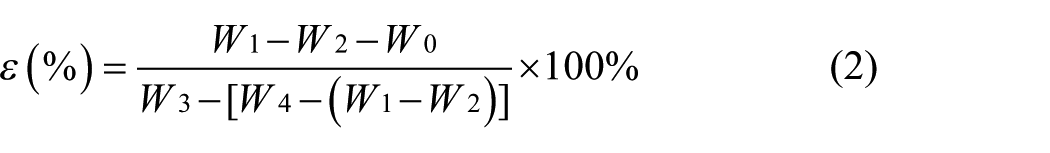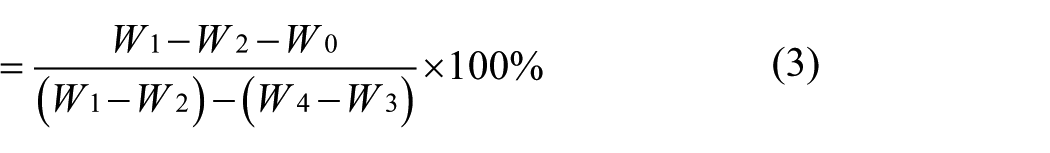Abstract
Introduction:
Bacterial cellulose (BC) aerogels have received more and more attention due to their renewability, biodegradability and other excellent properties in recent years. Modification of BC aerogels using different methods would expand their applications. However, many problems exist for these modifications, such as a low grafting ratio, the larger dosage of metal catalyst required and so on. Activator regeneration by electron transfer (ARGET) for atom transfer radical polymerization (ATRP) is a novel ATRP method which could significantly reduce the amount of metal catalyst required and achieve a high grafting ratio.
Methods:
Novel nanostructured BC aerogels containing epoxy groups were prepared by the ARGET ATRP method. BC aerogels were functionalized with initiating sites by reaction with 2-bromoisobutyryl bromide (BiBBr), and followed by ARGET ATRP reaction with glycidyl methacrylate (GMA) which was catalyzed by copper(II) bromide (CuBr2) and N,N,N',N,'N"-pentamethyldiethylenetriamine (PMDETA), and then reduced by vitamin C. BC aerogels containing epoxy groups (BC-g-PGMA) were obtained after freeze-drying. The influence factors of the solvent ratio of N,N-dimethyl formamide (DMF)/toluene, monomer concentration, the concentration of CuBr2, the molar ratio of vitamin C (Vc)/CuBr2,reaction temperature and time on the grafting ratio were investigated.
Results:
The results showed that the optimal DMF and toluene volume ratio was 2:1, the optimal monomer and CuBr2 concentration were 2 mol/l and 1.5 mmol/l. The optimal molar ratio of PMDETA/CuBr2 and Vc/CuBr2 were 4:1 and 1:1. The optimal reaction temperature and time were 60°C and 9 h. Scanning electron microscopy (SEM) images showed that GMA was strongly adhered onto the surface and inside of the BC pellicle.
Conclusions:
GMA was self-grown on the BC surface and achieved the high grafting ratio of 1052.7% under optimal conditions. The BC-g-PGMA aerogels containing the epoxy groups will provide wider application prospects in drug release, enzyme fixed, functional materials and other fields.
Introduction
Cellulose aerogels with the advantages of both green regeneration cellulose materials and porous aerogels are the third generation of aerogels which are different from the inorganic aerogels and organic polymer aerogels.1–3 Cellulose aerogels are widely used as catalysts, adsorbent materials and thermal insulation materials.4–6 Bacterial cellulose (BC) has gained more and more attention in recent years due to its biodegradability, environmental friendliness, reusability and other excellent properties. Its application could be further expanded by surface modification through different methods such as physical and chemical modification methods. However, many problems exist for these modifications, such as a low grafting ratio, the larger dosage of metal catalyst required and so on.
