Abstract
Introduction:
Three-dimensional (3D) collagen scaffold models, due to their ability to mimic the tissue and organ structure in vivo, have received increasing interest in drug discovery and toxicity evaluation.
Methods:
In this study, we developed a perfused 3D model and studied cellular response to cytotoxic drugs in comparison with traditional 2D cell cultures as evaluated by cancer drug cisplatin.
Results:
Cancer cells grown in perfused 3D environments showed increased levels of reactive oxygen species (ROS) production compared to the 2D culture. As determined by growth analysis, cells in the 3D culture, after forming a spheroid, were more resistant to the cancer drug cisplatin compared to that of the 2D cell culture. In addition, 3D culturing cells showed elevated level of ROS, indicating a physiological change or the formation of a microenvironment that resembles tumor cells in vivo.
Conclusions:
These data revealed that cellular response to drugs for cells growing in 3D environments are dramatically different from that of 2D cultured cells. Thus, the perfused 3D collagen scaffold model we report here might be a potentially very useful tool for drug analysis.
Keywords
Introduction
Over the past decades, although the speed of identification of potential therapeutic drugs has been greatly elevated with progress in scientific research, the number of drugs that finally get through clinical trials and reach the market is very low. 1 Most candidate drugs evaluated in the lab show either low clinical efficacy or intolerable toxicity. 2
Among the many reasons behind this, one apparent explanation is that conventional cell-based in vitro models for drug screening are based on the responses of cells cultured in two-dimensional (2D) monolayers on flat, firm plastic plates,3,4 which is dramatically different from physiological conditions. For example, tumor growth in vivo has a much more complex structure that contains cancer cells, stromal cells and extracellular matrix (EMC).5,6 These components not only connect to each other, but also form the microenvironment to support cancer cell growth, invasion, 7 as well as protecting tumor cells from damage.8–10
To better mimic the in vivo cell growth environment, three-dimensional (3D) cell culture systems have been developed that take advantages of both the simplicity and ease of use of the 2D cell culture as well as some properties resembling the in vivo animal model. Recent studies employing cancer cells in a 3D collagen scaffold revealed that various growth factors were expressed and retained in the microenvironment, favoring cancer cell growth and expansion11–13 and providing certain inhibitory effect against cancer drugs.14,15 As a research tool, the 3D cell culture technique has been used in many areas, including tissue engineering, tumor treatment, regenerative medicine, and stem cell biology.
One key feature of 3D cell cultures is that the spheroid shape is likely to favor the formation of a hypoxic microenvironment that would affect cell metabolism. Metabolism reprogramming is a hallmark of cancer. While the molecular mechanisms remain unclear, an increased level of reactive oxygen species (ROS) in cancer cells has emerged as an important event that plays a crucial roles both during carcinogenesis and for drug resistance.16–19 In this context, a 3D cell culture provides a better situation that resembles the redox state similar to cancer cells growing in vivo.
Due to the potential of utilizing the 3D model in drug discovery and mechanism study, more complex 3D models that can mimic and accurately replicate the function or physiology of living tissues are highly required. Here, we present a perfused 3D scaffold model for tissue cell culture. In this model, the collagen scaffold acts as the EMC environment, and perfusion resembles the blood circulation in vivo. With this perfused 3D collagen scaffold model, we investigated the cellular response to cancer drugs, as well as the cellular level of ROS to determine the differences of cells growing in the 3D environment in comparison with traditional 2D cell cultures.
Materials and methods
Cell lines and 2D cell culture
The human breast cancer cell line MCF-7 and human glioblastoma cell line U118-MG were cultured in Dulbecco’s Modified Eagle Medium (DMEM), supplemented with 10% (v/v) bovine serum and 100 U penicillin/ml and 100 µg streptomycin/ml in a humidified incubator at 37°C with 5% CO2.
