Abstract
This paper presents a study on the synthesis and characterization of hydroxyapatite that can be used to produce dental implants or serves as a replacement for other body hard tissues. The bioceramic material (hydroxyapatite) was derived from bovine bone samples after they were de-fatted and processed to produce particles measuring less than 250 μm. Processed bone powders were calcinated at 750℃ to obtain calcium phosphate compound (hydroxyapatite). This indicated complete elimination of the organic phase of the bone. The synthesized hydroxyapatite was physico-chemically characterized using Fourier transform infrared spectroscopy (FTIR), an optical microscope and scanning electron microscopy (SEM). Hydroxyapatite powder was sintered into a block and then characterized mechanically by measuring its hardness using a Rockwell hardness testing machine. The hydrophilicity and hydrophobicity of the sintered blocked specimens were studied. The average wettability of the substrate, which was a characteristic of the contact angle, was found to be approximately 31.73 degrees. This contact angle value is a measure of the material hydrophilicity. The biostability/corrosive resistance of the sintered samples was simulated in vitro in phosphate buffered saline solution (pH 7.4) by incubating in an optical incubator shaker (set at 60 revolutions per minute) to mimic physiological conditions. The results showed that hydroxyapatite can be extracted economically from a natural source such as bovine bone and can be employed as a restorative biomaterial for dental implants and hard tissue replacement.
Introduction
The main goal of modern restorative dentistry is to restore patients with dental disease or injury to normal function, health, speech, and esthetics, regardless of the disease, atrophy, or injury of the stomatognathic system. Dental implants are ideal options for people with oral health who have lost a tooth due to periodontal disease, injury, or some other reason. Dental implants (considered as an artificial tooth root) are biomaterial (metal or ceramic) anchors surgically positioned in the jaw bone (in other words, surgically traumatized bone) underneath the gums to support an artificial crown where natural teeth are missing. 1
Hydroxyapatite is a calcium-phosphate-based ceramics biomaterial that has been found to be crucial for the synthesis of dental implants or other hard tissues due to its biocompatibility, osteophilic nature and its easy incorporation into bone tissues. Hydroxyapatite has been used successfully for many years to replace and augment bone tissue since it was discovered that calcium phosphate plays a major role in the inorganic phase of hard tissues, bone, and teeth. 2 Hydroxyapatite is synthesized in the laboratory by a wet chemical precipitation reaction. This method of preparing hydroxyapatite is very expensive, thereby limiting the use of laboratory-synthesized hydroxyapatite for hard tissue replacement as it requires a large quantity of material.3,4 However, hydroxyapatite can be sourced naturally and economically from bones such as bovine bones and fish bones, fish shells, oyster shells, chicken egg shell, and corals. For sustainable development, waste should be recycled, reused, and channeled toward production of value-added products. Bovine bone, which is a biowaste, is an economical source of hydroxyapatite for hard tissue replacement in medical and dental therapy. 2
Materials and methods
Materials
The materials used for the preparation of hydroxyapatite are presented in Table 1. The source and reason for choice of these materials are also presented in Table 1.
Materials – sources and reason for choice.
Preparation of bovine bone powder
The method adopted for the extraction of hydroxyapatite followed the procedures of Khoo
The process was followed by sun drying of the bovine bones for 3 weeks to evaporate the absorbed moisture. The dried bovine bones were crushed into small pieces using a mortar and pestle and then pulverized using a grinding/milling machine. Finally, the powders were sieved to separate off particle sizes of less than 250 μm. Figure 1 shows the bovine bone samples at different stages of preparation.
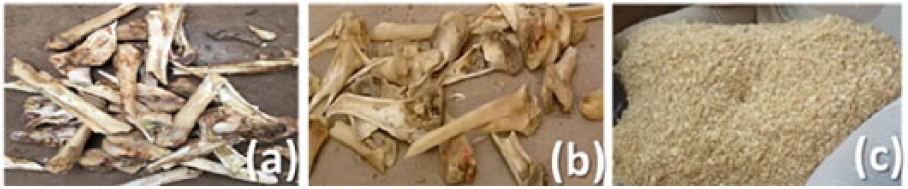
Bovine bone samples: (a) as sourced; (b) washed and cleaned; and (c) pulverized.