The atom transfer radical polymerization (ATRP) method has been widely used for surface modification of cellulose-based materials due to its versatility and compatibility. Activator regeneration by electron transfer (ARGET) is a new type of ATRP with advantages that greatly reduce the dosage of metal catalyst required. The reaction can be conducted in the existence of limited amounts of air due to a large excess of the reducing agent in this system.7–8 ARGET ATRP has also been used for the modification of cellulose substrates. Hansson et al. 9 reported surface modification of a cellulose substrate using various monomers such as methyl methacrylate, styrene and glycidyl methacrylate by ARGET ATRP. There is also research on the surface modification of BC using trimethylchlorosilane and acrylate polymers by the ARGET ATRP method.10–12 Glycidyl methacrylate (GMA) is an epoxy group-containing monomer which can react with the carboxyl group (-COOH), hydroxyl (-OH), amino (-NH2), and many other groups. 13 Preparation of BC substrates containing epoxy groups is one of the important ways to broaden their applications. However, using the method of ARGET ATRP to prepare the BC aerogel composites containing epoxy groups has not been reported previously.
In this paper, the BC aerogels containing epoxy groups were prepared using the ARGET ATRP method. The influencing factors of N,N-dimethylformamide (DMF)/toluene, monomer concentration, the concentration of copper(II) bromide (CuBr2), the ratio of N,N,N',N,'N"-pentamethyldiethylenetriamine (PMDETA)/CuBr2 and vitamin C (Vc)/CuBr2, reaction temperature and time on the graft ratio were investigated. The morphology of the BC aerogels before and after surface modification was viewed by scanning electron microscopy (SEM).
Materials and methods
Materials
The materials used were as follows: BC membrane (Hainan Yida Food Industry Co. Ltd., China); tetrahydrofuran (THF) with molecular sieves in addition to water, 4-(dimethylamino) pyridine (DMAP), CuBr2, and PMDETA for the analysis of purity (Shanghai Aladdin Biological Technology Co. Ltd., China); 2-bromoisobutyryl bromide 98% (BiBBr; Shanghai Aladdin Biological Technology Co. Ltd.); and triethylamine (TEA), Vc, DMF, anhydrous ethanol, and methyl glycidyl ester of acrylic (GMA), for the analysis of purity (National Medicine Group Chemical Reagent Co. Ltd., China).
Preparation of BC aerogels
BC membranes were washed successively with water and 1% (w/v) aqueous sodium hydroxide at 80°C for 1 h, then washed with deionized water to remove all bacterial product impurities, and then cleaned pellicles were obtained. Finally, they were dried to a constant weight by vacuum freezing.
Immobilization of ARGET ATRP initiators on BC aerogels
BC aerogels (160 mg) were added into a round bottomed flask containing DMAP (150 mg, 1.2 mmol) in anhydrous THF (50 ml) at room temperature. Then, 2.4 ml of TEA and 1.8 ml of BiBBr were added dropwise into the ice-cold reaction mixture. The mixture was purged with N2 for 15 min and put into a water bath oscillator at 60℃ keeping the reaction for 24 h. The initiator-modified BC (BC-Br) was thoroughly washed in anhydrous ethanol. Then it was dried to a constant weight by vacuum freezing.
Grafting of GMA onto BC-Br aerogels
The lyophilized BC-Br sample was added to a round bottomed flask containing CuBr2 (8.4 mg, 1.5 mmol/l) and PMDETA (31.4 μl, 0.15 mmol) in an anhydrous mixed solvent of DMF (16.6 ml) and toluene (8.3 ml) at room temperature. Then, 6.6mg (0.0375 mmol) of Vc and 7.11 g (0.05 mol) of GMA were added to the reaction mixture. The mixture was purged with N2 for 15 min and put into a water bath oscillator at 60℃ keeping the reaction for 9 h. Finally, the product was washed in deionized water, and then dried to a constant weight by vacuum freezing. Thus, the grafted sample of BC-g-PGMA was obtained.
The graft ratio was calculated as
where W0 and W1 denote the weight of BC-Br and BC-g-PGMA, respectively.
Characterization methods
The specific surface area (BET) was measured by a Micromeritics AccuSorb ASAP2460 BET, using the adsorption of N2. The sample was degassed at 80℃ for 6 h.
