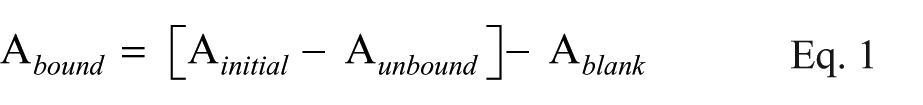Abstract
Introduction:
Magnetic nanoparticles (MNPs) are one of the most useful particulate systems in analytical applications such as specific aptamer selection. Proteins are the most noted targets of aptamer selection. Generally, covalently immobilized protein coated MNPs are more stable structures.
Methods:
In this study, coagulation factor VIII (FVIII) was immobilized on MNPs. A silica coating provided isocyanate functional groups was considered to interact covalently with reactive groups of the protein, resulting in a stable protein immobilization. The reactions was run in dried toluene. At end, these MNPs were applied for affinity determination of a previously selected FVIII specific aptamers.
Results:
Immobilization of 1 mg FVIII (~ 3 nmol) on 5 mg particles was achieved with no significant particle aggregation. Using a fluorescence-based method, affinity measurement resulted in a calculated dissociation constant of 120 ± 5.6 nM for the FVIII-specific aptamer to the FVIII-coated MNPs.
Conclusion:
The final product could be a suitable protein-coated solid support for magnetic-based aptamer selection processes.
Keywords
Abbreviations
FVIII Coagulation factor VIII
FVIIa Coagulation factor VIIa
FTIR Fourier transform infra-red
Kd Dissociation constant
MNP Magnetic nanoparticle
PBS Phosphate buffer saline
PVA Polyvinyl alcohol
SELEX Systematic evolution of ligands by exponential enrichment
TEOS Tetraethyl orthosilicate
TEPI 3-(triethoxysilyl) propyl isocyanate
Introduction
Nanoparticles, especially magnetic nanoparticles (MNPs), are important in analytical systems in medical research fields due to their valuable features such as small size, simple preparation, significant magnetic response, and, therefore, easy separation from the reaction mixture. Recently, different forms of iron-based magnetic nano-composites have been fabricated and applied in various fields of analytical, environmental, and medical sciences.1–6 Preparation of Fe3O4 nanoparticles has been reported by diverse methods, including, but not limited to, simple sonochemical synthesis using FeCl2 as a single precursor and without using inert atmosphere (resulting in a polyvinyl alcohol (PVA)-Fe3O4 nanocomposite), surfactant-free hydrothermal reaction, chemical sol-gel and auto-combustion processes, electro-spinning methods, and precipitation reactions at room temperature and ambient atmospheric conditions.7–12
Functionalization of MNPs by immobilization of biomolecules such as proteins on magnetic particles has numerous biotechnological applications, such as target purification, cell separation, immunoassay, magnetic resonance imaging, drug delivery, and other clinical and analytical applications.13–17 Naked iron oxide MNPs are prone to oxidization and aggregation instabilities, thus different types of polymer or non-polymer based coating have been applied to improve stability and applicability of Fe3O4 MNPs. 18 Proteins can also act as a functional coating on different nanocarriers such as MNPs to stabilize and improve their bioactive features. 19 In general, coating of Fe3O4 MNPs with protein can be achieved using chemical intermediates that induce covalent binding of protein functional groups to the surface hydroxyls of iron MNPs. For example, epichlorohydrin has been used to facilitate an epoxy cross linking between protein and MNPs. 20 Some other studies stabilized naked iron MNPs with polymer coatings such as silica, and then added functional groups to interact with proteins for covalent binding. In this regard, enzyme immobilization has been studied extensively. 21 Oriented attachment of proteins to MNPs is also highly important, especially in enzyme immobilization or in the design of a nanocarrier-conjugated protein therapeutic agent, since this type of immobilization can result in maintenance of protein activity after attachment. In this regard, different ligand coating strategies, such as agarose coating for LSL fusion proteins, His tag specific aptamer coating for his tag fusion protein, and protein A coating for monoclonal antibody attachment have been evaluated.22–24 Silica coating on MNPs has been applied extensively in various fields of research. 25 Different silica derivatives with functional groups (e.g. amine, phosphonate, or isocyanate) facilitate attachment of biomolecules to nanocarrier systems.26–28 3-Aminopropyltriethoxysilane (APTS) is one of the silica derivatives that has been used widely to introduce amine groups onto the MNP surface to facilitate covalent attachment of proteins.29,30 3-(Triethoxysilyl) propyl isocyanate (TEPI) is another silica derivative with an N=C=O group that can be functionalized as a nanocarrier for biomolecule attachment.31,32
In this study (Figure 1), functional MNPs were synthesized using TEPI as the substrate for a second layer silica coating. The isocyanate groups on the surface of the MNPs facilitated the covalent attachment of coagulation factor VIII (FVIII) to the nanoparticles. The FVIII-coated MNPs showed promising results in FVIII-specific aptamer affinity measurements, and could be applied to specific aptamer selection processes using systematic evolution of ligands by exponential enrichment (SELEX).
