Abstract
Background:
Sickle cell disease (SCD) management requires comprehensive care coordination and specialised resources, particularly in endemic regions. We describe the implementation and outcomes of a unique model, the Integrated Centre for Sickle Cell Disease (CID), in French Guiana, a region with high SCD prevalence.
Design and methods:
We established a comprehensive SCD care centre incorporating dedicated clinical space, specialised staff, and integrated emergency services. The model included 24/7 specialist availability, priority admission protocols, and a day hospital facility for managing acute complications.
Results:
The CID currently serves 740 patients (280 children, 406 adults, including 23 elderly patients aged 60–72 years). In 2022, the centre documented 4673 patient visits, comprising 2209 consultations and 417-day hospitalisations. Of these day hospitalisations, 229 were for vaso-occlusive crises, 147 for erythropheresis, 13 for therapeutic bleeding, and 28 for plasmaphaeresis. Most notably, following CID’s establishment, emergency room deaths were eliminated, compared to 2–3 annual deaths previously reported due to acute splenic sequestration. The centre maintained this achievement over eight consecutive years.
Conclusions:
The CID model demonstrates successful implementation of comprehensive SCD care in a resource-limited setting. The complete elimination of emergency room deaths over 8 years, along with improved patient retention and follow-up, suggests that this model could serve as a template for other regions with high SCD prevalence. The success of this approach highlights the importance of integrated, specialised care in managing SCD effectively.

Significance for public health
Patients with sickle cell disease (SCD) require care coordination, complex medication regimens, sustainable transportation means to access treatment centres, dedicated social support, and regular follow-up care, making hospital management of SCD challenging and complex. Implementation of team-based dedicated care and support systems can help in the better management of SCD. A dedicated comprehensive care including health and ancillary services is required to address the needs of patients with SCD. Such a dedicated centre has been established in French Guiana. Since establishing this unique model, no emergency room deaths have been reported in the last 8 years. In addition, improved patient retention and follow-up have been observed, leading to better treatment outcomes. Similar models can be implemented in other regions with higher SCD cases for better treatment and management outcomes.
Introduction
Sickle cell disease (SCD) is a complex inherited blood disorder that affects millions of individuals worldwide. Although SCD is more prevalent among those whose ancestors originated in sub-Saharan Africa, Saudi Arabia, India, the Caribbean and countries in the Mediterranean region like Turkey, Greece, and Italy, 1 migration of people with high rates of sickle cell trait (SCT) and SCD from these regions has contributed to the global spread of the disease, especially to North America and the Caribbean. 2 SCD is characterised by abnormal, crescent-shaped red blood cells that can cause a multitude of health complications, often requiring frequent hospitalisation and specialised care to manage symptoms, prevent crises, and address complications effectively. 3
The hospital management of SCD faces numerous challenges, ranging from inadequate pain management to a limited understanding of the disease’s unique needs.4,5 These challenges are compounded by the complex nature of care coordination, which includes managing intricate medication regimens, ensuring sustainable transportation to treatment centres, providing dedicated social support, and maintaining regular follow-up care. 4
The American Society of Haematology (ASH) 2020 guidelines for sickle cell disease emphasise the importance of comprehensive care delivery, recommending dedicated healthcare teams and specialised centres equipped with multidisciplinary expertise to manage both acute and chronic complications. The National Institutes of Health (NIH) guidelines align with ASH recommendations, emphasising preventive care through newborn screening, prophylactic penicillin, and comprehensive vaccination schedules, while also stressing the need for coordinated care transitions from paediatric to adult healthcare services. Both guidelines strongly advocate for standardised protocols for pain management, hydroxyurea therapy, and regular screening for common complications, highlighting the critical role of specialised centres in delivering evidence-based care.6,7 In regions like French Guiana, where primary healthcare serves as the foundation of the system, particularly through Centres délocalisés de prévention et de soins (CDPS) in remote areas, the limited availability of healthcare professionals, especially in general practice and specialised fields, further complicates care delivery. 8
To address these challenges and improve the overall quality of care, a holistic approach to hospital management for SCD is essential. This comprehensive approach encompasses a wide range of considerations, including the coordination of multidisciplinary teams, patient education and empowerment, pain management strategies, psychosocial support, and the integration of innovative technologies and treatment modalities. By adopting a holistic approach, hospitals can not only enhance the well-being and outcomes of these patients but also minimise the burden on healthcare systems and improve resource allocation. In this article, we not only focus on a plausible holistic approach to managing SCD in a hospital setting but also share our experiences and best practices to provide comprehensive support to SCD patients during their hospital stay.