The porosity of BC aerogels was obtained using the alcohol absorption method. 14 The BC aerogel (weight of W0) was placed in a weighing bottle with a certain amount of ethanol, and the weight of the pycnometer full of ethanol was W1. After removing the wet sample, the weight of the weighing bottle was W2. The wet weight was W1−W2, the sample absorbing the ethanol weight was W1−W2−W0 and the pore volume was (W1−W2−W0)/ρ, where ρ was the density of ethanol. Filled with ethanol, the pycnometer weight was W3, the wet sample was placed into the pycnometer, and then filled with ethanol (weight of W4). The wet sample discharge of the weight of ethanol was W4−W3 and the apparent volume of aerogels was {W3 − [W4− (W1−W2)]}/ρ. The aerogels porosity, ε, was calculated as follows:
SEM micrographs were obtained using a Hitachi S-4800 SEM. The modification and pristine BC aerogels were coated with gold ions using an ion coater under 20 mA current and 20 s coating time conditions.
Results and discussion
Characterization of BC aerogels
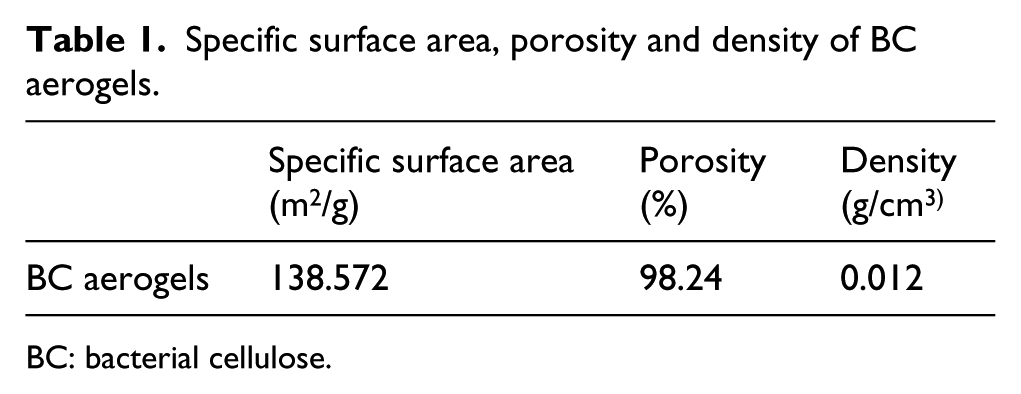
The specific surface area, porosity and density of the prepared BC aerogels before modification were as shown in Table 1. As can be seen from Table 1, the specific surface area of the prepared BC aerogels was as high as 138.572 m2/g and the porosity was 98.24% which was beneficial for their subsequent modification. It also possessed a low density of 0.012 g/cm3.
Specific surface area, porosity and density of BC aerogels.
BC: bacterial cellulose.
The diameters of the fibers of BC and BC-g-PGMA aerogels.

BC: bacterial cellulose; BC-g-PGMA: BC aerogels containing epoxy groups.
Preparation of BC-g-PGMA
Figure 1 shows linear kinetic plots for GMA grafting on BC aerogels. The BC aerogel macromolecule was the initiator that was grafted by polymerization with GMA. As can be seen from Figure1, the ln([M]0/[M]) increased linearly with time which indicated the character of controlling and living of the polymerization reaction of GMA grafting on the BC aerogels.
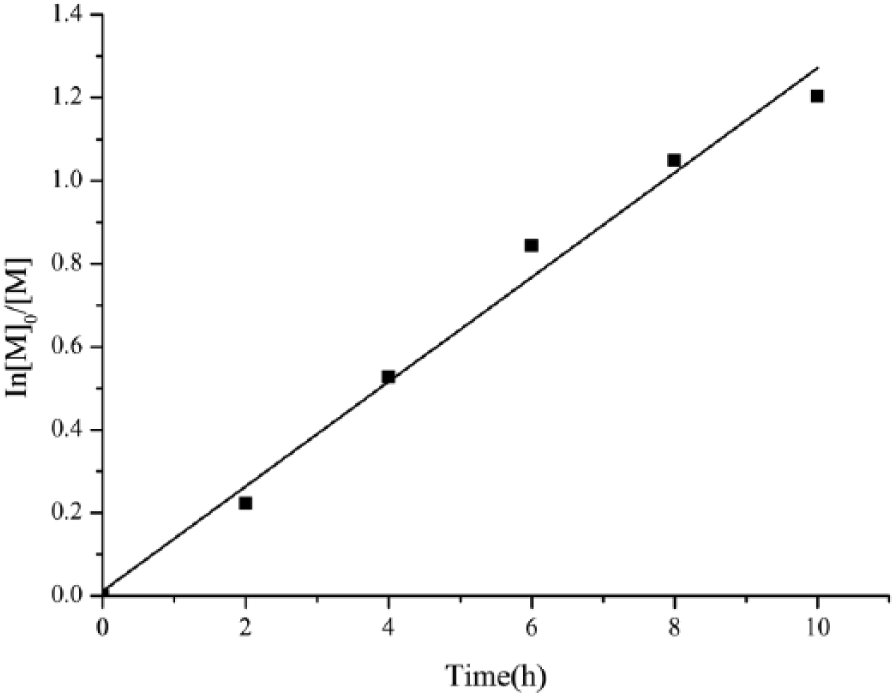
Semi-logarithmic dependences of monomer consumption versus time for grafting polymerization of GMA.
