Abstract
The doping of silicon (Si) has been proved to improve the bioactivity of Ca-P ceramics. In light of this thinking, in the present study, Ca-P coatings with La2O3 by addition of 10 wt% SiO2 and 10 wt% diatomaceous earth (DE) were fabricated by laser cladding on Ti6Al4V, respectively. Coating doped without Si was also fabricated as the comparison group for the experiment. The effect of two different Si sources on the surface morphology, microstructure, microhardness, and bioactivity was systematically studied. The experimental results show that the Si-doped coating is of rough surface morphology, and the addition of DE significantly reduces the number of cracks and improves the microhardness. The X-ray diffraction results reveal that the amount of bioactive phase tricalcium-phosphate (TCP) and hydroxyapatite (HA) reaches maximum in the DE-doped coating. After soaking in simulated body fluid (SBF), the precipitate of bone-like apatite in the DE-doped coating is significantly higher than that of the other coatings.
Introduction
Recently, the doping of silicon (Si) into calcium-phosphate (Ca-P) coatings has been extensively investigated for biomedical applications, particularly for bone regeneration. Bioceramic doping of small concentrations of Si has been demonstrating to improve the bone extracellular matrix production and the osteoblast proliferation. 1 Si-doped coatings could easily form a Si-OH functional group in body fluids and generate a more electronegative surface, improving the formation of bone-like apatites.2,3 Hench and Paschall 4 reported that the incorporation of SiO2 enhanced the strength of a hydroxyapatite (HA) coating by promoting the crack deflection or crack arrest.
SiO2 can be obtained by synthesis, and it is often found in nature. As far as biogenic sources are concerned, diatomaceous earth (DE) is the largest source of biogenic amorphous Si and the most abundant Si on Earth.5,6 It is composed of fossilized 10–150 µm diatom shells, which are rich in macropores; the size of the macropores extends from the micrometric to the nanometric domain, as well as containing a few mesopores and micropores. 7 Since DE is of the unique structure of diatom shells, coupled with high SiO2 content, it has a low density, high specific surface area, and good adsorption. 8 This kind of Si source has drawn wide attention because of its inexpensive cost. In addition, DE contains essential ions for the human body, such as Na, Mg, Al, K, and Fe, whereas pure SiO2 does not contain the essential ions mentioned above. 9 It is already proved that DE-doped coating significantly favors osteoblast proliferation and activity in comparison with the coating doping only SiO2 due to the presence of minority impurity elements in the coating. 1
Si-doped bioceramic coatings have recently been fabricated by techniques such as plasma spraying, 10 magnetron sputtering, 11 pulsed laser deposition, 12 and laser cladding.13,14 Of these methods, laser cladding shows broad prospects in the biomedical field owing to the high deposition rate and flexibility of the process. Moreover, it improves the bioactivity and biocompatibility of the tissue–implant interface due to its ability to achieve higher wettability of the implant surface by controlling the laser processing parameters.15,16
Rare earths are often used as effective additives to improve the microstructure of coatings. 17 On the other hand, rare earth elements, especially rare earth oxide as a catalyst, has characteristics such as high activity, low cost, and good selectiveness. 18 Lanthanum (La), as a rare earth element, has neither side effects nor it is noxious in humans under the use of the appropriate amount. In our previous studies, La2O3-doped Ca-P coating was fabricated by using the method of laser cladding on Ti alloy.18–22 Bioactive phases tricalcium-phosphate (TCP) and HA were obviously catalyzed by La2O3 during laser cladding process, while there is almost no TCP and HA in the coating doped without La2O3. In the electronic structure of the La atom the D5 rail is empty, which provides an electron transfer orbit and can be used as an electron transfer catalysis “station” so that La2O3 has high catalytic activity. A small amount of La2O3 reacts with oxygen, calcium, and other elements to form high melting point compounds, some of which can be used as nucleation cores to increases the nucleation rate of TCP and HA. 22 The amount of TCP and HA reached maximum in coatings with a Ca/P ratio of 1.4 when the content of La2O3 is 0.6 wt%; at the same time, the coating is of best bioactivity and biocompatibility. 22
In present paper, Ca-P coating containing 0.6 wt% La2O3 by addition of two different Si sources (DE and SiO2) was fabricated by laser cladding on Ti6Al4V. The comparative study for the effect of the two types of coatings on the surface morphology, microstructure, microhardness, and bioactivity was systematically analyzed and discussed.
