the stainless-steel mesh was considered as a variable in this experiment. Nanobubble size was affected through the crisscross of multiple-layer and high-density stainless-steel mesh. One of the controllable variables in this study was the three-segment rotor speed. A contact tachometer was used to measure the rotor speed; each segment was measured ten times, and the mean for each segment was considered as the representative value (Table 1).
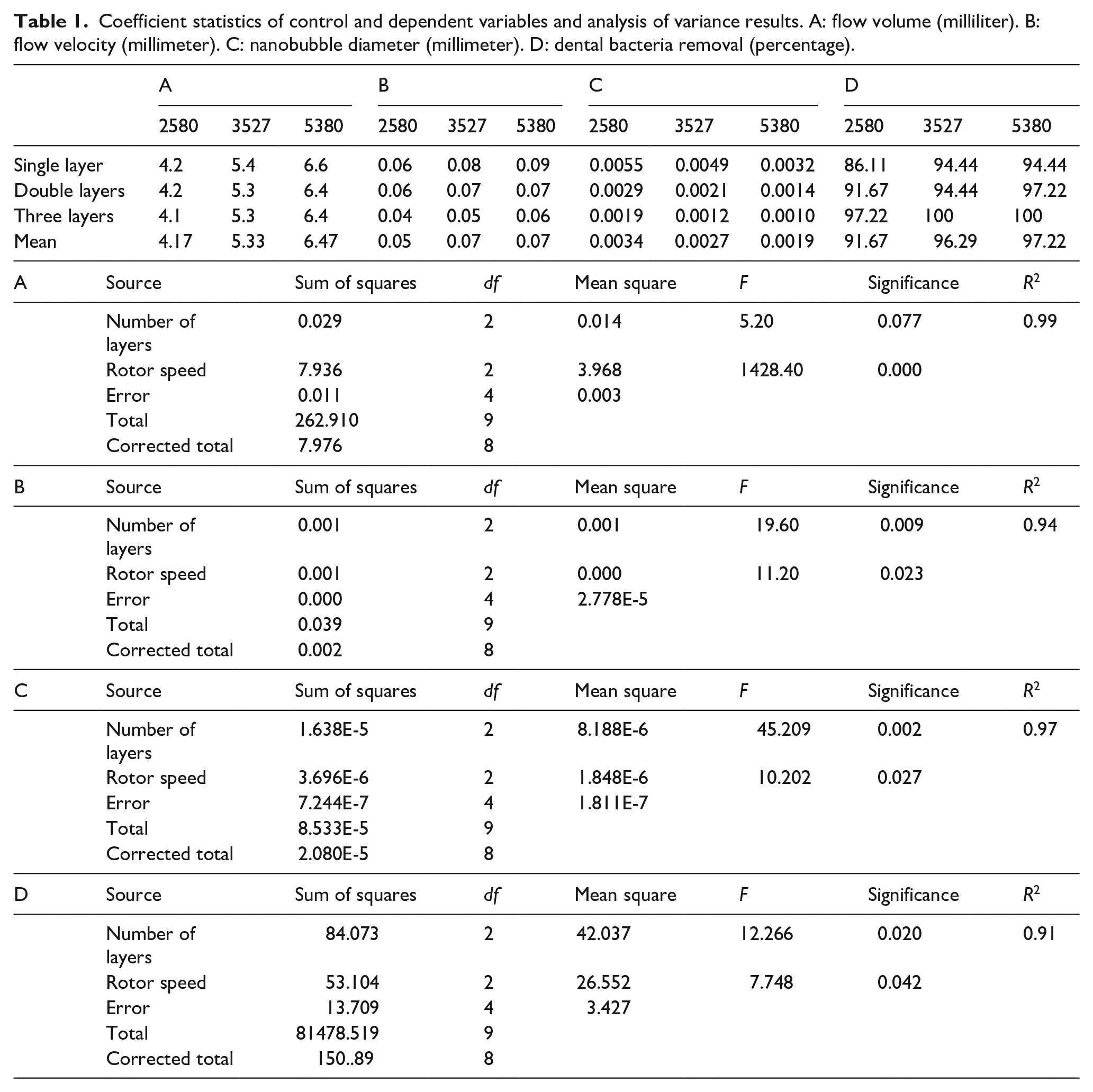
Coefficient statistics of control and dependent variables and analysis of variance results. A: flow volume (milliliter). B: flow velocity (millimeter). C: nanobubble diameter (millimeter). D: dental bacteria removal (percentage).
