Abstract
Introduction:
Concrete structures in shaft linings are apt to deteriorate prematurely and therefore prompt restoration is required. In considering this, desulphurization fly ash and machine-made tuff sand are employed to fabricate a phosphate-based rapid repairing material.
Methods:
The long-term efficiency of the material is evaluated based on combined factors, so drying shrinkage, interfacial bonding strength, corrosion resistance, and combustibility of the specimens are tested and researched in this paper.
Results:
Experimental results showed that, under a dry circumstance, the material goes through a minor expansion at an early stage. It goes into a stage of rapid contraction after one day and a stable contraction after seven days. After 28 days, the total deformation is 67 micro-strains. On the other hand, the fabricated material manifests an excellent mechanical property. The one hour bending strength and compressive strength were 9.2 MPa and 32.6 MPa, respectively. A long-term mine water flushing simulation demonstrates that only 10% bending strength is lost and the corrosion resistance coefficient stays above 0.8, so a very good corrosion resistance is thus achieved.
Conclusions:
What is more, this repairing material retains its stability even at a high temperature of 1000°C, revealing its good thermo-stability. All these prominent properties make it a good prospective material for real restoration applications.
Introduction
Shaft lining is such a crucial part of the whole coal mine system that mastering the working state of the shaft lining is fundamental to ensure coal mine safety. Concrete is widely used as the structural material for shaft lining. Unfortunately, due to complex geological and environmental conditions, these shaft linings tend to be subject to the interaction of continuous loads and chemical erosion, which ultimately destroys the concrete structure. 1 Therefore, it is important to repair the damaged shaft linings quickly to improve their durability and safety. Cement-based repair materials are generally used as concrete repair patches. However, these repair materials will cause a prolonged interruption of the service of the coal mine due to the slow development of early strength. Furthermore, these materials can generate cracks and shelling, thereby decreasing concrete structure durability. Solfo aluminate cement has a rapid setting period and high strength within hours, but it is vulnerable to weathering and easy to form surface cracks, which are not resistant to high temperature, and even cracks when it is not cured. The stable hydration product of solfo aluminate cements under high temperature is calcium monosulfate aluminate hydrate, which will translate into delayed ettringite under subsequent room temperature and cause strength deterioration of the cement. Plus, the concrete structures in coal mines often suffer from high temperature sudden events. 3 Expansive agents seem to be able to prevent cracking and spalling, but the effective expansion only occurs under sufficient water curing. The shrinkage fall after expansion is greater than that of the specimens without expansive agents if subsequently located in dry environments, which will cause serious cracking. 4
Recently, Magnesium phosphate cementitious material5–6 was prepared from the chemical reaction of dead-burned magnesia oxide (MgO), potassium dihydrogen phosphate (KH2PO4), and some retarders in a given proportion used in the same manner as Portland cement. Magnesium phosphate cementitious (MPC) material has been considered as an ideal rapid repairing material due to its early strengthening, high adhesiveness and good durability,6–7 fire resistance, and slight expansibility. But the MPC also has some shortcomings, such as high production cost, 5 which can restrict its development to some extent.
Meanwhile, with the development and promotion of circulating fluidized bed boiler and desulfurization technology, large amounts of desulfurized fly ash have been produced. The desulfurized fly ash is a mixture of ordinary fly ash, desulfurization products, and remained desulfurizer. Therefore, it has a variety of adverse chemical compositions for sufficient soundness in cement and concrete, such as high content of SO3, free CaO, and large amounts of additional materials (insoluble residue (IR) and loss on ignition (LOI)). 8 As result, desulfurized fly ash is mostly disposed of by means of piling up in the open-air at present, which uses a great quantity of land and causes serious damage to ecological environments.9–13
Compared to the silicate cementitious materials, the phosphate cementitious materials have a low requirement for raw material. It is believed that by adding desulfurized fly ash into the phosphate rapid repairing material the costs will be reduced, which provides new thought for resource utilization of desulfurized fly ash.