Methods
Study design and setting
This article describes the implementation and outcomes of a comprehensive SCD management model at the Integrated Centre for Sickle Cell Disease (CID) in French Guiana.
Model development and implementation
The CID was established as a dedicated comprehensive care centre to address the high burden of SCD in the region. The centre implemented a multidisciplinary approach incorporating the following key components:
Staffing and resources
The centre is led by a programme director/lead specialist responsible for developing care protocols, executing audits, and staff training. The core team includes dedicated nursing staff trained in acute and chronic pain management, social workers and patient coordinators, dedicated psychologists, nurse coordinators for patient education, round-the-clock SCD specialists accessible via the emergency department.
Physical infrastructure
The centre operates as a classic comprehensive SCD centre with dedicated clinical space and staff, specialised infusion space, day hospital facilities, priority admission circuit for emergency room access, dedicated consultation areas.
Care delivery model
The CID implemented several key operational elements: structured referral pathways from physicians, emergency departments, and user associations, patient screening and segregation protocols based on disease severity, comprehensive day hospital services including routine check-ups, therapeutic education, and counselling, integration with national bone marrow transplantation programmes, participation in regional and national research initiatives.
Data collection and outcomes assessment
The centre maintains comprehensive records of patient visits, hospitalisations, and clinical outcomes. Key metrics tracked include number of patient visits, day hospitalisation rates, emergency room visits, mortality rates, patient retention, and follow-up rates.
Setting up a dedicated SCD centre especially in highly endemic regions
The importance of comprehensive care for SCD has been acknowledged for long, as is evident by the implementation of the National Sickle Cell Disease Control Act in 1972. 9 Following this, the National Heart, Lung, and Blood Institute (NHLBI) initiative created several SCD centres in the United States; these centres focussed on comprehensive care, research, and engagement with organisations that are community-based, primarily for children with SCD. 10 The initiative successfully decreased child death rates by screening newborns and providing penicillin prophylaxis. However, the lack of funding led to the closure of these centres over time. 10 One of the most important challenges in countries with high SCD prevalence is the lack of appropriate healthcare infrastructure to manage these patients. This may be due to the prevalence of other tropical and endemic infections (such as in African and sub-African regions, Caribbean), lack of appropriate healthcare systems (India), and lack of awareness and knowledge of the disease (Saudi Arabia).2,11,12 One such territory is French Guiana (a French overseas territory located in South America), which is dominated by the Afro-Caribbean population. Sandwiched between Brazil on one side and little-known Suriname on the other, French Guiana is the only European territory in the Amazon with about 50% of its population living below the poverty line; this territory is home to most endemic and/or epidemic infections such as malaria, leishmaniasis, Chagas disease, histoplasmosis, or dengue fever. 13 According to the 2010 report of the Association French Institute for Screening and Prevention of Childhood Disabilities (AFDPHE), one in 227 people living in French Guiana had SCD and 10% of the population had SCT. Hospital data between January 2010 and July 2011 showed that the male/female sex ratio was 0.75 and the mean age of hospitalised patients was 28.5 years; about 67% presented an SS phenotype, 10% Sβ thalassaemia (Sβ° and Sβ+) and 23% SC. According to a recent study, it is estimated that there were about 1200–2000 patients with SCD in French Guiana (overall population 300 000) with frequent haemoglobin S haplotypes like Benin (65.9%), Bantu (20.5%), Senegal (7%), Cameroon (4%), Arab Indian haplotypes (1%), and atypical (6%). 14 Most SCD hospitalisations were due to vaso-occlusive crisis (VOC, 86%) and infections (16%) with 80% on patient-controlled analgesia with morphine. Reduced temperature and lower humidity were two independent risk factors for increased VOC cases (p = 0.0128 and 0.0004, respectively) and 15.5% of patients had severe VOC associated with the influenza epidemic. Between January 2010 and December 2016, 49.2% of visits to the emergency room were SCD-related. 15
Since the spleen is one of the organs affected by VOC, patients with SCD are prone to grave bacterial infections due to asplenism. 16 Complicated pregnancies, malnutrition, priapism, iron overload, retinopathy, renal disease, pulmonary hypertension, and chronic pain are some of the other complications related to SCD.10,17 Furthermore, delayed growth and puberty, opioid use (and misuse) for chronic and acute pain management often lead to stigmatisation of these patients, all of which negatively affect the care-seeking behaviour and psychological well-being. 18 The varied complexity of the disease demands a multidisciplinary holistic approach towards treatment and management, which involves not just primary physicians, hospital staff, and caregivers, but also specialty doctors (Figure 1).