Perfused 3D cell culture and maintenance
Cell suspensions of MCF-7 or U118-MG in DMEM were mixed 1:1 with collagen (v/v); the final concentration of collagen is 1.5%. These mixtures were injected into the perfusion 3D culture instrument called TissueFlex microbioreactors (Figure 1(a)), which are molded multiwell units formed from a gas-permeable biocompatible polymer at a concentration of 5 × 104 cells/well. As a 2D control, the cells were grown in 24-well plates at a concentration of 5 × 104 cells/well. The cell culture media or drugs to be tested or evaluated are individually perfused through inlet tubing by a syringe pump (Harvard Apparatus, Holliston, USA) to the bioreactors, where the 3D cell culture is growing in the wells similar to that of a 96-well plate. The nutrient-exhausted media are collected through the outlet tubing.
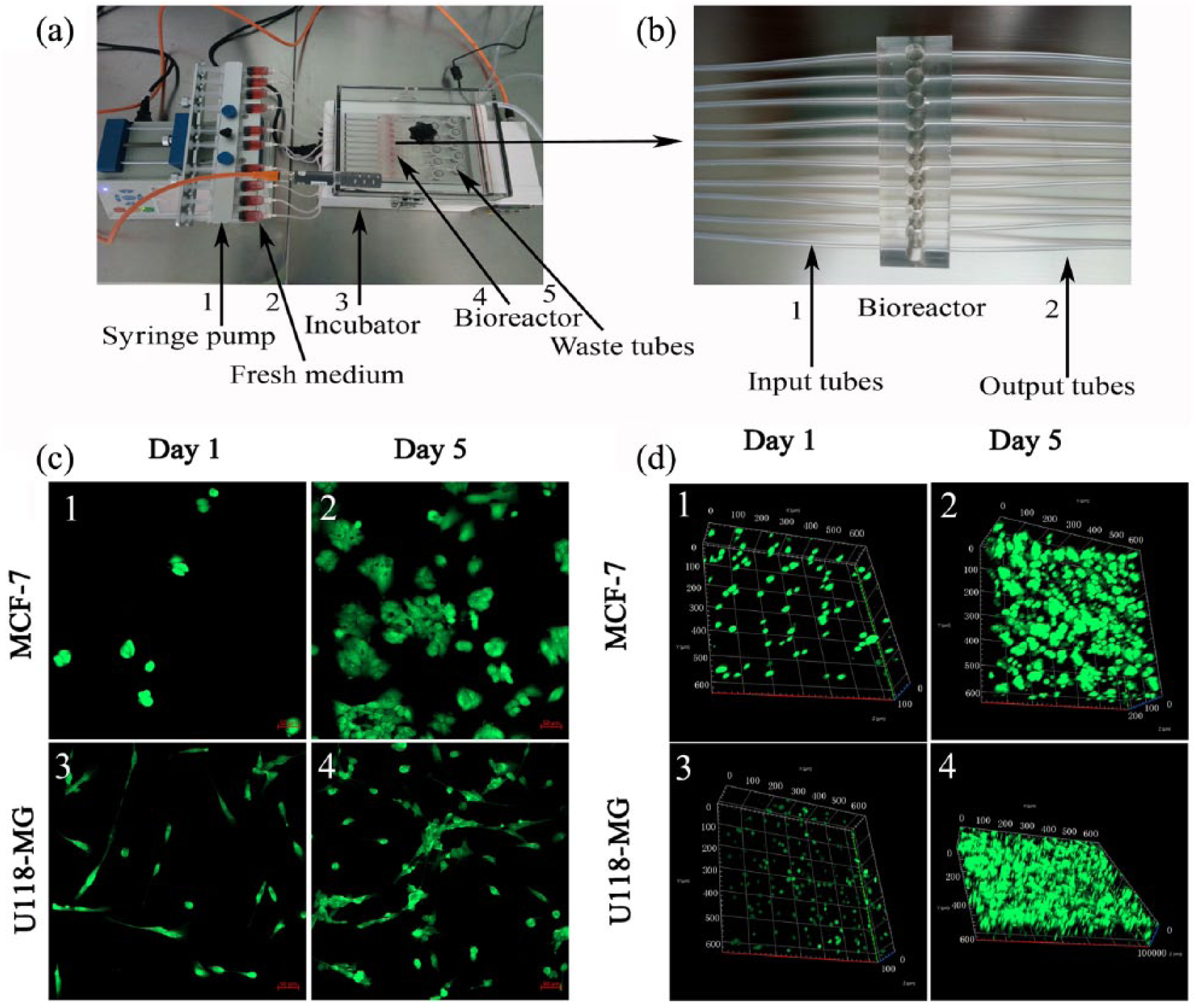
3D cell culture instrument and living staining of cells. The 3D culturing instrument (a) is composed of syringe pumps (1), incubator (3), bioreactor (4), and input and waste tubes (5). Fresh media (2) is under the control of the syringe pumps. (b) The enlarged image of input and output tubes. (c) and (d) The different morphology of cells (green) in 2D and 3D cultures. Two cancer cell lines (MCF-7 and U118-MG) were stained with Calcein-AM and EthD-1 at days 1 and 5 of cell culture to show morphology difference and cell viability (living cells are green and dead cells are red).(c) Cells cultured in traditional 2D model; (d) cells cultured in perfused 3D collagen scaffold model.