Materials and methods
Materials
On one hand, HA powder is expensive, so it is unrealistic for it to be widely used in production. On the other hand, bioceramic coating with pure HA cannot be obtained by laser cladding because of the decomposition of HA under high temperature. For this reason, a certain proportion of CaHPO4 • 2H2O and CaCO3 could be pre-coated on the substrate, which can obtain HA cheaply and efficiently by laser cladding.19,23 The coating materials were Ti powder (45–50 μm, AR), CaHPO4·2H2O, CaCO3, La2O3, SiO2 (1–5 μm, AR), and DE powders (90.62 wt% SiO2, 3.53 wt% Fe2O3, 2.52 wt% Na2O, 1.64 wt% Al2O3, K2O, MgO, CaO, P2O5, AR). The powders were purchased from Kermel Chemical Reagents (Tianjin, China). Medical titanium alloy (Ti6Al4V) was adopted as the substrate (purchased from the Northwest Institute for Non-ferrous Metal Research (Shanxi, China)).
Preparation of Ca-P coatings
A TFL-H10000 10 KW CO2 laser instrument was used in this experiment. Since there is a great difference between the line expansion coefficient substrate and coating powders (CaHPO4·2H2O and CaCO3), there exists larger thermal stress between substrate and coating in the process of solidification after laser cladding, which further causes cracks in the coating, leading to a decrease in bonding strength. 19 Considering that the linear expansion coefficient of Ti powders is 8.5 × 10−6 k−1, which is very close to Ti6Al4V (8.8 × 10−6 k−1), Ti powders could be gradiently doped in coating powders and the method of gradient design was adopted.24,25 The ratio of Ca/P of hydroxyapatite is 1.67 under a normal formula. Due to the loss of P during laser irradiation, the atomic ratio of Ca/P in precursor powders was designed to be 1.4. The gradient composition design is given in Table 1.
Composition of three types of gradient coatings.

DE: diatomaceous earth.
Firstly, a first gradient coating was coated on Ti6Al4V; after cleaning the samples as cladded, the second gradient layer was cladded. Similarly, the third gradient layer was treated finally. The optimized technological parameters are as follows: output of power P = 1.8 kW; laser beam size D = 4 mm; scanning velocity V = 240 mm·min−1. The schematic presentation of the laser cladding process is presented in Figure 1. Figure 2 shows the macroscopic morphology of the sample surface after laser cladding. The cladding coatings were cut into 10 mm × 10 mm × 3 mm and then cleaned by petroleum ether, acetone, alcohol, and distilled water for 15 min successively.

Diagrammatic sketch of laser cladding.

Macroscopic micrographs of the surface of (a) coating doped without Si source, (b) SiO2-doped coating, and (c) diatomaceous earth-doped coating.
Surface morphology
The surface morphology of Si-doped (SiO2 and DE) coatings and coating doped only with La2O3 was observed using scanning electron microscopy (SEM) (SUPRA-40, Carl Zeiss AG, Germany).
X-ray diffraction
X-ray diffraction (XRD, X’Pert Powder PANalytical B.V., Netherlands) was employed to analyze the phase structure of the coatings. In the phase analysis, the radiation source was CuKα.
Cross-section morphology
To study the effect of different Si sources on the crack susceptibility of gradient coatings, the cross-section of samples was ground using metallographic sandpaper then polished. The cross-section morphology of coatings was characterized by an optical microscope (OM, OLYMPUS GX51).
Microhardness
Upon grinding the cross-section after cutting to remove scale, the microhardness of the gradient coating was determined with a microhardness tester (DHV-1000Z, Shuangxu Electronics Co., Ltd, Shanghai, China). The measurement of the microhardness was performed sequentially from ceramic layer to the substrate at a distance of 0.2 mm. The experiment was replicated three times.
Bioactivity
Samples were immersed in simulated body fluid (SBF) (5 ml) for 7 and 14 d to assess their formation ability of the initial apatite of the coating. The SBF solution was prepared by the method proposed by Kokubo and Takadama. 26 The SBF solution was changed once a day to keep the composition of the solution unchanged. In vitro bioactivity tests were performed at 37°C. At selected times of immersion (7 and 14 d), the samples were taken out, gently washed with deionized water, and dried in a drying oven. The surface of the coating was observed by SEM and the elemental composition was investigated by energy-dispersive X-ray spectroscopy (EDS). The phase analysis of coatings before and after soaking was characterized by an X-ray analyzer.