Effect of solvent on grafting ratio
The ATRP reaction equilibrium constant was greatly influenced by the solvent. The catalyst and reducing agent have different solubility in different solvents, so the solvent is vital for the ARGET ATRP grafting polymerization reaction. Figure 2 shows the solvent effect on grafting ratio, with an increasing proportion of DMF/toluene, the grafting ratio increased first and then decreased. This is probably due to the hydrogen bonds formed between BC and solvents with an increasing amount of DMF. Hydrogen bonds are beneficial for GMA molecules entering the fibers by swelling the BC which results in the grafting ratio increasing. However, when the DMF concentration continued increasing, the solubility parameter decreased and slowed down the reaction rate. Therefore, the optimum DMF and toluene volume ratio of 2:1 was obtained after repeating the experiment twice.
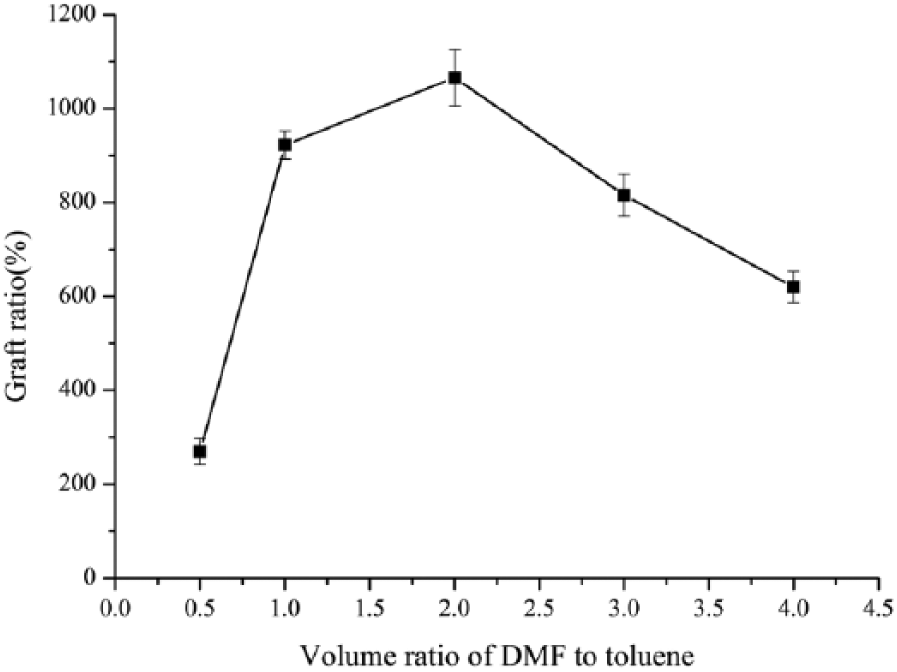
Effect of solvent on graft ratio.
Effect of monomer concentration on grafting ratio
Figure 3 shows the effect of monomer concentration on the grafting ratio. It can be seen that the grafting ratio increased with the increasing monomer concentration. The increasing monomer concentration allowed more GMA monomer to react with BC-Br when free radicals remained constant in this system. However, when the monomer concentration was higher than 2 mol/l, BC aerogel morphology was destroyed, so the optimum monomer concentration of 2 mol/l was obtained after repeating the experiment twice.