Drug treatment and cytotoxicity analysis
Cells under 2D and 3D culturing conditions were treated with different concentrations of cisplatin to compare their response to drug toxicity. For 2D cell cultures, MCF-7 and U118-MG cells were transferred into 24-well plates and split into two groups, then exposed to a 24-hour treatment with cisplatin (0, 10, 20, 40, 80, 160, 320 µmol/L) either immediately after seeding or after 4 days of growth. For perfused 3D cell cultures, MCF-7 or U118-MG cell suspensions were mixed with collagen, and then planted into the TissueFlex microbioreactors. Exposure to cisplatin was the same as above. After cisplatin treatment, cell viability was assessed by using a cell counting kit (CCK-8; Sigma Aldrich, Saint Louis, USA) according to the manufacturer’s instructions. Briefly, CCK-8 solution was injected into the sample wells and incubated for 1 h; the fluorescence intensity was quantified through optical density measurement at 450 nm using a microplate reader (Spectra Fluor Plus, Olympus, Tokyo, Japan). Cell viability inhibition percentage was calculated as 1 minus the ratio between the treated group and untreated group.
Cell death determination
The number of live and dead cells in culture were counted by fluorescent probes (Calcein-AM and EthD-1) with a Live/Dead Viability-Cytotoxicity Kit (Invitrogen, Carlsbad, USA), which is based on the detection of cell membrane integrity. Dead cells allow the penetration of ethidium homodimer-1 (EthD-1), giving red fluorescence, while living cells allow the penetration of non-fluorescent acetomethoxy derivative of calcein (Calcein-AM) and convert it into the green fluorescent calcein. Staining was performed according to the manual. Briefly, samples were washed three times phosphate buffer saline (PBS) followed by incubation with 100 μL of dyes (4 μM) for 30 min. Then the samples were imaged using a fluorescence microscope (Olympus, Tokyo, Japan) after washing three times in PBS.
Scanning electron microscopy analysis to detect the micromorphology of cells and collagen scaffold
The micromorphology of cells in traditional 2D cell culture or 3D collagen scaffolds was examined with a scanning electron microscope (SEM) (Model SU-1510, Hitachi, Tokyo, Japan) at two growing stages (days 1 and 5). Cells and collagen scaffolds were fixed in 4% glutaraldehyde overnight. After washing three times with PBS and one time with distilled water, cells were dehydrated in graded ethyl alcohol (30%, 50%, 60%, 70%, 80%, 90%, 100%, each for 5 min), followed by drying in air with hexamethyldisilazane. Samples were then mounted on stubs and sputter-coated with 10 nm gold and subjected to imaging with the SEM.