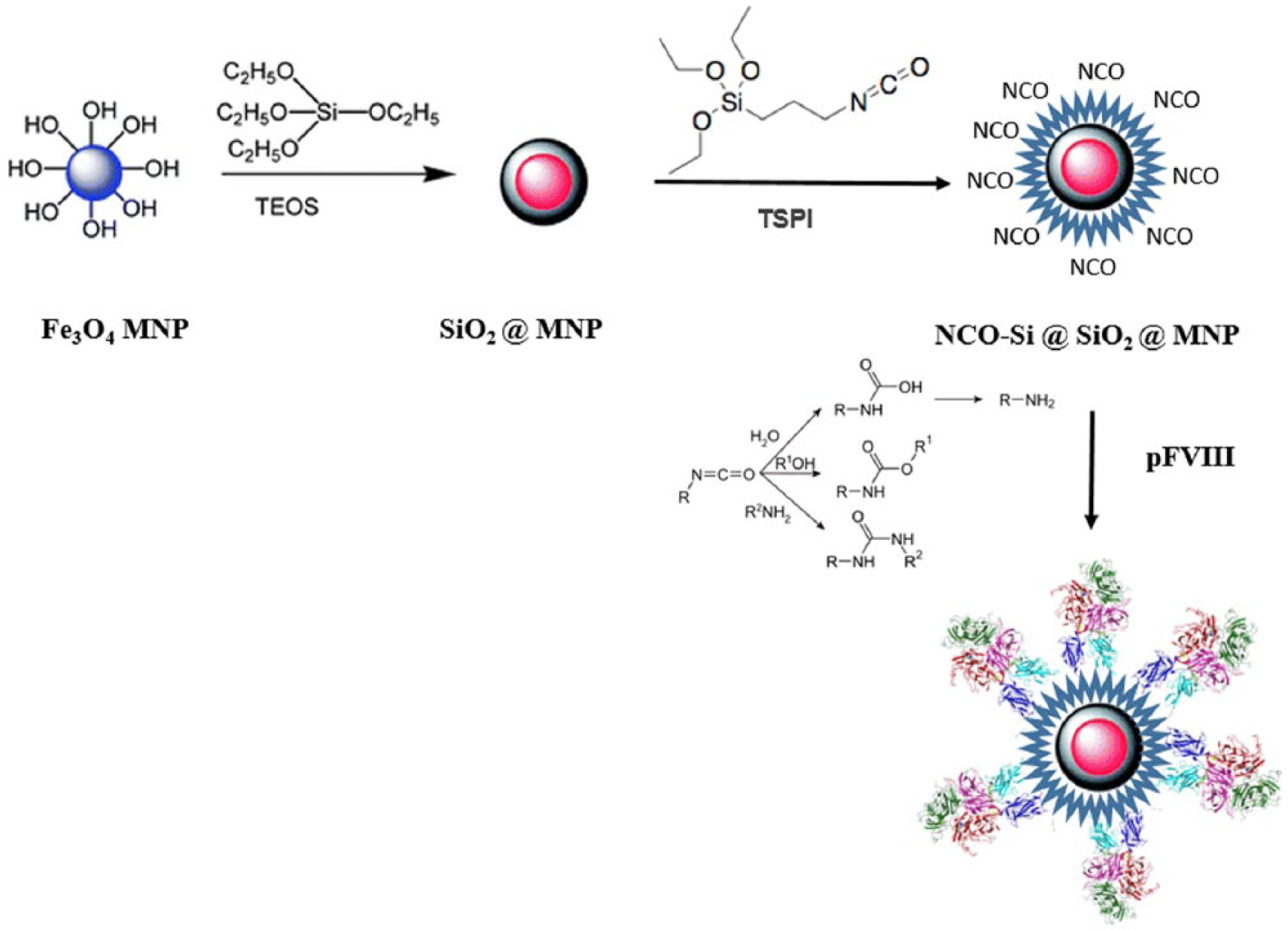
Schematic presentation of the coagulation factor VIII (FVIII) coating on magnetic nanoparticles (MNPs).
Materials and Methods
Chemicals
The plasma FVIII sample was Haemoctin® 500 (Biotest, Birmingham, UK) supplied from the Iranian Blood Research and Fractionation Company. The iron salts, FeCl3·6H2O and FeCl2·4H2O, and PVA 15000 were supplied from Merck chemicals (Darmstadt, Germany). Electrophoresis chemicals, tetraethyl orthosilicate (TEOS) and TEPI were purchased from Sigma (St. Louis, MO). Better Bradford Protein Assay Kit of BioBasic (Markham, ON, Canada) was used for protein quantification.
Synthesis of Fe3O4 nanoparticles
PVA 15000 (1 g) was dissolved in 15 mL of deionized water and then FeCl2·4H2O (0.9 g, 4.5 mmol) was added. A solution of FeCl3·6H2O (1.3 g, 4.8 mmol) in water (15 mL) was added to the previous mixture. The reaction was left stirring for 0.5 h at 80°C. Then, NaOH (1.0 mol/L) was added dropwise under vigorous stirring to produce a black solid product when the reaction medium reached pH 10. This was followed by heating the reaction for 2 h at 60°C in a water bath; the black magnetite solid product was separated using an external magnet, washed with ethanol three times, then dried at 80°C for 10 h. 33
Silica coating of Fe3O4 nanoparticles
Core–shell Fe3O4@SiO2 nanospheres were prepared by a modified Stober method. 34 Briefly, Fe3O4 (0.50 g, 2.1 mmol) was dispersed in a mixture of ethanol, deionized water, and TEOS (in the volume ratio of 50:5:0.20). Then, 5.0 mL of NaOH (10% w/v) was added to the mixture. This solution was stirred vigorously for 30 min at room temperature. The final product, Fe3O4@SiO2, was separated by an external magnet, washed with ethanol three times, then dried at 80°C for 10 h. The second silica layer was coated using TEPI for addition of isocyanate as reactive groups on the MNP surface. A 500 mg sample of the Fe3O4@SiO2 was dispersed in 20 mL dried toluene and stirred vigorously for 30 min at room temperature. Then 50 µL of TEPI was added dropwise with vigorous stirring for 8 h. Finally, functionalized Fe3O4@SiO2 nanoparticles were separated by an external magnet, washed three times with dried toluene, the, dried at 60°C for 10 h.