Extraction of hydroxyapatite using thermal decomposition
One hundred grams of bovine bone powder of particles sizes ranging from 0 to 250 μm was placed in an alumina crucible and then heated in a tube furnace at a rate of 10°C/min from room temperature to 750°C for 6 h. The thermally decomposed samples were then cooled slowly to room temperature following the procedure of Barakat
Preparation of sintered specimens
A square prism mild steel mold (20 mm×20 mm×30 mm) was filled with hydroxyapatite powder to produce a sintered sample specimen for characterization of the hydroxyapatite. The hydroxyapatite powders were mixed with liquid paraffin (2 ml) and organic oil to achieve porosity and proper lubrication of the mold. The powder was then pressed in a mild steel mold using a hydraulic press at two different pressures: 150 and 170 MPa. Finally, the blocks were sintered to 1300°C in a furnace.
Physico-chemical characterization
Fourier transform infrared spectroscopy (FTIR) measurements were carried out using a spectrophotometer (Schimadzu’s UV-2550, Japan), in the transmission mode in the mid-infrared range with wavelengths ranging from 400 to 4500 cm−1. Then, 2 mg of each of the powders was mixed and pressed with 300 mg of KBr to produce a pellet for the analysis. The surface morphology of the powder samples and sintered block were obtained using a scanning electron microscope (Aspex 3020, Pennsylvania, USA).
In vitro corrosion and biostability study of hydroxyapatite
The initial masses of the samples were determined before incubation in glass test tubes containing 15 ml of phosphate buffered saline (PBS) (at different pH). A hydrolytic stability/corrosion test was carried out in a digital incubator shaker (Innova 4300, New Brunswick Company Inc., NJ, USA) at 60 revolutions per min (rpm) at 37°C. This environment mimics the physiological conditions during localized implantation. The biostability/corrosion was characterized by weight loss by calculating the rate of material erosion/dissolution from the absorption of PBS after 3 months using Equation (1) as given below:

where dM is the change in mass M, at time t, and k is the kinetic rate constant of erosion.
The absorbance of the fluid was examined using ultra-violet spectroscopy by determining the absorbance of the media (PBS). The absorption in the visible range directly affects the perceived color of the chemical involved.
Wettability test
This measures the tendency of the body fluid to spread on or adhere to the surface of the sintered hydroxyapatite block in the presence of other immiscible fluids. The method employed to carry out this test was the contact angle method. The experimental setup required a lighting system in addition to an optical system. The optical components include a basic digital camera and an optical lens with a known focal length. The optical lens was placed between the camera and the sample, while the sample is placed in a black box with a 40-W lamp mounted behind the samples to shine light on the liquid drop. The setup was guided by the Lenses formula, presented in Equation (2):

where
With the aid of a micropipette, 5 μ

where γ1 is the energy of the solid–liquid interface, γ2 is the surface energy of the solid, γ3 is the surface energy of the liquid, and Ɵ is the contact angle.
Contact angle measurement using Gwydion software.
Mechanical characterization
The hardness of the sintered hydroxyapatite samples was determined with a Rockwell hardness testing machine (HR 150A, Jinan Precision testing, Shangdong, China), and the effect of particle size range on the hardness of the sintered hydroxyapatite was determined.
Results and discussion
Effect of calcination time and temperature
Figure 3(a) to (c) shows samples of bovine bone before and after it was calcined. The figures reveal changes in the physical appearance of the bone powder as it transformed from raw bone to hydroxyapatite due to the elimination of the organic phases during thermal treatment in the furnace. This indicates the effect of` calcination time and temperature on raw bovine bone powder. Figure 3(b) shows the color change of the raw powder from yellowish brown to light grey after 3 h exposure at 650°C. Further heating to 750°C for 6 h resulted in a sample that appears to be completely white (Figure 3(c)). This confirms the presence of calcium phosphate compound, that is, hydroxyapatite.

Images showing (a) raw bone powder, (b) semi-decomposed bone powder, and (c) hydroxyapatite.
