Abstract
In order to improve osseointegration, polyetheretherketone (PEEK) interbody fusion cages are frequently surface coated. The bonding strength of the coatings is mostly investigated under unrealistic loading conditions. To close this gap, in this study, uncoated and coated cages were loaded in a clinical setup in order to investigate their real resistance against abrasion.
Six uncoated, six calcium phosphate (CaP) nanocoated, and six titanium (Ti) nanocoated PEEK cages were tested in this study. The experimental setup was designed to mimic cage impaction into the intervertebral disc space using polyurethane (PU) foam blocks as vertebral body substitutes. The cage surface was inspected before and after impaction, and their weight was measured.
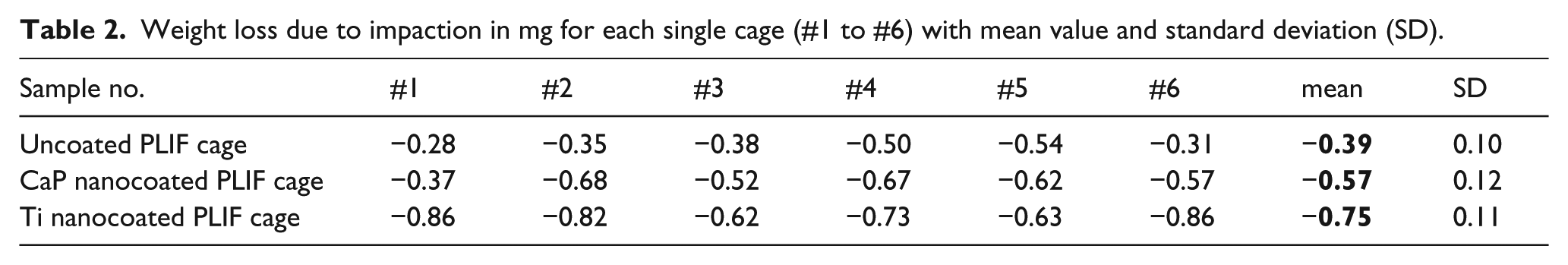
Impaction resulted in abrasion at the tip of the ridges on the implant surface. The mean weight loss was 0.39 mg for the uncoated cages, 0.57 mg for the CaP nanocoated cages, and 0.75 mg for the Ti nanocoated cages. These differences were statistically significant.
In conclusion, differences between the three cage types were found concerning the amount of abrasion. However, all three cages lost less weight than a comparative Ti plasma spray coated cage, which showed a mean weight loss of 2.02 mg. This may be because the plasma spray coating is significantly thicker than the two nanocoatings. If compared with the permitted amount of weight loss derived from an FDA guidance document, which is approximately 1.7 mg, the wear of the Ti plasma spray coated cage is above this limit, whereas the wear for all other cage types is below.
Keywords
Introduction
Numerous spinal interbody fusion cages are made of polyetheretherketone (PEEK). The advantages of this biomaterial are its good biocompatibility, its relatively low modulus of elasticity (3.5 MPa versus 100–110 MPa for titanium, Ti ), 1 which reduces the risk of bone resorption and stress shielding, and its radiolucency, which enables easier assessment of fusion on radiographs. One of the major disadvantages of PEEK is its poor osseointegration, especially in case of smooth implant surfaces. As a consequence, fibrous tissue layers are often observed at the PEEK-bone interface. 2
To counter this disadvantage, PEEK cages are frequently surface treated or coated. Various types of coatings have been developed such as the Ti plasma spray coating, which is among the most frequently used coatings in the field of medical implants.3–5 But various other coatings also address the poor osseointegration of PEEK such as nanocoatings with calcium phosphate (CaP) or Ti, 6 hydroxyapatite coatings, 7 or carbon coatings. 8 All these coatings have been intensively investigated concerning their biological behavior using cell culture or animal models. However, the mechanical safety of the coatings, especially their bonding strength to the PEEK substrate and their resistance against abrasion, has rarely been investigated.
