Abstract
The success of hyaluronic acid (HA)-based dermal fillers, with more than 2 million minimally invasive procedures conducted in 2016 in the US alone, is due to their hygroscopic properties of biocompatibility and reversibility. The type and density of HA cross-linkage, as well as the manufacturing technology, may influence not only the in vivo persistence but also the safety profile of dermal fillers. 1,4-Butanediol diglycidyl ether (BDDE) is the cross-linker used in most market-leading HA fillers; 1,4-butanediol di-(propan-2,3-diolyl) ether (BDPE) is the major impurity obtained from the HA–BDDE cross-linking (HBC) process. In this work, a new process to obtain high purity HBC fillers was developed. A new HPLC-MS method was validated for the quantification of BDPE content in HBC dermal fillers. In vitro cytotoxicity of BDPE was evaluated in fibroblasts (IC50 = 0.48 mg/mL). The viscoelasticity was monitored during the shelf-life of the HBC-10% hydrogel and was correlated with in vitro hyaluronidase resistance and in vivo residence time in a rabbit model. This analysis showed that elasticity is the best parameter to predict the in vivo residence time. Finally, a series of parameters were investigated in certain marketed dermal fillers and were compared with the results of the HBC-10% hydrogel.
Introduction
Injectable dermal fillers are space-filling gels and are available globally for replacing the hydrodynamic volume of the extracellular matrix, thus reducing the clinical effect of aging. 1 In 2016, 76.9% of all injectable Food and Drug Administration (FDA)-approved dermal fillers in the US were based on hyaluronic acid (HA), 2 due to its hygroscopic properties of biocompatibility and reversibility, with more than 2 million minimally invasive procedures having been conducted. 3 HA used in dermal fillers is commonly cross-linked to improve the in vivo residence time from 6 to 18 months. 4
Although HA-based dermal fillers have a low overall incidence of long-term side effects, occasional adverse outcomes such as foreign-body-type granulomatous reaction (incidence ranges from 0.02% to 0.4%) have been documented.5,6 Factors such as injection volume, process impurities, and physical properties of the filler affect granuloma formation. 7 The unreacted cross-linking agents or their byproducts are often toxic compounds, which should be extracted from the gels before they are applied. 8
The most commonly used cross-linker in HA dermal fillers on the US market is 1,4-butanediol diglycidyl ether (BDDE),
9
where the cross-linking reaction occurs between the diepoxide and two primary hydroxyl groups derived from different residues of
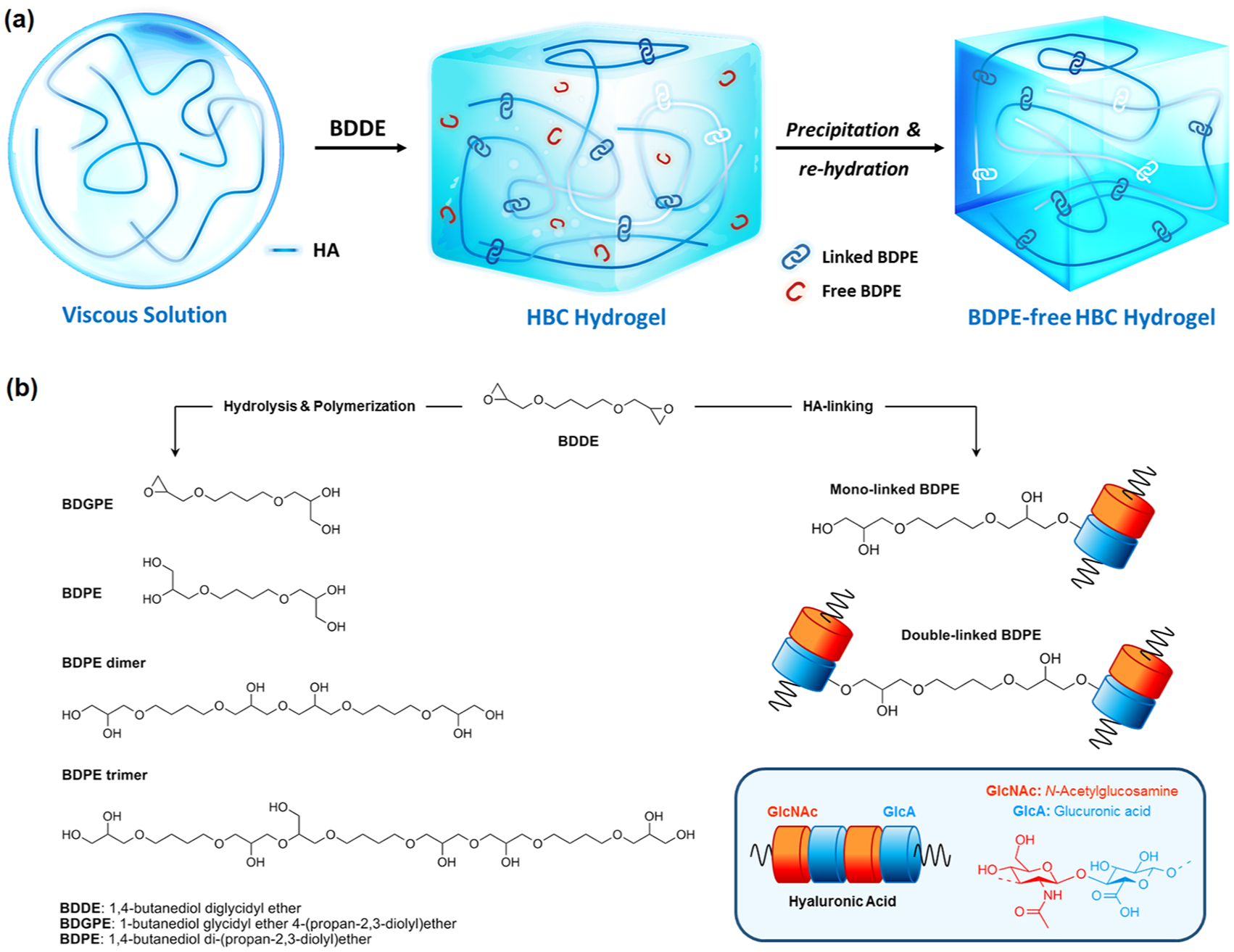
(a) Scheme of HBC synthesis and purification. (b) Chemical structures of BDDE, partially or totally hydrolyzed BDDE (BDGE and BDPE respectively), polymerized BDDE (BDPE dimer and trimer) and BDPE mono- or double-linked to HA.