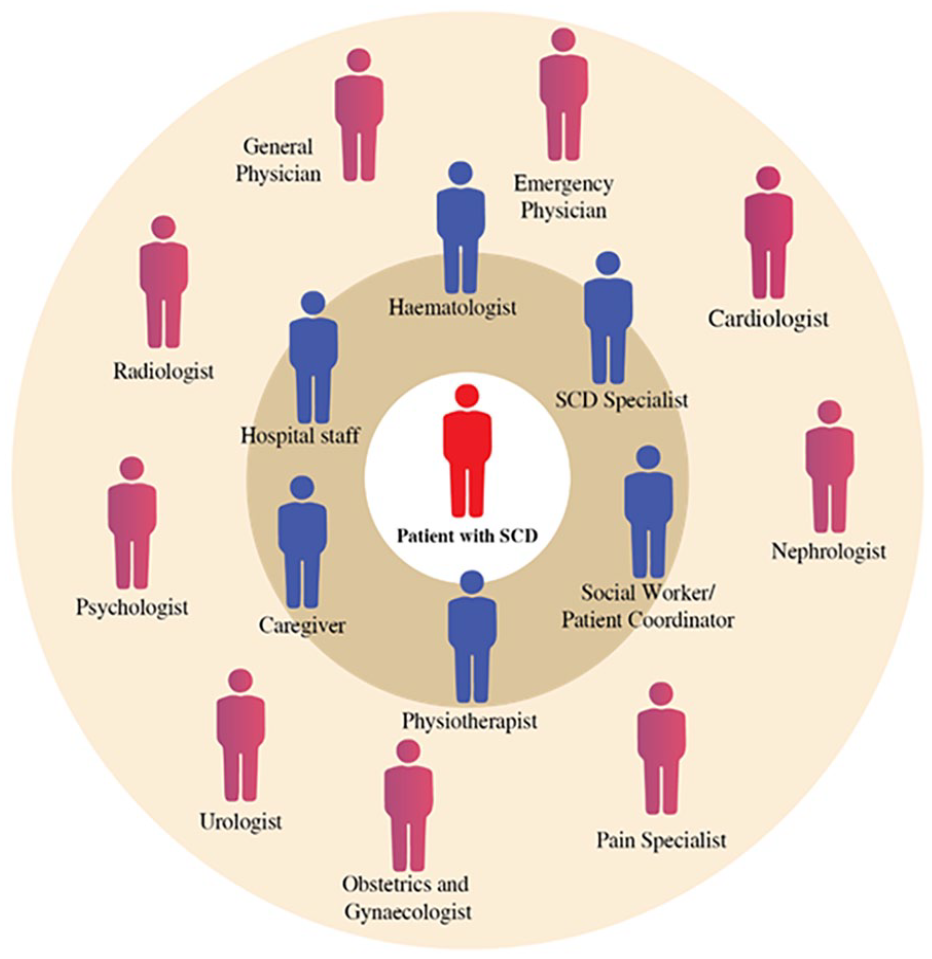
Multidisciplinary healthcare team framework for comprehensive sickle SCD patient care management.
Given the burden of this disease in French Guiana, it was imperative to initiate the Integrated Centre for Sickle Cell Disease (CID) to provide comprehensive management of SCD in these patients. Earlier studies have demonstrated how dedicated SCD care centres can result in better cost-effectiveness than episodic; emergency department (ED) based care.19–22 The objective of this centre was to establish a comprehensive SCD programme that would result in improved health outcomes and quality of life for patients, including reduced acute care visits and hospitalisations. The centre strengthened the emergency care and management of SCD by developing guidelines and protocols for SCD management and training emergency physicians to handle such emergencies adequately.