Computer numerical control machining was used to manufacture the stainless-steel nozzle (length 16 mm, outer diameter 6 mm, and inner diameter 5 mm), and a high-density stainless-steel mesh was combined through processing. Single, double, and triple layers of high-density stainless steel were respectively combined to form three types of nozzles. The mesh count of the high-density stainless-steel mesh was 165 × 800 meshes (provided by the May Chun Company), as shown in Figure 1. In combination with the three rotor speed variables, nine combinations were designed in this study.
Measurement of intermediate variables
First, the flow volume, the square glass tub (200 mm × 200 mm × 200 mm), and a measuring cup were used to collect 10-seconds of total volume, and the unit volume was calculated. The measurement was repeated 10 times, and their means were calculated. A high-speed camera was used to film the generation of the nanobubbles by fixing the test device’s nozzle onto the square glass tub (length/width/height: 200 mm/200 mm/200 mm) with 5 mm glass thickness. Reverse osmosis high-pressure water was used to decrease redundant impurities that could affect the nanobubbles. The square glass tub was filled with water in advance to a height of 10 cm, which was 2 cm higher than the nozzle water outlet. Before switching the test device on, the rotor speed was set, and filming was initiated 3 seconds after the device had been initiated. Filming began 3 seconds after the device was powered because larger bubbles were generated within the first second due to thrust and air effects and disintegrated these large bubbles into smaller bubbles, which scattered into the water. Therefore, the 3-second buffer time allowed the nanobubbles to enter a normal condition. 10-minute intervals must be left between each filming session before starting the next session to minimize the effects caused by residual nanobubbles in the water. During the process, each variable combination was filmed for 1 second (1000 frames/second), which was played back slowly in the software (MS35K DVR 4S). Among 1000 photographs, the 500th photograph of each 100 photographs was selected, and a total of ten pictures were collected. The mean of the bubble diameters in these ten photographs was measured. In addition, the intermediate variable of nanobubble flow velocity was measured using the 500th photograph of the 1st second and the 500th photograph of the 2nd second. The nanobubbles were tracked along the screen, and the selection was based on nanobubble size directly generated from the nozzle. The film was slowly played three times, after which the nanobubble was selected for measurement, and the means were calculated as representative values. After correspondence with actual nozzle size ratio, the distance of the same nanobubble between the 1st second and 2nd second was measured and calculated with the unit of millimeter/s. The results of the three intermediate variable measurements are shown in Table 1.
Materials and methods
A method for identifying bacterial colonies was used to understand the nanobubble generated under each combination variable and their effects on oral bacteria removal. The related procedures of material preparation and equipment required for this experiment were as follows:
(1) Oral bacteria culture procedure: samples were collected from patients with a clinical periodontal disease, and culture was transferred on agar plates. Subsequently, after which the optical density value was measured. In addition, the relationship between adhesive colonies and time was determined in advance by immersing the denture in a bacterial suspension and measuring the adhered bacteria after 15, 30, 60, and 120 minutes of immersion. After repeated testing, it was concluded that 30 minutes was the optimal immersing duration.
(2) The dental tray was manufactured by the dental laboratory using medical silicone. The tray had a hardness of 40°, with holes drilled to accommodate the water jets; a total of 14 holes with increasing sizes were drilled along the edge so that the nanobubbles could stream toward the end of the dental tray. Finally, the tray was immersed in 75% alcohol for sterilization.
(3) The denture used for testing was an ordinary adult’s maxillary denture comprising 14 teeth.
(4) Good manufacturing practice certified central supply department sterilized cotton swab.