It is known that bonding on PEEK is difficult,9–11 whereas the adhesion to metal substrates is significantly higher. 3 For patients treated with coated cages, a failure of the coating–PEEK interface bears significant risks such as inflammatory reactions or loss of bony integration. Despite these general risks, 12 the risk of coating abrasion in a specific clinical setting has not been investigated in detail so far.
In the present study, comparative impaction tests were carried out with a PEEK posterior lumbar interbody fusion (PLIF) cage, which either had no coating, a CaP nanocoating, or a Ti nanocoating. The purpose was to investigate whether the impaction of the cages into the disc space would result in abrasion of the coating and whether there are any differences between the coatings.
Material and methods
Three groups of PLIF cages were tested in this study. The cages in all three groups had the same geometry and were made of a PEEK substrate (Table 1, Figure 1). The cages in two of the three test groups were additionally surface coated either with a CaP nanocoating (osteoCon PLIF cage) or a Ti nanocoating (TSC PLIF cage), whereas one cage group was uncoated (reCreo PLIF Cages). Six samples were tested in each of the three cage groups.
Three groups of posterior lumbar interbody fusion cages were tested in this study: an uncoated PEEK cage, a calcium phosphate nanocoated PEEK cage and a titanium nanocoated PEEK cage.


Uncoated PLIF PEEK cage (left), CaP nanocoated PLIF PEEK cage (middle; the coating is not visible to the naked eye), titanium nanocoated PLIF PEEK cage (right) used for testing.
In clinical practice, impaction of the intervertebral fusion cage into the disc space is often required. This situation was simulated in the present study similar to Kienle et al. 13 Two vertebral body substitutes made of PU foam grade 40 pcf (Sawbones, Malmoe, Sweden) were manufactured with planar and parallel surfaces. This material is commonly used as a cortical bone substitute and the planar surface was chosen to mimic the surface of the endplates with mild disc degeneration. 14 These vertebral body substitutes were fixed in a pneumatically driven axial loading device (Figure 2).

Test setup for the impaction test. The two vertebral body substitutes (PU foam grade 40 pcf) were fixed in an axial preload device. A preload of 390 N (Fax) was applied to the two substitutes. The impaction force (Fim) was realized using a drop weight that impacted the insertion instrument at a speed of 2.6 m/s. To ensure proper alignment of the insertion instrument and the drop weight, an instrument guidance and a guiding rod were used.
The insertion instrument was connected to the cage, which was positioned in between the two vertebral body substitutes with its tip at a standardized starting position for impaction. Care was taken to align the insertion instrument along the posterior to anterior axis of the disc space. For this purpose an instrument guidance was used, which allowed a sliding movement of the handle of the instrument in the anterior direction of the simulated disc space.
Using the axial preload device, a constant axial preload (Fax) of 390 N (1 kN, Burster, Germany) was applied to the vertebral body substitutes with the tip of the cage in between, to simulate the axial preload that acts on the spine during impaction.
Impaction (Fim) was realized using a drop weight (526 g) that hit the cage at a speed of 2.6 m/s. The drop weight was guided using a guiding rod to ensure vertical alignment above the insertion instrument throughout testing.
The impacts were repeated until the cages were fully inserted into the disc space (Figure 3).

Test procedure of the impaction of the titanium nanocoated PEEK PLIF cage (representative of all three cage types).
The amount of wear was quantified through weight measurements on a microbalance (CPA225D-0CE, Sartorius, Germany) before and after impaction. In order to be able to weigh the specimens under standardized conditions each specimen was cleaned in an ultrasonic cleaner and dried in an exsiccator before weighing. The mean weight loss was calculated for each cage group. The statistical significance of the differences between the cage groups was evaluated using the Student’s t-test with at a 5% level of significance.
Impaction-related changes on the cages’ surface were visualized using macro photos.
Results
All cages were fully impacted into the disc space after either four or five hits.
The macroscopic examination after impaction showed that the tips of the ridges where the implant was in contact to the vertebral body substitute were abraded (Figure 4). This was the case for all three cage types. In the case of the Ti nanocoated implant, the coating was additionally worn off the slopes of the ridges.