Comprehensive SCD care requires a multidisciplinary, well-planned, and organised team-based approach. The CID at French Guiana adopted the following holistic model to integrate all the service sectors relevant to SCD management under one roof (Figure 2).
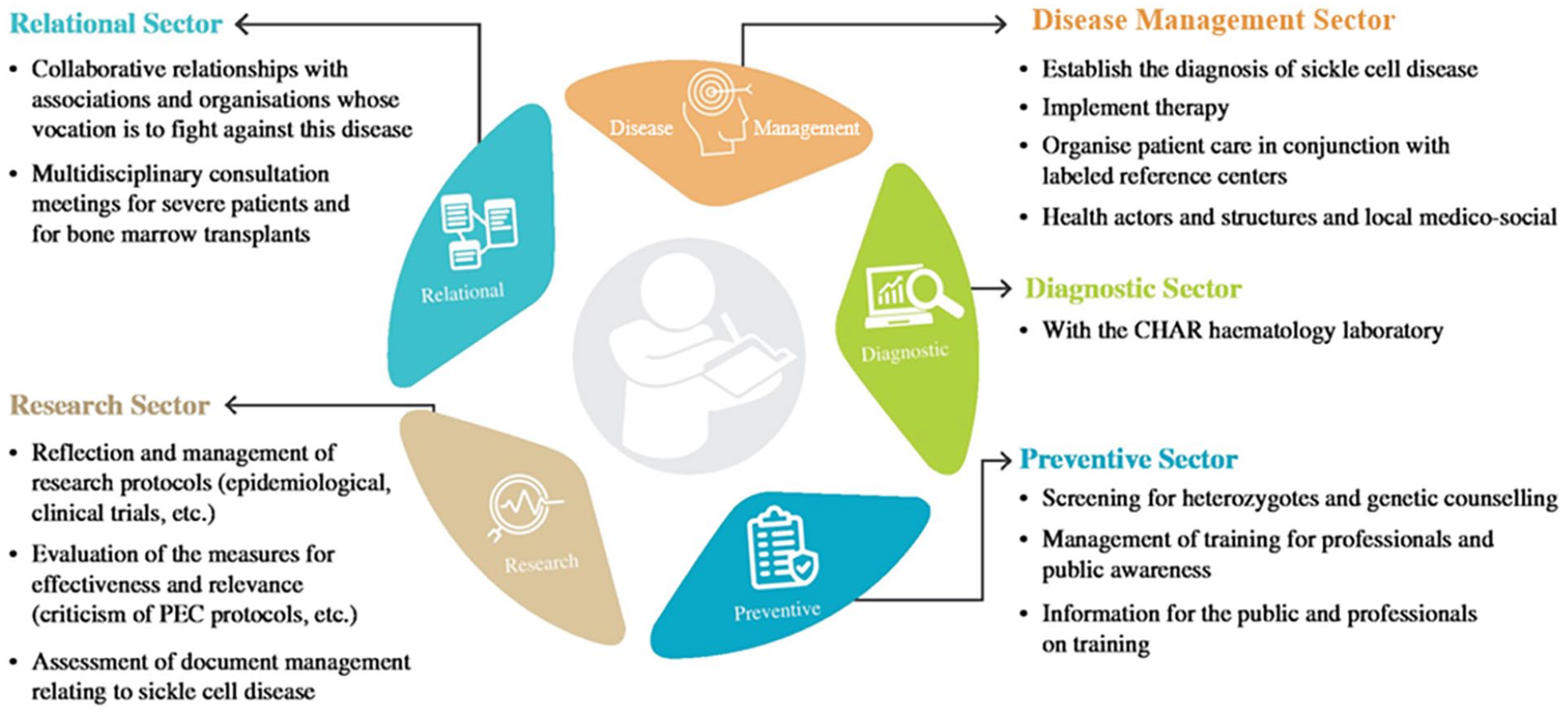
Comprehensive management of SCD at CID.