A great deal of research has investigated the physical and mechanical properties of MPC material. 6 , 14–16 However, there has been relatively little research attention given to the utilization of desulfurized fly ash into MPC material, and even less attention to its long-term performance, especially the durability in the special service environment of shaft lining structures.
Compared with engineering in the atmospheric environment, shaft lining structures tend to be subject to the interaction of multiple damaging factors, as previously described; for example, internal air environment and external corrosion in the rock or soil stratum by groundwater. Groundwater contains many corrosive substances, such as SO42-. Moreover, inner walls of shaft linings that are exposed to high temperatures and dry environments even suffer from burning effects.
Therefore, this paper is going to investigate the long-term behaviors and durability of phosphate-based rapid repairing material (MKPC) for shaft lining structures, through the testing of interfacial bond strength, drying shrinkage, corrosion resistance, and combustibility.
Experimental programs
Materials
In the experiments, dead-burned magnesia oxide (MgO), potassium dihydrogen phosphate (KH2PO4, or KDP), borax (Na2B4O7.10H2O), fine aggregate, and desulfurized fly ash (DFA) were mixed together as the MKPC.
The binders used in this work included dead-burned MgO, KDP and DFA. Among these are:
1) Dead-burned MgO (M) that was produced by a company in Xinghua, Jiangshu province, China. It was a pale yellow powder, which calcinated at 1853.15 K (2912°F) for four hours before being ground to powder. Its particle size distribution was ascertained by sieving: the percentage of the total mass in particles > 0.16 mm (0.0063 in), 0.16-0.08 mm (0.0063~0.0032 in), and < 0.08 mm (0.0032 in) was respectively 2.9%, 37.2%, and 59.9%. The chemical composition can be found in Table 1.
2) KDP (P), industrial grade white crystalline powder with 98% KH2PO4, which was provided by a company in Ningbo, Zhejiang province, China. Its granularity: over 95% (80–100 mesh).
3) DFA that was produced by Ningbo Changfeng Thermoelectric Co. Ltd, Zhejiang province, China. Its water demand ratio: 106%; the SO3 content: 2.26%; and loss on ignition: 1.02%. The chemical compositions are given in Table 2.
4) Borax (B): analytical reagent grade colorless transparent crystalline powder, which was provided by a company in Wuxi, Jiangsu province, China.
5) The fine aggregate (S): crushed tuff sand with a fineness modulus of 3.0 and stone powder content of 8%, which was produced by a company in Ningbo, Zhejiang province, China. Its maximum particle size: 5 mm (0.197 in).
6) Silane water repellent (SWR): industrial grade white emulsion with 40% active substance, which was provided by a company in Wuhan, Hubei province, China.
Chemical composition of dead-burned magnesia wt %.
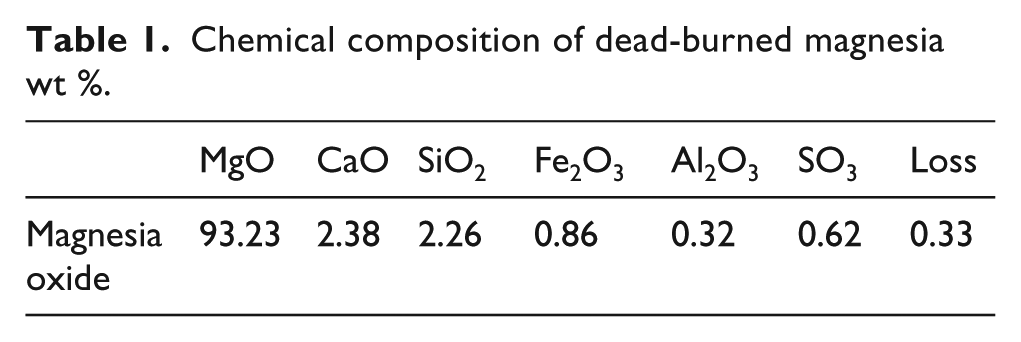
Chemical constituents of desulphurization ash wt %.