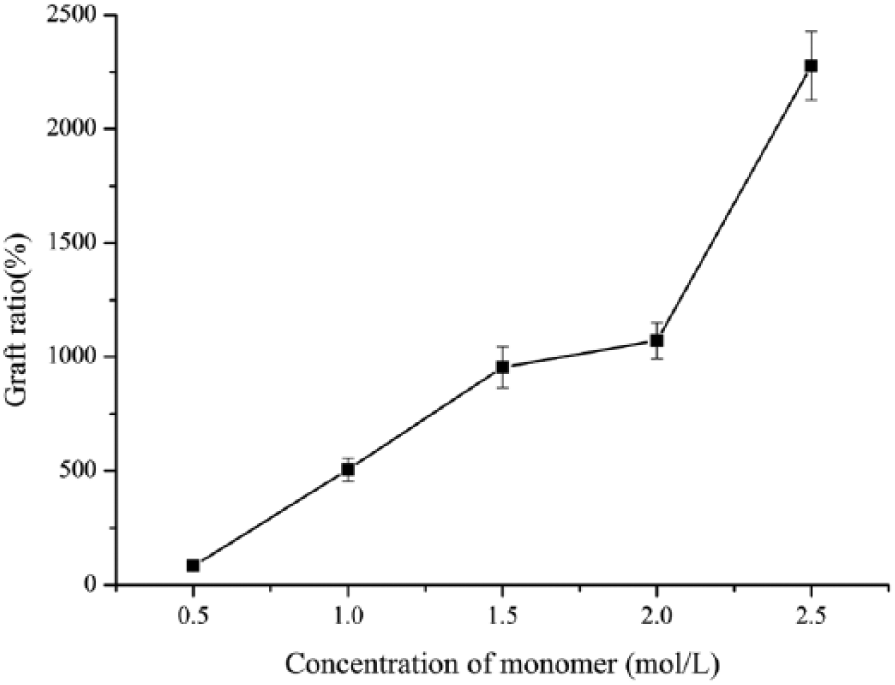
Effect of GMA on graft ratio.
Effect of concentration of CuBr2 on grafting ratio
Figure 4 shows the influence of CuBr2 concentration on grafting ratio. The grafting ratio increased with the increase in CuBr2 concentration. However, the grafting ratio began to fall when the concentration of CuBr2 was up to 1.5 mmol/l. The reason is as follows: the increase of the catalyst CuBr2 concentration promoted the generation of more macromolecular free radicals, while the macromolecular initiator concentration was kept at 6.4 mg/l. 15 However, when the catalyst concentration was higher than 1.5 mmol/l, continuing to increase the concentration of catalyst would lead to the formation of the dormant species due to most free radicals being captured by the catalytic systems. Therefore, the optimal concentration of CuBr2 of 1.5 mmol/l was obtained after repeating the experiment twice.
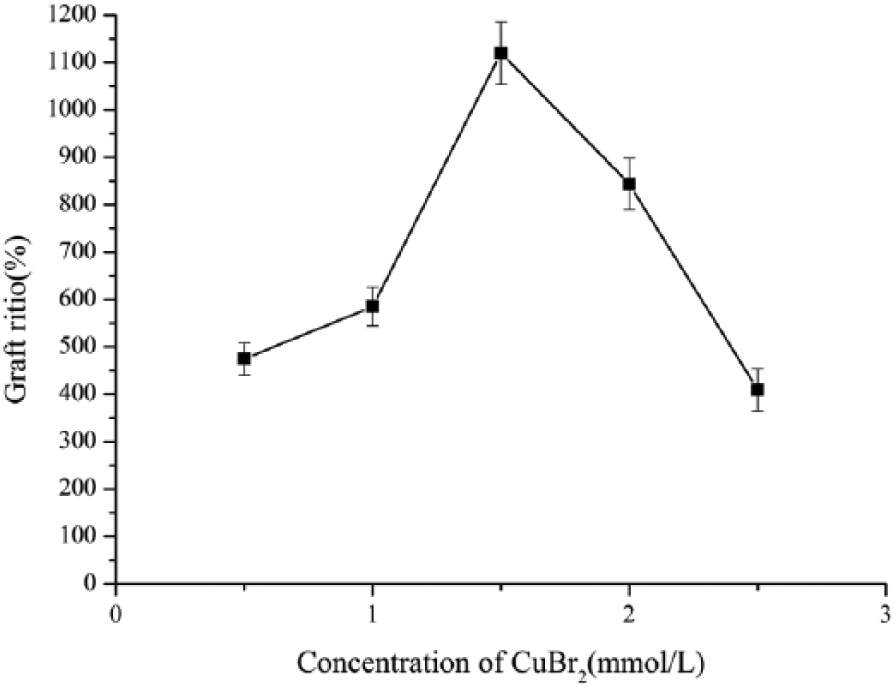
Effect of catalyst concentration.