FTIR results
FTIR spectra analysis in the range of 4000 to 500 cm−1 was employed to characterize the different functional groups present in the bone powder and hydroxyapatite. Figure 4 shows the FTIR spectra of bone powder and synthesized hydroxyapatite, respectively. Differences between the spectra of raw bone powder and the decomposed sample at 750°C were due to changes in the chemical bonds that occurred during the thermal treatment. Two peaks at 2929.8 cm−1 and 1669.7 cm−1 corresponding to NH and amide bands were observed in the raw bone spectra, but were eliminated in the heated sample. This agrees with the results of Hosseinzadeh
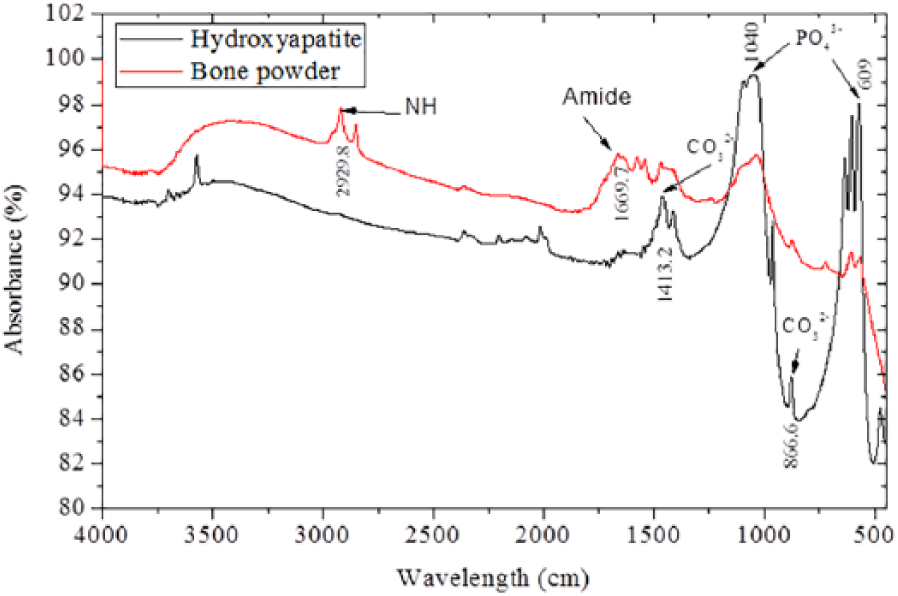
FTIR spectra of bone powder and hydroxyapatite.
This can also be confirmed through visual observation, as the color changes from yellowish brown to white.
7
The FTIR spectrum also revealed all characteristic absorption peaks of hydroxyapatite, showing the presence of phosphate (PO43−), hydroxyl (OH−), and carbonate (CO32−). These spectra are clearer in the decomposed sample, because the calcination process has eliminated the cross-linked structures in the raw bovine bone. The first indication of hydroxyapatite is in the form of a strong band at about 609 and 1040 cm−1, which is associated with the presence of a phosphate group (PO43−) in the decomposed sample. Barakat
The raw bovine bone particles exhibit a wide band near 3200 cm−1, which corresponds to the absorbed water molecule in the sample. The sharp narrow band at 643.80 cm−1 and the band at 3575.73 cm−1 are related to the hydroxyl group, which indicated the presence of hydroxyl group at 3436 cm−1. 7 The results of these FTIR analyses show that heating raw bovine bone powder at 750°C for 6 h will produce a calcium phosphate compound with characteristic hydroxyapatite phases.
Surface morphology of the samples
The SEM micrographs in Figure 5 (a) to (d) show the morphology of raw bone powder as well as the changes at the surface of the decomposed powder as the calcination temperature increases. The morphologies of the raw bone powder as it transformed during thermal treatment (Figure 5(a) to (c)) are similar to those observed by Khoo

SEM micrographs of: (a) raw bone powder, (b) semi-decomposed powder, (c) hydroxyapatite, and (d) sintered hydroxyapatite.
