Abstract
Introduction:
Micro/nanostructured materials have attracted a great deal of attention, and many strategies have been developed to fabricate micro/nanostructured materials.
Methods:
Amine-functionalized micro/nanostructured Fe3O4 with different sizes was synthesized conveniently in organic media. The chemical structures of as-synthesized products were characterized by FTIR, TEM, SEM, and XRD.
Results:
The ligand binds to the Fe3O4 core by hydrogen bond between the oxygen atom on the surface of Fe3O4 and the hydrogen atom in molecular ethylenediamine. Their magnetic properties were also investigated.
Conclusions:
First, there is no need to control the reaction under a nitrogen atmosphere, and just one salt is used as an iron source. The growth and the surface modification of Fe3O4 crystalline nucleation happen at the same time. Second, monodispersed Fe3O4 micro/nanospheres were prepared without additional surfactant or external magnetic fields. Third, this method is preferred compared with the conventional organic phase method, as the reaction condition is milder and less pollutant will be produced.
Introduction
Magnetic nanoparticles are typically referred to as those materials that possess a magnetic core chemistry (often either Fe3O4 or γ-Fe2O3), and their sizes, at least in one dimension, range from 1 nm to 100 nm. Much effort has been devoted to magnetic micro/nanocomposites during recent years due to their unique physical and chemical properties that make them useful for potential applications in a wide range of fields, including magnetic separation, 1 chemical sensors, 2 data storage current information technology,3,4 catalysis,5,6 biomedicine/biotechnology,7-15 environmental remediation,16-18 and solar cells. 19 The usefulness of magnetic nanoparticles—not only the practical but also the potential—is mainly derived from their small size and high surface area to volume ratio.
As one of the most important iron oxides, magnetite (Fe3O4), a mixed-valence compound, is defined as a polynuclear cluster with two or more metal centers bridge-linked by a ligand. 20 All electrons of Fe3O4 are delocalized in the whole cluster. Fe3O4 nanoparticles have been of great interest, not only because of the fundamental properties caused by their multivalent oxidation states. 21 In previous research, Fe3O4 was utilized as an artificial peroxidase to catalyze the reduction of H2O2, such as in metal hexacyanoferrates. 22 Fe3O4 nanostructures with good stability can be very useful to assist an effective separation of proteins, cells, nuclear waste, and catalysts.
Many strategies have been developed to synthesize micro/nanostructures. These approaches can be broadly divided into four categories: template-free synthesis, sacrificing template strategies, and template methods including hard and soft template methods. 23 Regarding magnetic Fe3O4 micro/nanostructures, some popular synthetic routes have been published during the last decades, including co-precipitation, microemulsion synthesis, micelle synthesis, thermal decomposition, hydrothermal/solvothermal, and/or reduction. All these methods have disadvantages. For example, the reaction needs to be controlled at high temperature (over 200°C) and high pressure for thermal decomposition and hydrothermal synthesis. Co-precipitation routes are very popular for making Fe3O4 nanomaterials. It is necessary to protect the resulting particles from being oxidized using inert gas for co-precipitation synthesis. And what is worse, the shape control is often not very good as two kinds of iron salt (e.g. FeCl3 and FeCl2) are needed in the co-precipitation method. Different iron salts result in Fe3O4 with different sizes. A surface-capping agent needs to be added during or after the co-precipitation reaction. As for microemulsion synthesis and micelle synthesis, despite the narrow size distribution and well-controlled morphology, one main drawback is that many poisonous wastes may be produced from the large amount of solvent used. 24
Compared with the synthesis, it is more crucial to maintain the stability of the magnetic nanostructures for a long time without agglomeration or precipitation. The stability is one of the most important requirements for almost any application of magnetic nanocomposites, including Fe3O4. An efficient approach to maintain chemical stability is to modify the nanocomposite surface by binding or coating a protective layer to the surface. The protective shell not only serves to prevent the core from degradation, but can also be employed for further modification with specific functional components such as drugs, binding sites, and catalytically active species.
