Abstract
Background:
Sickle cell disease (SCD) remains a significant public health challenge among tribal communities in India, causing high morbidity and mortality. Despite expanded hospital-based care, clinical outcomes have shown limited improvement due to persistent barriers in care delivery.
Design and methods:
A formative, mixed-methods study was conducted at Kasturba Hospital, SEWA Rural, Gujarat, to refine an SCD care model tailored for a low-resource setting serving a tribal population. Quantitative analysis of SCD registry data (July 2021–June 2022) assessed intervention coverage, adherence, and outcomes. Qualitative data from in-depth interviews with patients, caregivers, and healthcare providers explored barriers and facilitators. The Consolidated Framework for Implementation Research (CFIR) guided gap identification, while Expert Recommendations for Implementing Change (ERIC) informed strategy selection. Literature review and expert consultation further refined the model.
Results:
Findings showed low clinic adherence, high hospitalization rates (64.4/100 person-years), and severe anemia mainly due to nutritional deficiencies. Hydroxyurea was prescribed to 56% of eligible patients but clinical outcomes remained poor. Key barriers included transport difficulties, long waits, stigma, limited health literacy, operational constraints, and insufficient supervision. Model refinements included expanding outreach clinics, introducing structured counseling and supervision, enhancing digital follow-up systems, and adding routine micronutrient supplementation. Literature and expert reviews affirmed these adaptations’ feasibility and relevance.
Conclusions:
Using an implementation science framework, this study optimized an SCD care model tailored to a marginalized tribal population. By strengthening outreach, care protocols, counseling, and digital tracking, the model addresses persistent delivery gaps, illustrating the value of evidence-informed, context-specific intervention refinement in resource-limited settings.
Introduction
Sickle cell disease (SCD) is a genetic blood disorder that causes anemia, pain episodes, and organ damage due to abnormal, sickle-shaped red blood cells. SCD remains an important contributor to childhood mortality and premature deaths in the adult population. 1 The disease is more common among the scheduled tribe (tribal) population constituting 8.6% (68 million) of the total Indian population. 2 The prevalence of sickle cell heterozygosity (SCD trait) and SCD varies between 1%–40% and 1%–12%, respectively, in tribal population.3–5 Newborn screening and comprehensive care including hydroxyurea treatment, pneumococcal vaccination, blood transfusion and patient education, effectively reduce morbidity and mortality due to SCD.6–9
The management of SCD and coverage of the above clinical interventions has remained inadequate among tribal populations due to challenges in healthcare delivery, including geographical isolation, poor transportation infrastructure, limited health system capacity, and socioeconomic barriers. 5 The basic facilities required to manage patients are usually absent or suboptimal, even in high SCD prevalent tribal pockets of India. 10 Therefore, there is a need for a dedicated center for SCD care to ensure adequate services for prevention and treatment, as per the World Health Organization (WHO). However, the model of a national control programme developed in high-resource countries cannot be replicated in low-resource settings. 11 There is a need to develop a facility-based clinical care model for SCD in low-middle-income countries (LMIC) such as India specifically focused on the tribal population.
Several hospital-based SCD care models have been developed to improve coverage of key interventions and patient outcomes.12–16 These typically include comprehensive outpatient services, hydroxyurea therapy, counseling, and standardized care protocols. As for instance, the Paris model emphasized early screening and adherence to national guidelines, 16 and a United Kingdom (UK) program highlighted multidisciplinary adult care. 13 Despite growing efforts, key gaps persist in global SCD care models—especially for rural and tribal populations. Programs in LMICs are often urban-focused, with limited outreach to high-burden areas. 17 Challenges such as underuse of hydroxyurea, weak follow-up systems, and staff shortages remain common. 18 Few models use structured frameworks to adapt care models to local contexts. 19 In India, models in Gujarat and Tamil Nadu included structured follow-up and hydroxyurea provision for symptomatic patients.14,15 However, most published studies lacked rigorous evaluation designs such as absence of baseline comparison, small sample sizes, or limited tracking of clinical outcomes, thereby making it difficult to draw firm conclusions about effectiveness of these models. Hence, these gaps underscore the need for context-specific, implementation-informed interventions.