Detection of intracellular ROS
Intracellular ROS was detected using the fluorescent probe 2ʹ,7ʹ-dichlorofluorescin diacetate (DCFH-DA, Beyotime, Shanghai, China) as previously described. 20 Briefly, samples were incubated with DCFH-DA (100 mM) for 1 h at 37°C and rinsed with PBS before fluorescence measurement. Fluorescence intensity of the samples was measured at an excitation wavelength of 488 nm and an emission wavelength of 525 nm using a fluorescence microplate reader (Spectra Fluor Plus, Olympus).
Statistical analysis
Data were expressed as the mean ± standard error, accompanied by the number of experiments (N = 3) performed independently, and analyzed by t test. Differences with p < 0.05(*) and p < 0.01(**) were considered statistically significant and high statistically significant, respectively.
Results
Construction of the perfused 3D collagen scaffold model
We developed a 3D cell culture instrument containing a pump, an incubator, and a bioreactor (Figure 1(a)). The 3D cell culture system uses collagen I as the scaffold to provide the supporting matrix for cell growth. The nutrient-rich cell culture media and drugs to evaluate are individually perfused through inlet tubing by a syringe pump to the bioreactors, and the nutrient-exhausted media are collected separately through the outlet tubing (Figure 1(b)). Therefore, the cells grow in a stable culture environment with the advantage of providing perfusion capability to better resemble the in vivo situation of tissues with blood vessels.
A comparison of 2D and 3D cell cultures is shown in Figure 1(c) and (d). For the 3D cell culture, cell suspensions of MCF-7 or U118-MG in DMEM were mixed 1:1 with collagen (v/v, the final collagen concentration is 1.5%) to ensure the cells formed the sphere phenotype. The cell phenotype and living/dead status were simultaneously determined by fluorescence staining as described above. Cells grew well under both 2D and 3D culturing conditions since no dead cells (red colored) were observed. Cells were maintained as a monolayer and showed the flat morphology in the 2D culturing condition (Figure 1(c)). Cells cultured on the 3D collagen matrix, however, formed a sphere shape gradually from day 1 to day 5 (Figure 1(d)).
The architecture of 3D collagen scaffold visualized by SEM analysis
Collagen was used as the scaffold to provide the 3D environment for cell growth. To better understand the differences of the growing conditions between cells on the 2D hard plastic surface and 3D collagen matrix, SEM analysis was performed. As evidenced with two cell lines, MCF-7 and U118-MG, cells growing on the 2D plane surface displayed a flat or spindle-like morphology, adhering tightly to the supporting substrate (Figure 2(a,b,e,f)). Cells growing in the 3D collagen scaffold were inserted into the matrix and wrapped by collagen (Figure 2(c,d,g,h)). Cell clusters could be observed but still maintained the sphere morphology under the 3D culture condition after 5 days (Figure 2(d,h)). Cells under the 2D culture condition, however, only grew aggregately and slowly reached confluence but no sphere-like shape (Figure 2(b,f)). The collagen scaffolds are porous, and media/liquid can easily flow through the matrix. Compared to the adherent growth of cells in a traditional 2D environment, the porous nature of this system provides the required space for cancer cells to proliferate. Together with the perfusion setup, the 3D cell culture system we presented here mimics well the structure and function of tissue and organs in vivo. Therefore, it can serve as an excellent in vitro tumor model, and has the full potential to mimic other organ types where appropriate cells are incorporated.