Results and discussion
Surface morphology of the coatings
Figure 2 shows the macroscopic morphology of the sample surface. It can be seen that the coating surface is corrugated, and corrugations are approximately the same pitch. This corrugation feature indicates the fluidity of liquid in the molten pool during laser cladding and the periodic changes in the melting process. The surface morphology of the coating given in Figure 3 shows that all bioceramic coatings are of a unique coral reef structure after laser cladding, which would provide a channel for bone tissues to grow into coatings. The bioactivity of biomedical materials is not only related to the chemical composition, but also to the surface morphology. 16 There is evidence that if materials with smooth surface are implanted, a thick layer of cystic tissue composed of fibroblasts arranged in parallel is formed around the contacted bone tissue without binding to the material, which tends to trigger inflammation and tumorigenesis, while materials with relatively rough surfaces induce preferentially osteoblast and epithelial cells.27,28

Scanning electron microscopy micrographs of the surface of (a) coating doped without Si source, (b) SiO2-doped coating, and (c) diatomaceous earth-doped coating.
XRD analysis
The XRD pattern given in Figure 4 shows that the phases of the coating undoped Si are mainly composed of CaTiO3, TCP, HA, and TiO2. The characteristic diffraction peak corresponding to Ca2SiO4 occurred in coatings with two different sources of Si. The characteristic peaks of bioactive phase TCP and HA exists at 2θ around 31–33°. From XRD, compared with coating doped only with La2O3, the characteristic peaks of TCP at 2θ ~31° (corresponding to the plane (0 2 10)) and HA at 2θ ~32.9° (corresponding to the plane (3 0 0)) increase after doping SiO2 and DE, and the peak in DE-doped coating is more obvious than that of SiO2-doped coating.

X-ray diffraction patterns of Ca-P coatings. The coating doped without a Si source is considered as the comparison group. HA: hydroxyapatite; TCP: tricalcium-phosphate; DE: diatomaceous earth.
The reason why the increase of characteristic peaks of bioactive phases with the addition of Si can be explained based on the work by Yoo et al. 29 The authors reported that the doping of an appropriate amount of Si can accelerate the transfer of the material and result in faster microstructural evolution. 29 Hence, it can be speculated that the addition of an appropriate amount of Si could accelerate the transfer of reactive particles during laser cladding and therefore promote the flow of the molten pool, which could promote the catalyze synthesis of TCP and HA in the molten pool. On the other hand, some Si doping could be used as nucleation cores to accelerate the nucleation of TCP and HA, so the characteristic peaks in the XRD pattern increase.
The possible reactions between CaHPO4·2H2O and CaCO3 under laser cladding could be characterized as follows 23
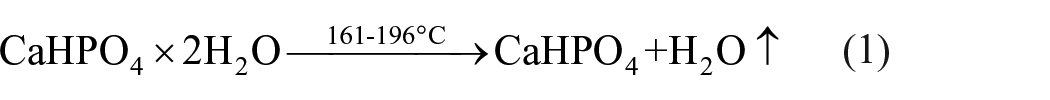




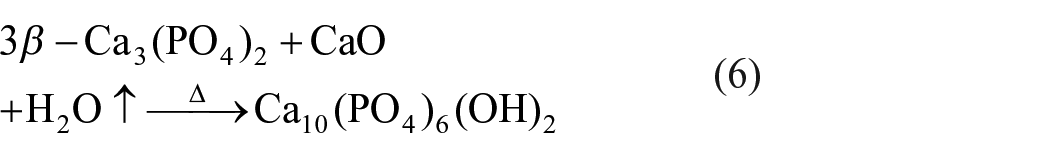
Because DE is of good absorptivity, there would be more H2O↑ absorbed on DE-doped coating in comparison with other two types of coatings. When more H2O↑ is absorbed on surface, the process of Equation (6) is accelerated, promoting the formation of HA.