Representative macro photos of the cage surfaces before and after impaction. Abrasion of the tip of the ridges (see close-up photograph on the right-hand side) was detected in all three test groups. Additionally in case of the Ti nanocoated cage, some areas were detected where the coating had almost disappeared.
The mean weight loss was between 0.39 mg for the uncoated PEEK PLIF cage and 0.75 mg for the Ti nanocoated PEEK PLIF cage (Table 2). The differences between each pair of the three test groups were statistically significant at the 5% significance level.
Weight loss due to impaction in mg for each single cage (#1 to #6) with mean value and standard deviation (SD).
Discussion
In this study, the resistance against abrasion was investigated of uncoated and coated PLIF cages using a setup that mimics an implant-specific clinical worst-case situation. Commonly, the adhesion of a coating to its substrate is quantified using standardized shear tests carried out on standardized test samples.15,16 These standard procedures are easily conductible, and allow direct comparison between different types of coatings, however it is not possible to transfer the results to a specific clinical setting. For example, if the results of such a test show that the shear bond strength is 47 MPa, it still remains unknown whether the coating of a specific implant with its specific surface geometry and size will withstand the shear loads it experiences in vivo. To address this disadvantage of standard testing, Kienle et al. proposed a setup that specifically simulates the impaction of a lumbar interbody fusion cage into the disc space. 13 Impaction was assumed to be associated with high loads on the coating and the coating-substrate interface. For that reason, this setup was also chosen for the present study. The main advantage of this approach is its close relation to the situation in vivo. Nevertheless, despite the realistic setup, it is still based on certain assumptions that should be kept in mind. 13
The results showed that even the uncoated cage lost weight (0.39 mg in mean), followed by the CaP (0.57 mg) and the Ti nanocoated cages (0.75 mg). These differences between the three cage types were small but statistically significant and can partially be explained by the coating thickness, which is smaller in the case of the CaP nanocoating compared with the Ti nanocoating. In order to better understand whether the amount of wear measured for the two nanocoated cages differs from that of a thicker coating, the results were compared with those of a Ti plasma spray coated PEEK PLIF cage. The coating thickness of this cage was in the order of 300 to 500 µm, which is more than x1000 the thickness of the nanocoatings. The size of the cage was identical to those of the nanocoated cages, only its surface geometry differed somewhat as it had little teeth instead of ridges. Impaction under the same conditions as described above resulted in a mean weight loss of 2.02 mg (±0.67 mg). It was mainly the tips of the teeth that were affected by abrasion. Similar results were reported by Kienle et al., 13 The weight loss of the Ti plasma spray coated cages was significantly higher than that of the nanocoated cages. There may be various reasons for this finding. First, a thicker coating can lose more material. The bonding strength between the different types of coating might also differ, and there are differences in surface roughness.
From a clinical perspective, however, the most important question is whether such abrasion has associated risks and possible complications for the patient. In general, where the coating has worn off, it can no longer improve osseointegration. Furthermore, there is evidence of inflammatory reactions caused by Ti wear particles. Local, mild, or medium inflammatory reactions have been reported in various animal and clinical studies.17–20 Inflammation seems to be associated with local osteolysis, bone resorption, implant loosening, and pseudarthrosis 21 and not only depends on the amount of particles but also on their size and shape. 17 However, today it is still unknown what amount of wear can be tolerated by the human body, which is dependent on the chemical composition of the wear particles, their size, and shape. In view of this lack of knowledge it is recommended to keep wear to a minimum and, thus, to use a coating that strongly adheres to the substrate and that is as resistant to abrasion as possible. Only then, the implant will preserve its biological function and prevent inflammatory reactions to abraded material.
Another approach to interpret the results is to use available FDA (U.S. Food and Drug Administration) acceptance criteria for comparison. The FDA guidance document on the testing of metallic plasma sprayed coatings on orthopedic implants allows a weight loss of 65 mg. This amount of wear, however, is generated through a much larger coating area than that of a lumbar PLIF cage. For the contact area estimated in the case of the PLIF cages tested in this study, the accepted amount of wear would be approximately 1.7 mg. The setup mentioned in this guidance document and the setup used in the present study are different, but those 1.7 mg may still serve as a rough estimation of the harmless threshold for a PLIF cage. In this case, the wear measured for the Ti plasma spray coated cage is above the limit, the wear for all other cage types is presented in Figure 5.