Considering the results of a systematic review of the available literature on HA-based dermal fillers cross-linked with BDDE,10,11 it is highlighted that BDPE content is not described in the literature. In this study, various downstream procedures for BDPE removal were evaluated. For the first time, a high performance liquid chromatography (HPLC)-mass spectrometry (MS) method was developed to quantify the BDPE content inside the dermal fillers. Cytotoxicity of BDPE and HBC at different MoD were determined in human fibroblasts. A dermal filler prototype was characterized and monitored for elasticity throughout the product shelf-life, and the elasticity was correlated with (i) in vitro resistance towards hyaluronidase degradation and (ii) in vivo residence time in rabbits. Finally, MoD, BDPE content, viscoelastic properties, and hyaluronidase stability were assessed for certain marketed dermal fillers with similar shelf-life and were compared with the properties of our new prototype.
Materials
Hyaluronic acid sodium salt (HA) and auto cross-linked polymer (ACP) sodium salt were provided by Fidia Farmaceutici S.p.A (Abano Terme, Italy). Bovine testes hyaluronidase (BTH; EC 3.2.1.35) was purchased from the European Directorate for the Quality of Medicines & HealthCare (EDQM, Strasbourg, France). The marketed fillers were purchased from different suppliers: Belotero Intense (Merz, Frankfurt, Germany), Restylane Kysse (Galderma, Uppsala, Sweden), Juvéderm Voluma (Allergan, Pringy, France) Teosyal Ultra Deep (Teoxane, Geneva, Switzerland). All the other reagents were supplied by Sigma-Aldrich (St Louis, MO, US) and were used without further purification.
Synthesis and downstream of BDDE cross-linked HA (HBC)
HA–BDDE cross-linked polymers (HBC) were synthesized from 700 kDa HA as previously reported. 12 Three HBC batches were prepared at the nominal BDDE derivatization degree of 10%, 15%, and 30% mol (vs HA r.u.). Each batch was split into three sub-batches (15 mL each): the first sub-batch was directly diluted to the final concentration of 25 mg/mL in phosphate-buffered saline (PBS; pH = 7) and was used as it was; the second sub-batch was dialyzed (dialysis membrane: Biotech RC Tubing, MWCO: 8–10 kDa) against PBS and then diluted to 25 mg/mL; the third sub-batch was precipitated with 40 mL of ethanol, washed three times in aqueous alcoholic solution (EtOH/H2O: 8/2), dried and rehydrated in PBS at 25 mg/mL. Finally, each HBC hydrogel was sterilized by steam for 15 min at 121°C in agreement with ISO 17665 “Steam Sterilization for Medical Devices.”