Cross-section analysis and effect of different Si sources on the crack susceptibility of gradient coatings
Our previous study has demonstrated that the rare earth/Ca-P gradient coating after laser cladding is comprised of a ceramic layer, an alloying layer, and substrate. 19 Figure 5(a) shows the cross-sectional morphology of the gradient coating (DE-doped coating). It is worth mentioning that the existence of the alloying layer in this kind of gradient coating is of important biomedical function, which could effectively prevent harmful ions in the substrate, such as Al and V, from invading tissues or organs. 20

(a) Cross-sectional morphology of diatomaceous earth (DE)-doped coating after laser cladding. The bonding interface between the ceramic layer and alloying layer of (b) coating doped without a Si source, (c) SiO2-doped coating, and (d) DE-doped coating.
Gradient biocoatings inevitably contain holes and cracks because laser cladding is a process of rapid melting and solidification.13,14 The bonding interface between the ceramic layer and alloying layer is shown in Figures 5(b)–(d). Since the ceramic layer in the coating doped without Si has large cracks, resulting in a deteriorative microstructure, it easily falls off the substrate(Figure 5(a)). Cracks in the Si-doped coating are significantly reduced. It is interesting to note that there exist only tiny cracks (vertical to the surface) and holes in the coating doped with DE (Figure 5(d)), which could improve the comprehensive properties of coating. In addition, a favorable environment for the growth of new bones would be provided by tiny cracks. 30 It takes less time for new bones to fill smaller cracks than larger cracks. 30 Overall, the doping of Si significantly improves the crack susceptibility of gradient coatings. In connection with this result, a past research by Yang et al. 2 reported that SiO2 addition could not only develop the grain boundaries and reduce the grain size, but also could suppress the cracks formation in the coating. Based on this research, our experimental results further illustrate that DE addition shows better effects on reducing the presence of cracks across the cross-section. The reason might be that the existence of some minority compounds (for example, MgO) could refine the grain size, which makes the coating denser and more uniform.
Microhardness analysis
The microhardness curves of the Si-doped coatings and coating doped without Si are shown in Figure 6. It is seen that the average microhardness of the coating is increased by doping 10 wt% SiO2 as well as 10 wt% DE, and the microhardness is more evenly distributed at distance from the surface to the substrate. In particular, the microhardness of the ceramic layer reaches the maximum after adding DE.

Microhardness analysis of the Ca-P coatings; the coating doped without a Si source is considered as the comparison group. DE: diatomaceous earth.
Si tends to inhibit the grain growth to generate materials with fine structures.31,32 In the work by Yang et al., 2 it was reported that in coatings produced by laser direct melting of Ca-P and SiO2/Ca-P precursors on Ti6Al4V, the presence of SiO2 in the precursor led to the formation of a smaller average crystallite size. The doping of synthetic SiO2 and a biological source of SiO2 (DE) suppresses grain growth, resulting in a dense microstructure and thereby improving the microhardness of the Si-doped coating. On the other hand, defects such as contraction cavities and cracks reduce the microhardness. 22 As mentioned above, the doping of DE reduces the initiation of cracks and, thus, the coating density is improved so that hardness of the coating is improved.
Bioactivity
When the composite coating is implanted into the human body, first and the foremost, the material is wetted by physiological fluid. 2 This further determines the activation of cells and stimulation of new bone tissue development. The in vitro bioactivity of coatings is assessed by the precipitation of an apatite-like mineral layer after soaking in SBF. HA is a natural mineral component of the human bone; this layer could form a chemical bond with human bone tissue. 15
Figure 7 shows a SEM analysis of the surface of coatings doped with 10 wt% DE, 10 wt% SiO2, and without a Si source. After 7 d of immersion (Figures 7(a)–(c)), bone-like apatite deposits are found on all bioceramic coatings. The deposits are chrysanthemum-like particles. Bone-like apatite with the maximum as well as largest size is covered on DE-doped coating (Figure 7(c)). As the soaking time increases, the surface is completely covered by a layer of bone-like apatite. After 14 d of soaking (Figures 7(d)–(f)), precipitates covered on the Si-doped and DE-doped coatings warp locally due to continuously increasing amounts of bone-like apatite; in particular, the DE-doped coating warps heavenly. The EDS analysis in site A shows that precipitates are phases in enriched calcium and phosphorus, and oxygen and carbon is also contained (Figure 8).