FVIII coating on functionalized MNPs
Haemoctin is a plasma-derived FVIII that comes in vials of 5 mg protein as a lyophilized powder. Each vial was reconstituted by adding 1 mL of PBS, pH 7.4. A highly concentrated stock solution of FVIII (5mg/mL) was prepared. Different mass ratios of protein to MNPs (0.5, 11, and 2) were prepared in 500 µL of dried acetonitrile and incubated in room temperature under mild shaking for 2 h. Then, protein-coated MNPs were separated using an external magnet and washed twice with 250 µL of PBS. Finally, residual protein in the supernatant was measured using a Better Bradford Protein Assay Kit, using acetonitrile-PBS (1:1 v/v) mixture as the blank.
MNP characterization
Particle size analysis was performed by the dynamic light scattering (DLS) method using a ZetaSizer model Nano ZS-ZEN3600 (Malvern, UK). Samples (at a concentration of 1 ng/mL) were analyzed in 0.1 M phosphate-buffered saline (PBS), pH 7, at room temperature. Particle morphology was evaluated by transmission electron microscopy (TEM) using a Zeiss EM10C 80 KV (Zeiss, Jena, Germany). X-ray diffraction (XRD) measurements were made at room temperature using Cu K radiation (Philips, PW1730). Chemical composition was analyzed using energy dispersive spectrometry (EDS) analysis using an XFlash 6130 detector (Bruker).
Fourier transform infrared (FT-IR) spectra were recorded (Perkin Elmer spectrometer, USA) at room temperature. UV–visible (UV–vis) absorption spectroscopic measurements were recorded using ScanDrop (Analytik Jena, Germany).
Affinity measurement of FVIII-specific aptamer using FVIII-coated MNPs
One previously selected FVIII-specific aptamer was considered in the affinity assay using recently developed FVIII-coated MNPs. 35 The fluorescent based method was applied for affinity determination. The FAM-labeled aptamer sequence was diluted to a set of 12 concentrations (from 0.5 to 700 nM) and incubated with 100 ng FVIII-coated MNPs in 500 µL binding buffer (Tris 40 mM, NaCl 100 mM, CaCl2 5 mM and MgCl2 5 mM, pH 7.4. A set of eight concentrations of labeled aptamer (0 to 1 µM) was prepared as standards to draw a calibration curve of fluorimetry. A non-specific 81-nucleotide FVIIa (coagulation Factor VIIa) aptamer was evaluated similarly in a cross-reactivity assay.
Magnetic separation was applied to separate bound from unbound oligonucleotides, followed by washing twice with 200 µL binding buffer. Albumin-coated MNPs were used as the blank. The fluorescence absorption of supernatant was then measured by fluorescence spectroscopy (F2500 fluorimeter, Hitachi, Tokyo, Japan). Excitation and emission wavelengths were set at 492 nm and 521 nm, respectively. Related concentration was calculated by standard curve and the concentration of bound oligonucleotides was extracted according to the fluorescence absorbance of samples considering Equation 1.
Statistics
The results were analyzed by GraphPad Prism 5 and the binding constant (Kd) was calculated according to the method of non-linear regression and binding-saturation, one-site total. The experiments were performed in triplicate. Mean ± SD was calculated using IBM SPSS Statistics, v21.
Results
Synthesis and characterization of MNPs
Fe3O4 MNPs were prepared by the co-precipitation method in alkaline conditions. A first layer of silica coating resulted in an improvement in MNP stability, as naked Fe3O4 MNPs were highly prone to oxidation and aggregation. A second functional silica layer was then added to Fe3O4@SiO2 MNPs. The particle size of these functional MNPs was 140 nm, compared to 390 nm for FVIII-coated MNPs. Functional silica coating of naked Fe3O4 MNPs resulted in an elevation in zeta potential to −1.46 mV, and the subsequent protein coating resulted in an even more negative zeta potential. The zeta potential of protein coated nano-composites was recorded as −8.32 mV. Particle size of naked and functional silica coated nanoparticles was also evaluated by TEM (Figure 2a and 2b, respectively), which confirmed a particle size of less than 12 nm.

Transmission electron microscopy (TEM) micrograph of naked (a), functionalized silica coated (b), and protein coated (c) MNPs.
The crystal structures of Fe3O4-, Fe3O4@SiO2-, and FVIII-coated final MNPs were analyzed by XRD (Figure 3). All the Fe3O4 peaks exhibited good agreement with the pattern for cubic crystalline magnetite Fe3O4. The sharp peaks that appeared at 2θ = 30.15° (276), 35.5° (774), 43.15° (224), 57° (307), and 62.60° (403) belonged to the nanocrystals of Fe3O4 structure. The XRD pattern of Fe3O4@SiO2 compared to the pattern of Fe3O4 showed similar peaks, with an extra weak peak at 21° (138) (3, 36, 37).