As mentioned above, it is still a great challenge to make narrow size distribution, biocompatible, and environmentally friendly magnetic nanocomposites in high yield. In this paper, size-controlled micro/nanostructured Fe3O4 modified by ethylenediamine was synthesized successfully in organic media using FeCl3﹒6H2O as the only iron resource. It is unnecessary to add additional surfactant to prevent the resulting products from conglomeration. Modified by ethylenediamine, the ability to connect with other components and the biocompatibility of Fe3O4 may be improved, which will increase its practical application areas. 25
Experimental
Chemicals and materials
FeCl3·6H2O (Shanghai Chemical Reagents Company) and anhydrous sodium acetate (Shanghai Chemical Reagents Company) was used. Ethylenediamine and glycol were both analytical grade, purchased from Sinopharm Chemical Reagents Company, Shanghai, China. All other chemicals and solvents were analytical grade and used as received with no further purification. Double distilled and sterilized water was used in all procedures.
Preparation of amine-functionalized Fe3O4 nanospheres
Fe3O4 nanospheres were prepared in organic solvent, briefly depicted in Figure 1. Under vigorous mechanical stirring, 4.0 g anhydrous sodium acetate, 2.0 g FeCl3·6H2O, and 10 ml ethylenediamine were added to a three-necked flask containing 30 ml glycol. The color of the reaction system changed to red from yellow with heat, then to deep black when the temperature reached 180°C. After stirring for 7 hours at a temperature of 180°C, the amine-functionalized magnetite Fe3O4 produced was separated from the solution by means of a magnet and washed several times with distilled water and alcohol. The obtained magnetite was subsequently dried in the oven at the temperature of 50°C.
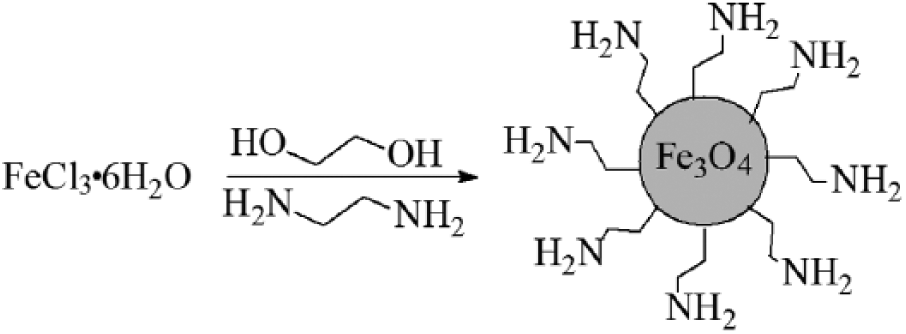
Synthesis of amine-functionalized Fe3O4 nanospheres.
Morphology and particle size of Fe3O4
The morphology of the Fe3O4 nanostructures as prepared was characterized by a Philips XL-30ESEM scanning electron microscopy (SEM). The particle sizes of Fe3O4 were observed under a Philips Tecnai-12 transmission electron microscope (TEM). The samples used for TEM observations were prepared as follows: nanoparticles were dispersed in a suitable amount of water which was vibrated ultrasonically for 10 minutes. Then a drop of water containing samples was spread over a copper grid. Finally, the copper grid was dried under air before the TEM observations were undertaken.
Magnetic measurement
The magnetization of dried Fe3O4 particles was measured with a Quantum Design MPMS7 superconducting quantum interference device (SQUID) magnetometer at 298 K.
X-ray powder diffraction
X-ray powder diffraction (XRD) of Fe3O4 was undertaken with a M03XFH (MAC Science) X-ray diffractometer, operated at a voltage of 40 kV and a current of 40 mA with Cu Kα radiation (λ = 0.154056 nm).
X-ray photoelectron spectroscopy spectra
The X-ray photoelectron spectroscopy (XPS) experiments were carried out on a Thermo Escalab 250 system using Al Kα radiation (hν = 1486.6 eV). The test chamber pressure was maintained below 2×10-9 Torr during spectral acquisition.