MoD analysis
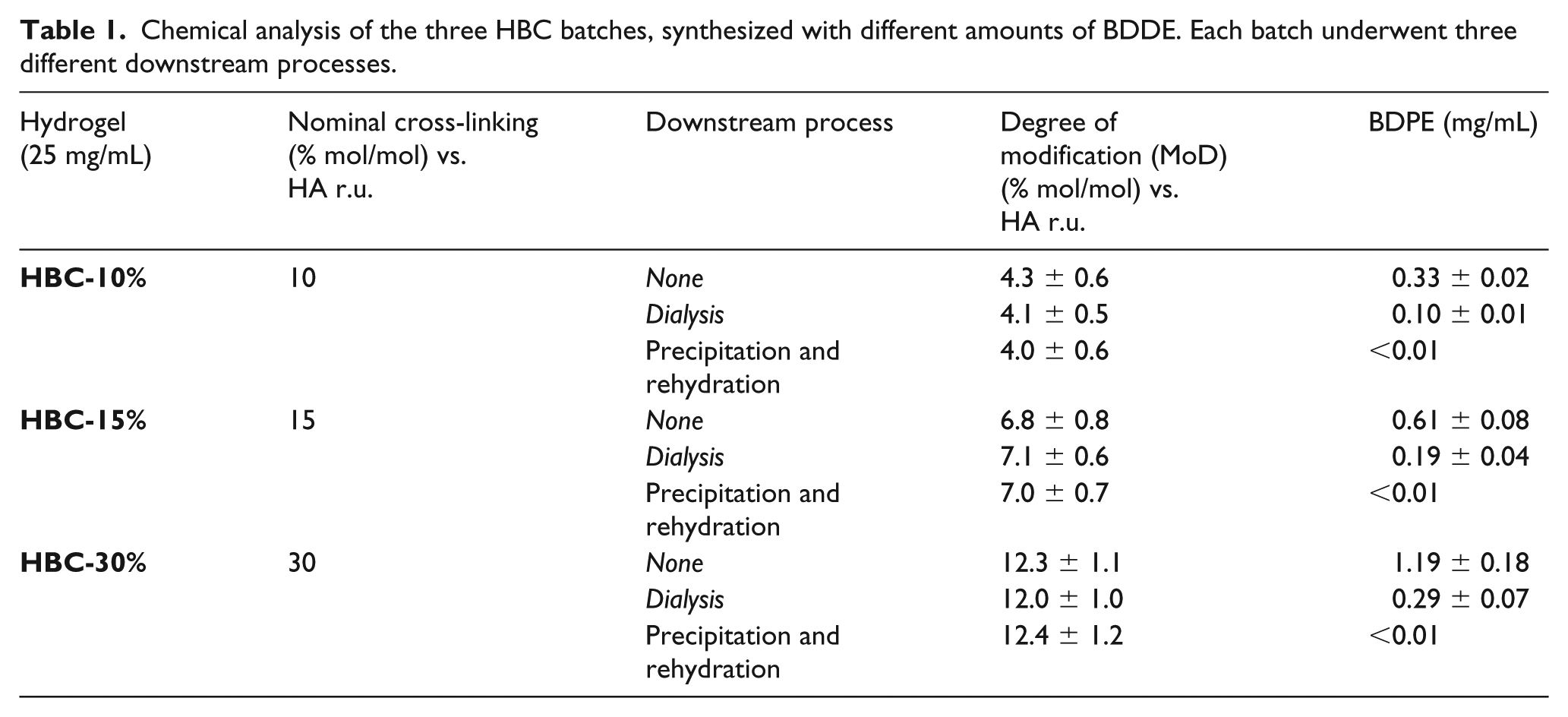
The cross-linking degree was measured using 1 H NMR spectroscopy using a Bruker Advance spectrometer operating at 300 MHz as previously described. 12 The results are tabulated in Table 1.
Chemical analysis of the three HBC batches, synthesized with different amounts of BDDE. Each batch underwent three different downstream processes.
BDPE analysis by HPLC-MS
HPLC analyses were performed on a chromatographic system model 1260 Infinity (Agilent Technologies), equipped with a UV–Vis Scan detector set at 216 nm and a quadrupole LC/MS detector (model 6120, Agilent): capillary voltage at 3000 V positive, drying gas flow at 11 L/min, nebulizer pressure at 35 psi and drying gas temperature at 350°C. A 1.7 µm Kinetex HILIC 100 Å (50 × 2.1 mm; Phenomenex) column was used. The mobile phase was NH4OH (0.1% w/w) pH = 7.2/acetonitrile (ACN; 2%, v/v). The analytical method was settled as follows: gradient: isocratic; flow rate: 0.6 mL/min; temperature: 30°C; injection volume: 10.0 µL; run time: 5 min; LC-MS: SIM mode at 261
Preparation of the calibration standard: 66.1 mg of BDDE was solubilized in 18 mL NaOH 1 M, stirred for 4 h at 80°C, neutralized with HCl 1 M and brought to the final volume of 50 mL with PBS buffer (pH = 7). The BDPE solution (1.558 mg/mL) was diluted at 1:100 with the mobile phase and characterized by HPLC-MS analysis (RT = 1.3 min; MS [M+Na+] calculated: 261.18 Da; experimental: 261.1 Da), and the absence of BDDE or BDDE dimer and trimer was assessed (see supplemental material). The initial solution was opportunely diluted at different concentrations for the linearity and recovery studies.
Preparation of an HA cross-linked sample (ACP) with a known amount of BDPE standard: 0.1 g of ACP gel (25 mg/mL in PBS pH = 7) was solubilized adding 0.2 mL of calibration standard at known concentrations (from 0.04 mg/mL to 0.14 mg/mL; final concentration from 0.2 mg to 0.7 mg of BDPE for 1.0 mL of ACP gel), and the ACP polymer was precipitated by adding 1 mL of ACN. After filtration (at 0.2 µm), the solution was diluted at 1:200 with the mobile phase and was analyzed by HPLC-MS spectrometer.