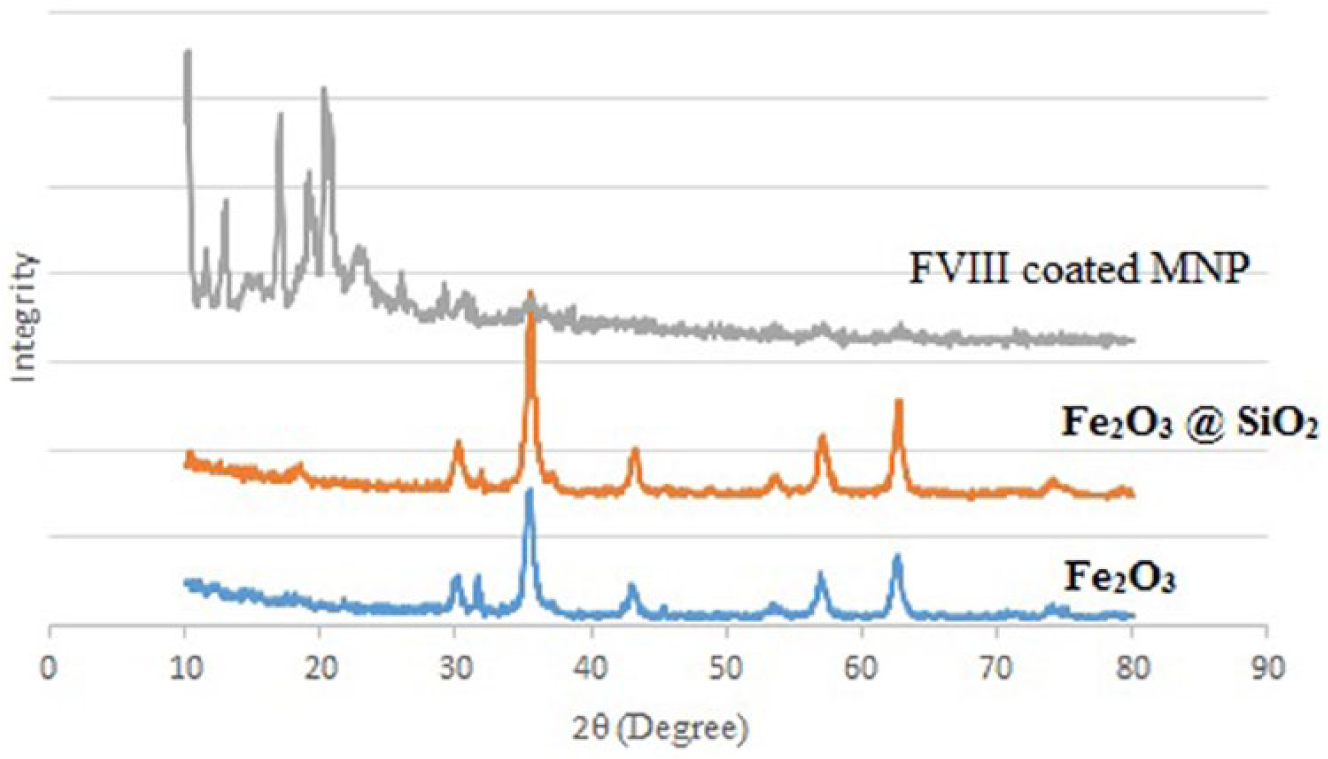
The X-ray diffraction (XRD) patterns of Fe3O4-, Fe3O4@SiO2-NCO- and FVIII-coated MNPs.
First and second layer of silica coatings were confirmed by Fourier transform infra-red (FTIR) spectroscopy (Figure 4). An absorption peak around 600 cm–1 indicated the stretching vibration mode of Fe–O and Fe–O–Si bonds, and an absorption peak around 1100 cm–1 typically related to the stretching vibration in Si–O–Si and Si–OH groups. The asymmetric vibration of Si–OH was observed at 956 cm–1 as reported in similar studies.36,38–40 Absorption peaks at 1590 cm–1 and 1630 cm–1 typically related to the stretching vibration nitrogen bound C=O, and the absorption peak at 2272 cm–1 was attributed to asymmetric and symmetric stretching of –N=C=O.37,38 Sharp peaks at 2800–3000 cm–1 were related to the stretching vibration of N–H bonds, which indicated relative interaction of the isocyanate group with H2O molecules in the medium, resulting in HN–C=O. Moreover, peaks above 3300 cm–1 are due to the adsorbed water in the sample. In addition, elemental analysis of functional silica-coated MNPs was performed by EDS analysis (Figure 5b). The chemical composition of these MNPs according to EDS analysis confirmed that the final product included Fe, O, Si, N, and C, which confirmed the production of desired functionally coated MNPs.