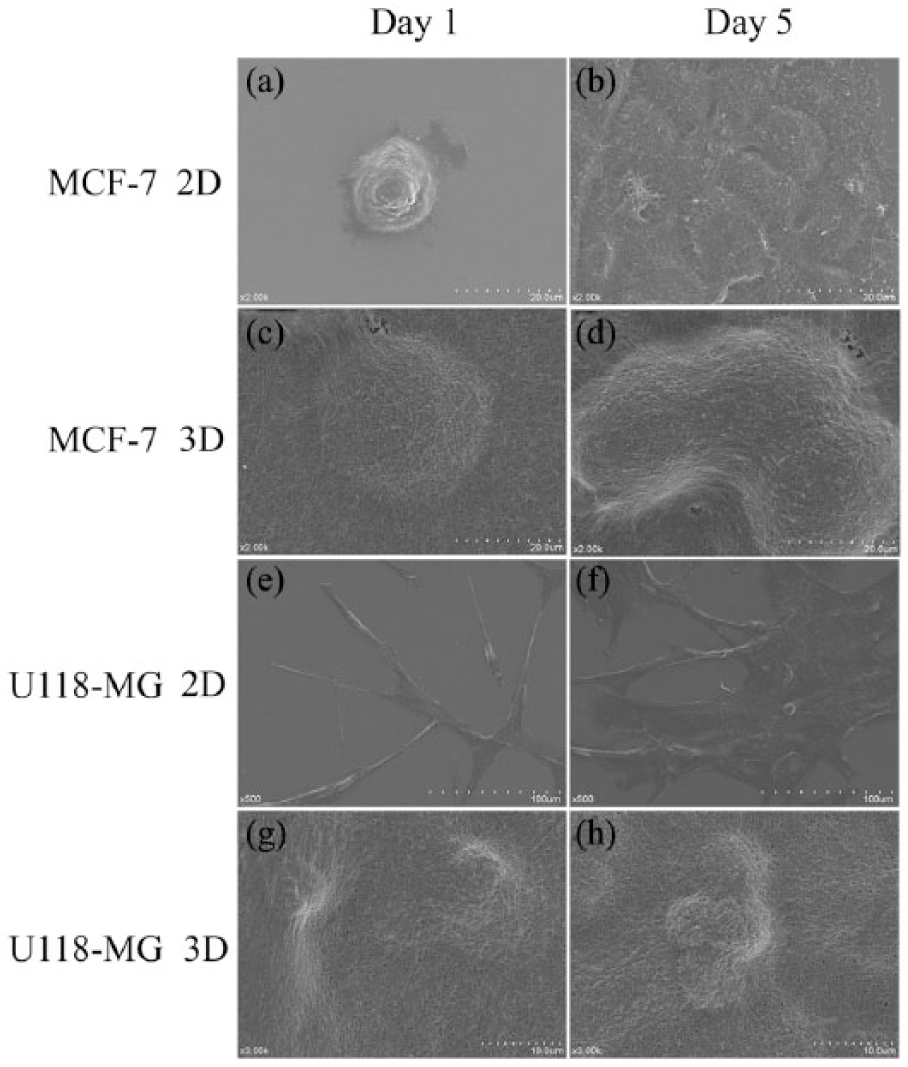
The morphologies of cells cultured under 2D and 3D conditions determined by scanning electron microscopy. (a), (b),(e), and (f) show 2D cultured cells, which displayed a flat or spindle-like morphology, adhering tightly to the supporting substrate at days 1 and 5. (c), (d), (g), and (h) are 3D cultured cells which were inserted into the matrix and wrapped by collagen, and maintained the sphere morphology at days 1 and 5.
Cells grown as tumor spheroid showed different anti-proliferative activity comparing to cells grown as monolayer in response to cisplatin treatment
Cells cultured in the perfused 3D collagen system maintained both cell–cell and cell–matrix interactions in a 3D manner, and formed a spheroid shape similar to tumor growth in vivo. In this context, it is interesting to determine how they respond to cancer drugs in comparison with 2D-grown cells, the most widely used model for cancer drug screening. Here, we compared the cellular response of 2D- and 3D-cultured cells to cisplatin. As shown in Figure 3, cell viability analysis was performed with two cancer cell lines MCF-7 ((a) and (b)) and U118-MG ((c) and (d)), The results showed that cells in perfused 3D collagen culture system at day 1 after seeding ((a) and (c)) were more sensitive to cisplatin than that in the traditional 2D culture. However, the results were reversed after 5 days of cell growth ((b) and (d)), showing that 3D-cultured cells became more resistant to cisplatin. The results were consistent across three independent repeats. Considering that the porous nature of the 3D matrix would allow better medium–drug circulation, the higher sensitivity of cells in 3D collagen at day 1 can be explained by the possibility of higher intake of the drug compared to the cells under the 2D culturing condition, which have up to half of the cell membrane surface (which is in contact with the bottom of the petri dish) that is not in direct contact with the drug. By day 5, however, the cells of the 3D culture have formed a spheroid, and the reduced sensitivity was either due to a possibly reduced drug intake or the tumor-like microenvironment.