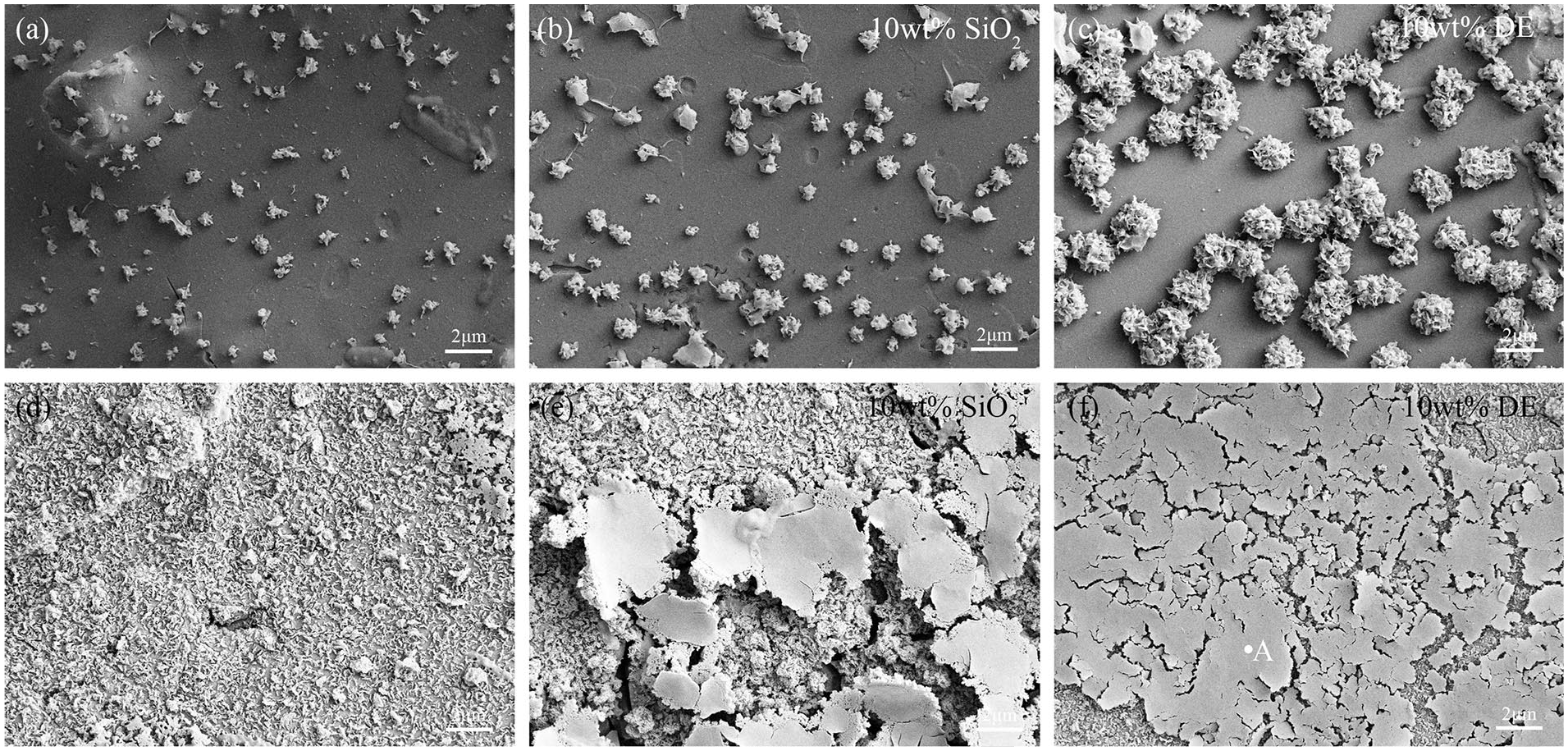
Surface micrographs of (a) coating doped without a Si source, (b) SiO2-doped coating, and (c) diatomaceous earth (DE)-doped coating after soaking for 7 d in SBF; (d) coating doped without a Si source, (e) SiO2-doped coating, and (f) DE-doped coating after soaking for 14 d in SBF. SBF: simulated body fluid.