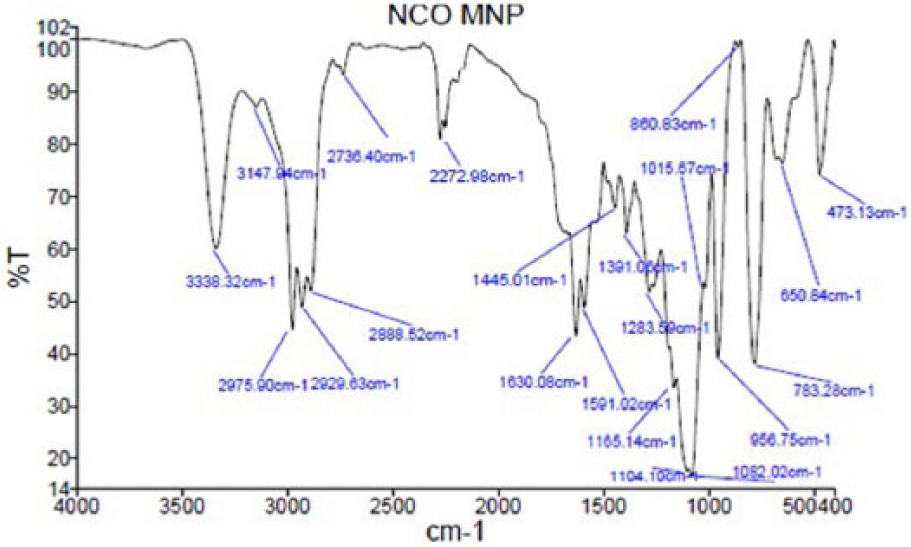
Fourier transform infra-red (FTIR) spectrum of functionalized silica coated MNPs.
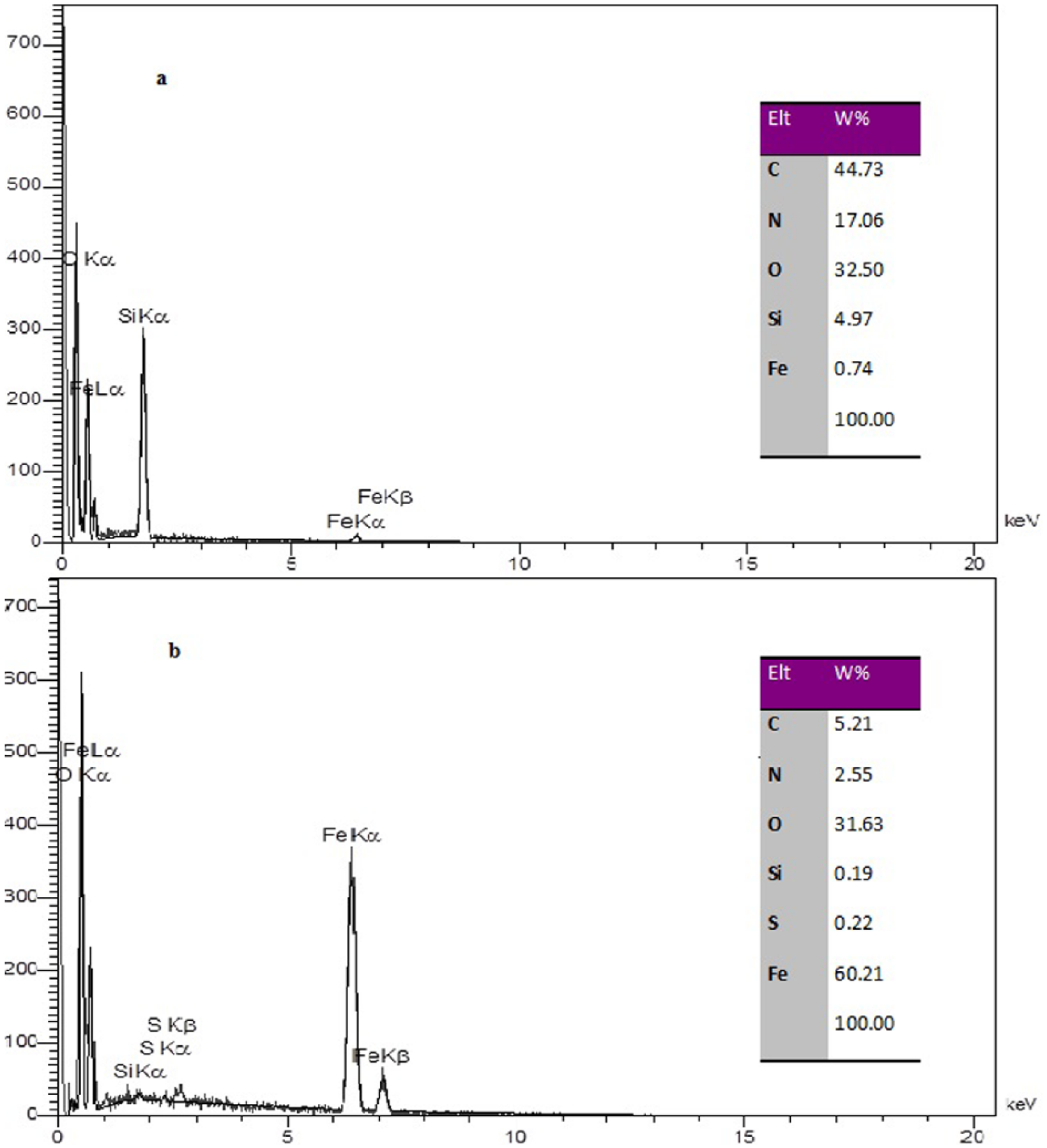
Energy dispersive spectrometry (EDS) curve of Fe3O4@SiO2-NCO (a) is FVIII-coated MNPs (b) is fe3o4@sio2-NCO.