Fourier-transform infrared test for amine-functionalized Fe3O4 nanospheres
The Fourier-transform infrared (FTIR) spectrum was detected by a Bruker Tensor 27 FTIR spectrometer with KBr disks.
Results and discussion
Synthesis of Fe3O4 nanospheres
During recent decades various kinds of monodispersed nanoparticles have been obtained from precursors such as acetate and inorganic salts in ethylene glycol. For this research, the reaction is also not a solid phase transformation, and the procedure includes four steps, as shown in Figure 2.
(1) Progressive dissolution of the solid precursor FeCl3·6H2O;
(2) Reduction of the dissolved species by the ethylene glycol;
(3) Nucleation and growth of the metal oxide particles from the solution;
(4) Metal oxide particles are reacted with the ligand ethylenediamine.
Therefore ethylene glycol acts not only as the dispersing medium, the solvent, but also as the reducing agent and the crystal growth medium for the metal particles. The anhydrous sodium acetate protects the resulting precipitation from conglomeration by repulsive electrostatic forces because the amine-functionalized Fe3O4 are positively charged in aqueous solution for the adsorption of Fe3+ on the surface. In addition, there may be sterical repulsion in the organic solvent.
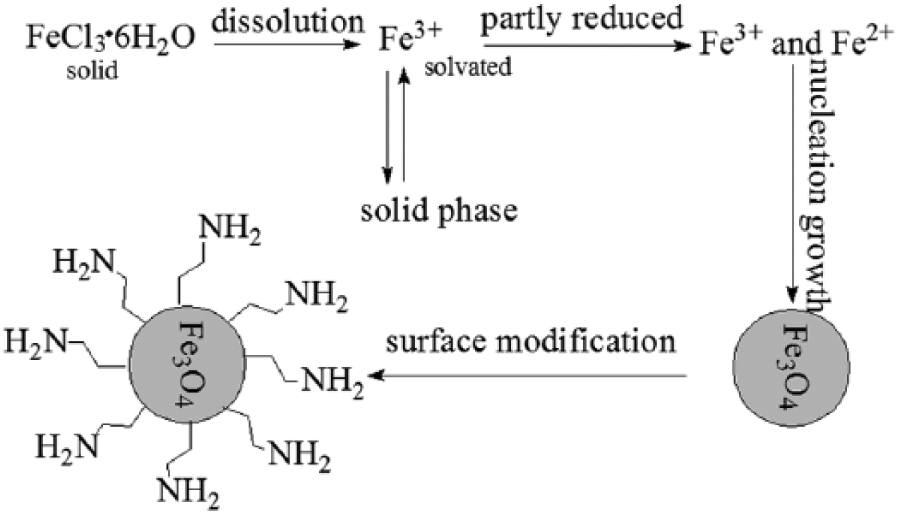
Mechanism of fabrication of amine-functionalized Fe3O4 nanospheres.
FTIR of amine-functionalized Fe3O4 nanospheres
Figure 3 shows the IR spectra of amine-functionalized Fe3O4 nanospheres. The strong IR band at 573 cm-1 was assigned to the characteristic absorption peak of Fe-O vibrations, while 877 cm-1, 1573 cm-1, and 137 cm-1 were characteristic of ethylenediamine, because no such bands were observed in the bare Fe3O4. As discussed above, it was easy to conclude that the Fe3O4 nanospheres were successfully functionalized with ethylenediamine.