Effect of the ratio of PMDETA/CuBr2 on the grafting ratio
Figure 5 shows the effect of the molar ratio of PMDETA/CuBr2 on the grafting ratio. It can be clearly seen that the grafting ratio increased with the increasing molar ratio of PMDETA/CuBr2 and reached the maximum when the ratio was 4:1. The reason for this was that the increasing amount of ligand increased the rate of the redox reversible equilibrium reaction of copper which facilitated the transfer of the bromine atom and formed more catalytic active sites. 16 However, the reaction rate decreased with a further increase of the molar ratio of the ligand to catalyst due to the ligand PMDETA having bidentate ligands. Therefore, the optimal molar ratio of the PMDETA catalytic system with CuBr2 was 4:1 which was the result of repeating the experiment twice.
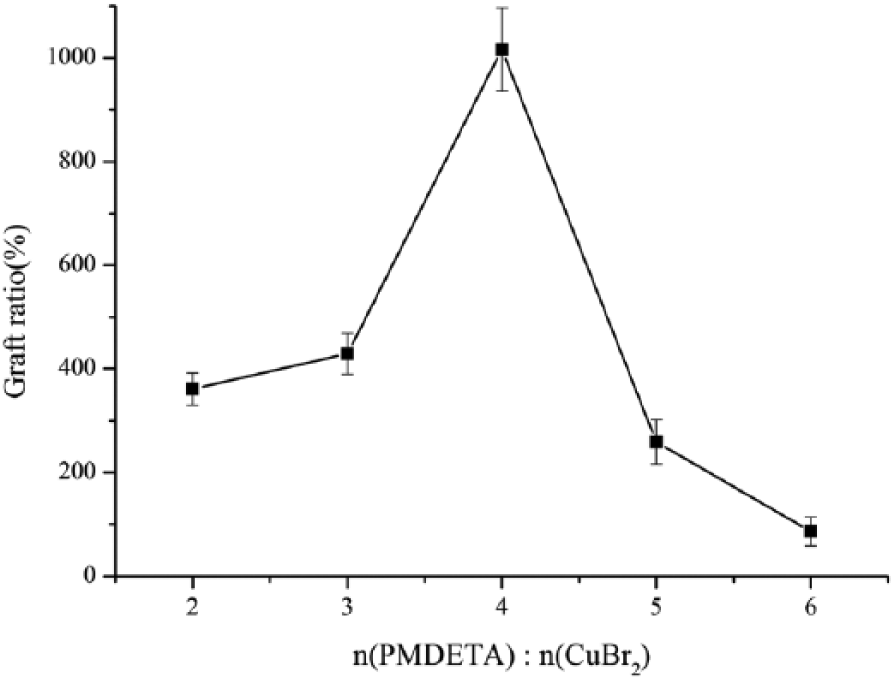
Effect of ratio of PMDETA/CuBr2.
Effect of ratio of Vc/CuBr2 on grafting ratio
Figure 6. shows the effect of the molar ratio of Vc/CuBr2 on the grafting ratio. It can be seen that the grafting ratio increased first with the increasing molar ratio of Vc/CuBr2 from 0.5 to 1.0, then decreased with further increasing. The reason is that the free radicals in this system increased due to the increasing concentration of Cu+ in the solution which arose from the increasing amount of Vc.17–18 However, when continuing to increase the amount of Vc, the increase of free radical concentration in the reaction solution led to the increasing side reactions of chain termination and slowed down the primary reaction rate. Therefore, the optimal Vc/CuBr2 molar ratio of 1:1 was obtained after repeating the experiment twice.