Sample preparation: 0.1 g of gel filler was solubilized by adding 0.2 mL of Milli-Q water, and the HBC polymer was precipitated by adding 1 mL of ACN. After filtration, the solution was diluted at 1:200 with the mobile phase and was analyzed by HPLC-MS spectrometer. The results are tabulated in Tables 1 and 2.
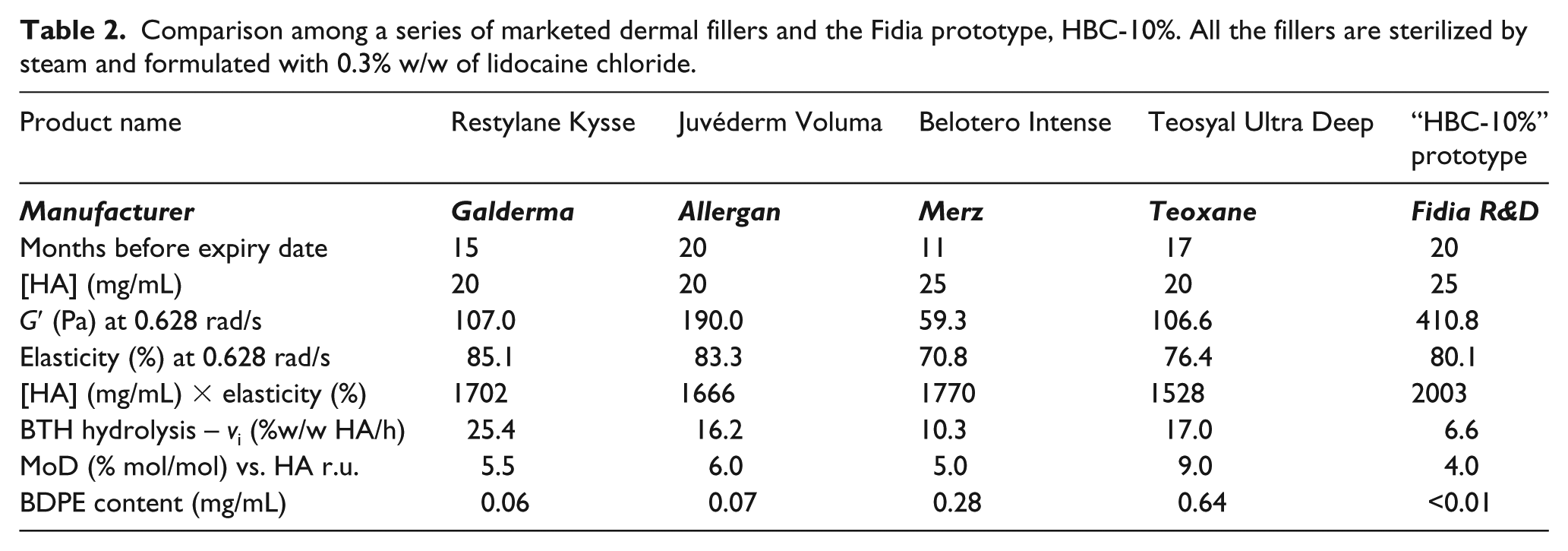
Comparison among a series of marketed dermal fillers and the Fidia prototype, HBC-10%. All the fillers are sterilized by steam and formulated with 0.3% w/w of lidocaine chloride.
Thermal stability study
A single batch of HBC-10% was purified by precipitation (as downstream process), and the powder was rehydrated at 25 mg/mL in PBS, transferred to several syringes and sterilized by steam. The syringes were stored at 55°C, and at fixed time points (0, 14, 30, and 45 days; t0, t1, t2, and t3, respectively), they were withdrawn and kept at 5°C, until they were investigated for the following tests:
- Rheological analysis
- Hyaluronidase assay
- In vivo residence time test
Based on preliminary data, the gel is stable at 5°C for at least 45 days (see S.I.).
Rheological analysis
Approximately 1 g of HBC gel sample (or marketed fillers) was analyzed using a Thermo Haake Mars II Rheometer at 25°C and analyzed as previously reported.
13
The
Elasticity (%) = [
Hyaluronidase assay
Each HBC-hydrogel (diluted to 1 mg/mL in PBS at pH = 6.4) was separately digested for 19 h at 37°C with BTH (10 U/mL) and analyzed by Viscotek TDA as previously reported. 13
Cytotoxicity assay
The biocompatibility of the tested compounds (HA, HBC-10%, HBC-15%, HBC-30%, and BDPE standard) was evaluated by means of a quantitative analysis according to the ISO 10993-5:2012 International Standard for the biological evaluation of medical devices. All HBC-hydrogels tested were previously depolymerized by thermal treatment for 14 h at 100°C, and all the samples tested were sterilized by filtration at 0.2 µm. The cytotoxicity study was performed using BALB/3T3 clone A31 mouse fibroblasts (ATCC® CCL-163) as previously described. 14 All the samples were tested in four replicates.
In vivo residence time test
This study (authorization of the Ministry of Health (IT) n°666/2017-PR) was conducted in accordance with the “Good Laboratory Practice” (GLP), following the “standard operating procedure” (SOP) at PRIMM S.r.l. (certification of Ministry of Health 018/2013). Five male rabbits (2–2.5 kg; age: 6–7 weeks; specific pathogen free) from the supplier establishment, Charles River Italy (Calco, Italy), were used in the study.