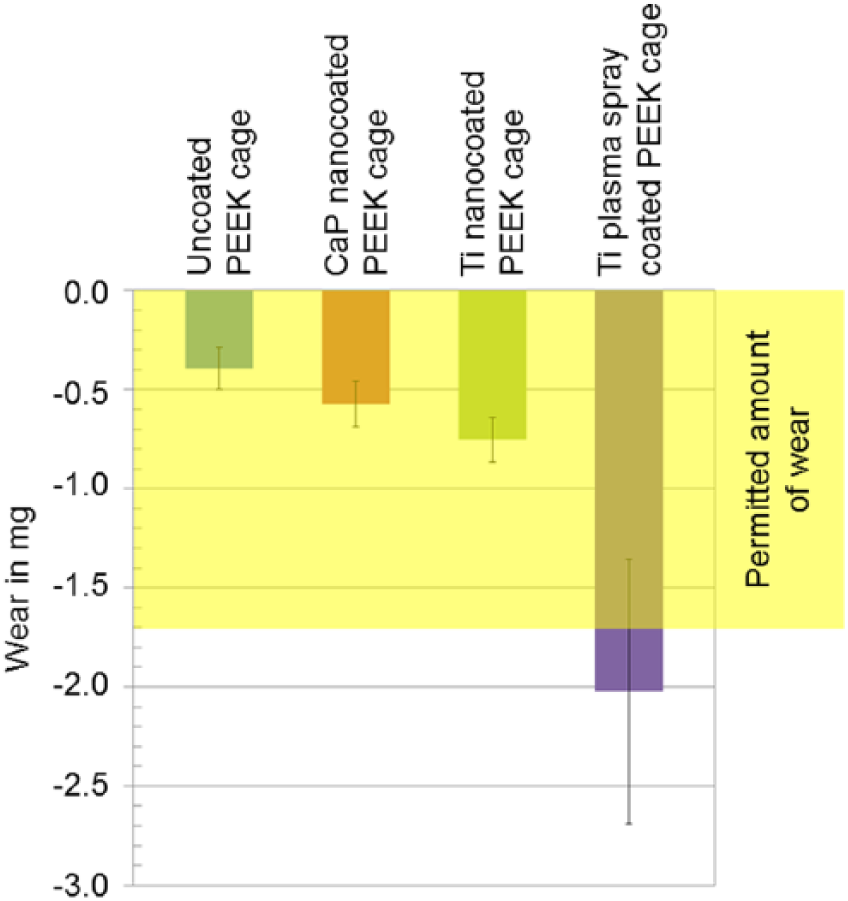
Mean wear of all tested specimens with standard deviation. The yellow area represents the permitted amount of weight loss (assigned to the implant surface, which was in contact to the vertebral body substitutes) derived from FDA Guidance Document 946.
Conclusions
In conclusion, all cages, including those without coating, lost weight due to impaction into the disc space. The amount of weight loss, which is equivalent to the amount of surface abrasion, was smallest for the uncoated PEEK PLIF cage with a mean values of 0.39 mg, followed by the CaP nanocoated cage (0.57 mg) and the Ti nanocoated cage (0.75 mg), and it was highest for the Ti plasma spray coated cage (2.02 mg). These differences depend on various factors, such as the bonding strength of the coating on the substrate and the coating thickness. From a clinical point of view, the most important question is whether the abraded material bears any risk for the patient. In general, abraded material can initiate inflammatory reactions with consecutive implant loosening. As long as there is no evidence of a harmless threshold for the amount of wear, it is advisable to choose a coating that strongly adheres to the substrate and that is as resistant to abrasion as possible.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Money was paid from Orthobion GmbH to SpineServ GmbH & Co. KG for services rendered during the conduct of the study.