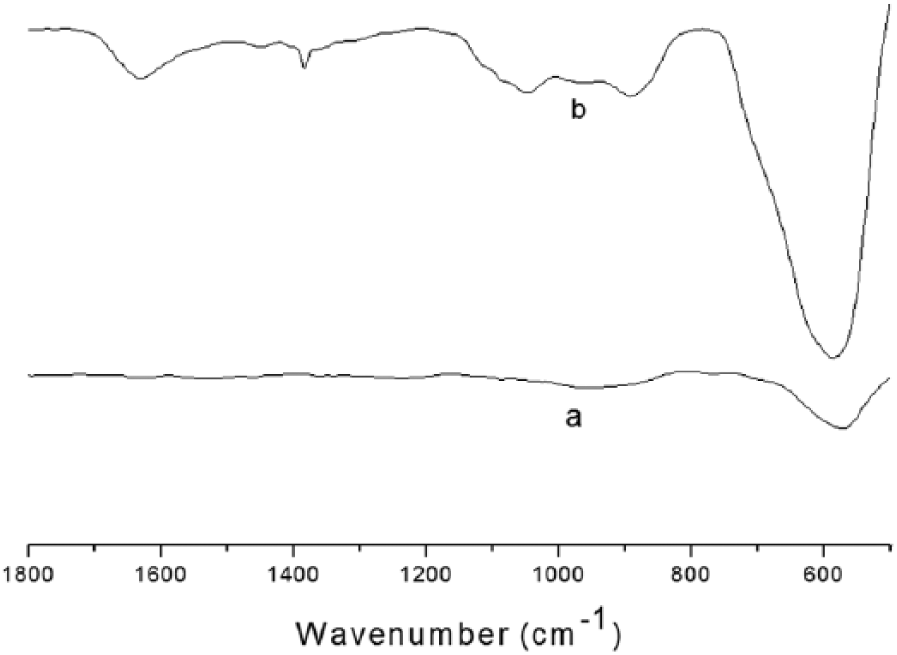
FTIR of bare Fe3O4 (a) and amine-functionalized Fe3O4 nanospheres.
SEM and TEM micrographs of amine-functionalized Fe3O4 micro/nanospheres
TEM micrographs of as-prepared amine-functionalized Fe3O4 nanospheres (Figure 4) were compared according to the amount ratio of ethylenediamine to FeCl3. TEM samples were formed when a drop of alcohol containing the nanospheres was carefully placed on the grid and dried in air. As Figure 4 shows, the diameters of Fe3O4 nanospheres increased with the ratio increase of ethylenediamine to FeCl3. In our experiment, the mass of FeCl3·6H2O was controlled constantly at 3 g. The amount of ethylenediamine was 5 ml for (a), 10 ml for (b), and 15 ml for (c). Finally, the diameters were ~50 nm, ~100 nm, and ~200 nm, respectively.

TEM images of magnetic nanoparticles prepared by changing the amount of ethylenediamine. (a) 5 ml, (b) 10 ml, (c) 15 ml. The amounts of FeCl3·6H2O were kept at 3 g.
SEM micrographs of as-prepared amine-functionalized Fe3O4 nanospheres are shown in Figure 5. From Figure 5, it is found that the sizes of the spheres are larger than those shown in Figure 4. This may be due to their conglomeration when they exist as solid state, which resulted in the large sizes observed by SEM.

SEM images of magnetic nanoparticles prepared by changing the amount of ethylenediamine. (a) 5 ml, (b) 10 ml, (c) 15 ml. The amounts of FeCl3·6H2O were kept at 3 g.
XRD of the as-prepared Fe3O4 micro/nanospheres
The phase structure and purity of the as-prepared Fe3O4 spheres were characterized by XRD. A typical XRD pattern of the as-prepared Fe3O4 micro/nanospheres is shown in Figure 6. All the diffraction peaks could be assigned to pure Fe3O4 with cell constant a = 8.375, which is in good agreement with the literature value. 26 No obvious impurity phase diffraction peaks were detected.