Components of an ideal SCD centre
Resources
At a minimum, any SCD centre should be led by a physician or specialist experienced in treating patients with SCD (programme director or lead specialist). The programme director or lead specialist should be responsible for developing care protocols, executing audits, training the staff, and sharing the overall responsibility for the management of the clinic. Additional roles of providing evidence-based pain management to patients with SCD as required may be undertaken depending on the expertise and specialisation. The programme director or lead specialist should undertake continuing professional development of relevance to this role and have an established plan for handling emergencies and absenteeism of other staff. The workforce required for any SCD centre includes at least one social worker or a patient coordinator along with dedicated nursing staff who are trained to offer acute and chronic pain management to patients, handle transfusions, and expedite timely access to specialists as required. A lead nurse may be employed who can help the programme director with the implementation of guidelines and protocols and conducting training and audits thereby improving the quality and oversight of the SCD centre. Consistent with this, the CID at French Guiana had several professionals working in conjunction with each other to help these patients with a better quality of life, optimal school and professional participation, and adequate social and family integration (Figure 3).

Multidisciplinary CID team for SCD management.
Having a dedicated psychologist and nurse coordinator for patient education in this SCD centre helped tremendously during the transition of paediatric patients to adult care settings and also counselling female patients for adopting appropriate contraception methods and preventing mother-to-child SCD transmission. The CID ensured the presence of an SCD specialist round the clock who could be reached via the emergency department. Moreover, continuous training was provided to emergency department staff to help them identify and deliver the most appropriate care to SCD patients.
One of the major concerns of any hospital is managing the attrition of healthcare professionals and training the newer staff and orienting them to the hospital protocols and processes. Countries in the Caribbean region are known to have high rates of overall emigration, especially from French Guiana and Saint Vincent. Majority of these emigrants are health workers, especially nurses and medical practitioners, thereby directly impacting the attrition rates. 11 This is one major challenge in French Guiana; however, people who come to work in Guiana receive 40% more salary compared to the conventional salary of French healthcare professionals. 23 Thus, incentivising to work as part of the dedicated task force of the CID, helped in retaining the special skilled staff. Incentivising was done in three ways: (1) financial incentive of 40% of their salary, (2) premiums for 5-year contracts, and (3) spousal hiring. Also, having a dedicated SCD centre coupled with resources having defined roles and responsibilities helped in the training and orientation of new staff for the smooth running of the centre.
Space
Several SCD centre models have been discussed in the past depending on the clinical space and availability of all resources; these include, classic comprehensive SCD centre (dedicated clinical space and staff), 19 embedded SCD care centre (clinical space is shared between programmes, to improve efficiency and reduce costs), 24 specialised medical home (comprehensive SCD care alongside primary care), 25 and hub and spoke model (wherein any one of the above centres serves as hub and patients are consulted remotely using telemedicine or through secondary clinics with limited clinical space). 26 Considering the vulnerability of patients with SCD to other infections, it is advisable to have dedicated clinical space and staff who will not come in contact with or are not shared with other patients. The CID in French Guiana is a comprehensive SCD care centre; patients are referred either by the attending physician, the emergency department (post-emergency consultations), or user association. All referred patients are screened and segregated into those requiring normal hospitalisation or ICU hospitalisations based on disease severity (Supplemental Figure 1).
A dedicated infusion space and a day hospital have been set up by the centre to decrease ED care and provide SCD-specific care in a timely manner (especially to manage pain crises). The day hospital staff is not just involved in screening patients for SCD and routine check-ups of these patients but also provides patients with therapeutic education and counselling. A priority admission circuit is also set up for quick admission of these patients to the emergency room; this includes an on-call CID physician who provides consultation in the emergency room as well as in the infectious disease unit and can also provide telephonic consultation if required. The CID also participates: (1) in the national RCP on bone marrow transplantation in patients with SCD, (2) as a regional reference structure on the management of SCD, and (3) in several epidemiological and clinical research projects at regional and national levels.