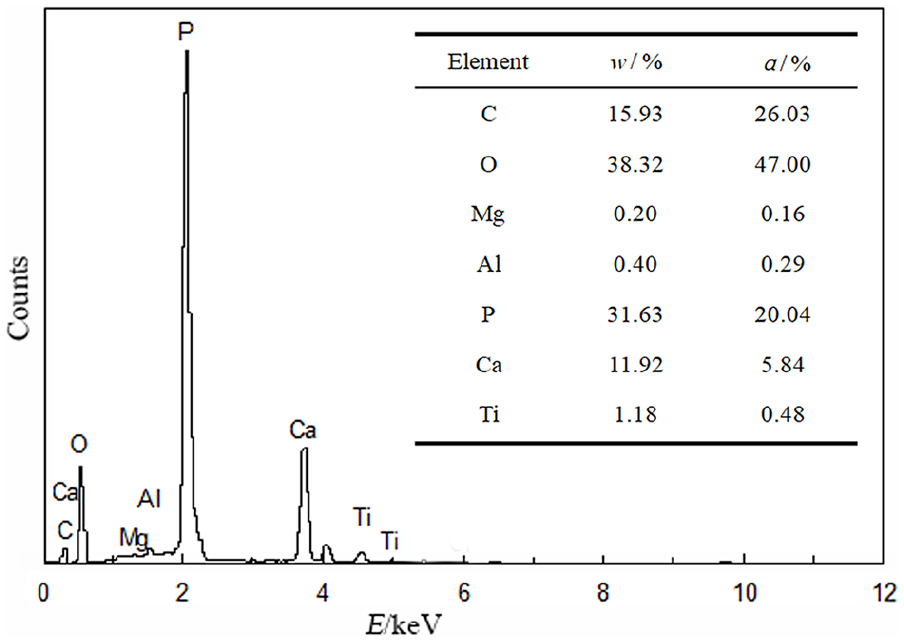
Energy spectra analysis of bone-like apatite on coating with 10 wt% diatomaceous earth.
Compared with the XRD pattern before SBF soaking, it is observed that bone-like apatite covers all samples after 7 and 14 d of soaking in SBF (Figure 9). After 7 d of immersion, for all coatings, the characteristic diffraction peak corresponding to HA becomes high. The characteristic peak of HA on the DE-doped coating is the highest compared with coating doped without Si after 14 d of immersion, which indicates that the amount of HA synthesized reaches maximum. The new phase Ca2SiO4 is synthesized in coatings with two different sources of Si during laser cladding. The work by Gou and Chang
33
reported that Ca2SiO4 possessed great bioactivity in the SBF environment due to the improved wettability of the coating. As mentioned with regard to the XRD analysis, the characteristic peaks of TCP are highest in the DE-doped coating. Indeed, TCP is an excellent degradable phase; when TCP degrades, it release Ca2+ and

X-ray diffraction patterns of different bioceramic coatings before (0 h) and after (7 and 14 d) immersion in SBF.
Compared with the coating doped only with SiO2, a higher content of minority elements, such as Na, Al, K, and Mg, is found in the DE-doped coating. The work by Solla et al. 9 reported that the Si–HA coatings grown by pulsed laser deposition using DE as a Si source presented more similar composition to the HA found in natural bone due to the existence of a mixture of different ions. López-Álvarez et al. 1 found that the Si–HA coating from DE grown by PLD significantly favored biocompatibility in comparison to the Si–HA coating from SiO2, due to minority compounds. In this study, it is confirmed that the DE-doped coating can deposit more bone-like apatite and therefore possesses better bioactivity. To summarize, the DE-doped coating is of better bioactivity and biocompatibility. This biomaterial is more suitable to be used as a potent bone substitute for clinical applications than the SiO2-doped coating.
Conclusions
The gradient Ca-P bioceramic coatings with La2O3 by addition of SiO2 and DE are fabricated by laser cladding on Ti6Al4V. XRD results show that the characteristic diffraction peak of bioactive phase TCP and HA is higher in Si-doped coatings; in particular, the peak of TCP and HA is highest in the DE-doped coating. The crack susceptibility of gradient coatings is deeply impacted by SiO2 and DE; when SiO2 and DE is added, the number of cracks is greatly reduced. At the same time, the microhardness is also significantly improved due to the density of the coating, and the DE-doped coating is of the best microstructure and microhardness. After immersion in SBF, the bone-like apatite layer is formed on the coating. The DE-doped coating has greater apatite precipitation than the other two types of coatings, exhibiting the best bioactivity.
Footnotes
Declaration of conflicting interests
The authors have no conflicts of interest to declare.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the NSFC (51663004) and the High-level Innovative Talents Plan of Guizhou Province (2015-4009).