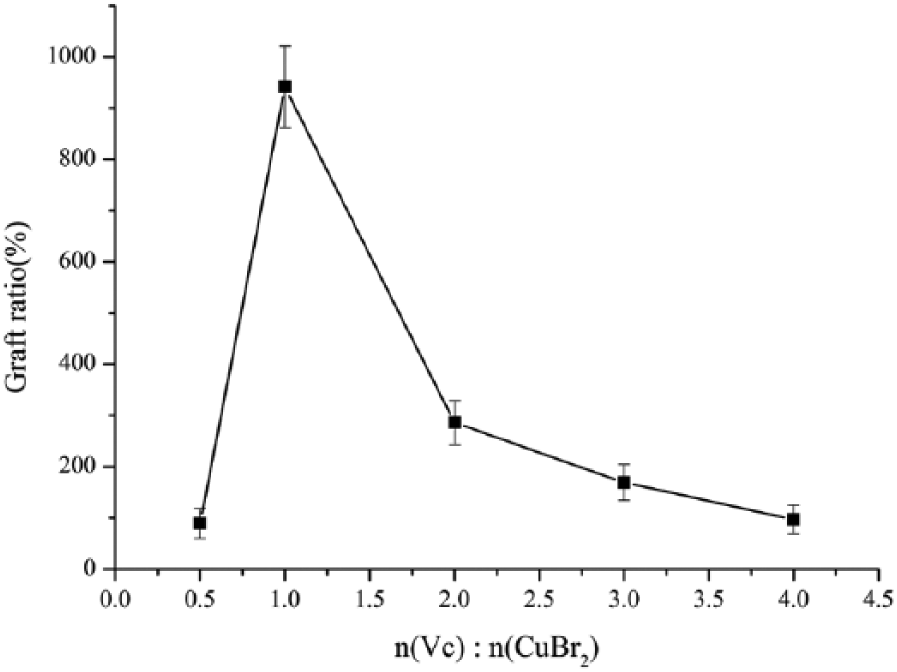
Effect of ratio of Vc/CuBr2.
Effect of reaction temperature on grafting ratio
Temperature is an important parameter in influencing the chemical reaction. As shown in Figure 7, the grafting ratio increased with increasing reaction temperature as well. The reason is that the chain growth rate and equilibrium constants of the atom transfer process increased with increasing temperature. At the same time, with the increasing temperature the reactivity of the halide was improved by accelerating the decomposition of the initiator which resulted in the increasing of free radical concentrations. However, the epoxy groups were easily to ring open affecting the reaction when the temperature was too high, the epoxy group’s loops opened easily which affected the reaction. Therefore, the optimum reaction temperature of 60℃ was obtained after repeating the experiment twice.
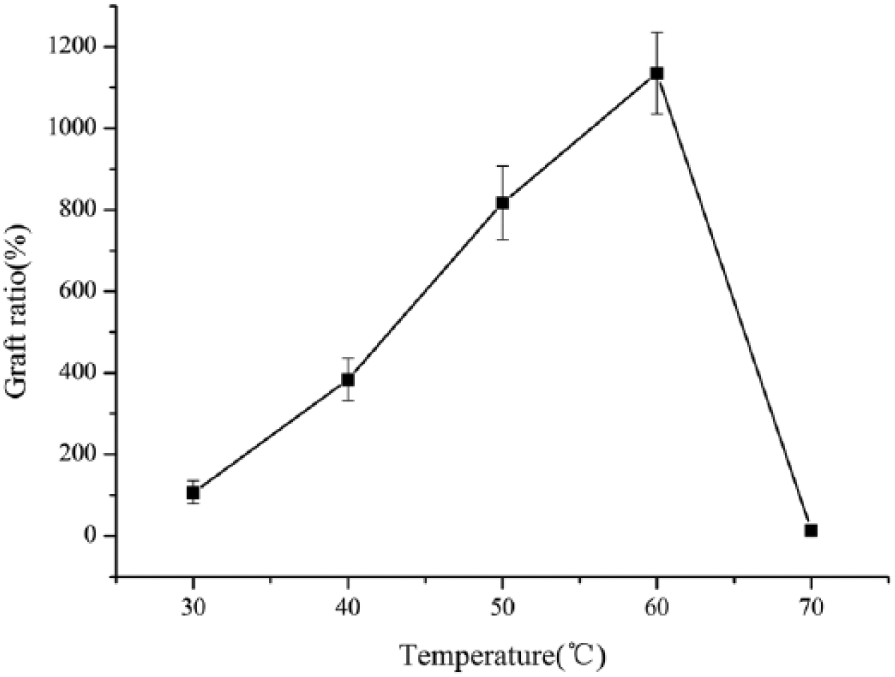
Effect of reaction temperature on graft ratio.