Methods
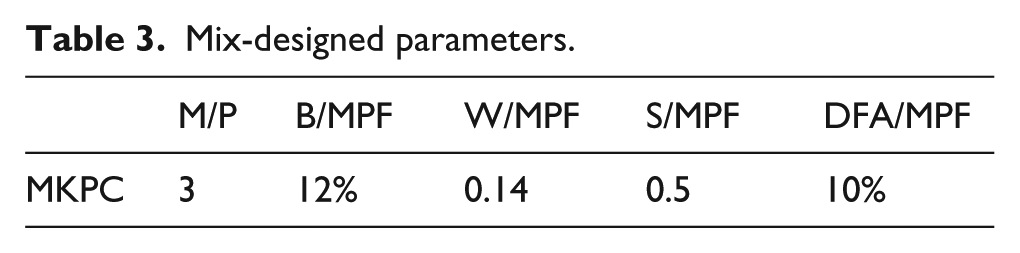
For the design of the MKPC, the MgO to KDP ratio (M/P), borax dosage (B/MPF), water to binder ratio (W/MPF), sand to binder ratio(S/MPF) and DFA addition (DFA/MPF) were chosen as key parameters. MPF is the total mass of MgO, KDP and DFA. The mix parameter is shown in Table 3, according to the previous work from the authors. 17 SWR of 0.2% MPF in weight was added.
Mix-designed parameters.
All raw materials were placed under room temperature conditions for 24 hours. Subsequently, all powdered materials, including MgO, KDP, borax, and DFA, were dry-mixed in an intensive mixer for 120 seconds with sand, and then the water was added and mixed for another 60 seconds.
After testing for the setting time, the initial flow, each mix was cast in to various steel molds and then compacted to measure the strength and long-term behaviors. Immediately after casting, all specimens were cured in room conditions (298.15–308.15 K (77~95°F), 55-65% RH) until the testing age. All steel molds were removed at an age of 0.5 hours.
The setting time was recorded by using a modified Vicat needle, according to the GB/T 1346 standard. The initial flow was measured, in accordance with GB/T 2419 standard, using a cone-shaped mold having a total height of 60 mm (2.364 in), a bottom diameter of 100 mm (3.94 in), and a top diameter of 70 mm (2.758 in). The compressive strength and bending strength testing, using specimens with the dimension of 40 mm × 40 mm × 160 mm (1.575 in × 1.575 in × 6.299 in), was conducted in accordance with the GB/T 17671 standard.
MKPC paste bar specimens with size of 25 mm × 25 mm × 280 mm (0.984 in × 0.984 in × 11.024 in) were used for the drying shrinkage test according to the JC/T 603-2004 standard. The length of MKPC paste specimens after the 0.5 hours hydration age, which was as the initial length of specimens (L0), was measured by a non-contact type laser sensor. The specimen length (Lt) after t-d hydration ages was measured. Then the linear deformation (εt) was calculated as follows
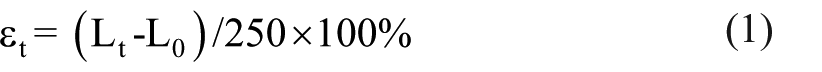
where 250 represents the effective length of the specimens.
Acidic mine water is distributed extensively in south China, 14 which is mainly sulfate-sodium. This mine water is therefore simulated by a solution of 3% Na2SO4 (mass density). After three months standard curing, C40 concrete specimens with aggregate size less than 4.75 mm (dimension: 40 mm × 40 mm × 160 mm (1.575 in × 1.575 in × 6.299 in)) were cut into two pieces. The broken concrete surface was dried and cleaned with a brush, and put into a mold with the size of 40 mm × 40 mm × 160 mm (1.575 in × 1.575 in × 6.299 in). Subsequently, the MKPC was poured on the mold. After three days, the steel molds were removed, and the specimens were divided into two sets: one set was kept under the simulated mine water, and the other set was cured in room conditions for contrastive analysis at the same stage. Then, the interfacial strength loss ratio (&t) was calculated as follows
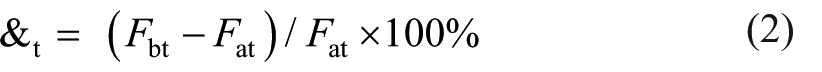
where Fbt is the interfacial strength after immersion in water for t days; Fat is the interfacial strength cured under the same days in room conditions.