The animals were locally anaesthetized with lidocaine chloride (LUAN), their backs were shaved, and four injection sites were marked with a pen. Then, 0.5 mL of HBC-10% was injected sub-dermally with a maximum of two injections/animal and at least in four replicates for each time point (see Figure 2). HBC-10% filler was injected at time zero (t0) and after storage at 55°C for 14, 30, and 45 days (t1, t2, and t3, respectively; Figure 2(a)). During the experiment, the appearance of inflammatory reactions (edema and/or erythema) or any other adverse reactions due to the filler injection were evaluated.
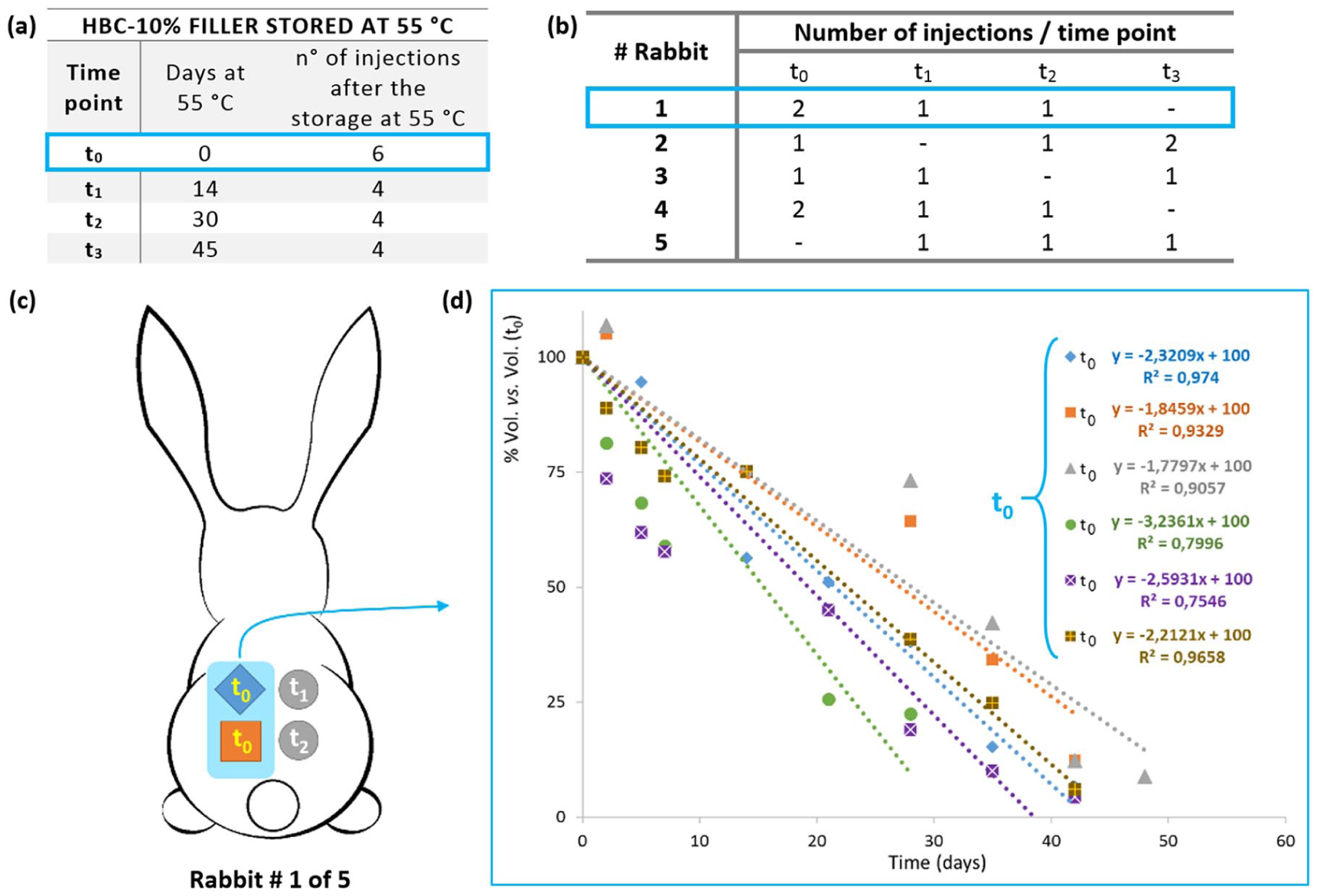
(a) List of samples tested in the in vivo residence time test. (b) Total number of injections at each time point per rabbit. (c) Schematic representation of the four injection sites on the rabbit#1 back: t0, t1, and t2 represent different time points of the HBC-10% filler. (d) On the right, the graph of the volume percent reduction vs. time for six replicates (6 injections) of the HBC-10% filler at t0 of the thermal stability study. The residence time value at t0 is 2.33±0.54% vol. vs vol. (t0)/day (
The bump volume in the injection site was measured weekly for two months, using the following formula:

where
For each bump, volume reduction was defined as the initial rate of the zero-order kinetic plot (volume % reduction
The mean of the rate constants of each stability time point was used to calculate the mean residence time value and standard deviation.