To improve the clinical outcomes of SCD patients, in 2014 a comprehensive hospital-based model was set in place at Kasturba Hospital, Society for Education Welfare and Action Rural (SEWA), rural Jhagadia, Gujarat. The prevalence of sickle cell disease is around 1.8% in this area. This comprehensive hospital-based SCD care model was developed on the basis of standard guidelines. 20 It had been evaluated in 2015 and 2021 to understand its effectiveness on coverage of proven interventions and clinical outcomes. The results suggested an improvement in coverage of proven interventions, although there was no improvement in clinical outcomes, including pain crisis, hospitalization and hemoglobin level.14,21,22 These findings highlighted the need to further refine the current SCD care model to improve clinical outcomes. The objective of this study is to conduct a formative evaluation of the current model and development of the refined/ optimized model in tribal area of Gujarat, India.
Material and method
Study setting
The study was done in Jhagadia block of Bharuch district in Gujarat. Gujarat, a western state in India, has a population of 60.4 million with 14.5% of the tribal population. 2 Jhagadia block has a population of 155,000, and 65% of the inhabitants are tribal.
The present study was conducted at Kasturba Hospital, a secondary care hospital run by one of the non-government organizations (NGO) SEWA Rural, beginning in 1980. It is a 250-bed hospital in the Jhagadia block of the Bharuch district and provides general and specialist outpatient and inpatient services. The hospital acts as the first referral unit for maternal and child health (MCH) services. 23
Current sickle cell disease care model at hospital
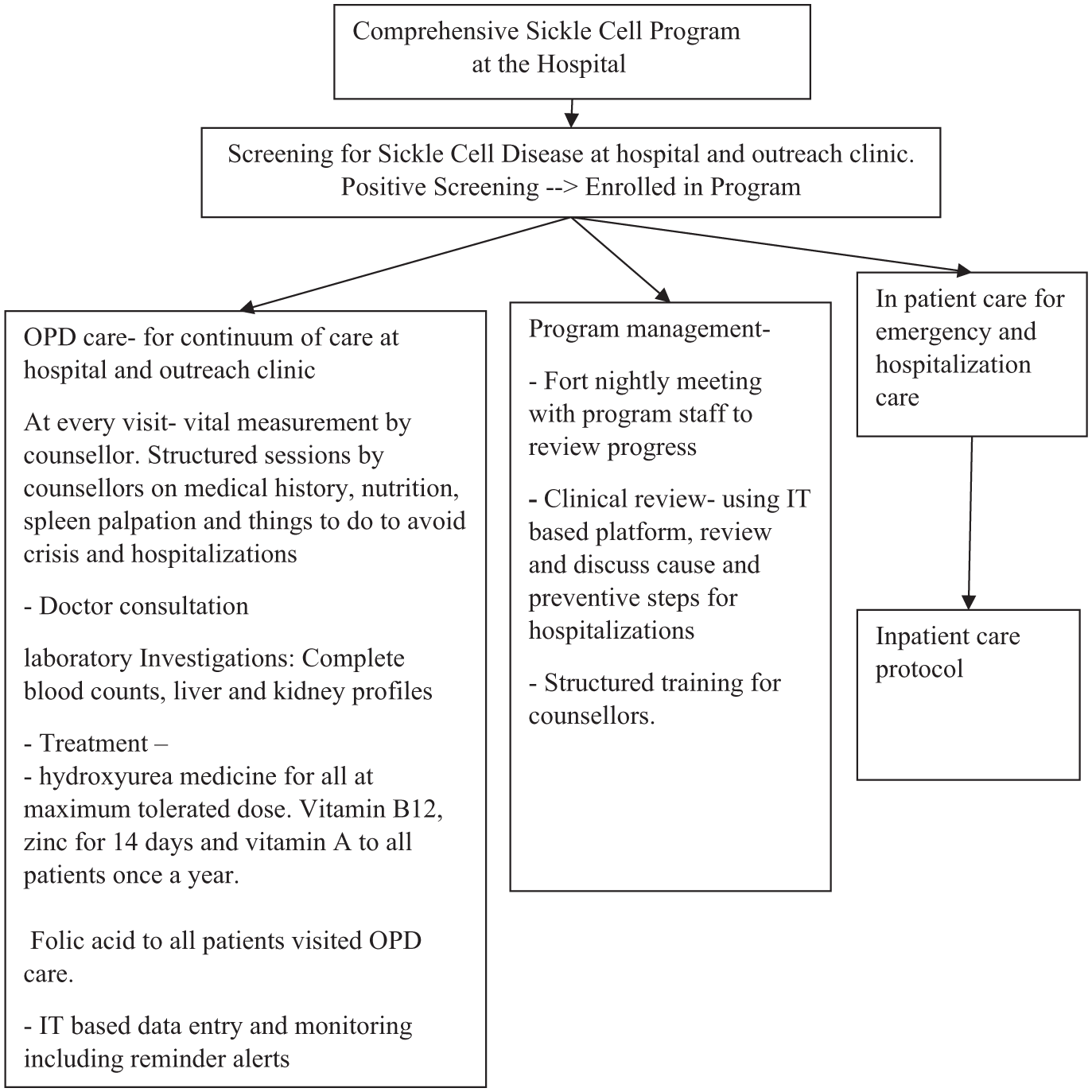
In study area, the sickle cell disease is prevalent among tribal population, so majority of SCD patients are from scheduled tribe population in this study. In February 2014, a comprehensive hospital-based program for SCD was launched to ensure continuity of care. The program included screening of newborn (within the first week of life), pregnant women and symptomatic adults by high performance liquid chromatography (HPLC), standardized outpatient/inpatient protocols, genetic counseling, and IT-enabled follow-up. The protocols have been updated periodically based on updated clinical guidelines. Initially all information were stored in paper-based register, but eventually, IT based system was implemented in 2016. The IT based system has been updated periodically over the years with additional features. All SCD patients were enrolled and followed up at every 3 months in a dedicated SCD clinic. Patients with complications or new complaints could return to the hospital at any time for unscheduled visits; no separate, more frequent review schedule was defined for these high-risk patients. Services included laboratory examination (including complete blood count, liver profile, and kidney profile), emergency care, inpatient management of pain crises, blood transfusions for severe anemia, pneumococcal vaccination, and hydroxyurea and folic acid therapy. Children under five received PCV-13 and penicillin prophylaxis (due to supply issues we used amoxycillin), adhering to the guidelines of Government of India. 