Experiment procedure for oral bacteria removal
The test device and the nozzle manufactured using a high-density stainless-steel mesh were connected, with an implicit space at the point of connection of the dental tray. A total of nine experimental groups with different variables and one control group, which did not undergo any cleaning procedures after being immersed in bacteria suspension and taken out to dry for 30 minutes, were evaluated in this study. Sterilized cotton swabs were used for sampling each tooth and were then smeared onto the culture petri dish in order (36 squares were drawn in advance at the back of the petri dish). The procedure for experimental groups was: the adult denture was disinfected in 75% alcohol, held for 5 minutes, and placed in a round container, which comprised 40 mL of oral bacterial solution (5 × 108 cfu/mL) for 30 minutes. Subsequently, the tooth side was faced up, and the denture was transferred into a sterilized round glass plate, where the denture was dried for 60 minutes. The denture was then transferred to another square container (200 mm × 100 mm × 60 mm). A sterilized round acrylic column (68 mm in height) was placed at the center of this container for placement of the denture to ensure that the denture is not overrun by waste water during the cleaning process, and the cleaning results are thus not influenced. Three-minute cleaning proceeded, and the denture was placed on another sterilized round glass plate with the teeth side up. The denture was allowed to dry for 15 minutes before sampling. The surface of each tooth was sampled with a 5-mm sterilized cotton swab. Each tooth had three surfaces, but it was easier to clean the bottom of the four incisors and two canines, given the smaller areas, which would not influence the cleaning effects; therefore, these results were not included. Actual sampling was collected from 36 surfaces (6 × 2 + 8 × 3 = 36). Therefore, sampling for 36 surfaces was performed on each of the nine culture mediums. After sampling, the mediums were placed in the culture box for 24 hours. The experiment procedure is shown in Figure 3. The cleaning effects are presented as the percentage of sterile surfaces, that is, if 30 squares presented with no dental bacteria, this indicates that the surfaces of 30 teeth could be cleaned thoroughly, and the cleaning effect would be 30/36 × 100 = 83.33%. The cleaning effects for each experimental group are summarized in Table 1-D. In addition to overall cleaning effect analysis, the study also divided the dentures into five regions as revealed in Figure 2 and conducted cleaning effect analysis accordingly. Three such groups were formed with eight surfaces each (buccal surface (premolars (PM)-BS)), occlusal surface (PM-OS), and lingual surface (PM-LI) of PM and molars) and two groups with six surfaces each (labial surface (IC-LA) and lingual surface (IC-LI) of incisors and canines). During the course of the experiment, the temperature of the bacterial console was controlled to prevent temperature changes in bacteria solutions from affecting the overall bacteria solution volume.

Experimental procedure of denture cleaning.

Tooth surface sampling regions after denture cleaning.
Analysis and discussion
Effects of controlling variables on intermediate variables and oral bacteria removal
Table 1-A–C summarizes the measurement results for flow volume, flow velocity, and the diameter of nanobubble generated by the combinations of the three-segment rotor speed for the nanobubble generator (2580, 3527, and 5380 revolutions per minute (rpm)) and the three stainless steel nozzles with different number of mesh layers. The coefficients in Table 1 indicated that flow volume was elevated with an increase in rotor speed and was less affected by an increase in number of mesh layers. The rotor speed and layers of mesh for the high-density stainless-steel nozzle were considered as independent variables and flow volume as dependent variables in the analysis. Analysis of variance (ANOVA) was conducted; the results implied that rotor speed had direct significance for flow volume (0.00 < 0.05; Table 1-A; p < 0.05 was considered statistically significant) and that the number of layers of mesh had less effect on flow volume.
The effects of controlling variables on flow velocity are summarized in Table 1-B. The flow velocity of the nanobubbles increased with an increase in the rotor speed and layers of mesh for the high-density stainless-steel nozzle. ANOVA results showed that variables controlled had reached significant effect on the jet flow velocity, both p < 0.05.
Table 1-C presents the results of nanobubble diameter measurement. The higher rotor speed and more layers of stainless steel mesh resulted in a smaller diameter of nanobubble. Furthermore, the ANOVA results also showed that the rotor speed was significantly (p = 0.046 < 0.05) related to the microtubule diameter.
The effects of control variables on bacteria removal
The coefficients in Table 1-D summarize the effects of design variables on the efficacy of oral bacteria removal. A high rotor speed and more layers of stainless-steel mesh resulted in a better bacteria removal effect. Some of the variable combination was able to achieve a 100% bacteria removal effect. Fewer number of layers combined with a low rotor speed was only able to achieve a 91% bacteria removal effect. ANOVA revealed that significant both high rotor speed and higher number of layers of mesh were effective for bacteria removal (p < 0.05).
Effects of intermediate variables on bacteria removal
Table 2 presents the results of regression analysis using flow volume, flow velocity, and nanobubble diameter as independent variables and bacteria removal percentage as the dependent variable. The nanobubble diameter was a factor associated with bacteria removal, and flow volume and flow velocity did not significantly affect bacteria removal.
Regression analysis for the influences of intermediate variables on bacteria removal effect.