Effect of reaction time on grafting ratio
Figure 8 shows the effect of reaction time on the grafting ratio. The grafting ratio increased with the increasing reaction time. However, the increasing rate was high at first, then leveled off after 9 h, and the grafting ratio reached a constant value. This is because the monomer concentration decreased gradually with the extension of reaction time when keeping the monomer concentration at 2 mol/l. Therefore, the optimum reaction time 9 h was achieved by repeating the experiment three times. The grafting ratio reached the maximum of 1052.7% under the optimal conditions of the above influencing factors.
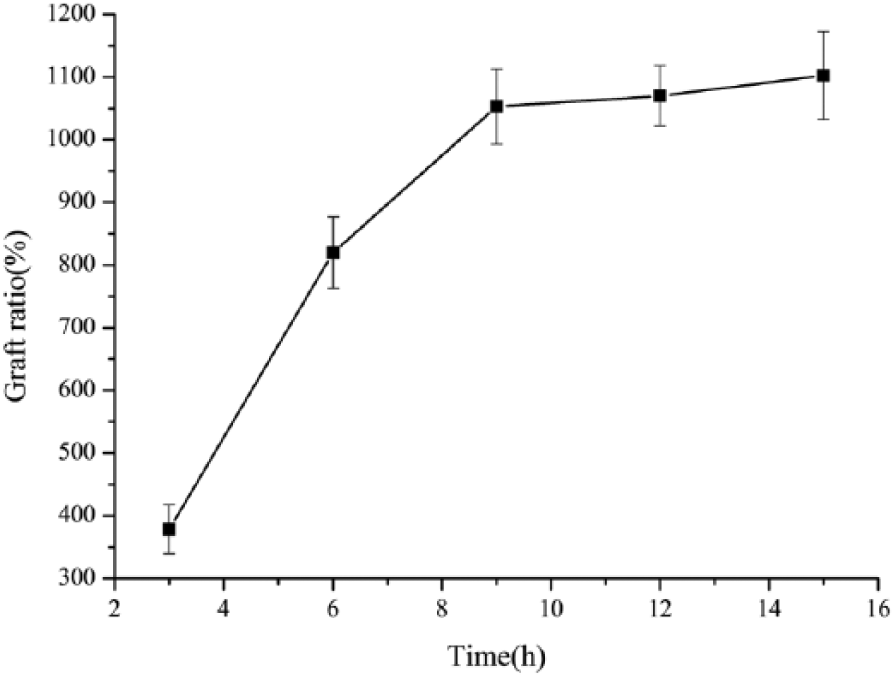
Effect of reaction time on graft ratio.
Scanning electron microscopy observations
Figure 9 shows the SEM images of BC aerogels and BC-g-PGMA. It can be seen that the BC aerogel fibers are relatively slender and homogeneous. The diameter of the fibers increased from 50 nm to 140 nm after modification which were as shown in Table 2. In addition, GMA made the fibers stick to each other and formed a coat membrane which also confirmed the successful grafting of GMA.

SEM of (a) BC, and (b) BC-g-PGMA.
Conclusions
In summary, novel nanostructured BC aerogels (BC-g-PGMA) containing epoxy groups have been successfully prepared using the ARGET ATRP method. The influence of the solvent ratio of DMF/toluene, monomer concentration, the concentration of CuBr2, the ratio of PMDETA/CuBr2 and Vc/CuBr2, reaction temperature, and time on the grafting ratio were investigated. The optimum reaction conditions for the surface modification of BC aerogels were obtained. The optimal DMF and toluene volume ratio was 2:1, the optimal monomer and CuBr2 concentrations were 2 mol/l, and 1.5 mmol/l. The optimal molar ratios of PMDETA/CuBr2 and Vc/CuBr2 were 4:1 and 1:1 respectively. The optimal reaction temperature and time were 60℃ and 9 h. The result of SEM images confirmed that GMA has successfully grafted onto BC aerogels. These novel nanostructured porous materials containing reactive epoxy groups could have potential applications in several fields such as drug release, enzyme immobilization and functional materials.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grants from the National Natural Science Foundation of China (grant numbers 21302001).