24 A trained counselor provided education on disease, complications, and adherence to care. (Figure 1). A web-based management information system (MIS) was developed by SEWA Rural which captured all data like socio-demographic details like age, gender, economic condition etc. at registration and follow-ups. Over time, the system was updated to include clinical decision support and monitoring features. Counselors inputted visit details and received automated alerts for missed appointments, prompting immediate phone calls to reschedule patients. Additionally, the system tracked due dates for key interventions, generating lists of patients due for hydroxyurea and children due for amoxycillin prophylaxis. To ensure data quality, a medical officer reviewed data monthly. Monthly reports generated by the MIS—tracking key indicators such as outpatient attendance, hospitalization rates, and loss to follow-up—were reviewed during team meetings to guide targeted outreach and program adjustments.

Baseline comprehensive sickle cell disease program implemented at secondary-level care hospitals.
Study design
The formative research was done using explanatory mixed method design.
To assess the gaps in intervention coverage, clinical outcomes, and care delivery barriers among SCD patients in the tribal population, we collected data from two sources: (1) Secondary data analysis from the current HMIS system; (2). Exploratory qualitative data from stake holders.
For a comprehensive assessment of the SCD care model, consolidated framework for implementation research (CFIR) was used. The CFIR, a widely used determinant framework in implementation science, guides comprehensive data collection and refinement of the model. 19 CFIR has five components viz, (1) the outer setting included access barriers, stigma, among patients with SCD; (2) the inner setting focused on clinic operations, human resources, and supervision; (3) individual characteristics captured patient and provider knowledge, beliefs, and motivation; (4) intervention characteristics addressed perceptions of hydroxyurea and counseling; (5) the process domain included planning, follow-up, and review practices. CFIR guided to map stakeholders for qualitative assessment and develop the study tools.
Based on formative assessment guided by CFIR framework, we refined hospital-based SCD care model. At the refinement stage, we applied the Expert Recommendations for Implementing Change (ERIC) framework to select targeted and context-appropriate implementation strategies. 25 ERIC framework suggested the strategies based on identified barriers such as task shifting, outreach services, and enhanced supervision. This refined model, along with others developed through a literature review, was further improved and ultimately validated by experts (Figure 2).

Overview of steps in the development process.