MKPC specimens with a size of 10 mm × 10 mm × 60 mm (0.394 in × 0.394 in × 2.362 in) were used for the sulfate attack test, referring to the GB 2420 standard. The corrosion resistance of MKPC was evaluated by use of a corrosion resist coefficient K, 15 which can be calculated as follows

where fb is the bending strength after immersion in water for t days; fa is the bending strength cured under the same number of days in room conditions.
In addition, in order to evaluate the high temperature damage resistance of MKPC, the specimens with size 40 mm × 40 mm × 160 mm (1.575 in × 1.575 in × 6.299 in) were used for the combustibility test. All specimens were demolded in 0.5 hours and then cured in room conditions for three days. Subsequently, the specimens were divided into two sets. One set was kept in a muffle furnace at combustion temperatures of 573.15 K (572°F), 873.15 K (1112°F), and 1273.15 K (1832°F) with 277.15 K/min (39.2 K/min) temperature increasing rate, two hours of constant temperature, and cooling to room temperature. The other set was cured in room conditions for contrastive analysis at the same stage.
Then the strength loss ratio (W) at high temperatures was calculated as follows
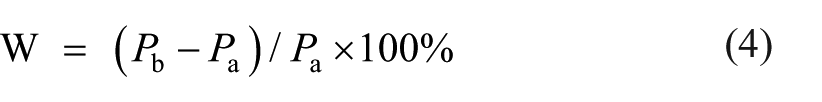
where Pb is the strength of specimens at high temperatures; Pa is the strength cured under the same number of days in room conditions.
Results and discussion
Basic properties
The basic properties were obtained for MKPC as shown in Table 4. The MKPC hardened quickly; the setting time was 25 minutes. It had high early strength in that 1-hour flexural strength and compressive strength reached 9.2 MPa (1334 psi) and 32.6 MPa (4728 psi), respectively. In addition, it had a 28-day compressive strength above 62.7MPa (9094 psi).
Performance test results of MKPC.

According to the construction requirement for an on-site rapid repairing material, 18 the setting time must be longer than 15 minutes for small area repairing and higher than 20 minutes for large area repairing.19–20 A set-retarding admixture could be useful in this case. Pure phosphonate, hybrid phosphonate, or traditional naftalene or acrylic admixture can represent an efferent tool to reduce water demand and prolong the pot life of MKPC. 21 In addition, the 1-hour compression strength should reach 20 MPa (2901 psi). Based on these regulations, the MKPC satisfies the requirements of rapid repair for shaft linings.
Long-term behavior
Drying shrinkage
The drying shrinkage of the rapid repairing material will significantly affect the long-term material performance. Therefore, based on the result of previous tests in this paper, the evaluation of the drying shrinkage for the MKPC was carried out and the results are shown in Figure 1. According to Figure 1, the drying shrinkage curve of MKPC could be divided into three stages, as follows.