Chemical-physical analysis of the marketed fillers
All the marketed fillers were analyzed with the methods described in previous paragraphs. Approximately 1.5 g of each marketed filler were analyzed singly (
Statistical analysis
The statistical analysis reported for the “in vivo volume reduction” set of data (see Figure 4) were performed using GraphPad Prism 5.0 applying the one-way analysis of variance with Tukey’s post-hoc analysis;
Results and discussion
BDPE analysis and validation
BDDE is the most common cross-linker used in HA dermal fillers on the US market, with a synthetic procedure that is well described in the literature. 12 However, to the best of our knowledge, an analytical method for quantifying the major byproduct (BDPE) is still lacking.
In the present study, we developed and validated a novel, simple, and fast method to identify and quantify BDPE, based on ultra-high-performance liquid chromatography (UPLC) with a quadrupole MS detector, using a hydrophilic interaction liquid chromatography column (HILIC). 15 The BDPE standard was obtained through quantitative BDDE hydrolysis in a sodium hydroxide solution. The reaction has been optimized to prevent the polymerization reaction: the absence of BDPE dimer and trimer, in addition to the absence of BDDE, were confirmed by MS analysis (see S.I.). The HPLC-MS analysis and the BDPE mass spectra are reported in Figure 3. The linearity, within the concentration range of 0.009–1.3μg/mL, was determined to have a correlation coefficient (R2) of 0.9994. The analysis was validated by estimating recoveries after the spiking with known amounts of BDPE to different samples of HA hydrogel cross-linked without BDDE (ACP, at 25 mg/mL). Average recoveries covering five points were within the range 94.1–101.6%, with an SD of 3.1% and with good repeatability (average recoveries in Run 2: 100.9±5.5%). The limits of detection (LOD) and quantification (LOQ) were 0.003 µg/mL and 0.01 µg/mL, respectively. The method exhibited good precision, accuracy, repeatability, specificity (see S.I.), and recovery.
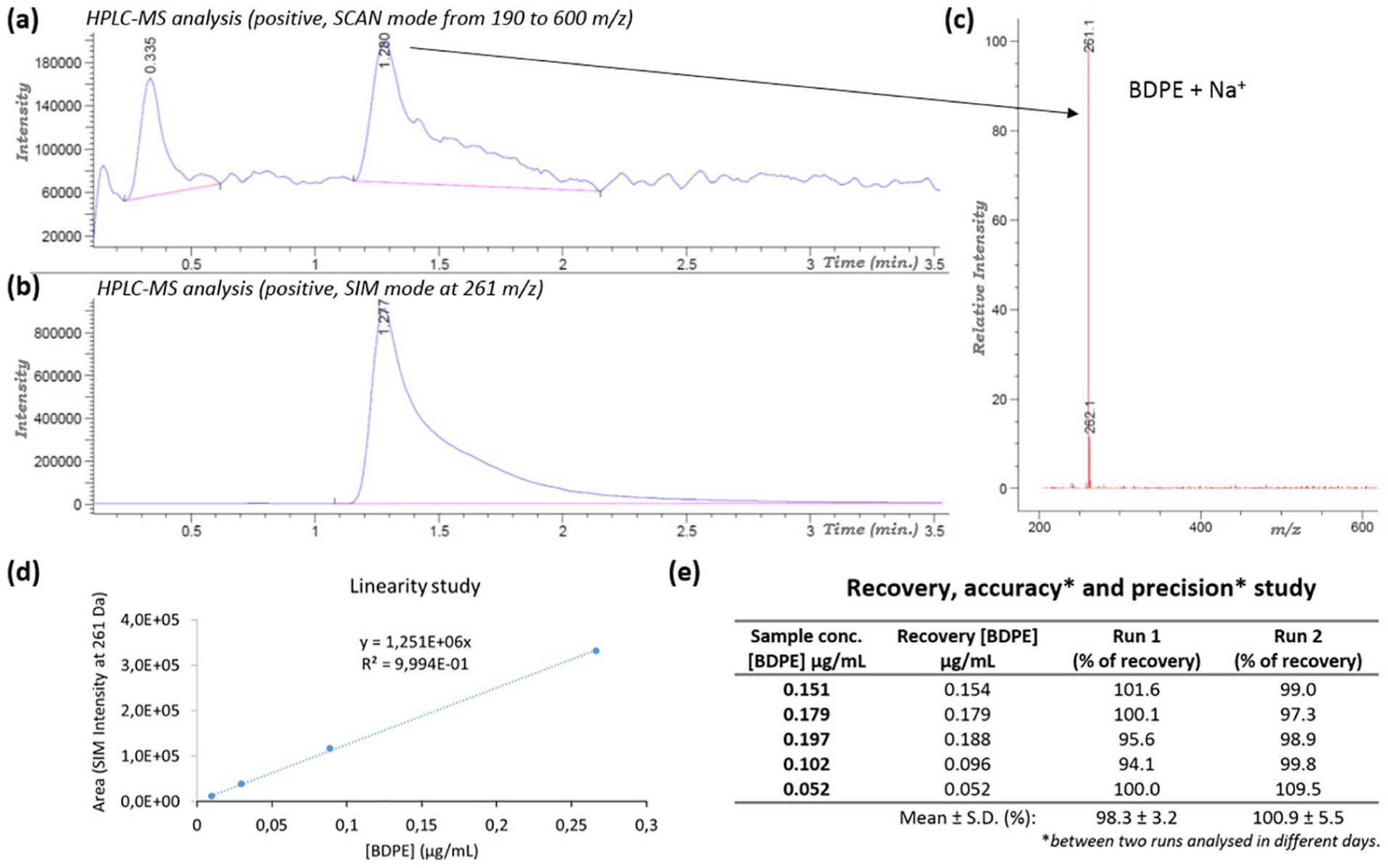
BDPE analysis method and analysis validation. (a) HPLC-MS chromatogram of standard BDPE in scan mode and (b) SIM mode at
Synthesis and characterization of HBC hydrogel at increasing cross-linking degrees and different downstream processes
In this work, a series of HBC derivatives were synthesized with increasing cross-linking degrees, and different downstream processes for BDPE removal were evaluated. Three batches of HBC were synthesized using an initial BDDE loading of 10%, 15%, and 30% mol (
Cytotoxicity evaluation of BDPE and HBC hydrogels at different MoD