Quantitative
In the first phase, we did a quantitative analysis of the sickle cell care program data to understand the clinical outcomes among patients registered under the SCD program. We obtained the data from SCD program database up to June 2022 in the form of an Excel spreadsheet and analyzed the outpatient data from July 2021 to June 2022. We included all patients who visited the outpatient clinic visit (OPD) at least once during last year. We excluded those patients who died or didn’t visit the clinic for last 1 year.
Outcome variables and statistical analysis
The patient profile was received from enrollment data and included age, sex, baseline severe status (yes/no). Baseline severity was defined as three crisis or three hospitalization or three blood transfusions within a year or presence of sickle cell related complication once a lifetime including avascular necrosis, splenic sequestration, hypersplenism, acute chest syndrome or stroke. To collect information on adherence to care and clinical outcomes following variables were evaluated - OPD follow up visits, hydroxyurea prescription and laboratory tests done among non-severe and severe patients, amoxycillin prophylaxis and vaccination status among children below 5 years, number of hospitalizations, cause of hospitalizations, and number of severe anemia cases (Hb <7.0 g/dl). Among patients with severe anemia, additional investigations were done to identify causes of anemia. These included serum ferritin and transferrin saturation for iron deficiency, and serum vitamin B12 levels. Results of these tests were extracted from the hospital laboratory information system and linked to the SCD registry for analysis.
We used SPSS version 25 software to analyze the data. The findings were presented using frequency and percentage. Based on the findings from the phase 1, the qualitative tools to understand gaps in the system (phase 2) guided by CFIR model were developed to understand factors associated with improvements in OPD follow up visits, hydroxyurea prescription and laboratory tests done among non-severe and severe patients, amoxycillin prophylaxis and vaccination status among children below 5 years and decrease in hospitalizations
Exploratory qualitative data collection
Mapping of stakeholders - The stakeholders included patients, care givers, and health care providers.
Qualitative assessment was done to understand the factors associated with adherence to care, gaps and good practices in the current system. We conducted interviews with 12 patients, 6 counselors and 2 doctors.
Patients were selected based on the following criteria. Patients with good adherence (taking hydroxyurea regularly, at least three follow-up visits at the OPD clinic within the previous year), poor adherence (zero OPD clinic visit within the previous year), good clinical outcome (no severe anemia, no crisis within the previous year), or poor clinical outcome (severe anemia or crisis or hospitalization within the previous year). We interviewed doctors and counselors from our hospital and the nearest government primary health center.
A semi-structured in-depth interview guide was developed separately for each participant category (patients, doctors, and counselors), based on predefined themes of barrier and facilitators in current system identified by the CFIR framework and findings from the quantitative phase. The guides included open-ended questions tailored to explore care-seeking behavior, barriers to adherence, service delivery gaps, and suggestions for improvement. Interviewers were trained in qualitative methods and followed the guide flexibly to allow probing and clarification based on participant responses. An experienced researcher conducted detailed interviews with patients and health care providers by visiting their respective homes or workplaces. All interviews were conducted in Gujarati language. The participants were informed about the purpose of the study, their voluntary participation, and confidentiality of responses. After this, written informed consent was obtained before each interview. Data collection continued until saturation was attained. Interviewer recorded all the interviews. Interviewer transcribed and translated all the interviews into English.
Data analysis
Thematic analysis framework was adapted to analyze the qualitative data analysis to identify the themes and patterns. The analysis focused on understanding and documenting the facilitators and barriers in care.
The researchers transcribed all the transcripts from the recordings, and all the transcripts were uploaded to the Max-QDA version 24 software. An expert reviewed all the transcripts and certified them. We developed the themes by reading the transcripts in the software and marked them accordingly. We also sent transcripts in the local and English languages and all interviews to an expert to certify that the transcripts were made as per the interviews. For patients, our purpose was to identify factors contributing to adherence and clinical outcomes. We identified the sentences in patient interviews via software and marked them as good adherence and poor adherence. All sentences were coded under the themes. The similar codes were merged into the sub themes. The qualitative component adhered to the Consolidated Criteria for Reporting Qualitative Research (COREQ) guidelines. A completed COREQ checklist is included as Supplemental File 1.
Ethical considerations
The study protocol was reviewed and approved by the Institutional Ethics Committee (IEC) of SEWA Rural (Ethical Approval Number: SR/IEC/2021/03/01). Informed consent was obtained from all participants prior to data collection.