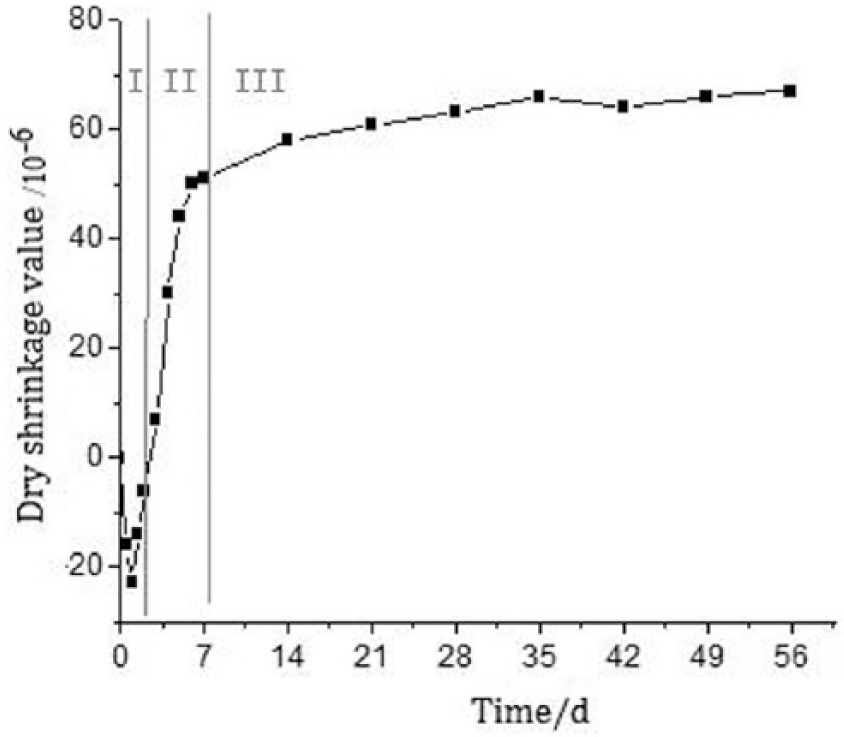
Dry shrinkage of MKPC repairing materials.
First, the slight swelling stage (before one day); in this stage, the maximum swelling is 20 micro-strain. Second, the rapid shrinkage stage (1–7 days); the shrinkage of seven days is 50 micro-strain, which is 75% of 28 days. Third, the stable shrinkage stage (after seven days); the shrinkage of 28 days is 67 micro-strain, which is consistent with experimental values existing in the literature.22–23 This indicates that the drying shrinkage property of MKPC is much lower than the common repairing materials 2 , 18 such as OPC mortar, 3000–5000 micro-strain, OPC concrete, 600–900 micro-strain, epoxy mortar, 700–1000 micro-strain, and polymers, 200 micro-strain. There is, therefore, better volume stability for the MKPC repairing material.
Figure 2 shows the results of interface strength between the repairing material and concrete under air curing conditions. Figure 3 shows the interface strength and its loss rate of specimens after the repairs are exposed the simulated mine water for seven days, 28 days, and 56 days. According to these two figures, the rapid improvement of interface strength could be obtained in the first day. The increase rate slowed down in the first to the seventh day, and then became smooth after seven days curing time. The 1-hour and 1-day interface strength can reach 3.3 MPa (479 psi) and 4.5 MPa (653 psi) respectively, which is 60% and 80% of the 56-day strength. And the 7-day and 28-day interface strength is 5.2 MPa (754 psi) and 5.4 MPa (783 psi), which is 93% and 96% of the 56-day strength respectively. For the C40 concrete, the 90-day compression and bending strength is 46.2 MPa (6701 psi) and 6.2 MPa (899 psi), respectively. Therefore, it can be found that the 1-day interface strength of MKPC could be 4.5 MPa (653 psi), which is 75% of the bending strength of the concrete.