In the present work, we evaluated the cytotoxic effect of the BDPE linked to HA and the major reaction by-product, i.e., free BDPE in solution. Specifically, a cytotoxicity assay (according to ISO 10993) in a mouse fibroblast cell line was performed exposing the cells to increasing concentrations of BDPE or HBC at different MoD (see S.I.). The HBC samples tested were purified by precipitation to completely remove the residual BDPE and BDDE and then were rehydrated and depolymerized by thermal treatment to reduce their viscosity. The stability of HA-linked BDPE under thermal treatments was already evaluated in a previous study. 12 Depolymerized HBC, even at the highest MoD, did not show any cytotoxic effect at the tested concentrations. In contrast, the IC50 value after BDPE treatment was 0.48 mg/mL. The half maximal inhibitory concentration (IC50) is a measure of the potency of a substance in inhibiting a specific biological or biochemical function. According to the FDA, IC50 represents the concentration of a drug that is required for 50% inhibition in vitro. These preliminary results showed a cytotoxic effect due to the presence of free residual BDPE; the effect was absent when BDPE was chemically linked to HA. However, the in vitro cytotoxicity assay is extremely sensitive, and the observed cytotoxic effect could be absent in an in vivo study; for this reason, further specific toxicity studies are needed. Nevertheless, this study confirmed the need to obtain HA dermal fillers without impurities.
Stability study of HBC hydrogels
In the development of a new dermal filler, hydrogel viscosity is the feature that determines the stiffness of the gel, the resistance to deformation and the ability to remain at the site of injection; however, this parameter changes during the lifetime of the filler.
16
To define which parameter is able to predict the clinical performances, the viscoelasticity was monitored throughout the shelf-life of an HBC hydrogel and was correlated with in vitro resistance towards hyaluronidase degradation and in vivo residence time in a rabbit model. The results are summarized in Figure 4. In detail, we synthesized a batch of HBC-10%; after precipitation (during the downstream process), the powder was rehydrated at 25 mg/mL in PBS, transferred to several syringes and sterilized by steam. The syringes were stored at 55°C with fixed time points of 0, 14, 30, and 45 days, following which they were withdrawn and kept at 5°C to prevent thermal degradation until the in vitro and in vivo tests were performed. The stability study was stopped on day 45; this time length simulated the degradation occurring after more than 2 years at 25°C.