The temperature and time (650°C and 3 h, respectively) at which the raw powder decomposed during calcination have a direct impact on grain growth, such that as the temperature was increasing, the particle grain size increased, as shown in Figure 3.3(b). Figure 3.3(c) also revealed that the hydroxyapatite particle grain size increased at higher calcination temperature and exposure time of 650°C and 6 h, respectively. This agrees with the observations made by Hoque
An SEM image of a pressed hydroxyapatite block made after sintering at 1300°C is shown in Figure 5(d). The figure shows that the powder particles became more compact and denser after sintering, thereby reducing the porosity of the sintered hydroxyapatite block. An liquid paraffin present was burnt off after serving as a binder for the fusion of the powder particles during the sintering process. The process of sintering resulted in more uniform grains, and finer and greater density of the samples. These properties are similar to the findings of Hosseinzadeh
Wettability
On dropping 5 µl of PBS on the surface of the sintered hydroxyapatite block, the bead (droplet) was absorbed immediately on contact with the surface. This is most likely due to the presence of pores. When the droplet volume was increased to 10 µl, the same condition was observed. Tripling the initial size of the droplets led to somewhat stable liquid on the surface with the orientation shown in Figure 2. The stability of the bead is most likely to be a function of the surface property (surface energy of the substrate) of the sample and size of the bead. The contact angles measured were observed to vary between 25.96 and 37.49 degrees.
The contact between the bead and the surface of the sample was used in the evaluation of the contact angle. The average contact angle was approximately 31.73 degrees, which is less than 90 degrees. This indicates that the sintered hydroxyapatite block surface is hydrophilic. The surface of the sample (hydroxyapatite) has a “liquid-loving” property, which may be due to the nature of chemical bonding present in hydroxyapatite (ceramic) – covalent bonds. De-Gennes. reported that hard solid like ceramics have high surface energy thereby resulting in a strong attraction for other surfaces in contact. 10 The PBS, on the other hand, is a molecular liquid with a low surface energy since it is a mixture of several compounds that are not chemically bonded. In general, this indicates that wetting on the hydroxyapatite surface is favorable.
According to Yuehua and Randall 11 small contact angles (< 90 degrees) correspond to high wettability, while large contact angles (> 90 degrees) correspond to low wettability. Therefore, since hydroxyapatite is hydrophilic, it shows that it would accommodate bio-fluid in the host body.
In vitro biostability and corrosion studies
As shown in Table 2, different masses of samples A to H of sintered hydroxyapatite were weighed before incubating them in a test tube that contained 1.5 ml of sterilized PBS. Samples A–D were dipped in a solution with a pH of 7.0, while samples E–H were dipped into a solution with a pH of 7.4. The masses of the samples were recorded on removal from the simulated physiological conditions (pH 7.0–7.4, 60 rpm, 37°C) after 3 months. For pH 7.0, the change in mass values for samples A, B, C, and D after the 3 months were 0.37 g, 0.384 g, 0.57 g, and 0.46 g, giving an average change in mass of 0.446 g, while for pH 7.4, the change in mass values for samples E, F, G, and H were 0.377 g, 0.384 g, 0.372 g, and 0.375 g, amounting to an average change in mass of 0.359 g. Therefore, the kinetic erosion rates were calculated using the average change in masses divided by the time of immersion.