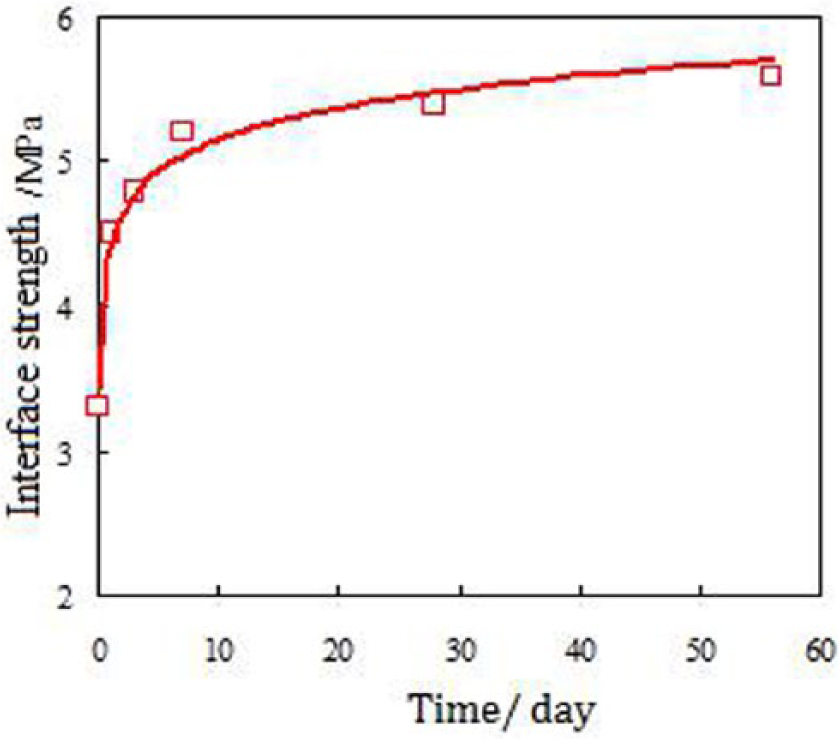
Interfacial strength at air curing.
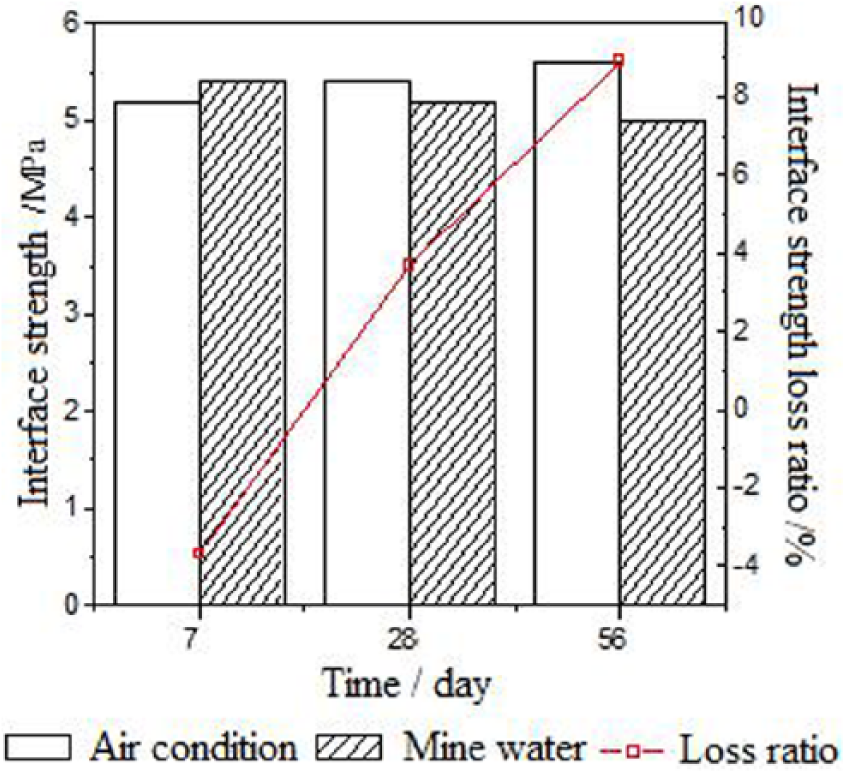
Interfacial strength and its loss ratio under the mine water.