Results
Quantitative
All patients were from scheduled tribe population, 556 (56%) were females and 684 (69%) of patients were unmarried. Most participants were residents from Bharuch district 641 (65%), followed by Narmada 298 (30%) and other districts 54 (5%). Regarding parental education, 592 (60%) had studied up to 1th–8thstandard, 179 (18%) up to 9th–10th, and 119 (12%) up to 11th–12th standard. Only 33 (3%) had diploma or higher education, while 46 (5%) had no formal education or were illiterate. The average (SD) age of patients at enrollment was 15.2 (10.7) years, and 18.7 (11.2) year currently. (Table 1). We didn’t do genotype for all patients but for some of the newborns genotype have been done in past and presented in different paper. 26 Alpha-thalassemia deletions were present in 95% of the newborns. In terms of beta-globin genotypes, the cohort was predominantly HbSS (98%), with HbS-β thalassemia accounting for less than 2%.
Sociodemographic characteristics for the patients registered at comprehensive sickle cell disease program in the tribal area of Gujarat, India.

Number.
Standard deviation.
These patients contributed to 671 person-years (PY) of follow-up, during which 481 hospitalizations occurred (64.4/100 PY), predominantly due to Vaso-occlusive crises and severe anemia. A small subset—90 (9%) of patients—accounted for nearly (224) 47% of all hospitalizations. Among them, 443 (47%) of non-severe SCD patients had at least one visit at OPD clinic. While 409 (66%) of severe SCD patients visited clinic regularly at every third month. Similarly, laboratory testing was done regularly at every third month among severe SCD patients. Total 17 patients died during this period. 556 (56%) of the patients received HU within past 6 months (Table 2).
Quality of care matrix for the comprehensive sickle cell disease program in the tribal area of Gujarat, India.
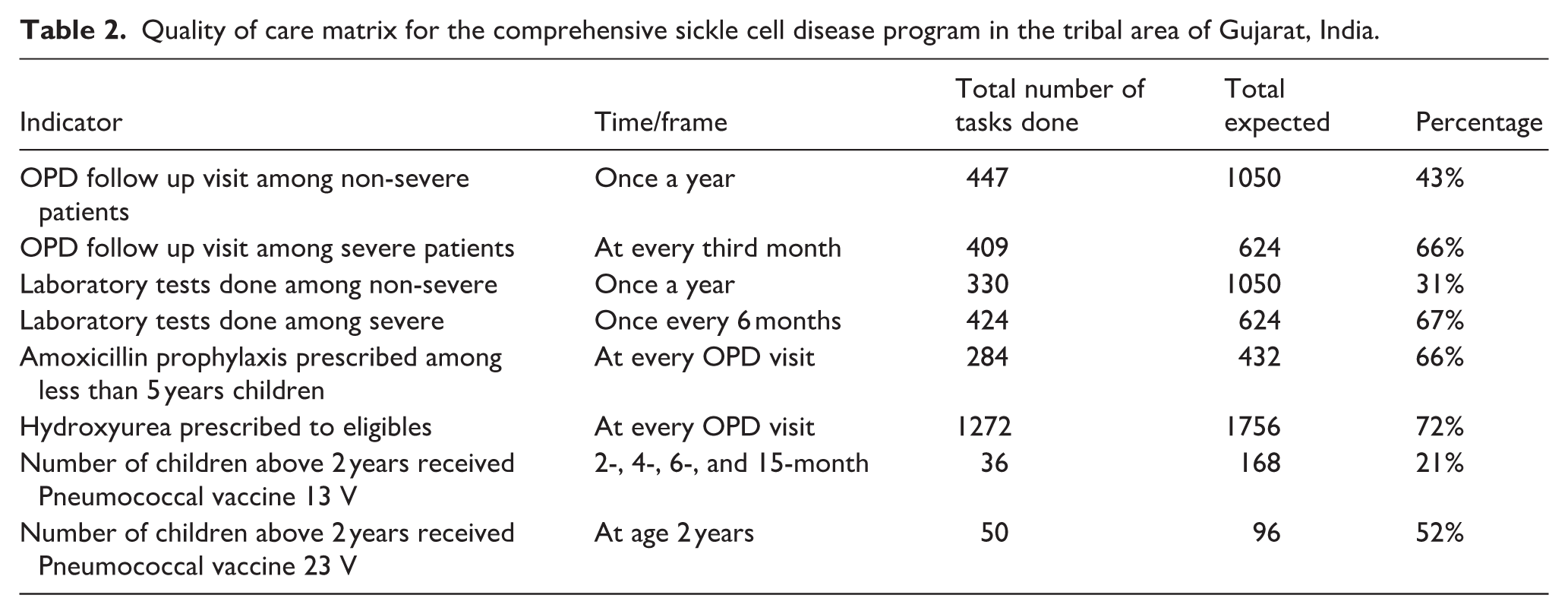
Among 96 cases of severe anemia (14.3/100 PY), causes were documented in 75 (78%), with iron deficiency 30 (40%), B12 deficiency 8 (11%), and SCD-related causes 36 (35%) being most common. hemoglobin levels improved to >7 g/dl in 52 cases (69%).