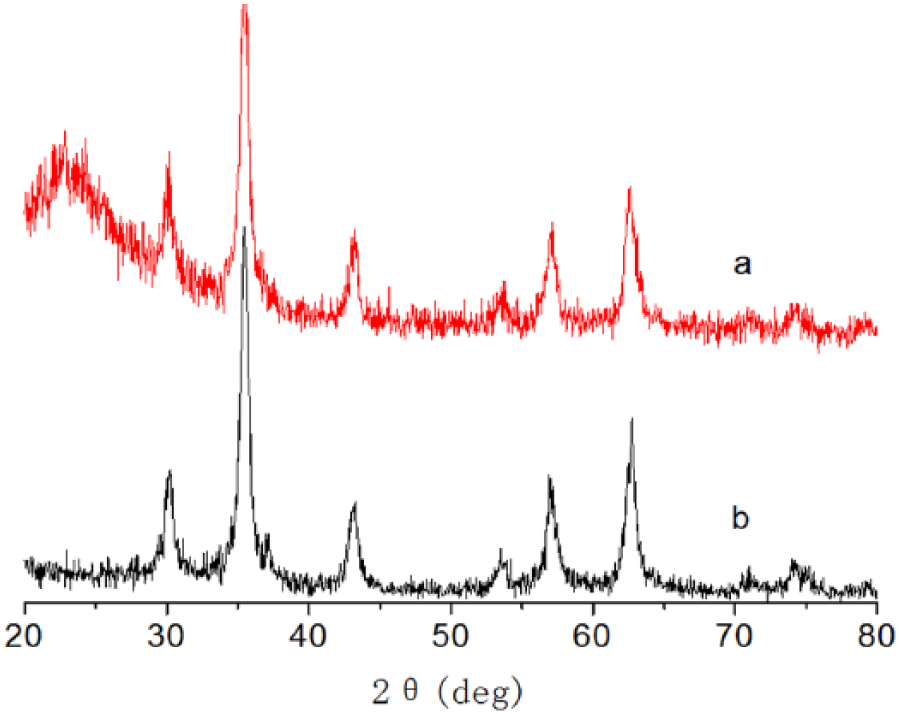
The presentation XRD pattern of amine-modified Fe3O4 micro-/nanostructures (a), bare Fe3O4 (b).
Magnetic properties of Fe3O4 micro/nanospheres
The value of bare Fe3O4 Ms is 37.3 emu/g, as Figure 7 shows. For the amine-functionalized Fe3O4, the value of Ms varies according to the amount of ethylenediamine. The values of Ms are decreased with the amount of ethylenediamine increase. According to Figure 8, the value of Ms is (a) 18.5 emu/g, (b) 16.1 emu/g, and (c) 15.5 emu/g. Since ethylenediamine is not magnetic, the magnetic properties of the composites are attributed to ferromagnetic properties of Fe3O4.
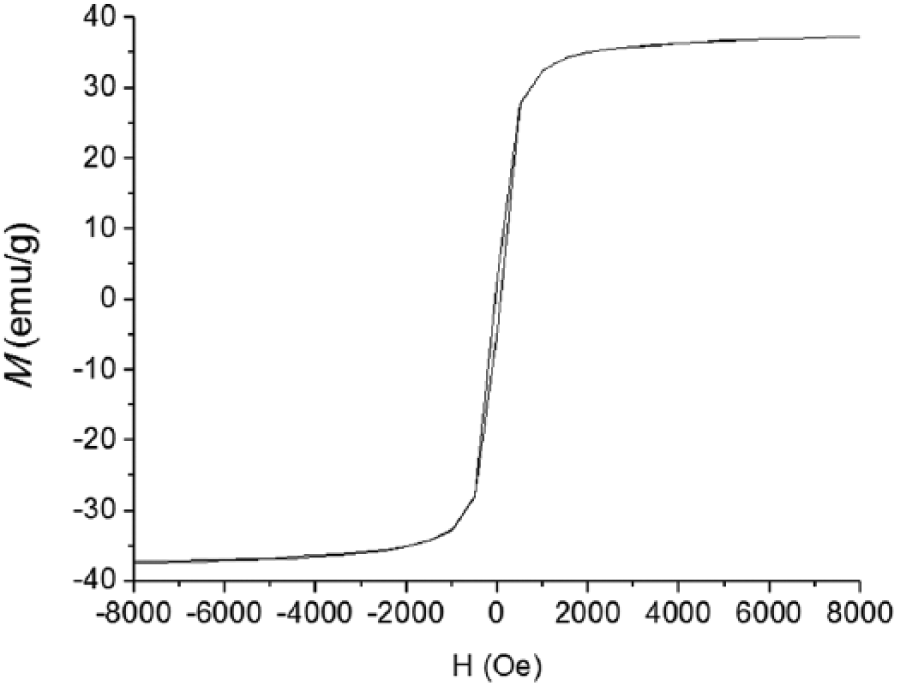
Magnetic hysteresis curves of bare Fe3O4 nanospheres without ethylenediamine.