Moreover, for the MKPC cured in the mine water conditions, the 7-day interface strength is higher than the air cured MKPC. A small drop occurred after 28 days, which is less than 10%. The proper reason could be hydration reaction between MgO and phosphates caused by mine water in the early hydration. At the same time, lower amounts of phosphates and hydras released in the early stage resulted in the improvement of early interface strength. However, the strength dropped once the hydration reaction ceased and increased the level of released hydras in the later stage.
Corrosion resistance coefficient K
Figure 4 shows the results of the bending strength and corrosion resistance coefficient for the MKPC under air and mine water curing conditions. According to Figure 4, strength loss would happen because of the corrosion of mine water; and the corrosion coefficient K is 0.95, 0.9, and 0.82 for 56 days, 180 days, and 360 days, respectively. According to the experiment results, the corrosion coefficient K is 0.98, 0.93, and 0.88 for 56 days, 180 days, and 360 days, respectively, under drinkable water conditions, which indicates that the corrosion resistance of the MKPC under mine water is lower than that under drinkable water. Referring to the standard GB 2420-81, the sulfate resistance is in good agreement with the rest results since the 12-month corrosion resistance coefficient is bigger than 0.8.
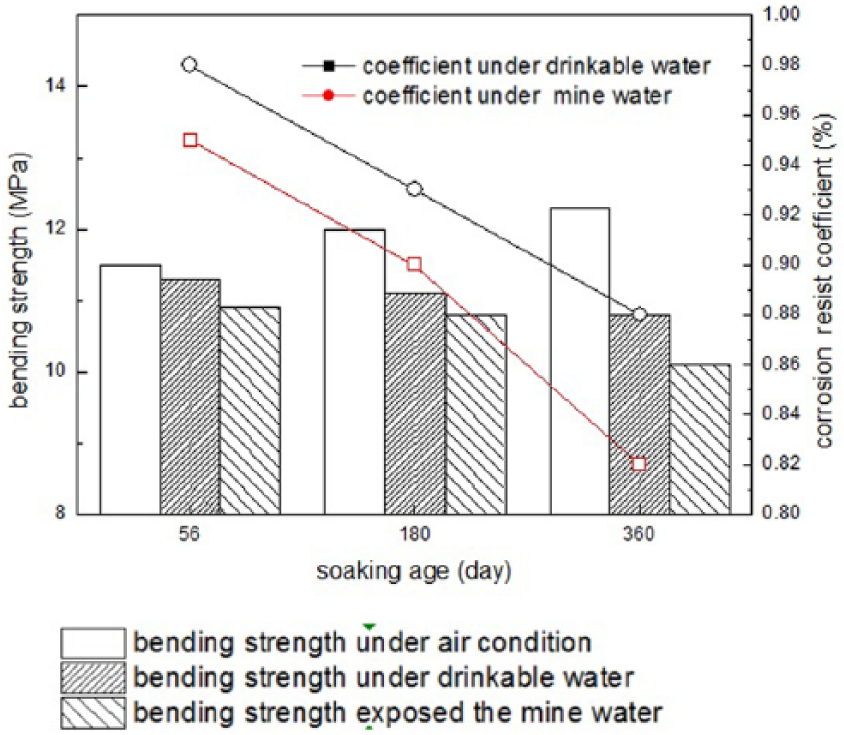
Bending strength and corrosion resistance coefficient of the MKPC under air or mine water curing conditions.
High temperature damage resistance
The same grade of Portland cement-based mortar was used to perform as the control group samples in this paper. The comparison between MKPC and the control samples was applied in order to evaluate the high temperature damage resistance of MKPC when mines burst into flames.
Figure 5 shows the appearance changes of MKPC and the control samples under different temperature conditions. According to Figure 5, the color of MKPC would change from light brown to black when the temperature increases from room temperature to 573.15 K (572°F). However, it would turn back to brown if the temperature reaches to 873.15 K (1472°F). There was less change of color for the control samples. Cracks were formed at 573.15 K (572°F) and become wider at 873.15 K (1472°F). Finally, the samples failed at 1273.15 K (1832°F).

Appearance changes of samples under different temperature conditions.