Qualitative
The findings were thematically analyzed and organized as per the CFIR.
Patients and caregivers identified transportation difficulties, long waiting times, and competing social responsibilities as major barriers to regular clinic attendance. The burden of travel from remote tribal areas and time spent waiting for lab tests and consultations reduced motivation to follow up.
“Vehicles are not available and the laboratory takes a long time.” — said Patient, female, 28 years “From morning to evening we sit in the clinic, it becomes boring.” — Patient, female, 21 years
Social stigma and limited community understanding of SCD further discouraged adherence, especially among adolescents.
“He doesn’t want to come to the clinic. He says, ‘Why only me?’ and gets angry if anyone talks about it.” Caregiver of 15 years old male patient
2.
The care model faced operational limitations, including high patient load, shared OPD space, and absence of structured supervision. Counselors expressed opinion about unstructured roles and limited training in counseling and patient assessment.
“We have clinic on Thursday, and the next day is full of inpatient work. We don’t get time for anything.” Said one counsellor from the hospital “We didn’t receive formal counselling training. We learned by observing.” Said one counsellor from the hospital
3.
Patients had poor understanding about hydroxyurea’s purpose and potential benefits, contributing to dropout and irregular follow-up.
“I take these pills as doctor advised us. I don’t know will this benefit to me or not”— Patient, male, 30 years “I was expecting relief from pain, but it didn’t happen, so I stopped attending SCD clinic.” Patient, male, 20 years
Patients who are aware about action of medicine, tends to take regular medications.
“She takes the medicine regularly at the same time every day. She knows that capsule prevents the pain” — Caregiver of 15 years old girl
4.
Some patients didn’t find medicine effective to reduce their pain.
“I came regularly for 2 years; I took all medicines prescribed by doctor but still feel pain. Nothing has changed.” Patient, male, 20 years
There was a felt need among some of the doctors to change the treatment protocols. They mentioned to provide micronutrients including B12, iron as part of routine care.
“The micronutrient deficiency including Vit B12 deficiency, iron deficiency and zinc deficiency is common in our area. That presents with anaemia, might be severe among SCD patients” mentioned by a doctor during the interview “As per recent guideline, hydroxyurea medicine supposed to provide to all SCD patients” mentioned by a doctor during the interview
5.
Patients appreciated the proactive calls by counselors. The phone calls lead to decrease the loss to follow up among patients.
“Whenever we miss the visit, counsellor madam calls us and reminds us, so we come regularly” said care giver of 10-year-old patient
Refining the intervention
Table 3 presents key interventions introduced to refine the care model into Model 2, based on implementation gaps identified using the CFIR framework and addressed through strategies drawn from the ERIC taxonomy. 25
CFIR-based mapping of gaps and interventions introduced in Model 2.

Each component of Model 2 directly responds to barriers uncovered in the baseline assessment. For instance, under the Outer Setting domain, limited access to care was addressed by establishing an outreach outpatient clinic. Thus, Model 2 increases the number of OPD clinic days per month but retains the planned three-monthly review schedule for each patient. In the Inner Setting, poor feedback mechanisms were countered by introducing an IT-enabled clinical review platform. The characteristics of individuals domain highlighted a need for improved health literacy, leading to the inclusion of structured counseling sessions during every visit. These practical solutions were identified by CFIR and matched with appropriate strategies from the ERIC taxonomy, such as “Change service sites; Increase access to care,” “Define professional roles ; Conduct ongoing training,” and “Audit and provide feedback; Use data experts to inform strategy.” 25
This combined CFIR-ERIC approach ensured that each change in Model 2 was evidence-informed that is, based on stakeholder insights and quantitative assessment, feasible to implementation, tailored to the SCD care model at SEWA Rural with Table 3 explicitly mapping each CFIR domain barrier to the corresponding intervention and ERIC strategy.