The magnetic properties of nanoparticles were evaluated qualitatively by external magnet, and exhibited a separation time of 30 s for 100 ng of functional silica coated MNPs and about 40 s for 100 ng of FVIII-coated MNPs.
FVIII coating on functional MNPs
Two factors, i.e. protein to MNP mass ratio and time, were evaluated in the binding assay. Binding efficiency was calculated according to the percentage of residual protein in the supernatant after magnetic separation compared to the starting protein mass. The best binding efficiency was achieved at a mass ratio of 0.5% (w/w) FVIII to MNPs (Figure 6). Nanoparticles aggregated extensively at high protein concentration (FVIII). This might be the result of the large size of FVIII, which could adsorb non-covalently to more than one nanoparticle at once. The reaction was run in acetonitrile with minimal H2O content. In addition, the results showed that an incubation time of 2 h could result in good binding and that a longer incubation time would not increase the binding efficiency significantly. The incubation temperature was set at 20°C, due to protein instability over long incubations at high temperature.
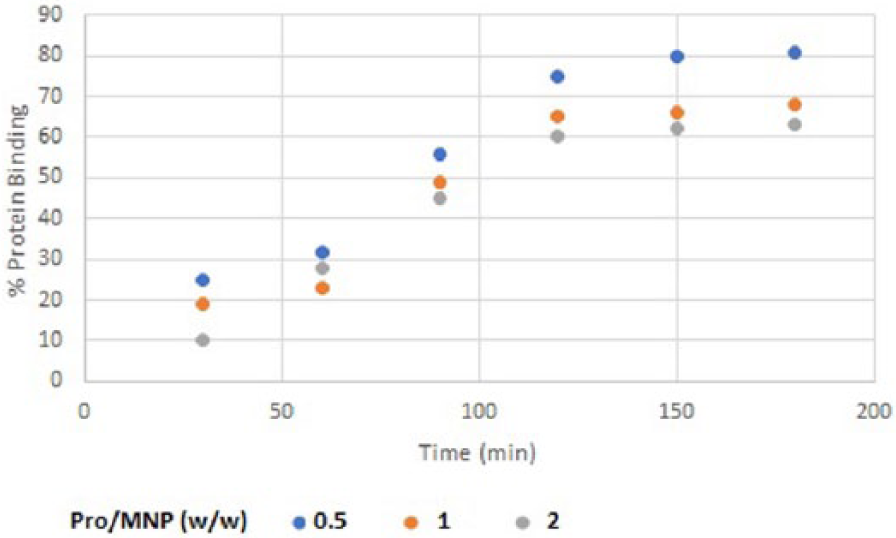
Percentages of FVIII binding to MNPs through 0–180 min.
FTIR analysis of protein-coated particles revealed that the plasma-derived FVIII was attached to the MNPs (Figure 7). Absorption peaks were detected around 2800–2900 cm–1 indicating the alkyl group-CH2, 1628 cm–1 for C=O of amide I, 1590 cm–1 for amide II, and above 3000 cm–1 for hydroxyl groups. Compared to the FTIR spectrum of non-coated MNPs (Figure 4), the two sharp peaks for amides I and II (1628 and 1590 cm–1) and a broad peak above 3000 cm–1 indicative of more hydration occurring around the protein shell, confirmed protein coating on MNPs. TEM analysis of the resulting particles confirmed the presence of nanoparticles with a size of about 20 nm (Figure 2c). The crystal structures of the final FVIII-coated MNPs compared to the XRD pattern of Fe3O4 and Fe3O4@SiO2 were presented in Figure 3. The sharp peaks in the XRD pattern FVIII-coated final MNPs between 10 and 20° were related to protein crystals on the surface of the MNPs, which resulted in weak presentation of the Fe3O4@SiO2 nanoparticle core.3,36,37 Elemental analysis of FVIII-coated MNPs performed by EDS (Figure 5a) confirmed that the chemical composition of the final product included Fe, O, Si, N, C, and small amount of S.
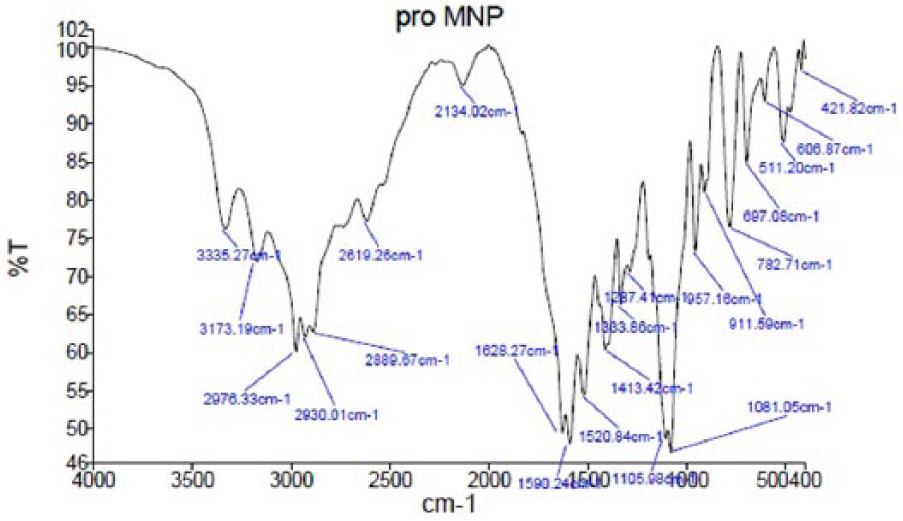
FTIR spectrum of FVIII-coated MNPs.