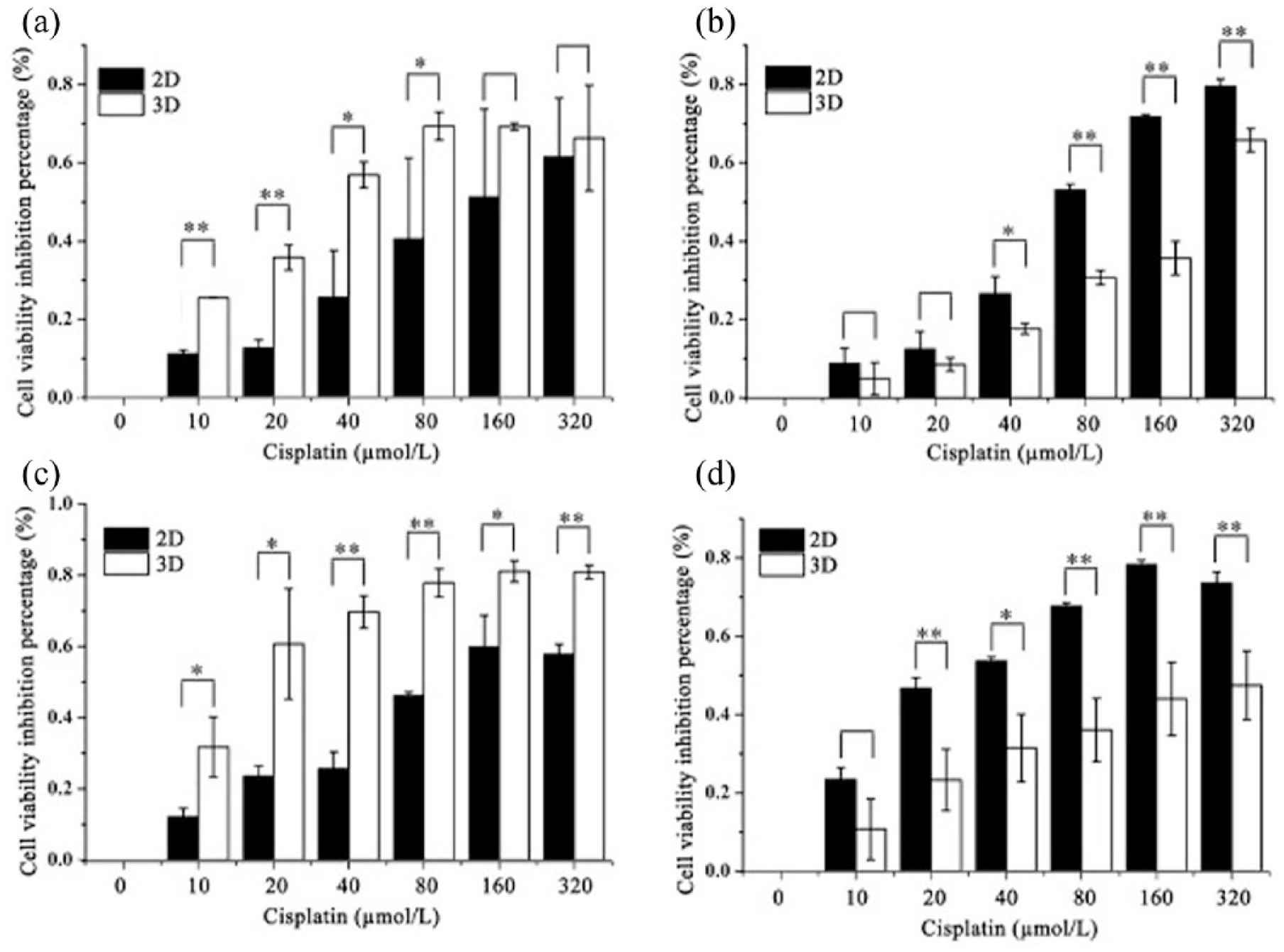
Drug sensitivity analysis of 2D- and 3D-cultured cells. MCF-7 ((a) and (b)) and U118-MG ((c) and (d)) cells cultured under traditional 2D and perfused 3D collagen scaffold culture at day 1 ((a) and (c)) and day 5 ((b) and (d)) were treated with increased doses of cisplatin for 12 hours. Cell viability was analyzed with CCK-8 assay. The bar represents the mean value of three independent measurements.