The masses of samples A–H before and after being subjected to simulated physiological conditions (pH 7.0–7.4, 60 rpm, 37°C) after 3 months.
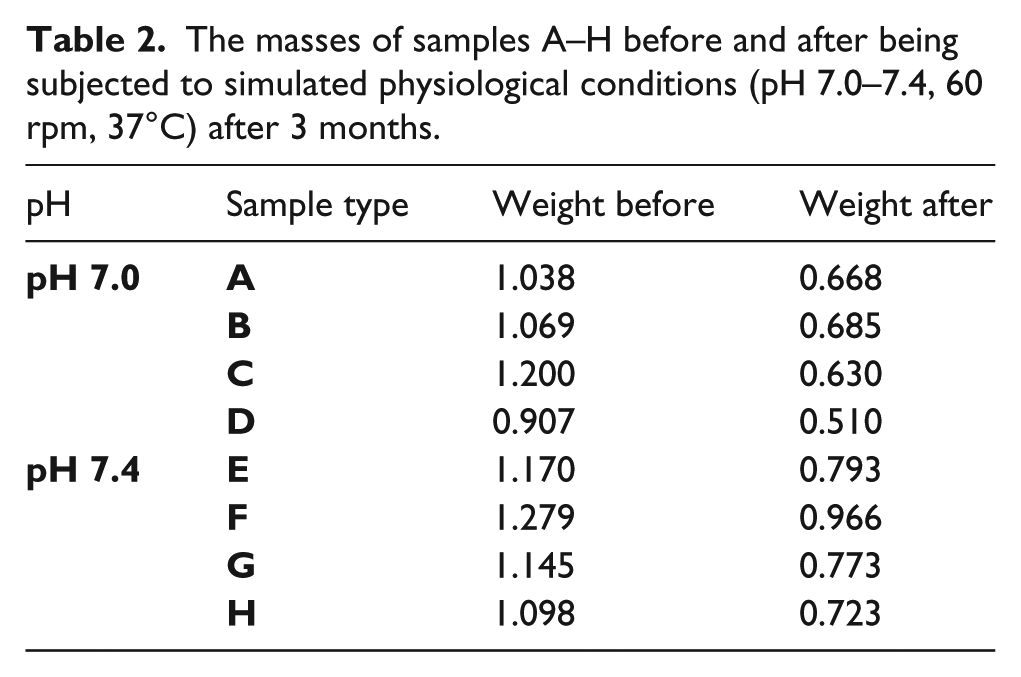
For pH 7.0,
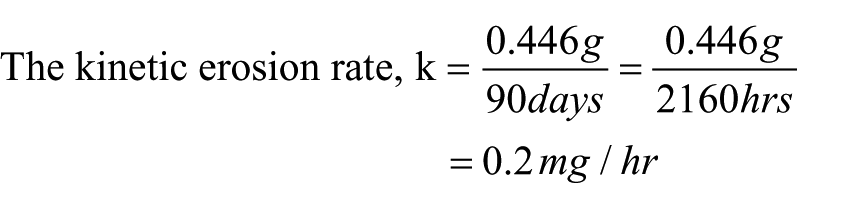
For pH 7.4,
There was a reduction in mass of the samples after 3 months, but this may be due to the presence of debris or unsintered particles at the surface.
The sintered hydroxyapatite block dissolved into the PBS solution thereby leaving a white residue at the base of the test tube. This resulted in the weight loss as observed. However, the weight loss may not have resulted from corrosion but may be due to other factors such as presence of unsintered surface debris. It can further be stated that hydroxyapatite is a biologically stable material that can withstand body physiological conditions. In order to avoid any kinetic erosion (if any) of the material, the surface can be modified by coating with other bone cements such as powdered hydroxyapatite (commercial grade). Thus, a bone-like apatite layer could be created on the surfaces of substrate with a bio-active ceramic such as CaO-SiO2-based glass as the source of the apatite-nucleating agent. 12 This will also improve the mechanical properties of the sample.
Hardness
Measurements of hardness of different samples of hydroxyapatite are given in Table 3. Sample A was mixed with a higher volume of liquid paraffin (20 ml) and sintered at 130°C, while Sample B was mixed with 10 ml of liquid paraffin and sintered at 1300°C. As shown, the average hardness of sample A (119.5 HRB) is higher than that of sample B (80.5 HRB). This revealed that the amount of binder (liquid paraffin) influences the sintering ability of the particles and thus the hardness.
Average hardness of samples.

Conclusion
Natural hydroxyapatite can easily be extracted from raw bovine bone by the thermal decomposition method. Extracted hydroxyapatite is useful for dental implants due to its biocompatibility properties, osseo-integration, and similar mechanical properties when compared with the human tooth. The results showed that calcinating at 650°C for 3 h produces semi-decomposed powder, while heating at 750°C for 6 h synthesizes hydroxyapatite from the raw bovine bone powder. It can be inferred that treatment temperature and time are key parameters in determining the composition of the extracted product.
An in vitro biostability/corrosion test showed that porous hydroxyapatite is stable under human physiological conditions as it is not absorbed in the simulated body fluid and also allows the osseo-integration of body tissues.
When comparing the hardness of sintered hydroxyapatite blocks with varying porosities, the block with less pores is found to possess hardness (119.5 HRB), while the block with more pores has less hardness (80.5 HRB), but the presence of pores will accommodate osseo-integration for use in dental or orthopedic applications.
It can be inferred from these observations that hydroxyapatite is an excellent candidate material for restorative dentistry and orthopedic applications due to its inertness to body fluids and good aesthetic appearance.
Footnotes
Acknowledgements
The authors are grateful to the technical staff of the Materials and Metallurgical Engineering Department, University of Ilorin and Kwara State University, Malete for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