Optimizing the intervention
To further optimize Model 2 and ensure contextual and scientific relevance, we conducted a targeted literature review of existing sickle cell disease (SCD) care models in India by June 2021. Databases searched included PubMed, Google Scholar, and the Cochrane Library. We found total 57 articles from all 3 databases. The most comparable program was identified in Tamil Nadu. 15 This model supported a comprehensive care model. Although that model, prioritized community-based screening and provision of hydroxyurea for all patients with a history of at least one vaso-occlusive crisis. By contrast, our baseline model restricted hydroxyurea to patients with severe disease.
We then engaged five national and international experts across hematology, public health, and program officers. Their input supported most proposed interventions and led to three key refinements 1 : emphasis on monitoring hydroxyurea adherence and toxicity, 2 incorporation of iron profile testing for anemia, and 3 use of a digital platform for follow-up and hospitalization review.
These insights were synthesized using the CFIR constructs (e.g. Intervention Characteristics, Characteristics of Individuals, and Process) and implemented through ERIC strategies like “promoting adaptability,” “engaging opinion leaders,” and “tailoring strategies to context.” This participatory process resulted in the final refined model of care (Table 3), illustrated in Figures 3 and 4.
Comparison of baseline and refined SCD care models.

Refined a comprehensive sickle cell disease program implemented at a secondary level care hospital.
Discussion
This study outlines the development and optimization of a hospital-based sickle cell disease (SCD) care model (Model 2) for a tribal population in India, using the CFIR framework for gap analysis and the ERIC taxonomy for strategy design. Earlier SCD programs—such as the door-to-door screening and hydroxyurea distribution initiative in Anuppur, 27 and PATH-led district-level integration of primary care services in Gadchiroli 28 demonstrated initial feasibility but were hindered by inadequate monitoring, medication supply inconsistencies, and weak supervision. Other regional screening efforts centered on primary healthcare platforms lacked structured treatment pathways and adherence support. 29 By contrast, our study is novel in that it systematically maps identified implementation barriers to targeted ERIC strategies, incorporates stakeholder and expert input, and integrates IT-enabled outreach and follow-up mechanisms to enhance scalability and contextual fit for a hospital-linked SCD care model.
The challenges of accessing hospital-based care in remote tribal settings observed in this study are consistent with previous reports from India and other low-resource settings. Poor transportation, high indirect costs, and limited awareness have been cited as persistent barriers to care among tribal populations. 10 Similar challenges have been documented in sub-Saharan Africa, where individuals with SCD face difficulty accessing care due to poverty and weak health infrastructure. 17 Our finding that patients did not perceive significant improvement in symptoms despite treatment highlights concerns related to suboptimal hydroxyurea dosing. Although hydroxyurea has proven efficacy in reducing SCD complications, 30 under-dosing due to safety concerns or lack of dose titration is common in Indian settings, as seen in recent trials and practice surveys.18,31 Furthermore, severe anemia frequently caused by nutritional deficiencies such as iron and vitamin B12 deficiency contributes significantly to complications in SCD.32,33 Our intervention aimed to address these clinical gaps through both optimized hydroxyurea dosing and the inclusion of nutritional supplementation, which may be critical to improving outcomes in this population.
The frequency of hospitalization is a well-established predictor of poor survival among individuals with SCD, particularly in those experiencing recurrent vaso-occlusive crises, severe anemia, and organ damage. 34 One unexpected observation in our study was the limited impact of initial hospital-based interventions on reducing rehospitalizations, especially among patients with severe symptoms. This finding aligns with global evidence suggesting that high-risk SCD patients require individualized, multimodal care plans that extend beyond standard protocols. 34 Post-discharge follow-up, nurse-led education, and EMR-based tracking have been associated with reduced readmissions. 35 In our context, the use of EMR contributed meaningfully to continuity of care. Our refined model incorporated these elements, along with targeted follow-up strategies, which may enhance long-term adherence and outcomes. These findings suggest that the quality and continuity of care—not just access—are critical determinants of intervention success, particularly in underserved tribal populations.