Impact of SCD care centre on disease burden
Several studies have demonstrated that patients treated in dedicated SCD hospitals have fewer emergency department visits, lower admission rates, and shorter hospital stays.19,27-29 One study reported a decrease in hospital admission by 43% and a 49% decrease in occupied bed days seen within 3 years in an SCD unit; furthermore, by the third year, 84% of patients with pain episodes were managed without the need for hospitalisation. 27 In CID, French Guiana, there were 4673 patient visits documented in 2022, which included 2209 consultations and 417 cases of day hospitalisations. Of these, only 229 had day hospitalisations due to VOC, while 147 cases were related to erythropheresis, 13 therapeutic bleedings, and 28 plasmaphaeresis (2 plasmaphaeresis per week). Prior to setting up the CID, there were at least 2–3 deaths in patients with SCD per year due to acute splenic sequestration, however, post-establishing CID, no emergency room deaths have been reported in the last 8 years. In addition, improved patient retention and follow-up have been observed, leading to better treatment outcomes. Hydroxyurea (HU) treatment is the gold standard that is followed, which is made available free of cost to the patients. Amongst the entire patient population, 20% of the SS and Sβ° patients are treated with HU, with a compliance of 80%. The CID also has patients on L-Glutamine (four patients with good compliance) and two patients on Voxelotor with poor compliance (treatment stopped after 3 months, without medical advice).
The CID currently attends to 740 patients with SCD, including 280 children and 460 adult patients; these include a small proportion of elderly patients as well (60–72 years old, n = 23). Children had more hospitalisations for VOC (60%) and acute chest syndrome (35%). Adults had more hospitalisations for infectious complications (50%; pneumonia, acute pyelonephritis) and chronic pain (30%). The majority of deaths occurred after the age of 18 years, wherein the causes of death were dengue, COVID-19 complicated by acute chest syndrome and multiorgan failure, and pulmonary embolism.
Strengths and limitations
The key strengths of this study include a comprehensive real-world implementation of an integrated sickle cell disease centre in a resource-limited setting, providing valuable insights for similar regions with high SCD prevalence. The longitudinal nature of the data, spanning multiple years, allows for meaningful assessment of the CID model’s impact on patient outcomes.
However, several limitations should be considered when interpreting these findings. The study is descriptive in nature and lacks a formal control group, making it difficult to definitively attribute improvements solely to the CID model versus other potential concurrent changes in healthcare delivery. The single-centre design, while providing detailed insights into the French Guiana experience, may limit generalisability to other settings with different healthcare systems or resource availability. Furthermore, while the study reports improvements in mortality and emergency room visits, it does not include comprehensive data on other important outcomes such as patient-reported quality of life measures, long-term complications, or detailed cost-effectiveness analyses.
Conclusion
Lifelong care is needed by children and adults with SCD, efforts to improve care are primarily focussed on children, while adults with SCD seek acute care, primarily for pain, in the emergency department. Therefore, there is a need for team-based comprehensive care including health and ancillary services to address the needs of children and adults with SCD. CID is a distinct model (training of emergency physicians, the implementation of paediatric and adult protocols, and the management of sickle cell emergencies, the implementation of emergency priority admission for sickle cell patients) that represents a novel approach for SCD management in French Guiana and could serve as a template for similar resource-limited settings in South America. Since the implementation of the CID model, an increase in the number of patients monitored, a reduction in overall mortality, an improvement in the quality of care, and a high level of patient satisfaction have been observed. Patients are thereby better-taken care of, which was confirmed by the association of sickle cell patients in French Guiana. The present model has successfully reduced the number of deaths in the emergency room. This is a successful model that can be implemented in other regions with higher SCD cases for better treatment and management outcomes.
Supplemental Material
sj-docx-1-phj-10.1177_22799036251356775 – Supplemental material for Breaking barriers: Enhancing hospital support for sickle cell disease management
Supplemental material, sj-docx-1-phj-10.1177_22799036251356775 for Breaking barriers: Enhancing hospital support for sickle cell disease management by Narcisse Elenga and Abdelaziz Fahem in Journal of Public Health Research
Footnotes
Acknowledgements
Writing assistance was provided by Turacoz Healthcare Solutions. This work was supported by Emmaus Medical Inc. We acknowledge that Dr. Fahem was affiliated with Emmaus Medical Inc. during the initial research and manuscript preparation; however, he is currently employed at Hemara Bio (
Author contribution
N.E and A.F contributed to the design and implementation of the research, to the analysis of the results and reviewing of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Elenega has received consultancy fees from Emmaus Medical.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