Oral bacteria removal conditions according to denture regions
In addition to the overall analysis of the efficacy of bacteria removal, the dentures were divided into five regions as shown in Figure 2, namely the PM-BS, PM-OS, and PM-LI of PMs and molars, the labial surface (IC-LA), and the lingual surface (IC-LI) of incisors and canines. A further analysis was conducted on the cleaning effect percentage of each region, as shown in Table 3; areas A–E, respectively, reflect the results of bacteria removal for each region. A three-layers stainless-steel mesh nozzle was combined with 3527 and 5380 rpm rotor speeds, and a 100% cleaning effect was obtained after cleaning. Conversely, single- and double-layered stainless-steel mesh nozzles with a lower rotor speed achieved poorer bacteria removal effects (86% and 91% bacteria removal effect). Regarding the regional cleaning effect on bacteria removal surface, region E was found to be completely cleaned: region E was IC-LI, the lingual surface of incisors and canines (6 total surfaces). Region D (IC-LA) the labial surface of incisors and two canines (a total of six surfaces), had a tooth surface that was not completely cleaned with the combination of a single-layered stainless-steel mesh nozzle and a rotor speed of 5380 rpm. Regarding the regional cleaning effect on bacteria removal surface, region B was PM-OS, the occlusion surface of PMs and molars (8 total surfaces), and regions C (PM-LI), both had multiple tooth surfaces that were not completely cleaned. Region A (PM-BS) had all tooth surfaces completely cleaned in the combination of three-layered stainless-steel mesh nozzles and rotor speeds of 3527 and 5380 rpm.
Number of surfaces related to bacteria removal for different denture regions: a stepwise regression analysis of influence of intermediate variables on denture regions (A: PM-BS, B: PM-OS, C: PM-LI, D: IC-LA, E: IC-LI).

Note: PM were regions of premolars and molars; BS: buccal surface; LI: lingual surface.
In the present study, a regression analysis was performed on the A–E regions, and we found that the intermediate variables affected region C, with results shown in Table 3. Furthermore, Table 3 clearly shows that the oral bacteria removal of both the regions was significantly affected by nanobubble diameter.
Conclusion
The present study used a high-density stainless-steel mesh to manufacture nozzles using designs of single, double, and triple layers. This was used in combination with a dental tray made of medical silicone, and nanobubble generated by the motor rotation of a nanobubble generator to perform cleaning experiments on oral bacteria in dentures. Overall, the results showed a 95% bacteria removal effect. Particularly, under design conditions of 3527 rpm and 5380 rpm rotor speeds with the triple-layered stainless-steel mesh, a 100% bacteria removal effect could be achieved. The double- and triple-layered stainless-steel mesh could also achieve a 97% bacteria removal effect with rotor speeds of 5380 rpm and 2580 rpm. A poorer bacteria removal effect also achieved 86%. The flow volume was only affected by the rotor speed, and this is in line with the common concept that the faster the rotor speed, the greater the flow volume. The flow velocity was affected by the two variables. The nanobubble diameter was both influenced by the number of layers of stainless-steel mesh and rotor speed, indicating that more layers of mesh or a higher rotor speed resulted in smaller nanobubble diameters. Among the two control variables, the rotor speed significantly affected the oral bacteria removal. However, nanobubble diameter could better affect the bacteria removal effect of these two regions. Overall, better effects of oral bacteria removal can be achieved by increasing the rotor speed. Rotor speed had more significant effects on flow volume, flow velocity, nanobubble diameter, and bacteria removal. The efficacy of the intermediate variables on bacteria removal was nanobubble diameter size. PM-BS, PM-OS, and PM-LI were regions of PMs and molars that were the most important occlusal areas of the human body. Only IC-LI was able to achieve a complete bacteria removal effect under any design conditions, whereas the low rotor speed was unable to achieve a complete bacteria removal. However, some combinations could not still achieve complete bacteria removal effect, possibly because of an insufficient control of the variable range; we aim to clarify this in the next stage of our study. The present study used a high-density stainless-steel mesh to create nanobubble, assuming that the nanobubbles were able to clean the deep oral pockets and adjacent surfaces of teeth and achieve complete oral bacteria removal. In this study, the cleaning method was used with a dental tray model, and dentures were regarded as study subjects to replace the human oral cavity. The bacteria culturing method was applied to conduct preliminary verification of dental bacteria cleaning, and thus this method is expected to be used in future studies on actual human oral cavities. This correlation is anticipated to be clarified, which may reduce the complications incurred by dental hygiene methods for long-term bedridden or post-operative patients.