ROS was significantly increased in cells of the 3D culture compared to those of the traditional 2D culture
It is well known that cancer cells generate more ROS than normal cells.21,22 To further investigate the changes that 3D culture brought to cells compared to a 2D cell culture, we determined the ROS production using the fluorescent dye DCF. As shown in Figure 4, ROS is strongly elevated in the cells grown under the perfused 3D collagen culturing condition (Figure 4(b,d)). ROS is also positive in cells cultivated in the traditional 2D model, but the level is lower (Figure 4(a,c)). These results suggest that in addition to forming the sphere-like shape similar to tumors, the physiology of cells in the perfused 3D collagen scaffold also changed toward the situation of the tumor microenvironment in vivo.
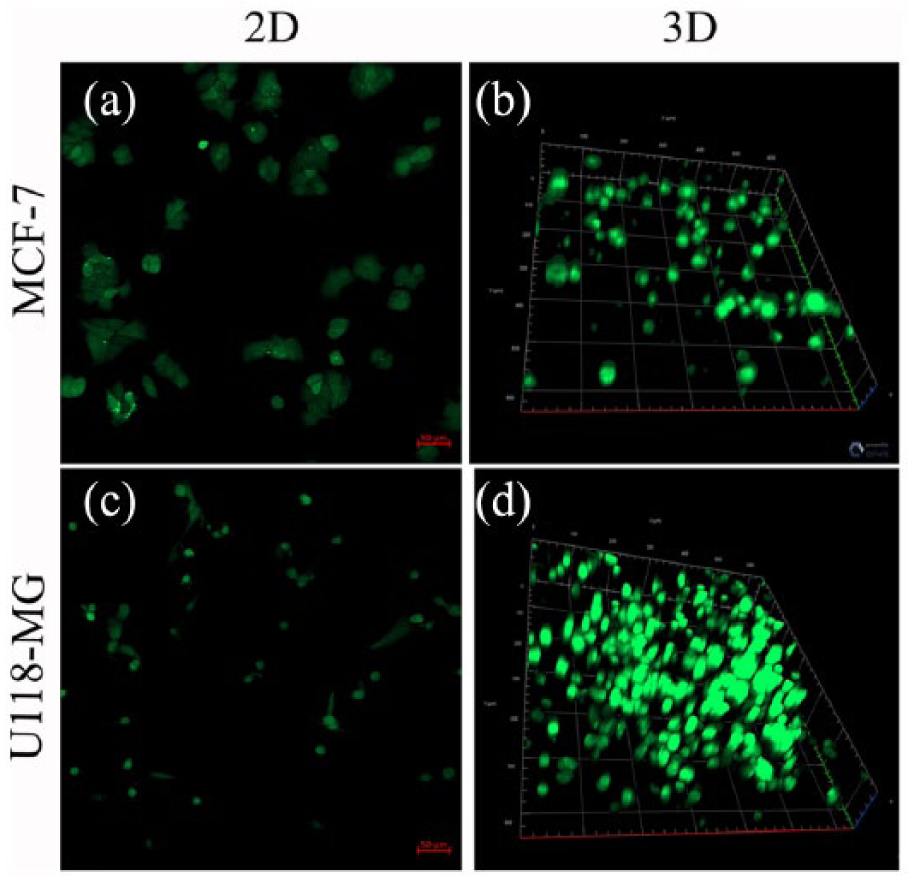
3D-cultured cells showed higher level of ROS production. The expression of ROS was determined using the fluorescent dye DCF in MCF-7 and U118-MG cells cultured under traditional 2D ((a) and (c)) or perfused 3D ((b) and (d)) collagen scaffold models respectively.