Stability of FVIII-coated MNPs
Release of FVIII from the protein-coated MNPs was assayed by incubation of the final product in binding buffer for 30 min to 72 h at 4°C. After incubation, particles was separated from buffering medium and the protein content of the supernatant was evaluated by Bradford assay. FVIII release from FVIII-coated MNPs was less than 10% mass of coated protein after 72 h incubation.
Aptamer affinity determination using FVIII-coated MNPs
The dissociation constant (Kd) of the FVIII-specific aptamer to the FVIII-coated MNPs was determined as 120 ± 5.6 nM, which was comparable to the Kd of the specific monoclonal antibodies for coagulation FVIII, exhibiting more affinity compared to the FVIIa aptamer with a Kd of 1.23 ± 0.9 µM (Figure 8).
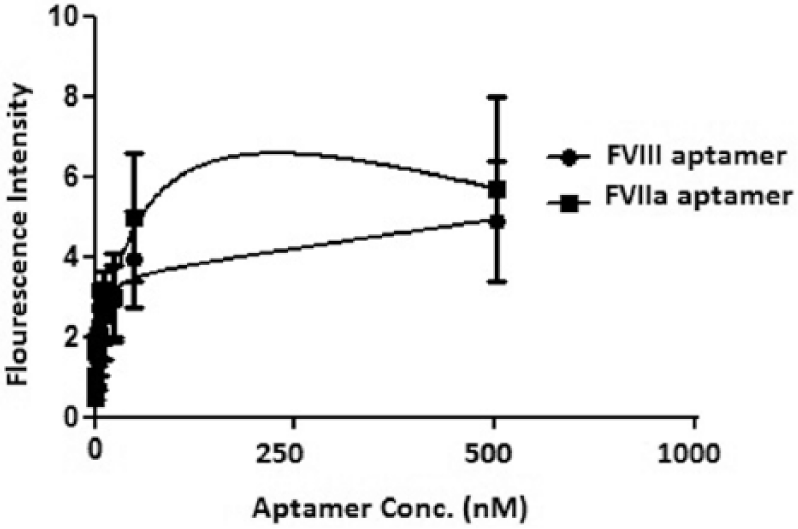
Nonlinear regression analysis of FVIII and FVIIa aptamer binding to FVIII-coated MNPs.
Discussion
MNPs show great promise as analytical materials due to their simple handling, possibility of various surface modifications, and large surface area.14,41 Magnetic nanostructures have been synthesized by different methods, which involve physical, chemical and biological approaches including precipitation from solution, co-precipitation, high temperature decomposition of organic precursors, gas-phase deposition, electron beam lithography, sol−gel synthesis, oxidation method, hydrothermal reactions, aerosol/vapor phase methods, and microbial incubation. Co-precipitation methods have proved most popular, due to their simplicity, productivity, ease of implementation, and need for less hazardous materials and procedures.16,42 Naked-iron-based magnetic nanostructures are chemically and physically unstable. Therefore, coating of iron MNPs with natural or synthetic polymers has been evaluated in this regard, exhibiting promising results.16,43 MNPs are a good solid support in the selection process of protein specific aptamers such as FluMag SELEX.44,45 Considering the importance of aptamers as a selective affinity ligands in analytical processes and protein purification methods, numerous investigations have focused on selection process improvement.46–49 In this regard, MNPs have a special place in aptamer technology.22,50,51 Accordingly, we evaluated the immobilization of FVIII on MNPs for application in the aptamer selection process. FVIII is a valuable therapeutic protein that has been purified from plasma or recombinant sources by different chromatography processes, especially affinity chromatography.52,53
In this regard, Fe3O4 MNPs was first synthesized by a co-precipitation method, and then a silica coating was applied to stabilize the iron-based magnetic core. Then, a second layer of functional silica was coated onto the MNPs to improve their reactivity to the NH2 or OH groups of proteins. The functional group isocyanate was selected in this study. Isocyanate is highly reactive to the different functional groups of reagents, thus improving the binding efficiency of target molecules to MNPs. As isocyanate can react with H2O molecules and result in NH2 terminal products, the coating reaction should be performed using dry non-functional solvents such as dried toluene or acetonitrile.31,54 Protein coating resulted in more negative zeta potential, as also confirmed by FTIR.