17
Rheological measurements of each time-point underline how well the exponential decrease in the
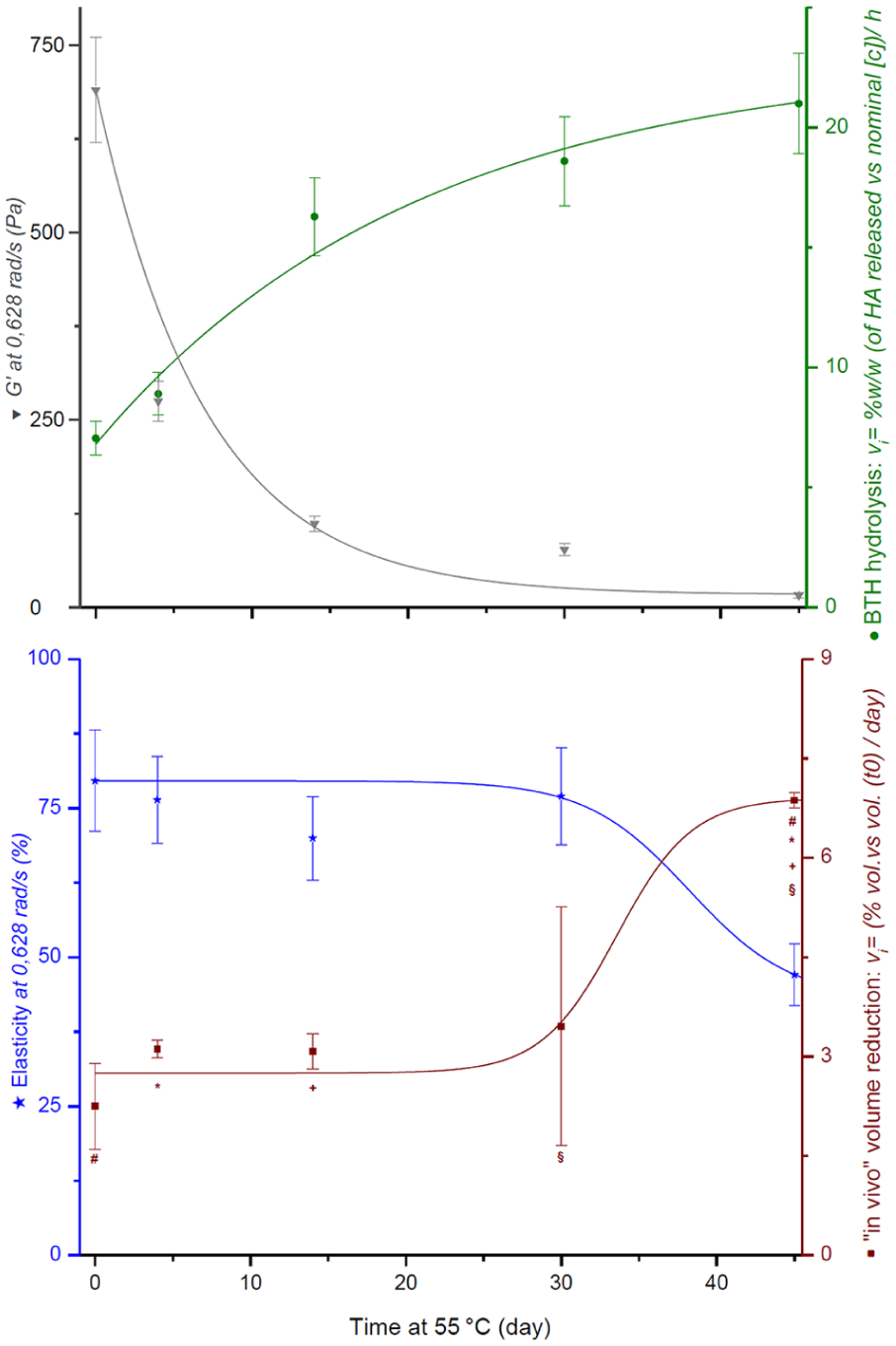
Thermal stability evaluation of the HBC-10% filler at 25 mg/mL, at 55°C for 45 days. Analyzed parameters: elasticity (blue line), elastic modulus (grey line), in vitro hyaluronidase degradation rate (green line) and early
Chemical and physical comparison of marketed dermal fillers
In Table 2, we compare a series of marketed dermal fillers, which were already designed for volume restoring of the face and/or the lips. All the fillers show good rheological behavior with high elasticity. Belotero Intense exhibited lower elasticity, but it was also close to the expiry date. The rheological parameters were supported by the high MoD, while the initial rate of BTH hydrolysis did not appear to be linked with elasticity, likely due to differences in HA concentrations, synthetic procedures and months before the end of the shelf-life. The BDPE content reflects different manufacturing technologies and purifications. However, it can be stated for none of these marketed products that BDPE is absent, as demonstrated via the downstream process (precipitation and rehydration) used to produce the prototype HBC-10%.
Conclusions
In the fillers obtained with the HA–BDDE cross-linking process, BDPE (double-linked 1,4-butanediol di-(propan-2,3-diolyl) ether) is the major impurity. In this work, a new HPLC-MS method with good precision, accuracy, repeatability, specificity, and recovery was developed to quantify the BDPE content in HBC fillers. A series of HBC derivatives was successfully obtained. Independent from the MoD, only the downstream process with precipitation and rehydration showed the complete removal of the unreacted cross-linker or its byproducts. In a cytotoxicity assay (according to ISO 10993), BDPE gave an IC50 value of 0.48 mg/mL, while depolymerized HBC (without residual BDPE), even at the highest MoD, did not show any cytotoxicity effect. To define which parameter is able to predict clinical performance, the viscoelasticity was monitored throughout the shelf-life of an HBC hydrogel (HBC-10%) and was correlated with in vitro resistance towards hyaluronidase degradation and
Supplemental Material
Supporting_Information_rev – Supplemental material for HA-based dermal filler: downstream process comparison, impurity quantitation by validated HPLC-MS analysis, and in vivo residence time study
Supplemental material, Supporting_Information_rev for HA-based dermal filler: downstream process comparison, impurity quantitation by validated HPLC-MS analysis, and in vivo residence time study by Cristian Guarise, Carlo Barbera, Mauro Pavan, Susi Panfilo, Riccardo Beninatto and Devis Galesso in Journal of Applied Biomaterials & Functional Materials
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