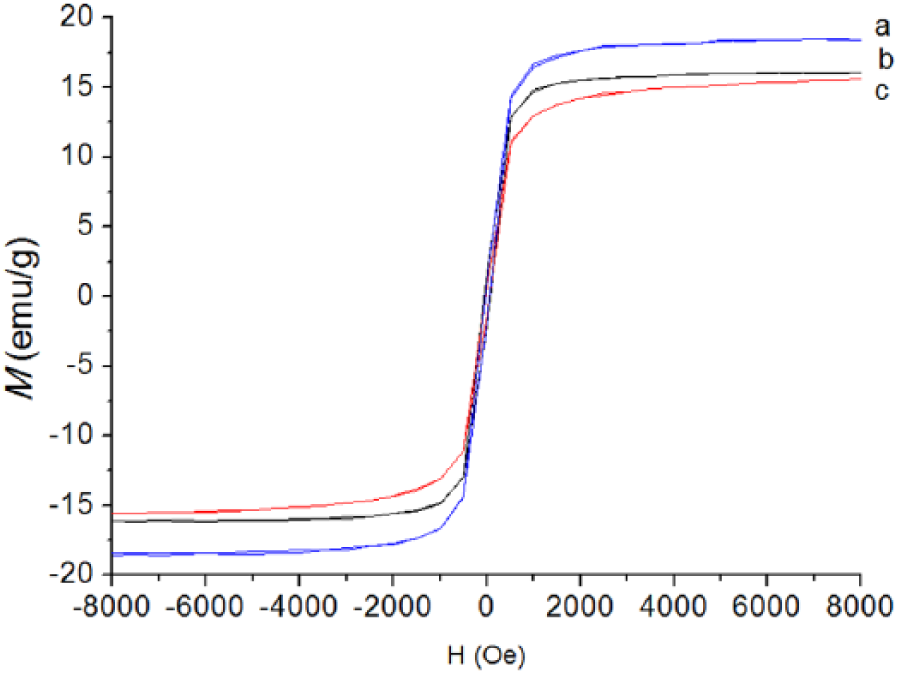
Magnetic hysteresis curves of as-prepared amine-functionalized Fe3O4 nanospheres with different amounts of ethylenediamine: (a) 5 ml, (b) 10 ml, (c) 15 ml. The amounts of FeCl3·6H2O were kept at 3 g.
Conclusion
In summary, micro/nanostructured Fe3O4 modified by ethylenediamine was synthesized successfully in organic media using FeCl3·6H2O as the only iron resource. Compared with traditional methods, this method has the following advantages. First, there is no need to control the reaction under a nitrogen atmosphere and just one salt is used as the iron resource. The growth and the surface modification of Fe3O4 crystalline nucleation happened at the same time. Second, monodispersed Fe3O4 micro/nanospheres were prepared without additional surfactant or external magnetic fields. Third, this method is preferable compared with the conventional organic phase method as the reaction condition is milder and less pollutant will be produced. Once the amino of ethylenediamine bonds to the Fe3O4 surface, not only is the oxidation of Fe3O4 prevented, but also the ability of Fe3O4 nanocomposites to bond to other components is enhanced, which will expand the applications for Fe3O4.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported by the Colleges Natural Science Foundation of Jiangsu (15KJA430003), the Production and Research of Jiangsu University of Technology (KYH16033) and Changzhou high-tech Key Laboratory (CM20153001).