Figure 6 shows the changes of compression strength and its loss rate for the MKPC and OPC at different temperatures. According to the results, the compression strength for both materials decreased by different degrees. The compression strength decrease rate of OPC is much lower than MKPC when the temperature is below 573.15 K (572°F). The loss rate for OPC and MKPC is 14.3% and 67%, respectively. This is consistent with experimental values existing in the literature. 24 However, the compression strength decrease rate of OPC will quickly rise up once the temperature increases to 873.15 K (1472°F). The strength loss of MKPC tends to be stable. Finally, the OPC samples fail at 1273.15 K (1832°F), but the MKPC still has integrity (72.5% loss rate) and the compression strength could reach to 18.7 MPa (2712 psi) at 1273.15 K (1832°F). Above all, the results indicate that, compared to the OPC repairing material, MKPC has better temperature stability, especially for the high-temperature stage.
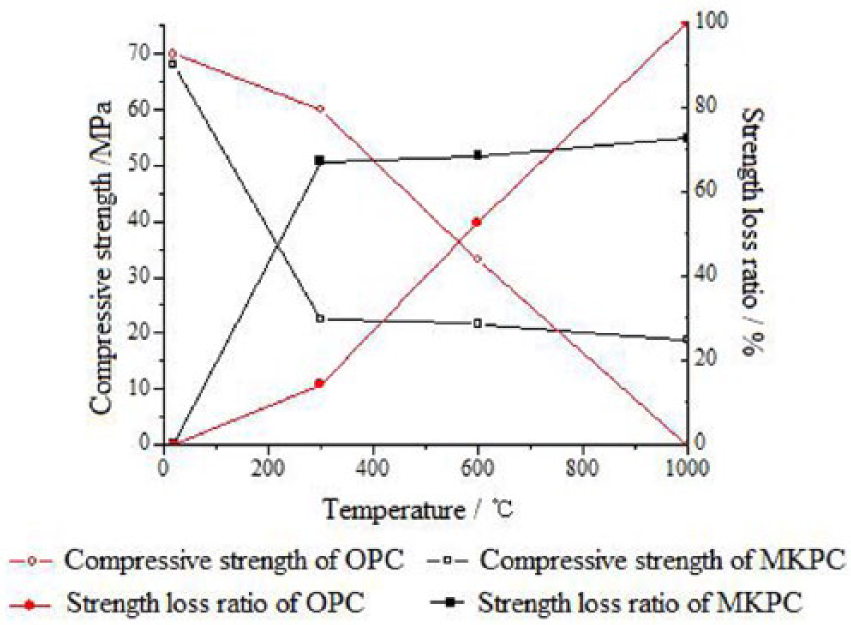
Compressive strength and its loss ratio of samples in different temperatures.
Conclusion
(1) The MKPC satisfies the requirement of rapid repair for shaft linings that are prepared with DFA and machine made tuff sand.
(2) The drying shrinkage curve of MKPC could be divided into a slight swelling stage (before one day), a rapid shrinkage stage (1–7 days), and a stable shrinkage stage (after seven days). Its shrinkage at 28 days is 67 micro-strains, which is much lower than the common repairing material.
(3) The rapid improvement of interface strength of MKPC could be obtained in the first day. The increase rate slowed down in the next 1–7 days, and then became smooth after seven days curing time. The 1-hour interface strength can reach 3.3 MPa (479 psi), which is 60% of the 56-day strength.
(4) A long-term mine water flushing simulation demonstrates that only 10% bending strength is deduced, with corrosion resistance coefficient stays of one year above 0.6, so a very good corrosion resistance is thus achieved.
(5) MKPC retains its stability even at a high temperature of 1273.15 K (1832°F), which thus reveals its good thermo-stability.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support from the Zhejiang Province Natural Science Foundation (LY15E080014), Ningbo Municipal Major Science and Technology Projects (2013C51004), and State Key Laboratory of Chemical Resource Engineering (No.CRE-2015-C-114).