A notable strength of this study is that it builds on previous work conducted at the same center, utilizing existing electronic medical records and stakeholder insights. The use of both quantitative and qualitative methods allowed for a comprehensive understanding of implementation barriers and facilitators. Notably, gap identification and intervention development were systematically guided by the CFIR and the ERIC compilation of strategies. This use of established implementation science frameworks enhances the credibility, transferability, and theoretical grounding of the intervention. The optimized model includes both clinical and programmatic improvements, and each step in the development process was described in detail, supporting transparency and reproducibility. Involving stakeholders from diverse cadres strengthened the feasibility and contextual relevance of the intervention. All qualitative interviews were conducted by a trained medical doctor, ensuring methodological consistency.
This study had certain limitations. First, the findings are based on a single-center model, which may limit generalizability. Second, some self-reported qualitative data may be subject to recall or social desirability bias. Third, the long-term impact of the refined model will require further evaluation. The next phase will evaluate the refined model’s impact on clinic adherence and clinical outcomes.
Conclusion
This formative research used mixed methods to refine a hospital-based SCD care model in a tribal region of Gujarat, guided by the CFIR and ERIC frameworks. Quantitative data revealed poor clinic adherence and high hospitalization rates, while qualitative insights highlighted barriers such as travel difficulty, long waits, and limited awareness. The refined model addressed these gaps through outreach clinics, task shifting, IT-enabled follow-up, and standardized protocols. The ERIC framework guided the selection of targeted implementation strategies. Expert consultations and literature review confirmed the model’s feasibility and contextual fit. This study demonstrates how implementation science can adapt evidence-based interventions for marginalized populations.
Supplemental Material
sj-docx-1-phj-10.1177_22799036261428569 – Supplemental material for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India
Supplemental material, sj-docx-1-phj-10.1177_22799036261428569 for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India by Kapilkumar Dave, Abhay Nirgude, Gayatri Desai, Sumeet Devare and Shrey Desai in Journal of Public Health Research
Supplemental Material
sj-docx-2-phj-10.1177_22799036261428569 – Supplemental material for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India
Supplemental material, sj-docx-2-phj-10.1177_22799036261428569 for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India by Kapilkumar Dave, Abhay Nirgude, Gayatri Desai, Sumeet Devare and Shrey Desai in Journal of Public Health Research
Supplemental Material
sj-docx-3-phj-10.1177_22799036261428569 – Supplemental material for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India
Supplemental material, sj-docx-3-phj-10.1177_22799036261428569 for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India by Kapilkumar Dave, Abhay Nirgude, Gayatri Desai, Sumeet Devare and Shrey Desai in Journal of Public Health Research
Supplemental Material
sj-docx-4-phj-10.1177_22799036261428569 – Supplemental material for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India
Supplemental material, sj-docx-4-phj-10.1177_22799036261428569 for A mixed-methods evaluation to optimize the existing hospital based sickle cell disease care model from a tribal area of Gujarat, India by Kapilkumar Dave, Abhay Nirgude, Gayatri Desai, Sumeet Devare and Shrey Desai in Journal of Public Health Research
Footnotes
Acknowledgements
We are thankful to local community members; and to the staff of the Kasturba Hospital for providing health care to SCD patients. We are thankful to Dr. Apoorva Pandey for her insightful comments to improve this manuscript.
Ethical considerations
The study proposal received permission from ethics committee of SEWA Rural, Gujarat (Ethical Approval Number: SR/IEC/2021/03/01). We removed the personal identifiers like name from the data files and de-identified data used for analysis.
Author contributions
Conceptualization: Kapilkumar Dave, Abhay Nirgude, and Shrey Desai. Methodology and Investigation: Kapilkumar Dave, Shrey Desai, Gayatri Desai, and Sumeet Devare. Formal Analysis: Kapilkumar Dave and Abhay Nirgude. Writing – original draft: Kapilkumar Dave and Abhay Nirgude. Writing – review & editing: Kapilkumar Dave, Abhay Nirgude, Gayatri Desai, Sumeet Devare, and Shrey Desai. All authors have read and agreed to the published version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Dataset used for the study is available and shared at: 10.6084/m9.figshare.26763388. The data can be viewed publicly.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