FVIII attachment to functionalized MNPs was performed during a 2-h incubation at room temperature as long incubations could result in protein instability events. 55 This type of coating was more stable than direct adsorption (our previous study). 56 Finally, FVIII-coated MNPs were applied in specific aptamer affinity measurement and the results confirmed the suitability of these particles for analytical approaches. Different methods could be considered for aptamer affinity measurement, including direct and indirect fluorescence based methods, colorimetric assays, surface-plasmon-resonance-based methods, enzymatic assays, etc. Among them, fluorescence-based methods have the advantages of a relatively fast, simple and high resolution methodology. 57 The affinity of FVIIa and FVIII aptameric ligands were previously studied using fluorescence methods. The reported Kd values of the FVIIa and FVIII aptamers against their specific protein ligands were 938 ± 149 and 925 ± 11.6 nM, respectively.58,59 In this study, these two aptameric ligands were applied in affinity measurement assays using FVIII-coated MNPs as a target immobilized solid support. The FVIII aptamer (77 nt DNA aptamer) is the specific ligand and the FVIIa aptamer (81 nt DNA aptamer) is the unrelated nonspecific aptamer (for cross reactivity analysis). Comparative study showed that the Kd calculated for the specific aptamer was lower than the Kd calculated for the nonspecific FVIIa aptamer. The results confirmed that immobilization of FVIII on the MNP surface did not change significantly the protein apatopes (aptamer binding domains); therefore, a specific aptamer could bind selectively with higher affinity (more than 100-fold higher) than the nonspecific ligand. In addition, magnetic separation of bound from unbound aptameric ligands could result in a more precise Kd calculation than the previously reported Kd from fluorescence-based methods using filtration separation. 59 According to the promising results obtained from application of FVIII-coated MNPs in affinity assays, these protein-immobilized MNPs could be valuable reagents for subsequent FVIII-specific aptamer selection through magnetic-based SELEX processes.
Conclusion
Magnetic analytical techniques are promising methods for in vitro development of nucleic acid-, peptide- or protein-based affinity ligands. In this regard, FVIII-coated MNPs developed using TEPI as a silica derivative for functional coating were evaluated in an aptamer development process (Figure 9). The final FVIII-coated MNPs can be considered as a target immobilized supporting agent for specific aptamer selection in subsequent studies.
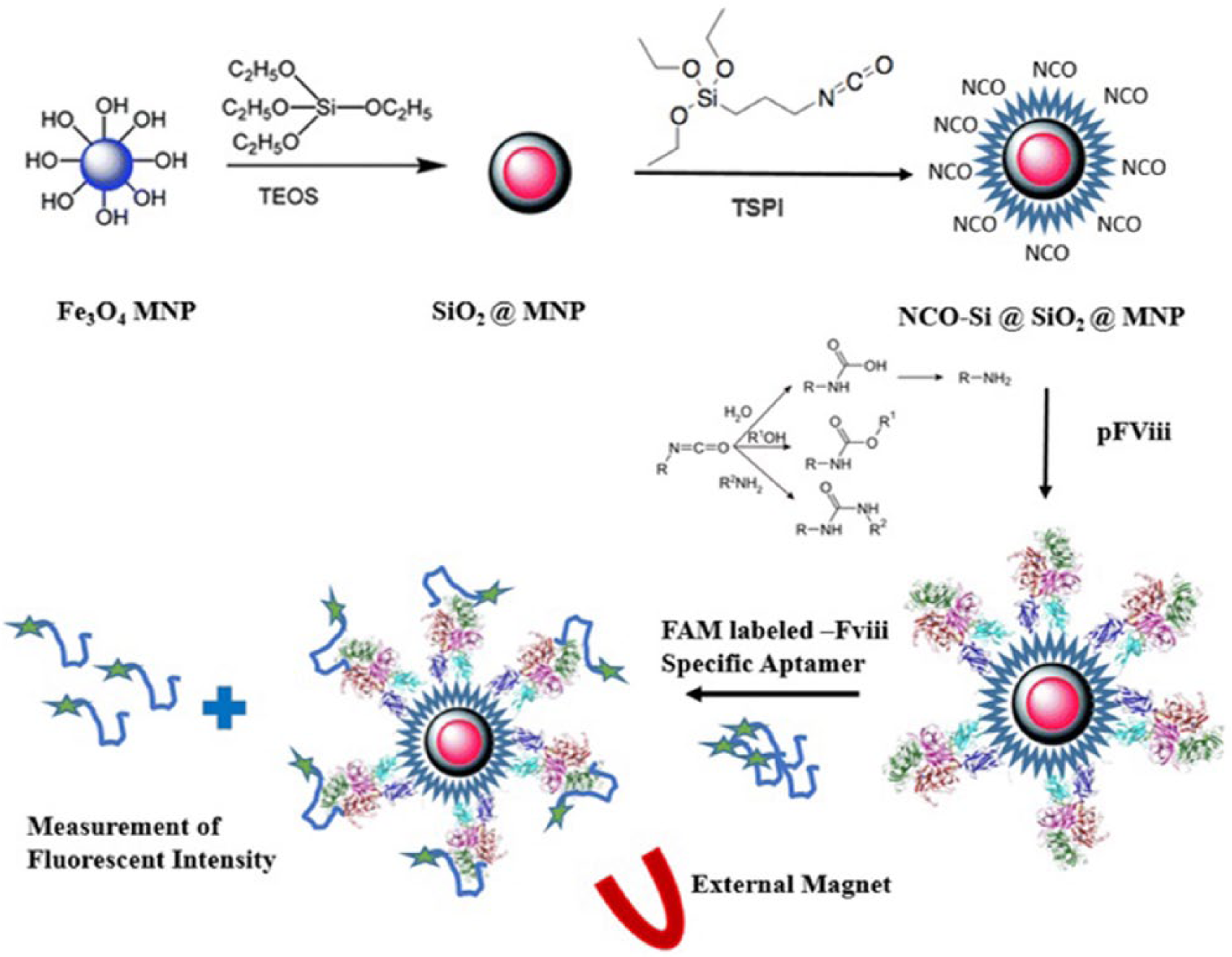
Graphical presentation of the study.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors gratefully acknowledge the financial support granted by the research deputy of Shahid Beheshti University of Medical Sciences (Iran).