Discussion
In this study, we developed and characterized a perfused 3D cell culture system and showed evidence that it can mimic the cellular environment in vivo, especially for tumor cells. Therefore, it has the potential to serve as an effective preclinical platform in cancer drug screening, and presents advantages not only over traditional 2D culture models, but also other non-perfused static 3D models.
Currently, 2D monolayer cell culture is the standard used for in vitro drug screening and toxicity analysis. Although it enables researchers to learn cellular responses with much reduced complexity and cost, its “oversimplification” has long been recognized as meaning it cannot fully reflect the physiological complexity of real tissue.3,4 Studies based on the use of in vitro monolayer cell-based assays might result in errors when predicting tissue-specific responses. Cells cultured in 3D scaffolds, however, are organized with consistent shape and maintain cell–cell and cell–matrix interactions similar to the in vivo situation. These changes not only affect cell survival, proliferation, and migration, but also influence cell response to drugs.
Although compared with traditional 2D cultures the in vitro functions of cells could be generally improved in a 3D culture,23,24 the efficient diffusion of nutrients and waste products across the matrix has been a problem associated with static 3D models. 25 In the current study, however, we overcame this obstacle by applying perfusion to the 3D culture, which simulating human blood circulation. This dramatically enhanced the diffusional capability as well as provided better control of the local microenvironment (e.g., better gas exchange and higher mass transfer). As a result, we demonstrate that cells in the perfused 3D collagen scaffold are easy to form into the sphere phenotype (Figure 2). As illustrated in two cancer cell lines, we provided evidence showing that this in vitro perfused 3D cell culture model can provide more accurate predictions of drug toxicity and efficacy, since cells grown and formed in this microenvironment more closely mimic in vivo conditions.
Our work revealed that cells in 3D collagen scaffolds at the stage prior to the formation of the sphere phenotype appear to be more sensitive to drug toxicity compared to 2D-cultured cells (Figure 3, day 1). However, the results were reversed following cell growth when the compact spheroids were formed (Figure 3, day 5). On the one hand, the response difference might be due to reduced uptake of cisplatin as a result of the compact sphere structure in which cells in the middle of a mass were somewhat more protected. On the other hand, it could be due to the formation of a microenvironment similar to a tumor in vivo. One piece of evidence supporting this idea is the observation of elevated ROS level in 3D cultured cells, which suggests that the cell metabolism is more active. No matter what the underlying mechanism is, it is clear that the cellular response to drugs for cells grown in a 3D environment were dramatically different to than in 2D-cultured cells.
The fact that ROS was more highly expressed in cells in the perfused 3D environment than in the 2D cultures (Figure 4) may be due to the formation of a hypoxic microenvironment. This is a phenomenon often found during the development of tumor tissue from tumor cells.21,22 Hypoxia can induce various factors like HIF-1a, VEGF-A, and so on. Early studies demonstrated that through increasing the levels of ROS, the expression of HIF-1a could be stabilized.16,17 HIF-1a then activates the activity of VEGF-A and facilitates the induction of angiogenesis.26–28
In summary, the perfused 3D collagen scaffold model we presented here resembles well tumor growth in vivo. Through the formation of a spherical and compact morphology, the capability of allowing continued flow of nutrients and removal of waste, similar to blood circulation, as well as the generation of a microenvironment that provides both a buffer zone external to cells and a barrier to exposure, this perfused 3D cell culture model provides a valuable tool for cancer research and oncology drug discovery over traditional 2D cell cultures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key Research and Development Program of China (Grant 2017YFD0400304), the National Natural Science Foundation of China (Grant 31171297) and Natural Science Foundation of Tianjin (17JCZDJC33600).
